Abstract
Introduction
Dietary serotonin increases urinary secretion of 5-HIAA. A falsely elevated 5-HIAA may lead to incorrect suspicion of a neuroendocrine tumour. Therefore, we determined the effect and duration of dietary serotonin on serum 5-HIAA concentration. We also studied the distribution of 5-HIAA in serum fractions.
Methods
We used serum samples from healthy volunteers (31 women and four men). All test subjects avoided serotonin-containing foods for three days before sample collection. They then ate either pineapple, banana, kiwi fruit, tomato or walnuts and additional blood samples were taken after 2, 4, 6, 24, 48 and 72 h. To study the distribution of 5-HIAA in serum, samples from a healthy individual, a test person who had ingested walnuts, and from a neuroendocrine tumour patient were fractionated by gel filtration chromatography. The fractions were analysed for 5-HIAA.
Results
Serum 5-HIAA concentration increased significantly (P ≤ 0.001) within 2 h after ingestion of serotonin-containing food. After 2 h, 5-HIAA concentration started to decrease and reached the baseline concentration within 24 h. A calculated half-life of 5-HIAA in circulation was 1.3 h. In fractionated serum, 5-HIAA was found not only in free form but also in the albumin and α2-globulin fractions.
Conclusions
The increase of serum 5-HIAA caused by dietary serotonin is significant but transient. Therefore, serotonin-containing foods should be avoided for one day before blood sampling. In serum, 5-HIAA is free and apparently bound to albumin. Minor amounts were also found in the α2-globulin fraction. Our liquid chromatography tandem mass spectrometry assay measures free 5-HIAA in serum.
Introduction
Neuroendocrine tumours (NETs) are derived from endochromaffin cells, and most of the tumours excrete serotonin. Serotonin is a heterocyclic amine synthesized from the amino acid L-tryptophan. 1 Approximately 50% of plasma serotonin is taken up by platelets by an active, but relatively slow transport mechanism. 2 The majority of non-platelet serotonin is metabolized by monoamine oxidase producing 5-hydroxyindoleacetaldehyde, which is further oxidized to 5-hydroxyindoleacetic acid (5-HIAA, about 99%) or reduced to 5-hydroxytryptophol (5-HTOL, about 1%). 3 Determination of urinary, plasma or serum 5-HIAA is used in the diagnosis and monitoring of the NETs.4–6
Certain foods like pineapple, banana and walnuts contain significant amounts of serotonin.7,8 Ingestion of these may increase plasma 5-HIAA concentration beyond the reference range and may interfere with the NET diagnosis or follow-up. The major part of dietary tryptophan is exploited for protein synthesis and only 1–3% is metabolized to serotonin. 9 Previous studies have demonstrated an increase in 5-HIAA concentrations after ingestion of serotonin-containing foods. Kema et al. 10 found a five-fold increase in urinary 5-HIAA, and Degg et al. 11 demonstrated an increase of plasma 5-HIAA after 2 h of food ingestion (n = 14). However, platelet serotonin content was unaffected by dietary serotonin in short-term ingestion. 12 To our knowledge, the duration of elevated 5-HIAA concentration in serum or plasma has not been studied earlier.
Generally, many small molecules bind to plasma proteins, either to specific binding molecules or non-specific carrier molecules. 13 Albumin is a substantial carrier protein for numerous endogenous and exogenous compounds. 14 Serotonin is known to bind to a receptor on platelets but binding of 5-HIAA to plasma proteins has not been reported.
In this study, we wanted to determine the duration of elevated serum 5-HIAA concentration caused by dietary serotonin in order to avoid unnecessary diet restriction for the patient. We also studied the distribution of 5-HIAA in serum fractions and found that our liquid chromatography tandem mass spectrometry (LC-MS/MS) assay determines serum-free 5-HIAA.
