Abstract
Background
Eales’ disease (ED) is an idiopathic retinal vascular disorder. It presents with inflammation and neovascularization in the retina. Adult men, aged between 15 and 40 years are more susceptible than women. Homocysteine has been implicated in other ocular diseases including age-related macular degeneration (ARMD), central retinal vein occlusion (CRVO) and optic neuropathy. The present study investigates the role of homocysteine in ED.
Methods
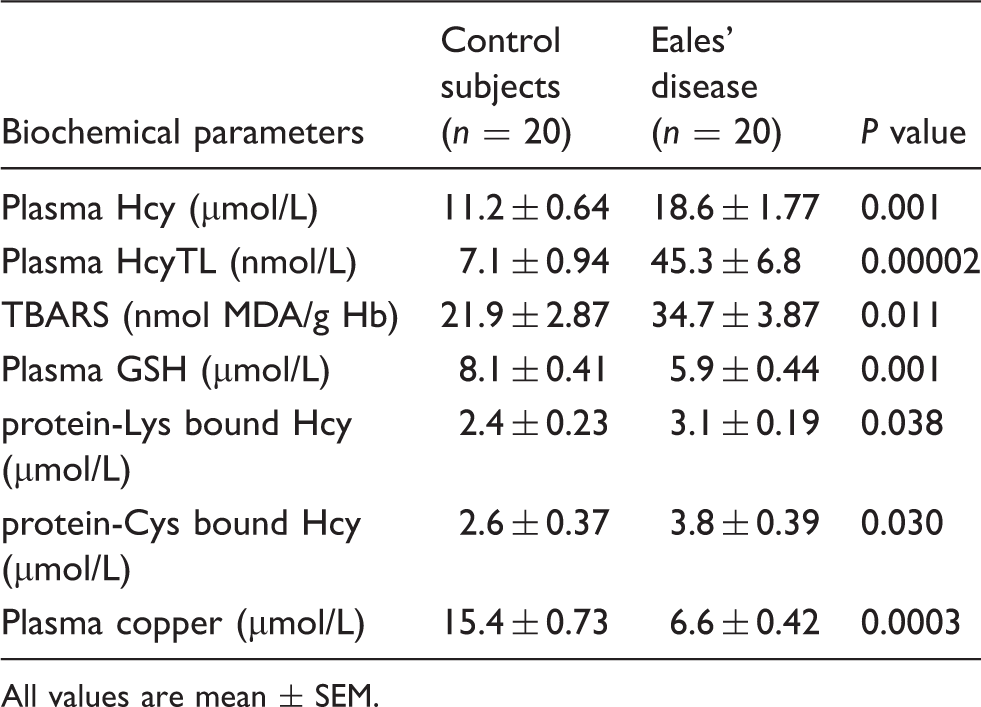
Forty male subjects, 20 with ED and 20 healthy controls, were recruited to the study. Their blood samples were used to measure thiobarbituric acid reactive substances (TBARS), glutathione (GSH), homocysteine, homocysteine-thiolactone, extent of homocysteine conjugation with proteins and plasma copper concentration.
Results
In the ED group, plasma homocysteine (18.6 ± 1.77
Conclusions
Increased homocysteine, and its metabolite thiolactone, is associated with the functional impairment of protein due to homocysteinylation in ED.
Introduction
Eales’ disease (ED) is an idiopathic retinal periphlebitis characterized by capillary non-perfusion and neovascularization. 1 It was first described by Henry Eales in 1880. 1 The disease is more prevalent in the Indian subcontinent than anywhere else in the world. 2,3 It occurs primarily in otherwise healthy adult men aged between 15 and 40 years, 4 initially presenting as retinal periphlebitis and later as retinal ischaemia that may lead to neovascularization. The biochemical studies undertaken earlier by our group have revealed increased oxidative stress, free radical accumulation, oxidative damage to DNA and protein and decreased antioxidant status in the blood and vitreous samples of individuals suffering from ED. 5
Homocysteine (Hcy) is a sulphur-containing amino acid with a free thiol (sulphydryl; SH) group, formed from methionine through
Recent studies have shown that the atherogenic effect of HHcy may be accounted for by homocysteine-thiolactone (HcyTL). 16 HcyTL is formed from Hcy, as a result of an error in the editing function of methionyl-tRNA, when it reacts with homocysteine instead of methionine. 17,18 Inadvertent reactions of HcyTL with proteins are harmful. 19 Protein homocysteinylation may contribute to an increased atherothrombotic effect in individuals with HHcy. 17 Besides, increased concentration of homocysteine results in abnormal chelation of the trace element copper resulting in copper deficiency, which is a leading cause of cardiac diseases. 20
To the best of the authors’ knowledge, there has been no substantial report on the association of homocysteine and homocysteine-thiolactone in ED. Hence, the aim of the present study was to investigate the plasma concentrations of Hcy and HcyTL which in turn modify the proteins by protein-Cys bound Hcy and protein-Lys bound Hcy. In addition to the above, the concentrations of the oxidative stress marker thiobarbituric acid reactive substances (TBARS), antioxidant glutathione (GSH) and copper (Cu) have also been investigated.
