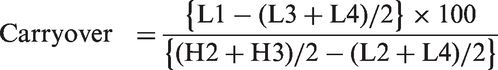Abstract
Background
We evaluated the performance of ultra-performance liquid chromatography-tandem mass spectrometry to measure serum 3-epi-25-hydroxyvitamin D3, 25-hydroxyvitamin D3 and 25-hydroxyvitamin D2 concentrations in 519 infant, paediatric and adolescent serum samples in Korea.
Methods
We used a Kinetex XB-C18 column and isocratic methanol/water (77.5/22.5, v/v) with 0.025% (v/v) high-performance liquid chromatography solvent additive flowing at 0.25 mL/min, yielding an 11 min/sample run time. A TQD triple quadrupole mass spectrometer in electrospray ionization positive ion mode with multiple reaction monitoring transition via an MSMS vitamin D kit was used to evaluate precision, carryover, ion suppression and linearity. Samples were prepared using the 4-phenyl-1,2,4-triazoline-3,5-dione derivatization method.
Results
Intra- and inter-run precisions were 1.23–13.28% and 1.02–10.08%, respectively. Group carryovers were −0.27% and 0.10%, respectively. There was no ion suppression. The calibration curve showed good linearity from calibrator Level 1 (11.75 nmol/L) to 6 (375 nmol/L) with R2 > 0.9999. The 3-epi-25-hydroxyvitamin D3 and 25-hydroxyvitamin D3 peaks were clearly separated in the extracted ion chromatogram. Infant serum samples 3-epi-25-hydroxyvitamin D3 concentrations were significantly higher than paediatric and adolescent concentrations.
Conclusions
The ultra-performance liquid chromatography-tandem mass spectrometry assay performed acceptably, clearly separating 3-epi-25-hydroxyvitamin D3 from 25-hydroxyvitamin D3. High 3-epi-25-hydroxyvitamin D3 concentrations were observed in infant but not in paediatric and adolescent serum samples.
Keywords
Introduction
Vitamin D is a fat-soluble vitamin that plays an important role in maintaining calcium homeostasis in the body. 1 Its deficiency is closely related with the occurrence of metabolic bone disease such as rickets in children. 2 It also regulates numerous cellular functions; accordingly, studies have revealed associations between vitamin D deficiency and the risk of metabolic syndrome, diabetes, autoimmune diseases and some types of cancer.3–8 Furthermore, vitamin D deficiency in infants due to an increase of breastfeeding has been highlighted, and vitamin D insufficiency is expected to be prevalent among school-aged children as well. 9
The analysis of 25-hydroxyvitamin D (25(OH)D) concentrations is used to diagnose hypovitaminosis D, as it is the precursor to active 1, 25-dihydroxyvitamin D (1, 25(OH)D) and has a longer half-life of three weeks compared with 24 h. 10 There are two types of 25(OH)D that can be found in the circulation: the endogenously derived 25-hydroxyvitamin D3 (cholecalciferol, 25(OH)D3) and 25-hydroxyvitamin D2 (ergocalciferol, 25(OH)D2), which is derived from plant sources and fish.11–13 25(OH)D3 is more potent and is normally present in higher concentrations in the body compared with 25(OH)D2. 10
The candidate reference method for 25(OH)D assessment, liquid chromatography-tandem mass spectrometry (LC-MS/MS), requires technical expertise, specialized equipment and expensive deuterated internal standards (IS). 10 Nevertheless, LC-MS/MS is increasingly popular for measuring 25(OH)D metabolites because of its high degree of accuracy and selectivity. 14
Recently, LC-MS/MS methods have identified an epimeric form of 25(OH)D3 that has been shown to contribute significantly to the total 25(OH)D3 concentration, particularly in infant populations. 15 Singh et al. 16 reported that 3-epi-25-hydroxyvitamin (3-epi-25(OH)D3) is found in 23% of infants and contributes 9–61% of the total 25(OH)D metabolites. On the other hand, another study reported that the commonly used immunoassays and LC-MS/MS-based methods for the measurement of 25(OH)D3 do not adequately detect or resolve the 3-epi-25(OH)D3 form. 17 In addition, few publications provide measurement information regarding this epimer form of 25(OH)D3.14,16–18 Although little is definitively known regarding the in vivo importance of 3-epi-25(OH)D3, clinical laboratories face the decision of whether or not to include 3-epi-25(OH)D3 in the measurement of total 25(OH)D.14–17
To address this issue, we evaluated the performance of an ultra-performance (UP) LC-MS/MS method with calibration traceable to National Institute of Standards and Technology (NIST) Standard Reference Material (SRM) and a PerkinElmer Kit (PerkinElmer, Turku, Finland) to separately measure the concentrations of 3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2. We then measured the concentrations of 3-epi-25(OH)D3 in 519 infant, paediatric and adolescent serum samples from Korean subjects using this method.
Materials and methods
Samples
The serum used in this study represented leftover material from samples drawn from 519 children who had visited the Department of Pediatrics, Ilsan Paik Hospital from November to December 2015. The selection criteria were age, sex and a surplus serum volume of >500 µL. We divided the population into three groups: (1) the infant group, with an age range <1 year; (2) the paediatric group, from 1 to 9 years old and (3) the adolescent group, from 10 to 19 years old. The male/female ratios of these three participant groups were 62/41, 88/121 and 88/119, respectively. All samples were protected from light and stored at −20℃ until analysis and then transferred to the LabGenomics Clinical Laboratories for analysis. Storage time of the specimens did not exceed two months. Institutional review board approval was obtained prior to the start of this study.