Material and methods
Test subjects
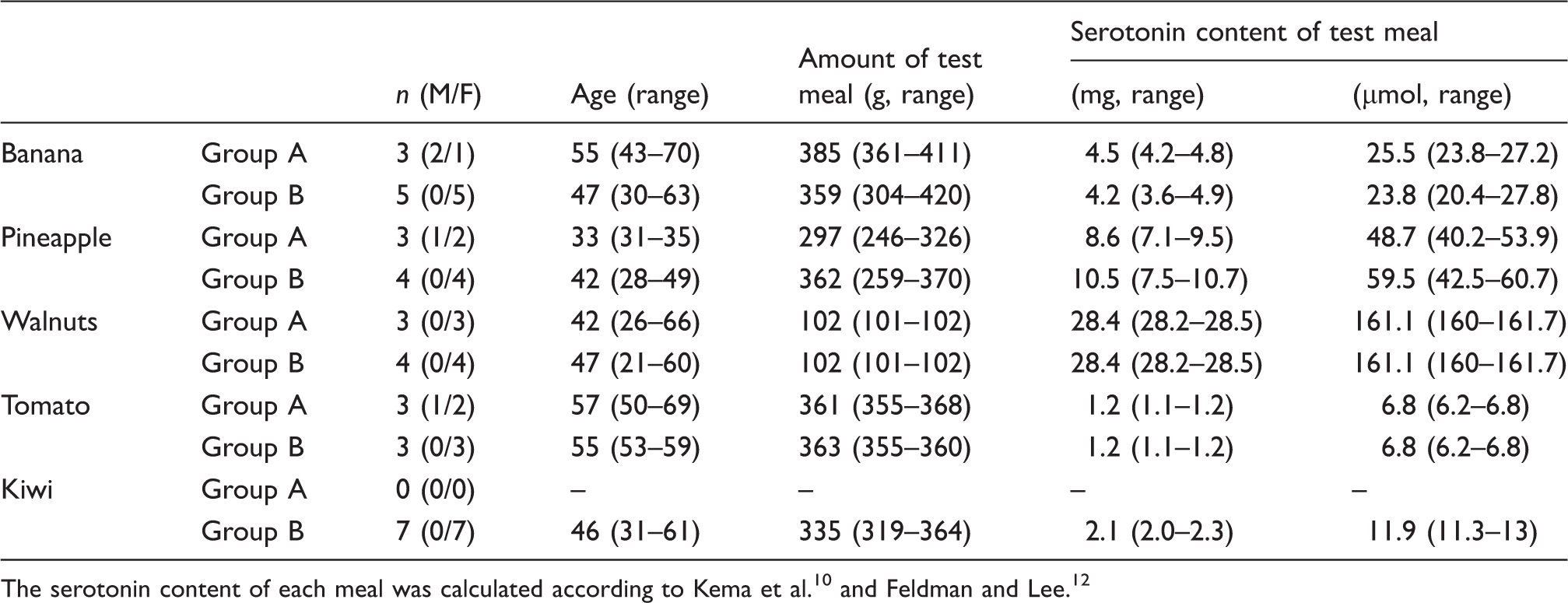
Characteristics of the test subjects and test meals.
The diet study
The foodstuffs were purchased from a local grocery store. All items were weighed, the net weight calculated without peelings and eaten uncooked. The first blood sample after the restricted diet was drawn at 8 a.m. into plain serum tubes. Then, individuals in Group A had their test meal within 20 min of the first blood sample. Individuals in Group B were allowed to eat their test meal freely during the first day. In addition, all individuals were allowed to have their normal meals, however, with serotonin restriction. Next samples were drawn at 10 a.m., noon and 2 p.m. only from individuals in Group A and from all individuals at 8 a.m. in the following three days. Serotonin content of the test meals was calculated (Table 1).10,12 The average circulation half-life (T½) of 5-HIAA was calculated according to formula T½ = tln(2)/ln(N0/N t ), where N0 refers to the original concentration and N t to the concentration at time point t. 15 The calculation was done at time points 2 h and 4 h in test subjects in Group A. An average serum volume of 3 L was used.
Distribution of 5-HIAA in serum
To study the distribution of 5-HIAA in serum fractions, we used serum samples from a healthy woman (36 years) after a three-day restricted diet, from a healthy woman (26 years) 2 h after ingestion of 100 g of walnuts corresponding to 29 mg serotonin and from a NET patient (man, 22 years). 6 Informed consent was obtained from the healthy individuals. This study was approved by the ethical committee of Helsinki University Central Hospital, Finland.
