Abstract
Background
Exclusion of macroprolactinaemia, a well-recognised interference, as the cause of hyperprolactinaemia is essential to avoid potential misdiagnosis and mismanagement of patients. We have derived gender-specific serum total and post-polyethylene glycol (PEG) precipitation monomeric reference ranges for the recently re-standardised Abbott Architect prolactin assay.
Methods
Prolactin was measured in serum samples obtained from males (n = 49) and females (n = 52) using the current Abbott Architect immunoassay pre- and post-PEG precipitation. Gender-specific reference ranges were derived for total and monomeric (post-PEG) prolactin. Routine patients’ samples (n = 175) with a serum total prolactin >700 mIU/L were screened for macroprolactinaemia to assess classification compared with our previous post-PEG precipitation percentage recovery-based approach.
Results
Reference ranges for serum total prolactin were 58–419 mIU/L (male) and 63–561 mIU/L (female). Male and female monomeric prolactin reference ranges were 32–309 mIU/L and 39–422 mIU/L, respectively. Mean (SD) post-PEG percentage recovery of the IS 84/500 prolactin standard was 80 (2.3)%. Of 175 patients’ samples screened for macroprolactinaemia, 149 had monomeric prolactin concentrations (median monomeric prolactin = 1035 mIU/L; median recovery = 83%) above the gender-specific reference range. Monomeric prolactin concentrations (median monomeric prolactin = 162 mIU/L; median recovery = 20%) in the remaining 26 were within the reference ranges. One patient classified as macroprolactin positive and another classified as macroprolactin negative would not have been identified as such using the previous recovery-based approach.
Conclusions
The use of post-PEG monomeric reference ranges not only identifies hyperprolactinaemia due solely to macroprolactinaemia but has the added advantage of identifying patients who have simultaneous true monomeric hyperprolactinaemia and elevated concentrations of macroprolactin.
Keywords
Introduction
Hyperprolactinaemia may manifest clinically with symptoms of hypogonadotrophic hypogonadism, infertility and galactorrhoea. The diagnosis is dependent upon the laboratory demonstration of hyperprolactinaemia in the appropriate clinical setting.
Prolactin is known to exist in a number of different molecular forms in the circulation.1–6 Monomeric prolactin (Mr = 23 kDa) is the biologically active and most abundant form (typically 60–90%) 5 found in healthy individuals and patients with true hyperprolactinaemia. However, prolactin may also be present in the serum in varying quantities as big-prolactin (typically 15–30%; 5 Mr = 60 kDa) and macroprolactin (typically 0–10%; 5 a prolactin-IgG complex of Mr = 150 kDa) which are thought to be biologically inactive. Despite its biological inactivity, macroprolactin remains immunoreactive and is detected to varying degrees by different immunoassay platforms.5–11 Depending upon the assay used, it has been reported that up to 26% of hyperprolactinaemia cases are due to macroprolactinaemia.5,6 Failure to identify macroprolactinaemia may lead to misdiagnosis and unnecessary intervention.6,11–16 Thus, when serum prolactin is raised, it is recommended that laboratories screen for macroprolactinaemia in order to differentiate between those patients with elevated concentrations of bioactive monomeric prolactin (true hyperprolactinaemia) and those whose hyperprolactinaemia is due to the presence of macroprolactin.12,13,17
Gel filtration chromatography (GFC), the gold standard for the measurement of serum monomeric prolactin, 5 is slow, costly and labour intensive and therefore impractical for routine clinical use. While polyethylene glycol (PEG) precipitation4,18–21 is the method most commonly used in UK laboratories to screen for macroprolactinaemia (UK NEQAS [Edinburgh] for Prolactin scheme Survey of Practice 2012), it has limitations. Co-precipitation of monomeric prolactin can lead to significant under recovery of up to 25%.19,22 Furthermore, PEG itself is known to cause interference in some prolactin methods.22–25
Prior to changing analytical platforms, our laboratory screened samples with total serum prolactin >700 mIU/L for macroprolactinaemia on the basis of percentage recovery post-PEG precipitation as is the current conventional approach. A recovery of >60% was considered to indicate genuine hyperprolactinaemia and that significant macroprolactinaemia was not present, a recovery of 40–60% (classed as equivocal) indicated that macroprolactinaemia may be present (sample referred for GFC) and a recovery of <40% was considered to indicate the presence of significant macroprolactinaemia. 18 However, due to the limitations of this approach, 12 it has been proposed that the results of macroprolactinaemia screening should be reported as absolute post-PEG prolactin concentrations together with an appropriate reference interval.21,26 Post-PEG precipitation monomeric reference ranges to define true hyperprolactinaemia have been reported previously for other prolactin assays. 22 We describe the derivation of gender-specific reference ranges for both serum total prolactin and monomeric prolactin following PEG precipitation for the recently re-standardised Abbott Architect assay.