Materials and reagents
The analytes measured using the MSMS vitamin D Kit (PerkinElmer) and their corresponding IS, calibrators and controls are as listed below. For 25(OH)D2 and 25(OH)D3, the IS were 2H3-25(OH)D2 and 2H3-25(OH)D3, and the calibrators and controls were 2H6-25(OH)D2 and 2H6-25(OH)D3, respectively. Each MSMS vitamin D Kit contained reagents for 480 assays. This kit was designed to be used with the MSMS vitamin D Tool Box (3076-0010), the MSMS vitamin D Derivatization Box (3078-0010) and the MSMS vitamin D Maintenance Set (3077-0010). The MSMS vitamin D Tool Box contained microplates and covers used during sample preparation. The MSMS vitamin D derivatization box contained MSMS vitamin D 4-phenyl-1,2,4-triazoline-3,5-dione (PTAD) derivatization reagent, MSMS vitamin D Quench Solution and MSMS vitamin D high-performance liquid chromatography (HPLC) solvent additive. The MSMS vitamin D maintenance set contained one vial of 25(OH)D2 and 25(OH)D3 that was used for instrument setup and maintenance. We purchased acetonitrile, methanol and HPLC-grade water from J.T.Baker (Avantor Performance Materials, PA, USA). Analytical reagent-grade formic acid was purchased from Wako Pure Chemical Industries (Osaka, Japan). Conical-bottomed 96-well plates were purchased from Corning, Inc. (Cambridge, MA, USA). A Thermo shaker (MB100-4A) and an Eppendorf centrifuge (5810 R; Eppendorf, Hamburg, Germany) were used.
Sample preparation using a derivatized assay procedure
We reconstituted the vial of MSMS vitamin D IS by adding 1.25 mL acetonitrile and then shaking gently for 30 min. The vials of MSMS vitamin D controls and calibrators were reconstituted by adding 2 mL deionized water and then shaking gently for 1 h. The vial of MSMS vitamin D derivatization reagent was reconstituted in 30 mL acetonitrile. Daily precipitation solution (DPS) was prepared by diluting the reconstituted IS 1:100 with acetonitrile containing 0.1% formic acid. We added 100 µL serum to the appropriate number of wells of a 500 µL V-bottomed microplate, and added 200 µL DPS to each sample well. The plate was covered with a plate mat and was shaken at 750 r/min at room temperature for 10 min. The plate was centrifuged at 2000 g at room temperature for 40 min. We removed the cover mat and transferred 150 µL supernatant to a 200 µL V-bottomed microplate. The plate was placed under a stream of high purity dry nitrogen gas, and 50 µL MSMS vitamin D derivatization reagent solution was added to each well. The PTAD derivatized reaction is explained in online supplementary Figure 1. Next, the plate was covered with aluminum foil and was shaken at 750 r/min at room temperature for 15 min. We carefully removed the aluminum foil and added 50 µL MSMS vitamin D quench solution to each well. The plate was again covered with aluminum foil and shaken at 750 r/min at room temperature for another 10 min, then loaded onto the autosampler at room temperature. A 50 µL aliquot was injected into the UPLC-MS/MS system for analysis.
UPLC-triple quadrupole MS/MS
A Kinetex XB-C18 column (100 Å, 2.6 µM, 2.1 mm × 150 mm) (Phenomenex, Torrance, CA, USA) and isocratic methanol/water (77.5/22.5, vv) with 0.025% HPLC solvent additive flowing at 0.25 mL/min were used. We used a Waters Acquity UPLC MS/MS and a TQD triple quadrupole MS/MS (Waters) and electrospray ionization (ESI) with a positive-ion, multiple reaction monitoring (MRM) mode and flow injection. The run time was 11 min per sample. Mass Lynx version 4.1 (Waters) was used for data analysis.
Method validation: Precision
The intra-run precision was evaluated by analysing six levels of calibrators and three control samples at low, medium and high concentrations of 25(OH)D2 and 25(OH)D3 in three replicates. To evaluate the inter-run precision, the run was repeated on five consecutive days (total: n = 15). The acceptable limit of the intra- and inter-run coefficients of variation (CVs) was <15%.
Method validation: Carryover
To investigate the carryover effect, two levels of quality control (QC) samples were used; the order of analysis was as follows: high1-high2-high3-high4-base1-base2-base3-base4. Each of the peak areas was designated as H1, H2, H3, H4, L1, L2, L3 or L4, and carryover was calculated using the following equation
Method validation: Ion suppression
Ion suppression was studied using the postcolumn infusion method, in which patient samples and the mobile phase were separately injected during continuous postcolumn infusion of 300 ng/mL maintenance solution, containing 3-epi-25(OH)D3, 25(OH)D2, 25(OH)D3, 2H3-25(OH)D2, 2H3-25(OH)D3, 2H6-25(OH)D2 and 2H6-25(OH)D3. The resulting MRM signals were monitored. The ion suppression owing to overlapping components manifested as a dip in the particular MRM trace.
Linearity of the calibration curve and QC
We followed the instructions in the MSMS vitamin D kit for calibration and QC. Quantitation of the measured patient samples was performed using the calibrators and IS provided with the kit. Kit calibrators consisted of charcoal-stripped human serum enriched with six increasing concentrations of 2H6-25(OH)D2 and 2H6-25(OH)D3. Calibrators were prepared using the same assay procedure as patient samples, including the addition of IS. Calibrator and IS signal areas were determined, and analyte/IS ratios were calculated. Calibrator analyte/IS ratios were then plotted (y-axis) against the lot-specific calibrator assigned concentrations (x-axis), and a calibration curve was generated using linear regression with 1/X2 weighting. The analyte concentrations in the patient samples were determined using the calibration curve values of slope (m) and intercept (b) according to equation (1)
Kit control samples (including three increasing concentrations of 2H6-25(OH)D2 and 2H6-25(OH)D3 in charcoal-stripped human serum) were also included for QC in each sample run.