Analysis of serum 5-HIAA
After blood collection, the samples were allowed to stand at room temperature for 1 h; centrifuged and separated serum was divided into aliquots and kept at −20℃ until 5-HIAA analysis within one week. Sample preparation and 5-HIAA analysis were performed by LC-MS/MS as previously described. 6 Briefly, serum samples were spiked with 5-HIAA-D2 internal standard and extracted by solid phase extraction with WAX µElution plates (Waters, Milford, MA, USA). Extracted samples were then quantified by LC-MS/MS employing an Atlantis HILIC Silica column (Waters), Agilent 1200 liquid chromatograph (Agilent Technologies, Santa Clara, CA, USA) and a 4000 QTRAP mass spectrometer (AB Sciex, Toronto, Canada).
Gel filtration
We used the ÄKTA purifier system (GE Healthcare Bio-Sciences AB, Uppsala, Sweden, www.gelifesciences.com) with a Superdex™ 200 10/300 GL column (GE Healthcare Biosciences) equilibrated with 1 mmol/L ammonium acetate – 150 mmol/L sodium chloride at a flow rate 0.5 mL/min. All reagents were of the highest analytical grade. The column was calibrated with synthetic 5-HIAA, human albumin and α2-macroglobulin (α2 M). Serum samples were centrifuged for 3 min at 10,000 g and 0.5 mL was applied onto the column. Absorbance at 280 nm was monitored, fractions of 0.5 mL were collected and assayed for 5-HIAA as above.
Statistical methods
The paired t-tests were performed by Analyse-it software for Microsoft Excel 2010 (Ver. 2, Analyse-it software Ltd., http://www.analyse-it.com).
Results
Effect of dietary serotonin on serum 5-HIAA
The basal serum 5-HIAA concentration after a 3-day restricted diet was 76 nmol/L (range 68–83 nmol/L, n = 35). Serum 5-HIAA concentration increased (P ≤ 0.001) to the highest within 2 h after ingestion of all serotonin-containing test meals (banana, walnut, tomato or pineapple) in all individuals (Group A, n = 12) (Figure 1). The increase was the highest (average 1797 nmol/L, range 1308–2538 nmol/L, n = 3) in samples from individuals who had eaten walnuts (corresponding to an average of 28.5 mg serotonin) and the lowest in samples from individuals who had eaten tomatoes (average 394 nmol/L, range 366–413 nmol/L, n = 3) or pineapple (average 465 nmol/L, range 354–556 nmol/L, n = 3) (corresponding to an average of 1.2 mg and 8.6 mg serotonin, respectively). An estimation of serotonin half-life in serum was 2 h as calculated from the appearance rate of 5-HIAA in serum samples. A decrease in serum 5-HIAA was seen within 4 h after ingestion of the test meal (mean 49%, n = 12). Serum 5-HIAA concentration reached the basal concentration after 6 h in individuals who had eaten tomatoes or pineapple. In individuals who had eaten walnuts or banana, the basal serum 5-HIAA concentration was reached in the next morning. The calculated half-life of 5-HIAA in circulation was 1.3 h. There was no difference (P ≥ 0.078) in serum 5-HIAA concentrations in Group B individuals, from whom samples were collected once a day, at any time point with any test meal.