Materials and methods
Chemicals
All the fine chemicals used in the study were purchased from Sigma Chemical Company (St Louis, MO, USA) unless otherwise specified. All other high-grade reagents were obtained from E-Merck chemicals (Mumbai, India). Water used in this study was purified through Millipore Water Purification System (Millipore Co., India).
Patients
Following approval from the Institutional Ethics committee (REC number 40-2006-P, 41-2006-P), 20 patients were recruited for study in the Sankara Nethralaya Eye Hospital, between August 2009 and August 2011.
Selection criteria
ED was diagnosed on the basis of the following criteria: periphlebitis of the retina, neovascularization and vitreous haemorrhage not associated with anterior uveitis, choriditis, parsplanitis or other retinal vascular diseases, which mimic ED.
Twenty patients with active vasculitis of unknown aetiology as diagnosed after detailed fundus examination by an ophthalmologist, and 20 healthy adult volunteers were recruited to the study based on the inclusion criteria. Active vasculitis in ED is characterized by the presence of serous exudates around the retinal veins with retinal oedema. In addition to the above, there was venous occlusion, neovascularization and fibrovascular scar formation with associated vitreous haemorrhage observed in all patients. All subjects were men aged between 15 and 40 years and were otherwise healthy. The study population were non-smokers, non-alcoholics and not on any vitamin supplements. Prior to blood collection, the patients were not on any corticosteroid treatment or antioxidant supplement. Informed consent was obtained from all the participants in the study. Blood samples were drawn into ethylenediaminetetraacetic acid (EDTA) containing tubes after the participants had fasted overnight. Plasma was separated immediately from blood cells by centrifugation at 3000
Determination of TBARS in erythrocytes
TBARS determination in erythrocytes was performed by the method described by Devasagayam
Determination of plasma GSH
Plasma GSH was determined by the method of Hu
22
with minor modifications.
23
The concentration of plasma GSH was expressed in
Determination of plasma homocysteine
Fasting plasma total Hcy analysis was done using a commercial ELISA kit (Bio-Rad, Hercules, CA, USA). The plasma concentration of Hcy was expressed in
Determination of protein-Cys bound Hcy and protein-Lys bound Hcy
Measurement of protein homocysteinylation was performed by the method of Jakubowski
HPLC analysis of protein-Cys bound Hcy and protein-Lys bound Hcy
Released Hcy from plasma proteins, i.e., protein-Cys bound Hcy and protein-Lys bound Hcy was analysed using HPLC as described by Coral
Determination of plasma homocysteine thiolactone (HcyTL)
Sample preparation for the determination of HcyTL has been described by Jakubowski. 26 Plasma Hcy-Thiolactone was determined by HPLC using a cation-exchange PolySULFOETHYL aspartamide column by Chwatko and Jakubowski. 27 The detection limit was 10 pmol/L HcyTL. The concentrations of plasma HcyTL were expressed in nmol/L.