Accuracy and traceability testing with NIST SRM 972a
To evaluate the accuracy and traceability to primary reference material, we purchased NIST SRM 972a of vitamin D metabolites in frozen human serum and measured its concentrations three times, repeatedly. The % bias of the measured value from the NIST target value was calculated.
Measurement of 3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2 with the UPLC MS/MS method and of 25(OH)D with the Advia Centaur vitamin D total assay method
3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2 were measured in 103 infant serum samples, 209 paediatric serum samples and 207 adolescent serum samples using an UPLC-MS/MS setup with the procedures and conditions as described above. The mean concentrations of each age group were compared with statistical assessment of SPSS Statistics 23 (IBM, Armonk, NY, USA). The mean bias of 25(OH)D using UPLC-MS/MS was calculated and assessed with SPSP Statistics 23. The mean bias was compared with the % total allowable error (TAE) of 25(OH)D, which is 25%. 25(OH)D concentrations in all 103 infant serum samples, 35 paediatric serum samples and in 32 adolescents serum samples were measured using the Advia Centaur vitamin D total assay (Siemens Healthcare Diagnostics Inc, PA, USA). The 35 paediatric serum samples and 32 adolescent serum samples were selected randomly regardless of their vitamin D concentrations as measured with the UPLC-MS/MS method. The Advia Centaur vitamin D total assay method is a competitive immunoassay that does not differentiate between 25(OH)D3 and 25(OH)D2, does not detect 3-epi-25(OH)D3 and does not exhibit any cross-reactivity with 3-epi-25(OH)D3 in detecting 25(OH)D.
Statistical assessment
SPSS Statistics 23 (IBM, USA) was used for (1) one-way analysis of variances (ANOVA) for comparison of the mean concentrations of 3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2 among the 519 infant, paediatric and adolescent serum samples corresponding to the respective age groups, (2) ANOVA for comparison of the mean bias of 25(OH)D using UPLC-MS/MS in each age group and (3) Pearson’s correlation in the methods comparison. P values ≤0.05 were considered to reflect statistical significance. MedCalc version 16.2.0 (MedCalc Software, Mariakerke, Belgium) was used to analyse Passing and Bablok regression and Bland–Altman plots in the methods comparison.
Results
MS/MS conditions