Methods
Prolactin reference intervals were determined using anonymised surplus serum obtained from samples (49 males and 52 females) received into the laboratory from our local primary care patient population. Specimens used in the study originated from non-pregnant adults with normal thyroid (thyroid stimulating hormone), liver (alanine aminotransferase, alkaline phosphatase, albumin, total bilirubin, total protein and calculated globulin) and renal (sodium, potassium, creatinine and urea) function. Blood samples were originally collected into Sarstedt serum gel Z/4.7 mL tubes using the Sarstedt Safety Monovette® system (Sarstedt Aktiengesellschaft & Co, Germany), left to clot at room temperature for at least 30 min, separated by centrifugation at 4754 g for 10 min, stored at 4℃ and analysed for prolactin within three days. Prolactin measurements were performed on an Abbott Architect i platform using the Abbott Architect LN 7K76 prolactin chemiluminescent microparticle immunoassay 27 (Abbott Laboratories, USA) which has recently been re-standardised. 28
Following measurement of total serum prolactin, the samples were PEG precipitated by a modified version of the method previously described by Suliman et al. 12 Serum (200 µl) was mixed with an equal volume of 25% (w:v) PEG 6000 (Sigma-Aldrich, St Louis, USA) dissolved in phosphate-buffered saline (PBS) pH 7.4 (Sigma-Aldrich; 137 mmol/L sodium chloride, 10 mmol/L sodium phosphate, 2.7 mmol/L potassium chloride). Following incubation at room temperature for 10 min, the precipitant was pelleted by centrifugation at 17,000 g for 5 min and the supernatant analysed for prolactin as described above. Post-PEG monomeric prolactin values were corrected to account for the dilution factor.
Recovery of prolactin post-PEG precipitation, using the above method, was assessed using the monomeric prolactin international standard IS 84/500 (National Institute for Biological Standards and Controls, Herefordshire, UK). The standard was prepared as previously described 22 but with the following modifications and analysed pre- and post-PEG precipitation (n = 9); the standard was first reconstituted in PBS and then diluted to a final concentration of approximately 900 mIU/L in prolactin naive (<12 mIU/L) double charcoal-stripped defibrinated serum (SCIPAC, UK).
The classification of hyperprolactinaemia using the newly derived post-PEG monomeric reference ranges was compared to the previous approach of percentage recovery using routine samples (n = 175) received by our laboratory (from different patients) for prolactin analysis with a total serum prolactin >700 mIU/L (131 females and 44 males). Percentage recoveries were calculated as follows: after correction of the dilution factor, the post-PEG prolactin values were multiplied by 1.2 (derived from international standard recovery experiment described above) in order to compensate under recovery of monomeric prolactin post-PEG precipitation. This was then divided by the original prolactin value and multiplied by 100.
Data analysis was performed using GraphPad Prism version 4.00 (GraphPad Software, San Diego, USA).
Results
Statistics for male and female sample data for total and post-PEG prolactin.
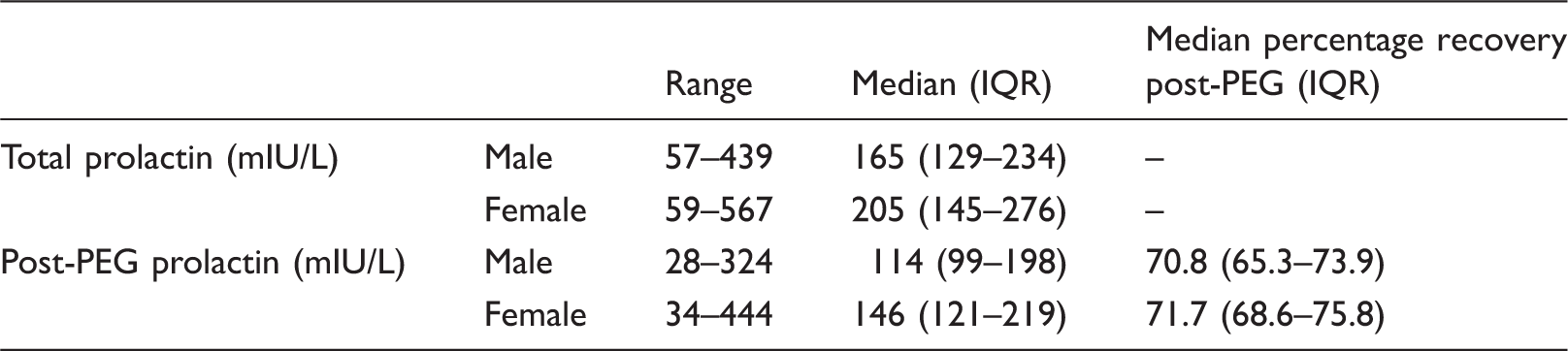
Gender-specific reference ranges for serum total and monomeric post-PEG precipitation prolactin for the re-standardised Abbott Architect prolactin assay.