Determination of plasma copper
Plasma samples measuring around 0.35 mL were digested with 1.0 mL of concentrated nitric acid:perchloric acid (Merck, India) mixture (5:1) by heating in a china dish for 30 min. The ash was finally dissolved in 2.0 mL of 0.2% nitric acid to prevent adsorption of minerals on the wall of the container. The copper concentration was measured by atomic absorption spectrophotometer (AAS) (AA700; Perkin Elmer, Santa Clara, CA, USA) as described previously
28
and expressed as
Statistical analysis
All values were expressed as mean ± standard error mean. With SPSS software (version: 16.0), the raw data were analysed for statistical significance using Student ‘
Results
Increased concentrations of plasma Hcy and HcyTL correlate positively with TBARS and negatively with GSH in ED
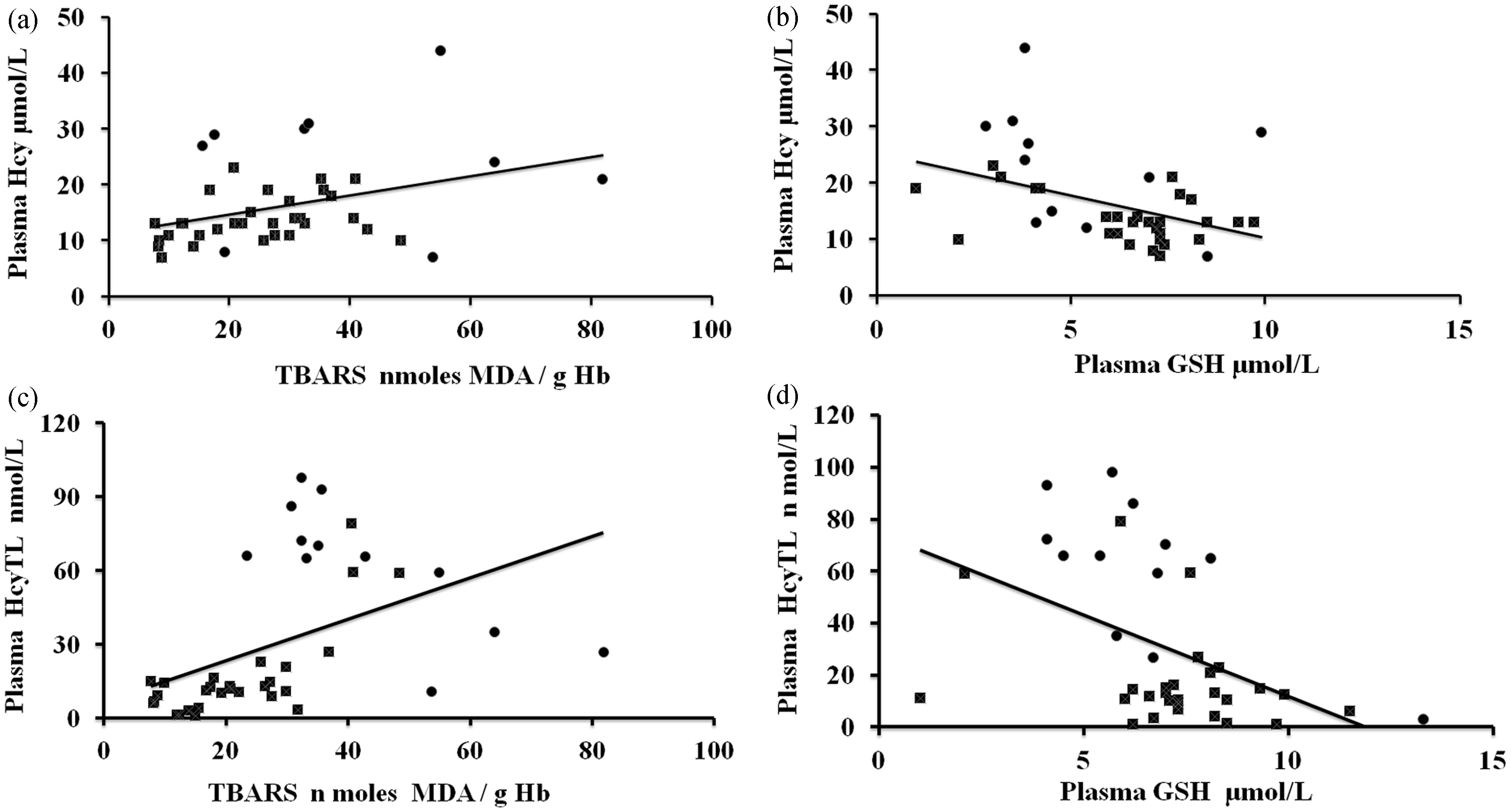
The concentrations of plasma Hcy, HcyTL, TBARS and GSH in patients and control subjects are given in Table 1 and Figure 1. There was a significant increase in the observed concentrations of erythrocyte TBARS in the ED groups compared to the control group. Results from our study indicated that increased concentrations of oxidative stress lead to increased concentrations of plasma Hcy and its metabolite HcyTL and a decrease in the concentrations of antioxidants GSH in ED patients when compared to controls (Figure 1). There was a significant, positive correlation between plasma Hcy and TBARS, plasma HcyTL and TBARS and significant negative correlation between plasma Hcy and plasma GSH, plasma HcyTL and plasma GSH (Figure 2).