The MS/MS conditions were optimized (e.g. capillary voltage, cone voltage and collision energy) for the ESI and the MRM of the analytes and IS. The conditions used for MS/MS are described in detail in online supplementary Table 1. The representative MRM chromatograms with the R form and S form of the analytes and the IS of 25(OH)D2 and 25(OH)D3 are shown in Figure 1. The 3-epi-25(OH)D3 form was clearly separated from 25(OH)D3 in patient serum samples and in the NIST SRM972a. The representative MRM chromatograms of 3-epi-25(OH)D3 in patient serum samples and in NIST SRM972a Level 4 are also shown in Figure 1.
Representative mass chromatogram showing the multiple reaction monitoring (MRM) transitions of the 2H3-25-hydroxyvitamin D3 (2H3-25(OH)D3), 25-hydroxyvitamin D3 (25(OH)D3), 3-epi-25-hydroxyvitamin D3 (3-epi-25(OH)D3) of the NIST SRM972a Level 4, 25(OH)D3, 3-epi-25(OH)D3 of the NIST SRM972a Level 1, and 25(OH)D3, 3-epi-25(OH)D3 of a sample from the infant group (age range <1 year).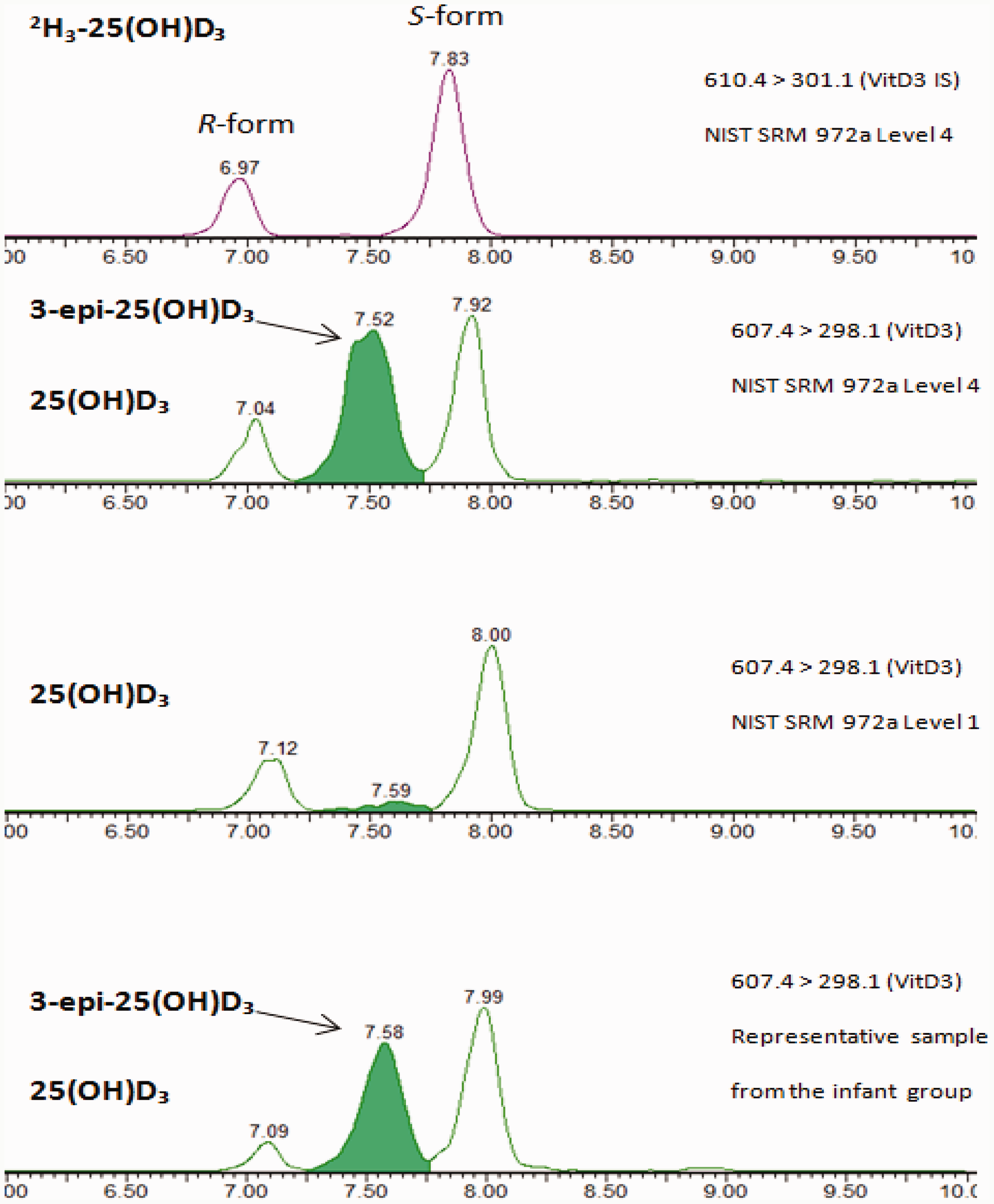
Method validation results: Precision
The intra- and inter-run precisions of 25(OH)D2 and 25(OH)D3 concentrations in calibrators and controls (nmol/L) measured with the UPLC-MS/MS method (five runs, three replicates/run) are shown in online supplementary Table 2. The intra- and inter-run CVs for 25(OH)D2 and 25(OH)D3 were 0.29–14.12% and 1.11–10.20%, respectively.
Method validation results: Carryover
The carryover was −0.27% and 0.10% for 25(OH)D2 and 25(OH)D3, respectively.
Method validation results: Ion suppression
There was a dip in the MRM trace of the patient sample. However, this did not occur during the detection time for 25(OH)D, but prior to it, which did not overlap the detection time of 25(OH)D determined using UPLC-MS/MS (online supplementary Figure 2). Therefore, no ion suppression was noted during the detection time.
Linearity of the calibration curve and QC results
The concentrations of the six levels of calibrators are shown in online supplementary Table 2. All the R2 values for the six levels of calibrators used to mean linearity were 0.9999. We used three levels of QC materials from the kit for each 96-well plate. The plates were judged valid, when all concentrations of these were in the range recommended by the manufacturer, and all the QC materials were within the range.
Accuracy and traceability testing with NIST SRM 972a
Bias between 25(OH)D2, 25(OH)D3 and 3-epi-25(OH)D3 values measured by the UPLC-MS/MS and the target values of the NIST SRM 972a material.
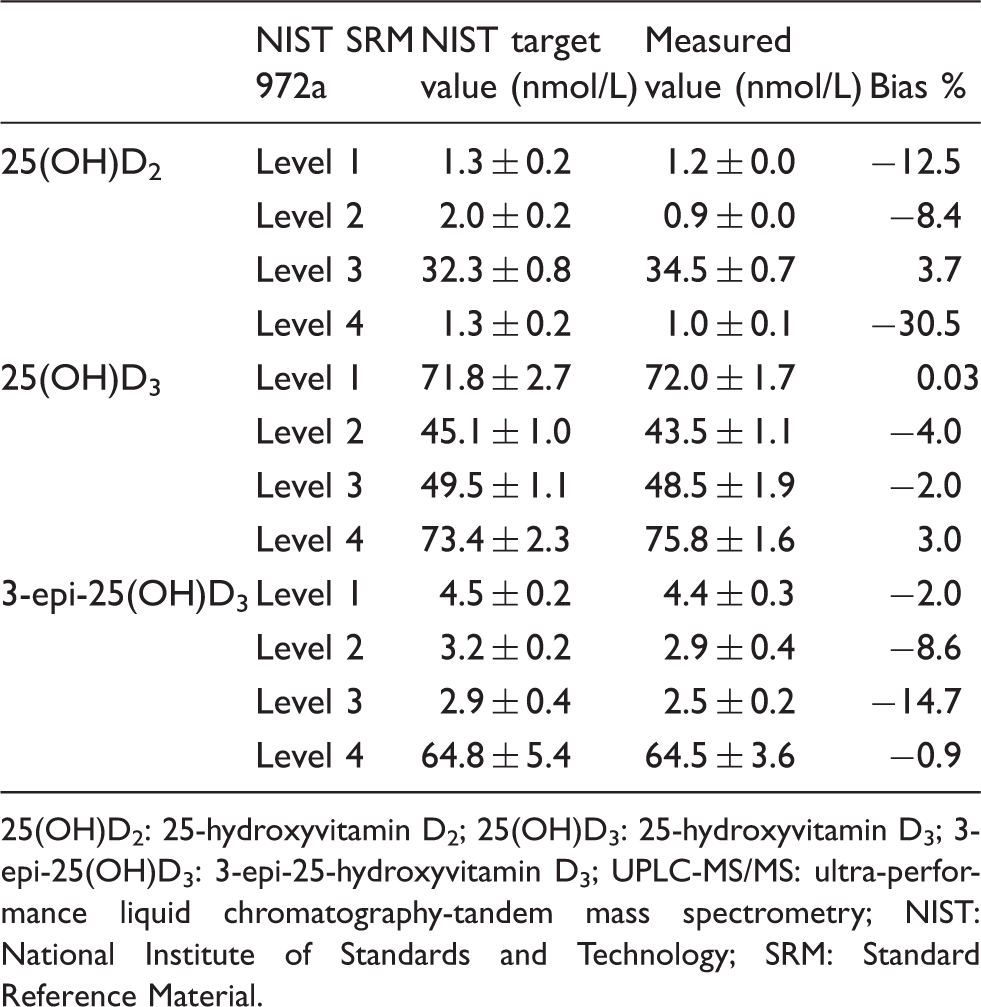
25(OH)D2: 25-hydroxyvitamin D2; 25(OH)D3: 25-hydroxyvitamin D3; 3-epi-25(OH)D3: 3-epi-25-hydroxyvitamin D3; UPLC-MS/MS: ultra-performance liquid chromatography-tandem mass spectrometry; NIST: National Institute of Standards and Technology; SRM: Standard Reference Material.