Serum 5-HIAA concentrations after serotonin-containing test meals. Time points 0, 24 h, 48 h and 72 h show the mean of combined Groups A and B (n = 35) where as time points 2 h, 4 h and 6 h show the mean of test Group A only (n = 12).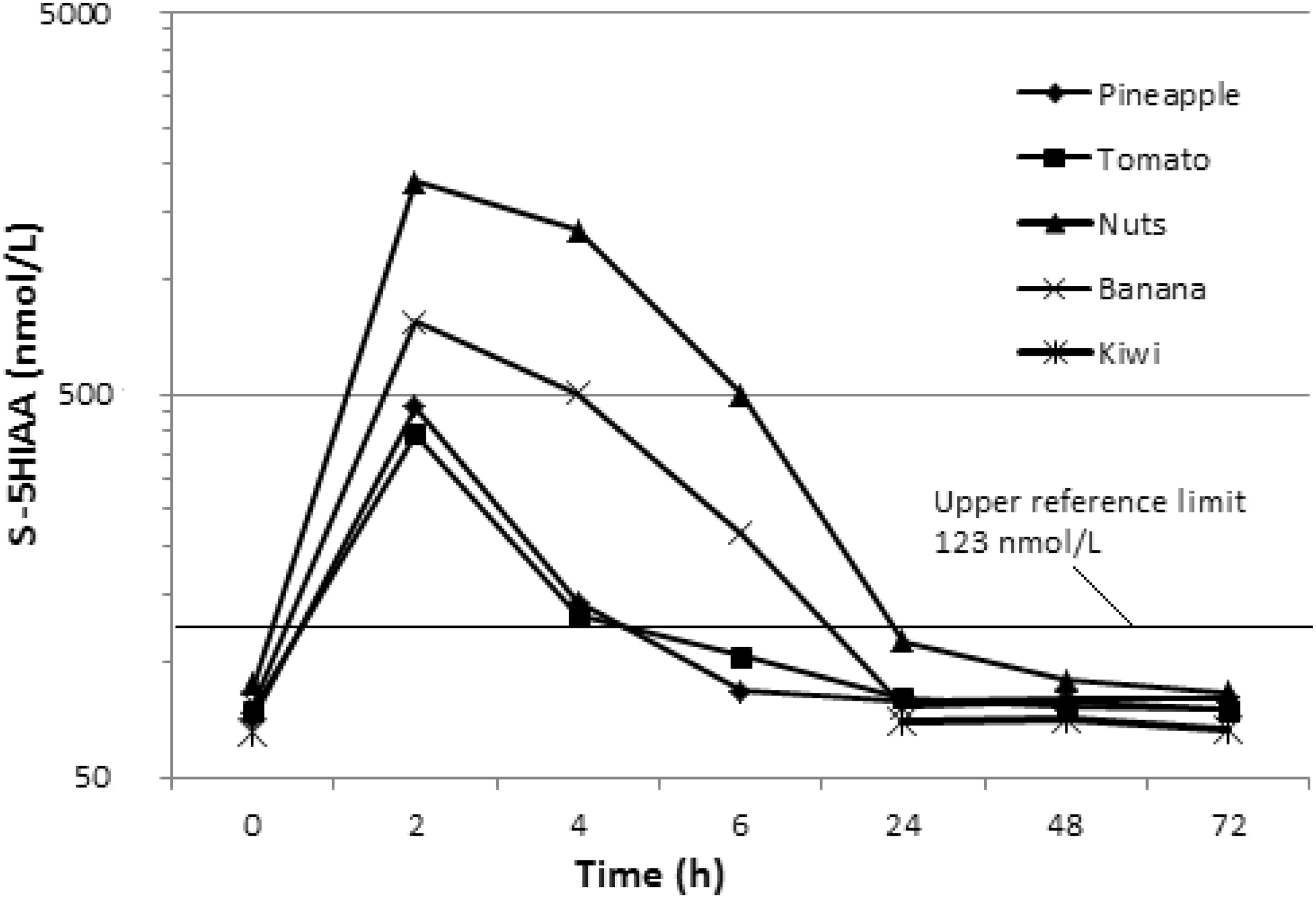
Distribution of 5-HIAA in serum fractions
In our LC-MS/MS assay, 5-HIAA and the internal standard elute at retention time 1.3 min.