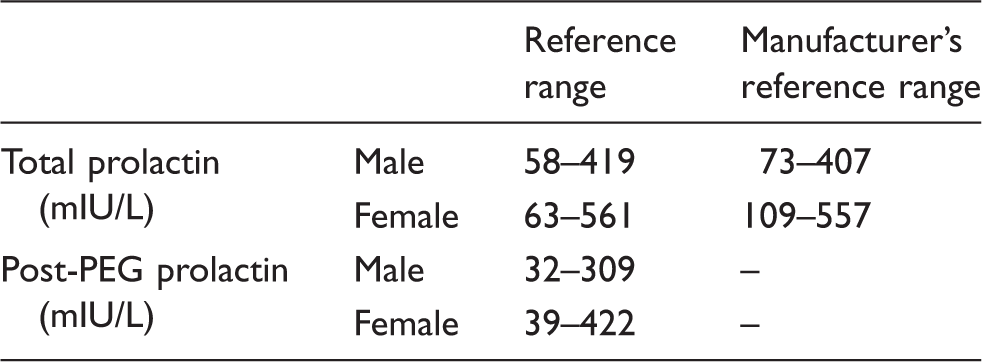
The 95% reference ranges (2.5th–97.5th percentiles) for serum total prolactin (pre-PEG) were 58–419 mIU/L and 63–561 mIU/L for males and females, respectively (Table 2). Male and female post-PEG monomeric prolactin reference ranges were 32–309 mIU/L and 39–422 mIU/L, respectively (Table 2). The observed differences between the male and female sample populations in the post-PEG percentage recoveries were not significant as determined using a Mann–Whitney test (P = 0.1003).
Mean (SD) recovery of the IS 84/500 monomeric prolactin international standard was 80.0 (2.3)%; mean (SD) pre- and post-PEG prolactin concentrations were 892 (25) mIU/L and 714 (20) mIU/L, respectively, with a percentage coefficient of variation of 2.8% in both cases. A factor of 1.2 was derived from this experiment for use in the percentage recovery calculation to correct for under-recovery of monomeric prolactin post-PEG precipitation.
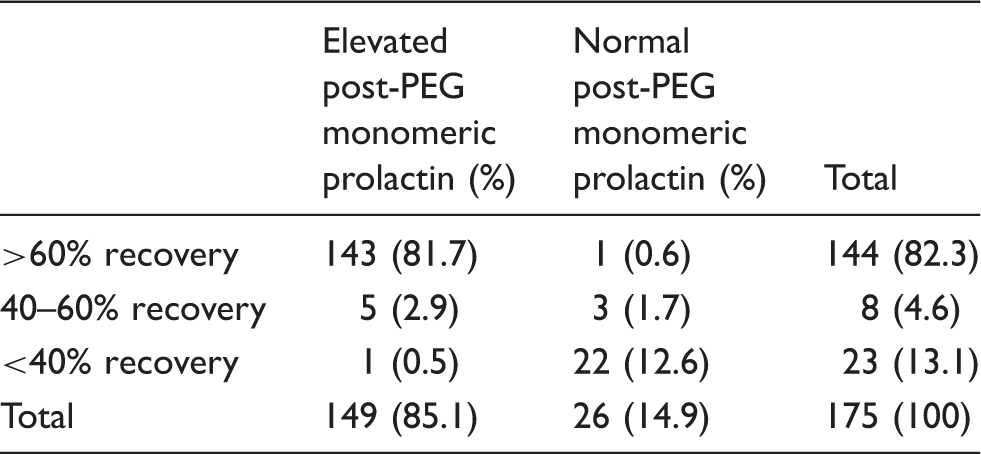
Comparison of macroprolactinaemia screening methods: percentage recovery versus monomeric post-PEG reference ranges.