Measurement of 3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2 in 519 infant, paediatric and adolescent serum samples with the UPLC-MS/MS method
Mean concentrations of 25(OH)D2, 25(OH)D3 and 3-epi-25(OH)D3 in each age group with ANOVA analysis.
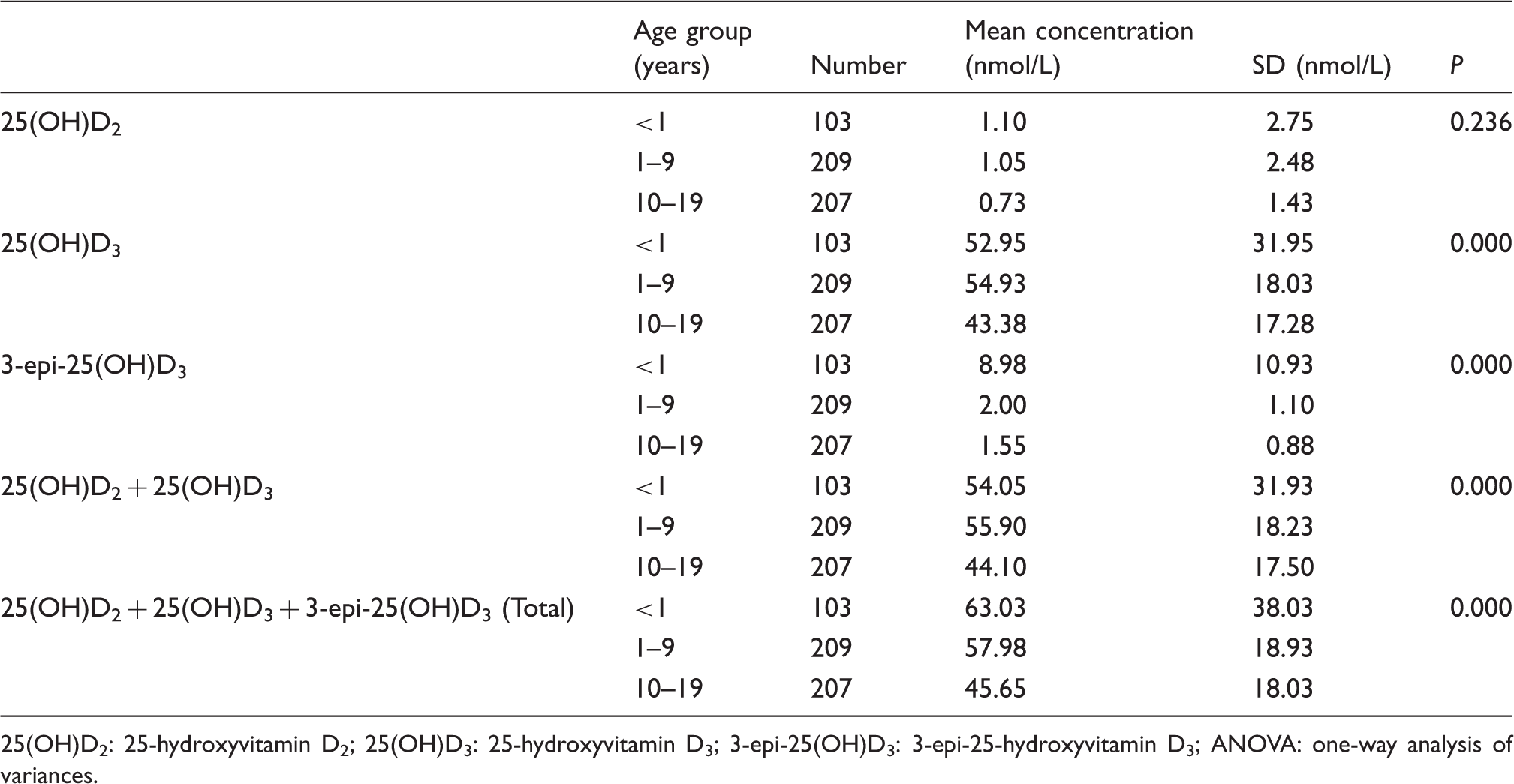
25(OH)D2: 25-hydroxyvitamin D2; 25(OH)D3: 25-hydroxyvitamin D3; 3-epi-25(OH)D3: 3-epi-25-hydroxyvitamin D3; ANOVA: one-way analysis of variances.