6
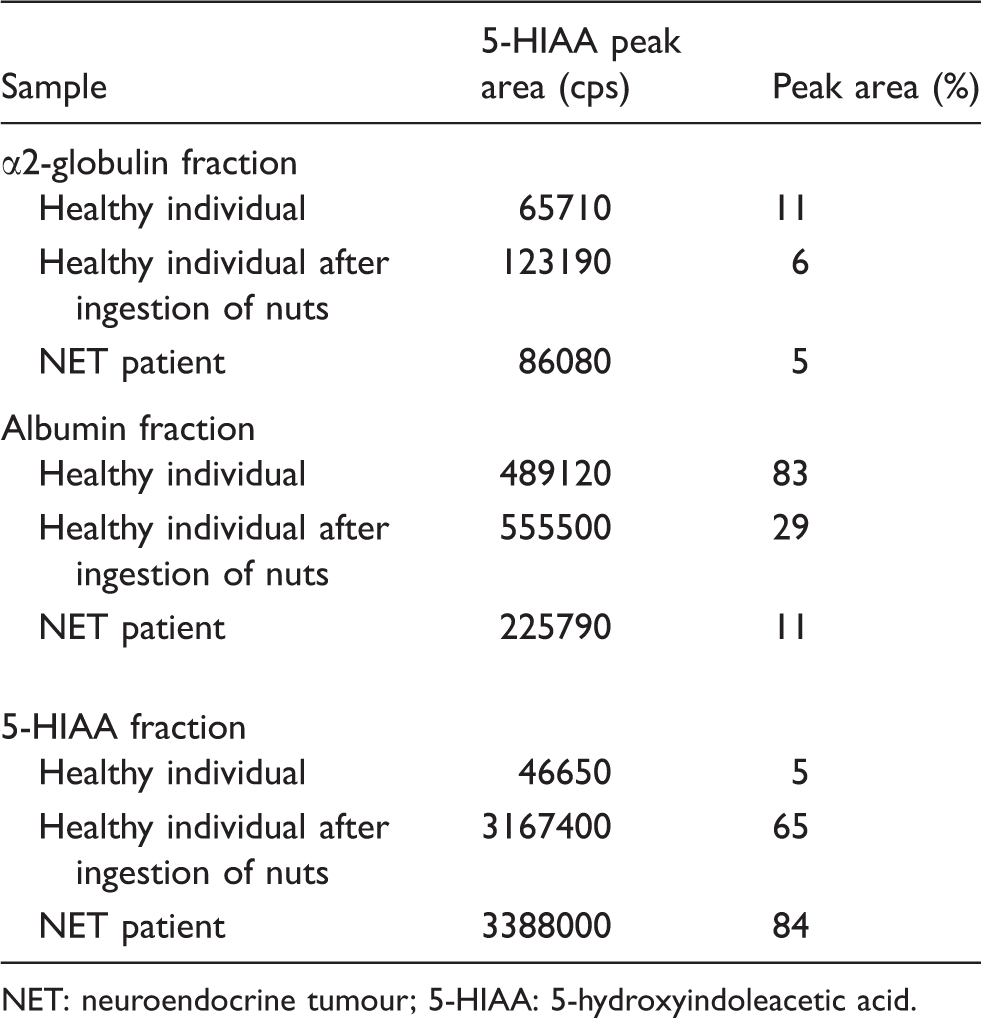
However, in the chromatograms of serum samples three additional peaks are seen at retention times 1.0, 1.9 and 2.5 min with the fragmentation pattern of 5-HIAA (Figure 2). In gel filtration, α2 M, albumin and 5-HIAA eluted at 9–11 mL, 14–17 mL and 22–26 mL, respectively. LC-MS/MS analysis of serum gel filtration fractions revealed that the 5-HIAA peaks eluting at 1.0 min, 1.3 min, 1.9 min and 2.5 min are derived from the background, from free 5-HIAA, and from α2-globulin and albumin fractions, respectively. Only the peak eluting at 1.0 min was detected in every gel filtration fraction.
