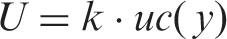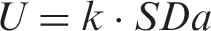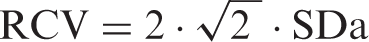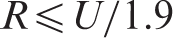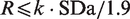Abstract
Background
Measurement uncertainty (MU) estimates can be used by clinicians in result interpretation for diagnosis and monitoring and by laboratories in assessing assay fitness for use and analytical troubleshooting. However, MU is not routinely used to assess the appropriateness of the analyte reporting interval. We describe the relationship between MU and the analyte reporting interval.
Methods and results
The reporting interval R is the smallest unit of measurement chosen for clinical reporting. When choosing the appropriate value for R, it is necessary that the reference change values and expanded MU values can be meaningfully calculated. Expanded MU provides the tighter criterion for defining an upper limit for R. This limit can be determined as R ≤ k·SDa/1.9, where SDa is the analytical standard deviation and k is the coverage factor (usually 2).
Conclusion
Using MU estimates to determine the reporting interval for quantitative laboratory results ensures that reporting practices match local analytical performance and recognizes the inherent error of the measurement process.
Introduction
The release and adoption of the latest 2012 revision of ISO 15189 has seen greater attention paid by many laboratories to measurement uncertainty (MU) and its estimation. 1 The previous version required laboratories to determine the uncertainty of results, where relevant and possible, allowing for individual interpretation and implementation of this requirement. Under the 2012 standard, laboratories ‘shall determine measurement uncertainty for each measurement procedure in the examination phases used to report measured quantity values on patients’ samples. The laboratory shall define the performance requirements for the measurement uncertainty of each measurement procedure and regularly review estimates of measurement uncertainty’. 1 MU estimation has thus become a required procedure for ISO 15189-accredited laboratories and is an increasingly common practice. MU estimates are used by clinicians in result interpretation for diagnosis and monitoring and by laboratories in assessing assay fitness for use and analytical troubleshooting.
In the Foreword to the Clinical and Laboratory Standards Institute document ‘Expression of Measurement of Uncertainty in Laboratory Medicine; Approved Guideline’, it states that ‘Uncertainty estimates … can be important in defining the measuring interval of measurement systems to ensure that the quality of results issued meets clinical requirements’. 2 However, MU is not routinely used to assess the appropriateness of the analyte reporting interval. This is evident from the fact that laboratories often use inappropriate reporting intervals and there has been no significant change in reporting intervals since the widespread adoption of MU in clinical laboratories. In this brief note, we describe the relationship between MU and the analyte reporting interval.
Methods and results
The expanded measurement uncertainty, U, defines the interval about the result of a measurement that may be expected to encompass a large fraction of the distribution of values that could reasonably be attributed to the measurand.
3
The calculated confidence interval encompasses the true value of the population parameter at a specified level of confidence.
4
This uncertainty can be communicated to users by reporting laboratory results together with the appropriate U as ± U, where x is the measured concentration of measurand y, and U is calculated using an agreed coverage factor (usually 2).
5
Reference change value RCV
6
Expanded measurement uncertainty U
Thus, U provides an upper limit for R. An additional factor of 1.9 is required to account for potential information loss due to rounding. With rounding, an apparent difference of 1 unit between results (e.g. 130–131) can hide an actual difference of up to 1.9 (e.g. 129.5–131.4) units.
7
Discussion
It should be noted that our definition of R refers to the reporting rather than the measuring phase of the analytical process. The measuring unit size used in the measuring phase must be smaller than the analytical standard deviation to allow accurate calculation of the standard deviation itself. Thus, in the example of serum sodium measurement used above, given SDa of 1 mmol/L, we would recommend measuring in 0.1 mmol/L increments but reporting using an R of 1 mmol/L. We appreciate that in practice, SDa rarely matches the decile (e.g. 0.01, 0.1, 1) increments used in most laboratory information system, so the closest decile should be selected. 7 When in doubt, the larger decile should be chosen over the smaller (e.g. 1 rather than 0.1), given that the use of SDa as the sole contributor to U undoubtably excludes other real sources of uncertainty, leading to a probable underestimate of U and hence an underestimate of the true value of R.
The approach given is a simplification of the more complex real world where some assays do not have one SDa (or CVa) but a precision profile that varies with the concentration of the analyte. Furthermore, for analytes that span several orders of magnitude, e.g. tumour markers, the reporting interval will vary according to concentration, and the same reporting interval should ideally not be used across the whole span of results. However, with many commonly reported analytes this analysis is valid and useful.
We have described a novel approach to the determination of the reporting interval for an assay, one that is determined by the uncertainty of the measurement process and therefore provides useful information to the clinician about the interpretation of the result. With widespread routine calculation of MU by clinical laboratories, this information is readily available and ensures that the local reporting practice matches the local analytical performance of the assay. We would advocate that laboratories use the MU process to assess the reporting intervals of the results and ensure that they are appropriate, given the inherent error of the measurement process.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
TB.
Contributorship
TB conceived original idea. Both authors developed concept, reviewed and edited the manuscript and approved the final version of the manuscript.