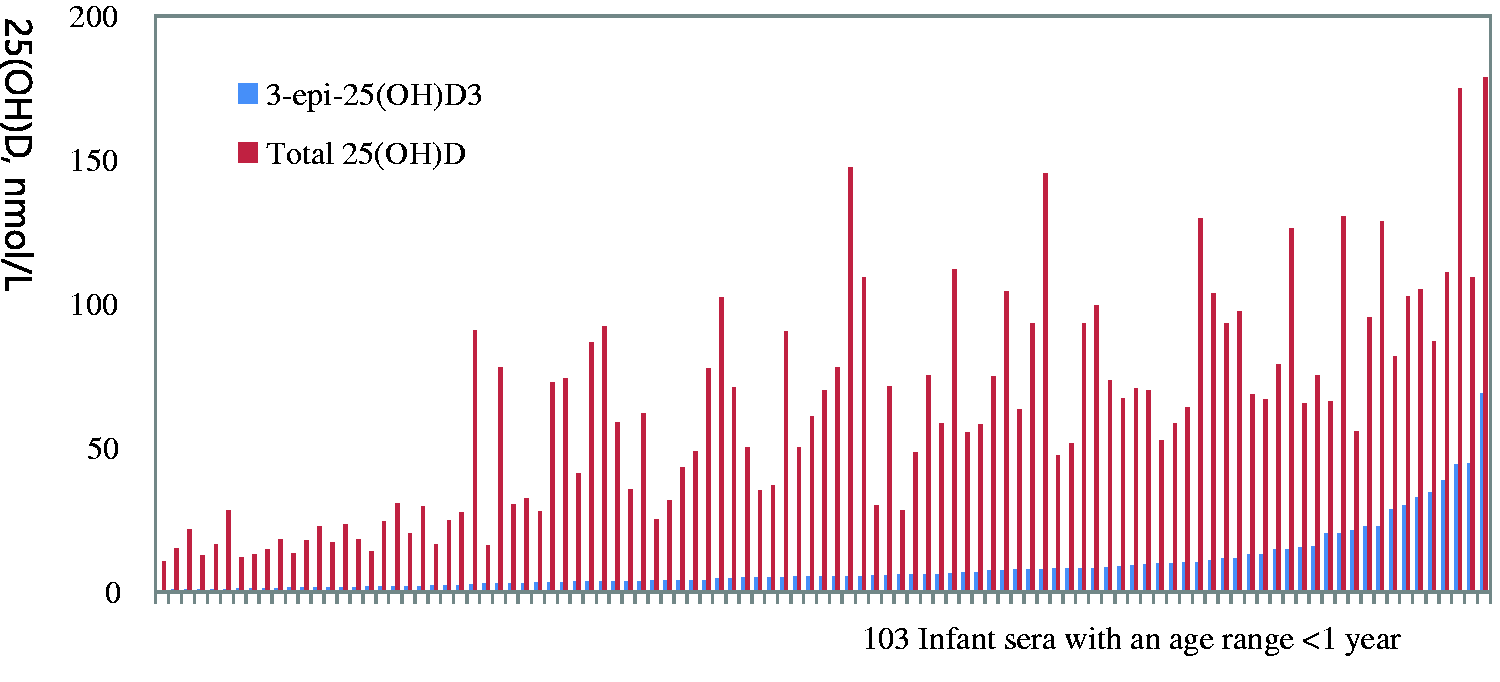
Histogram of the 3-epi-25(OH)D3 and total 25(OH)D concentrations in 103 infant serum samples (age <1 year) to show the range of concentrations.
Mean bias of 25(OH)D in each age group with ANOVA analysis.
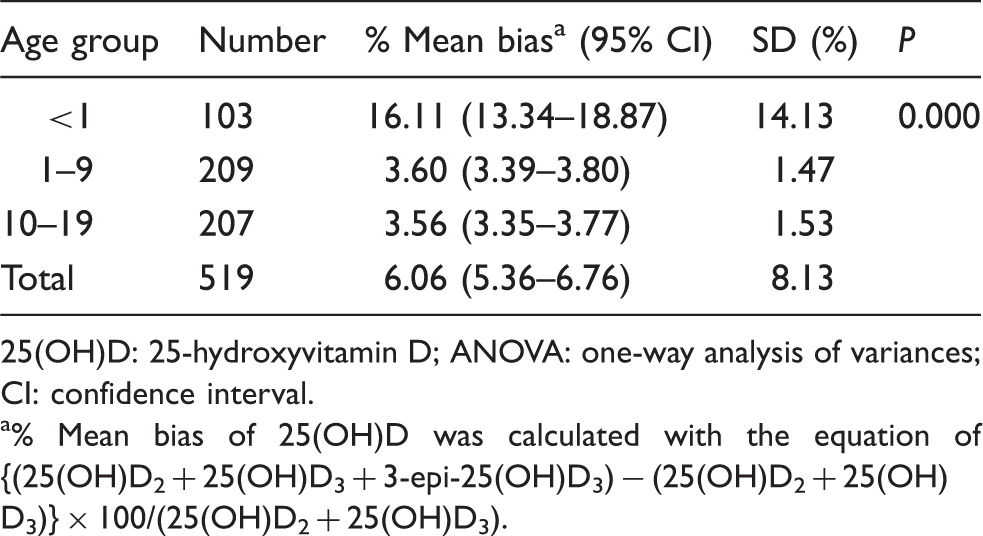
25(OH)D: 25-hydroxyvitamin D; ANOVA: one-way analysis of variances; CI: confidence interval.