Overlaid chromatograms of 5-HIAA (black) and IS (grey) of a healthy subject serum sample containing 88 nmol/L of 5-HIAA.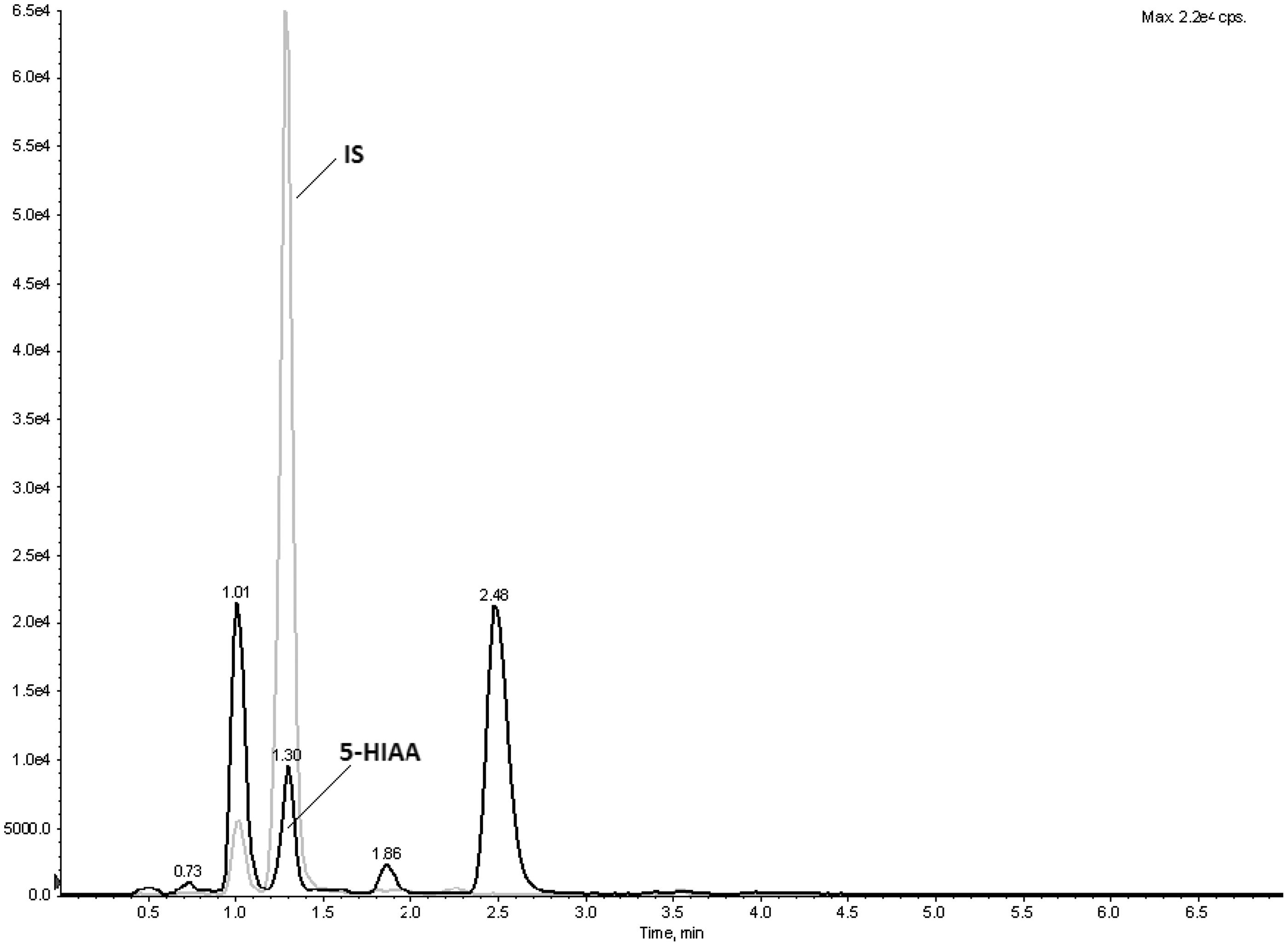
The peak areas and relative 5-HIAA proportions corrected by recovery in serum gel filtration fractions.
NET: neuroendocrine tumour; 5-HIAA: 5-hydroxyindoleacetic acid.
Discussion
We show here that dietary serotonin causes a significant but transient increase in serum 5-HIAA reaching the highest measured concentration in 2 h. Even a very marked elevation of 5-HIAA concentration, i.e. 2000 nmol/L declines to the basal concentration within 24 h with a half-life of 1.3 h. To our knowledge, this is the first study showing that a diet restriction for several days before blood collection is not necessary for the diagnosis of NET.