Discussion
Screening hyperprolactinaemic samples for macroprolactinaemia, a well-recognised cause of interference in prolactin assays, is important to prevent potential misdiagnosis and mismanagement of patients. Calculation of the percentage recovery post-PEG precipitation is the current conventional approach, 18 with a >60% recovery indicating genuine hyperprolactinaemia, 40–60% recovery indicating that macroprolactinaemia may be present (i.e. an equivocal result requiring further investigation by GFC) and a <40% recovery suggesting the presence of significant macroprolactinaemia. However, this approach lacks specificity and can be misleading12,22,23 as, although rare, it is possible for a patient to have hyperprolactinaemia due a simultaneous excess amount of both macroprolactin and monomeric prolactin. For example, the percentage recovery may be low (<40%), yet the patient still has supraphysiological concentrations of bioactive monomeric prolactin which may warrant further investigation. Data from the UK NEQAS prolactin EQA scheme also suggested that the Abbott method is more susceptible to macroprolactin interference compared to other assays (UK NEQAS [Edinburgh]). Fahie-Wilson and coworkers22,26 advocated a move away from the use of potentially misleading percentage recoveries in favour of absolute post-PEG prolactin concentrations together with appropriately derived reference ranges. Beltran et al. 22 previously reported serum total and post-PEG monomeric prolactin reference ranges for commonly used immunoassay platforms. The Abbott Architect method has subsequently undergone re-standardisation, 28 thus necessitating revision of these ranges. The use of post-PEG monomeric ranges also accounts for any interference by PEG in the prolactin assay which has been reported previously.22–25
In this study, we derived local gender-specific reference intervals for both serum total prolactin and monomeric prolactin following PEG precipitation for the current generation Abbott method.27,28 While at the upper limit of normal, our derived total prolactin reference ranges were consistent with those stated by the manufacturer, 27 at the lower limit of normal both our male and female ranges were lower than those stated by Abbott. Furthermore, both our serum total and monomeric prolactin reference intervals were wider than those published by Beltran et al. 22 for both males and females. Although Abbott stated that prolactin results may be 15–25% lower 28 with the re-standardised assay, this was not observed in this study. Furthermore, Abbott made no adjustment to the reference range stated in the kit insert following this re-standardisation.27,28 Our observed mean percentage recovery of the IS 84/500 monomeric prolactin standard material (80%) for the current Abbott assay was identical to that reported by Beltran et al. 22 for the previous version of the assay.
The comparison between the old (percentage recovery) and new (monomeric prolactin) approaches to macroprolactinaemia screening revealed two cases that would have been classified differently depending upon the approach used. On the basis of percentage recovery, they would have been incorrectly classified as either macroprolactinaemia (despite an elevated monomeric prolactin) or true hyperprolactinaemia (despite a normal monomeric prolactin). One sample (male) had a monomeric prolactin concentration (470 mIU/L) above the upper limit of our reference range (309 mIU/L), yet the percentage recovery was <40% (36%). In the other case (female), the monomeric prolactin concentration (414 mIU/L) was below the upper limit of our reference range (422 mIU/L) despite a percentage recovery of >60% (69%). However, it should be noted that, in the absence of GFC analysis, the absolute distribution of prolactin species in each of the patients’ samples was not known and other interference could not be excluded. In addition, eight samples were classified as having intermediate recovery (40–60%) which, under our previous protocol, would have had to be referred to an external laboratory for GFC analysis.
Following this study, we adopted the approach of routinely screening patients’ samples with a total serum prolactin >700 mIU/L for macroprolactinaemia using the monomeric reference ranges derived in this study to differentiate between genuine hyperprolactinaemia and macroprolactinaemia. Patients’ specimens with a total serum prolactin >700 mIU/L are screened for macroprolactinaemia because values lower than this are thought less likely to be clinically significant.
In conclusion, the use of post-PEG precipitation monomeric prolactin reference ranges not only simplifies result reporting and interpretation for the requesting clinician but also avoids potential misdiagnosis and mismanagement. Furthermore, the change in method has also provided a small cost saving by reducing the number of samples referred for GFC analysis.
Footnotes
Acknowledgements
The authors would like to thank the staff of Southend University Hospital Biochemistry Department for advice and donation of samples.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not needed.
Guarantor
RG.
Contributorship
SJW, MC and CF conceived the study. MC and SJW researched the literature, designed the studies, processed and analysed the data, and wrote the first draft. All authors reviewed and edited the manuscript, and approved the final version of the manuscript.