% Mean bias of 25(OH)D was calculated with the equation of {(25(OH)D2 + 25(OH)D3 + 3-epi-25(OH)D3) − (25(OH)D2 + 25(OH)D3)} × 100/(25(OH)D2 + 25(OH)D3).
There were 24 cases, where the epimer have confounded interpretations. In these cases, according to the cut-off value of 50 nmol/L, the 25(OH)D concentrations over 50 nmol/L were regarded as ‘optimal’, 25(OH)D concentrations between 27.5 and 50 nmol/L were regarded as ‘insufficient’. Following this classification, 11 the rate of misclassified cases was 7.8% (8/103), 2.9% (6/209) and 4.8% (10/207) in infant, paediatric and adolescent serum samples, respectively. All of these cases were misclassified as ‘optimal’, when their non-epimeric 25(OH)D concentration indicated ‘insufficient’. In the paediatric and adolescent groups, all of these cases showed that % of 3-epi-25(OH)D3 which were calculated with this equation of {100 × (3-epi-25(OH)D3/total 25(OH)D)}were less than 5%. Meanwhile, in all of the misclassified cases in infant serum samples, % of 3-epi-25(OH)D3 was more than 10%.
Comparison between the two different methods of analysis
Passing Bablok regression analysis of UPLC-MS/MS compared with 25(OH)D Advia Centaur method.
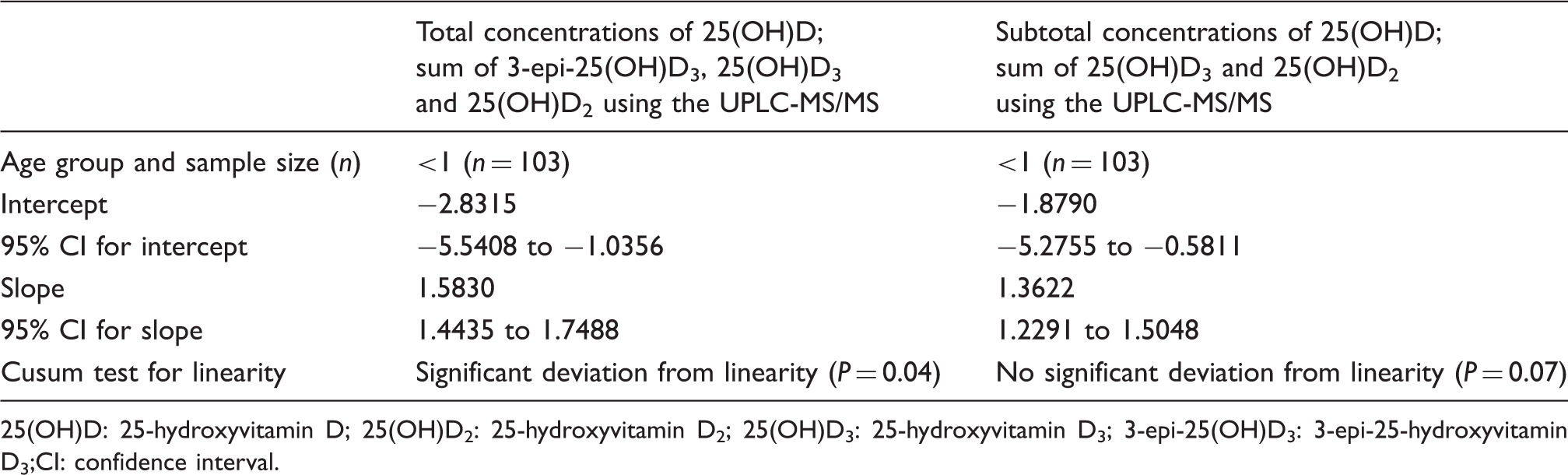
25(OH)D: 25-hydroxyvitamin D; 25(OH)D2: 25-hydroxyvitamin D2; 25(OH)D3: 25-hydroxyvitamin D3; 3-epi-25(OH)D3: 3-epi-25-hydroxyvitamin D3;CI: confidence interval.
Discussion
We evaluated the performance of the UPLC-MS/MS method to separately measure the concentrations of 3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2 in the serum samples of 519 infants, children and adolescents.
The precision, carryover and ion suppression evaluation results were acceptable for measurement. We used the PTAD derivatization method in this study, which improves the ionization efficiency and MS/MS signal intensity of the analytes. This reaction of PTAD results in the subsequent formation of a new pair of 25(OH)D diastereomers (online supplementary Figure 1), and the signals of both the 6S- and 6R- isomers are then combined for the quantitative determination of 25(OH)D2 and 25(OH)D3 in the samples.
The C-3 epimerization pathway of vitamin D3 was first observed by Reddy et al. 19 in neonatal human keratinocytes. The C-3 epimerization pathway leads to the conversion of the configuration of the hydroxyl group at C-3 of the A-ring and produces 3-epi-25(OH)D3 from 25(OH)D3. 20 Because both 3-epi-25(OH)D3 and the native form of 25(OH)D3 have the same molecular weight and fragmentation pattern, the presence of 3-epi-25(OH)D3 would lead to an overestimation of 25(OH)D3 in various methods currently used for vitamin D measurements that do not separate this isomeric form. 21 Furthermore, existing LC-MS/MS methods that were developed for C-3 epimer separation require long chromatography separation times, extensive sample preparation or high-resolution mass spectrometer.15,21–23
In this study, we separated the 3-epi-25(OH)D3 form successfully using an UPLC-MS/MS method with a PerkinElmer Kit and a Kinetex XB-C18 column for 11 min of run time. This method offers an improvement in sample preparation step and in the throughput compared with previous methods with longer run times. For the evaluation of the accuracy and traceability to primary reference material, we measured NIST SRM 972a and calculated the % bias of the measured value from the NIST target value. The NIST SRM 972a Level 4 was fortified with 3-epi-25(OH)D3, and the concentrations of 3-epi-25(OH)D3 in Level 4 were accurately measured with −0.92% bias by this method.