Previous studies show that serotonin-containing foods increase urinary 5-HIAA excretion. Feldman and Lee 12 reported that fruits (banana, plantain, kiwi and pineapple) and walnuts (corresponding to ∼3–280 µg/g of serotonin) increased urinary 5-HIAA on average five-fold during a day but there was no change in platelet serotonin concentrations. The increase was dose-dependent and the highest urinary 5-HIAA was caused by ingestion of walnuts. No increase in platelet serotonin, but a 6 - to 20-fold increase in urinary 5-HIAA during 2–4 h was reported by Kema et al. 10 In this study, test subjects had a mixed diet containing 330 g of serotonin-containing fruits and 140 mL of pineapple juice at 8 a.m. with extra juice (280 mL) at 10 a.m. and second test meal containing 50 g of banana at noon. In a study by Degg et al. 11 plasma 5-HIAA was increased after 2 h ingestion of serotonin-containing meal (n = 14). Our serum 5-HIAA results are in line with these reports. We found a dose-dependent 5 - to 20-fold increase in serum 5-HIAA concentration 2 h after ingested dietary serotonin. In the study of Kema, increased urinary 5-HIAA declined to the basal concentration after 19 h. We observed that serum 5-HIAA reaches the highest concentration 2 h after the intake and decreases (mean 49%) already after 4 h. Even a very high serotonin intake followed by a marked elevation of serum 5-HIAA is cleared from the circulation within 24 h. We calculated a half-life of 1.3 h for 5-HIAA in serum. Due to the short observed half-life of 5-HIAA, the highest 5-HIAA concentration may in fact occur in less than 2 h.
Test subjects in Group A ingested the test meals on an empty stomach. Therefore, the observed absorption rate of ingested serotonin in our study may reflect the maximal rate. The individual absorption rate may vary according to nutritional status as has been shown, e.g. with ethanol. 16 Despite the fact that the test subjects had a test meal that exceeds the amount normally ingested (i.e. three bananas or 100 g of walnuts) at one time (Group A) or freely during one day (Group B), serum 5-HIAA concentration was on the basal concentration by the next morning also in Group B. Urinary 5-HIAA collection guidelines 8 advise to avoid serotonin-containing foods for three days before sampling. Our results suggest that an overnight restriction of serotonin-containing foods is sufficient. However, until further studies, we advise our patients to avoid serotonin-containing foods for one day before blood sampling.
We determined the distribution of 5-HIAA in serum fractions in samples with a low 5-HIAA concentration, a transient elevation caused by dietary intake of serotonin and a prolonged elevation caused by a serotonin-secreting NET. In circulation, elevated 5-HIAA concentrations mainly remain in the free fraction (65% and 84%, respectively) and the proportion bound to the α2-globulin or albumin fraction is rather constant. At low concentrations, 5-HIAA is mainly (83%) found in the albumin fraction. However, our study is limited and cannot rule out a possible slight difference in the distribution during long term and transient elevation of 5-HIAA. To our knowledge, the distribution of 5-HIAA in serum has not been studied earlier. Albumin has a very high capability to bind ionic compounds 17 and in fact, it is known to bind non-specifically several molecules in the circulation. 18 Taken together, our results suggest that elevated 5-HIAA remains unbound in serum, and our LC-MS/MS assay measures free 5-HIAA in serum samples. Our 6 reference range is based on free 5-HIAA in serum. Whether a reference range based on total, i.e. free and protein-bound 5-HIAA performs better than that based on free 5-HIAA remains to be elucidated.
In conclusion, we found that the dietary serotonin results in a dose-dependent elevation of serum 5-HIAA within 2 h. Elevated 5-HIAA is rapidly cleared from the circulation with a half-life of 1.3 h and thus, a diet restriction prior to blood sampling for no longer than one day is necessary. In serum, 5-HIAA is both free and bound to proteins in the albumin and α2-globulin fractions.
Footnotes
Acknowledgements
The authors thank volunteers for sample donation.
Declaration of conflicting interests
None declared.
Funding
This work was supported in part by the Research Grants from Academy of Finland, Sigrid Juselius Foundation and Helsinki University Central Hospital.
Ethical approval
This study was approved by the ethical committee of Helsinki University Central Hospital, Finland (211/13/03/00/14).
Guarantor
OI.
Contributorship
OI and NT planned the study; NT, AJ and OI performed the study; NT wrote the draft paper and OI, AJ, EH and RR revised the manuscript. All authors contributed to the study and approved the final version of the manuscript.