The presence of 3-epi-25(OH)D3 in infants was confirmed in this study. The mean 3-epi-25(OH)D3 concentration of infant serum samples was significantly higher than those of paediatric and adolescent. This result is similar with those obtained in previous studies. Singh et al. 16 reported the detection of C-3 epimers only in children <1 year of age; they did not detect C-3 epimers in children 1–18 years old, nor in adult groups. Similarly, Keevil 24 found that the prevalence of 3-epi-25(OH)D3 is higher in neonates <1 year of age and approaches adult concentrations in children after just 1 year. Yang et al. 23 introduced a high-throughput measurement of 25(OH)D via LC-MS/MS with separation of the C-3 epimer interference for paediatric populations. That data demonstrate that the separation of 3-epi-25(OH)D3 in adult samples is not required owing to its low prevalence and concentration. However, because of the presence of significant concentrations of 3-epi-25(OH)D3, younger paediatric samples require a method that can separate this from the native form of 25(OH)D3 for accurate quantitation and clinical management. On the other hand, Stepman et al. 18 suggested that the prevalence of 3-epi-25(OH)D3 in serum of infants is considerable, and that even in adults, the concentrations of this form should not be neglected, because they found that in some adults, 3-epi-25(OH)D3 was present in considerable amounts, although the relative content in serum from adults was typically less than that seen in children. However, these findings were determined using repository samples; thus, the authors mentioned that the results should be confirmed by studies on freshly obtained samples.
In the current study, the total concentrations of 25(OH)D, which meant sum of 3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2 in the infant group (n = 103) determined using the UPLC-MS/MS and the total 25(OH)D determined using the Advia Centaur method deviated significantly from linearity (P = 0.04), whereas the sum of 25(OH)D3 and 25(OH)D2 concentrations without 3-epi-25(OH)D3 using the UPLC-MS/MS method compared with the total 25(OH)D determined by the Advia Centaur did not (P = 0.07). This could be explained by the presence of considerable amounts of 3-epi-25(OH)D3 in the infant group. As the Advia Centaur method exhibits no cross-reactivity with 3-epi-25(OH)D3, 25 the results between the two methods were comparable when the UPLC-MS/MS values for 25(OH)D excluding 3-epi-25(OH)D3 were used. This is consistent with previous studies.10,25 On the other hand, the LC-MS/MS method, which cannot separate 3-epi-25(OH)D3, would be expected to overestimate the concentration of 25(OH)D in the infant group, owing to the considerable amounts of 3-epi-25(OH)D3 in their circulation.
There is a limitation in our study. Flynn et al. 21 demonstrated enhanced 3-epi-25(OH)D3 signal relative to equimolar 25(OH)D3 during infusions and in spiked human serum. They insisted enhanced signal caused overestimation of 3-epi-25(OH)D3 concentrations when quantified using 25(OH)D3 calibrators as an indirect quantitation method, and the 3-epi-25(OH)D3 signal enhancement was dependent on mobile phase composition. Even though we used the 25(OH)D3 calibrators as an indirect quantitation method of the 3-epi-25(OH)D3, we did not examine the signal response from equimolar concentrations of the epimer and of 25(OH)D3 in our assay to determine whether this is a factor in our quantitation. So, we cannot rule out the possibility of overestimation of the amount of epimer present especially in the infant group.
The clinical utility of measuring 3-epi-25(OH)D3 is at present unclear. However, considering the high concentration of 3-epi-25(OH)D3 in the infant group, which would substantially increase the total 25(OH)D concentration measured by LC-MS/MS, or by certain immunoassays that could not differentiate the epimeric forms, certain cases might arise wherein an infant patient deficient in vitamin D might be misclassified as sufficient owing to the inclusion of the epimer concentration. In this study, in the 24 cases where the epimer have confounded interpretations, the misclassification rate of infant serum samples was more higher than those of paediatric and adolescent serum samples. This result upholds the previous study by Strathmann et al., 17 where 9% were misclassified in infant group, while 3% were misclassified in children and adult group (1–94 years). Furthermore, in the paediatric and adolescent groups, all of these cases showed that % of 3-epi-25(OH)D3 was less than 5%. Meanwhile, in all of the misclassified cases in infant serum samples, % of 3-epi-25(OH)D3 was more than 10%, which demonstrates that C-3 epimer interference would be more affecting in interpretation especially in infant group. Thus, for infant populations <1 year of age, it would be preferable for the concentrations of 3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2 be reported separately regardless of the clinical relevance of 3-epi-25(OH)D3.
In summary, this is the first study regarding the separation of 3-epi-25(OH)D3 in a Korean paediatric population. The performance of the UPLC-MS/MS assay for measuring 3-epi-25(OH)D3, 25(OH)D3 and 25(OH)D2 was acceptable. Further study would be needed to determine the clinical importance of 3-epi-25(OH)D3 in various populations in Korea.
Footnotes
Acknowledgements
The authors are grateful to Ji Young Shin, M.T. for technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was obtained from Ilsan Paik Hospital, Reference No. 2015-10-023-001.
Guarantor
AD.
Contributorship
Sung Eun Cho and Sollip Kim contributed equally to this work. All authors contributed to study design. All authors reviewed and edited the manuscript and approved the final version.