Concentrations of Hcy, HcyTL, TBARS and GSH among ED and healthy controls. Statistical significance between control ( Correlations between plasma Hcy, plasma HcyTL, TBARS and plasma GSH. (a) Positive correlation between plasma Hcy and TBARS ( Concentrations of biochemical parameters measured in control and Eales’ disease subjects. All values are mean ± SEM.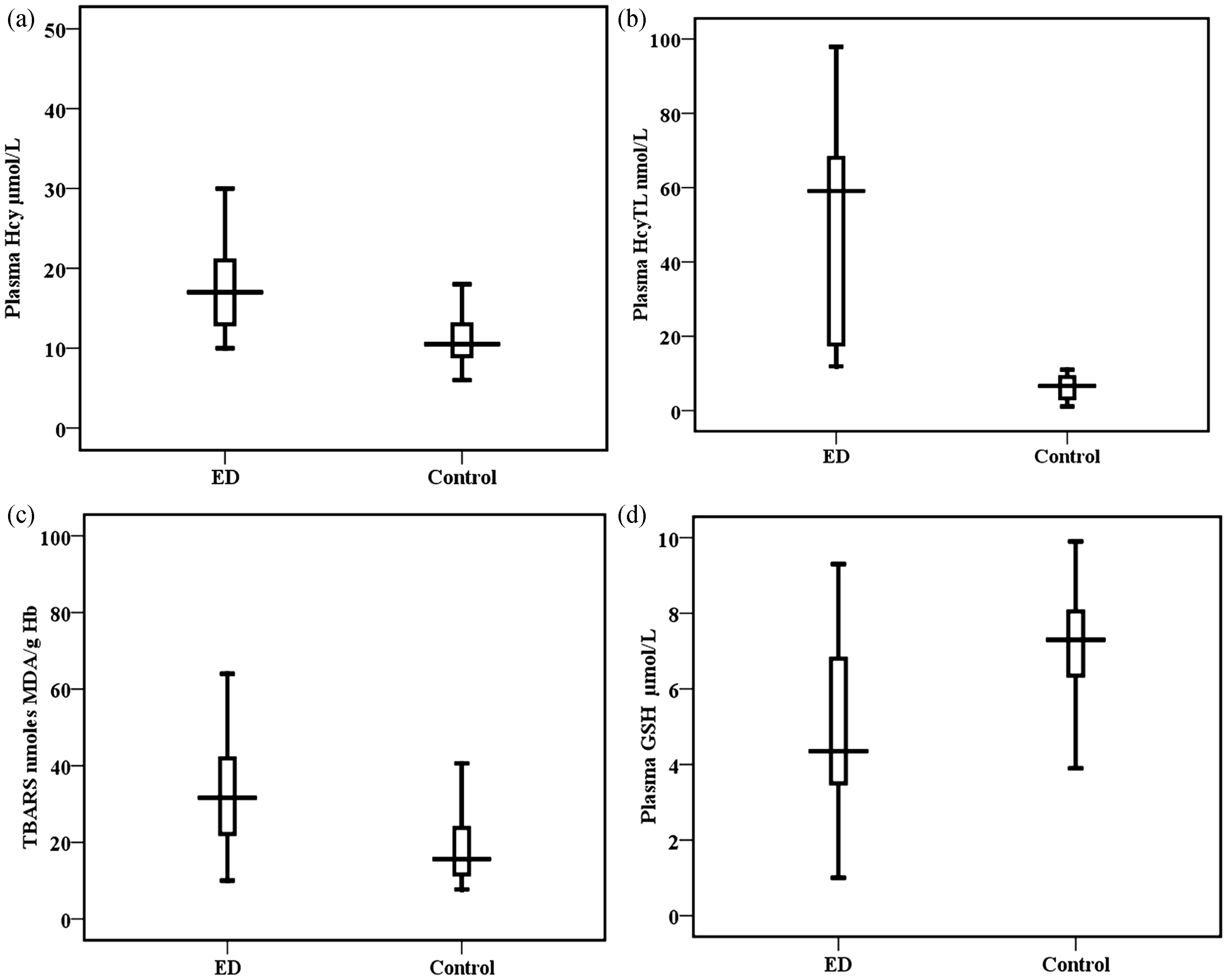
Structural damage to proteins due to Hcy and its metabolite HcyTL in ED
HcyTL is a product of Hcy. Increased HcyTL in ED may be due to increased Hcy in the patients. HcyTL has the ability to form isopeptide bonds with protein lysine and cysteine residues, which impairs or alters protein functions.
17
We estimated lysine and cysteine bound Hcy in the plasma proteins of study subjects by HPLC. We found increased concentrations of protein-Cys bound Hcy and protein-Lys bound Hcy in ED patients compared to controls. There was a positive correlation between plasma HcyTL, and protein-Cys bound Hcy and protein-Lys bound Hcy (Figure 3).
Comparison of protein-Cys bound Hcy and protein-Lys bound Hcy among ED and healthy controls. Statistical significance between control (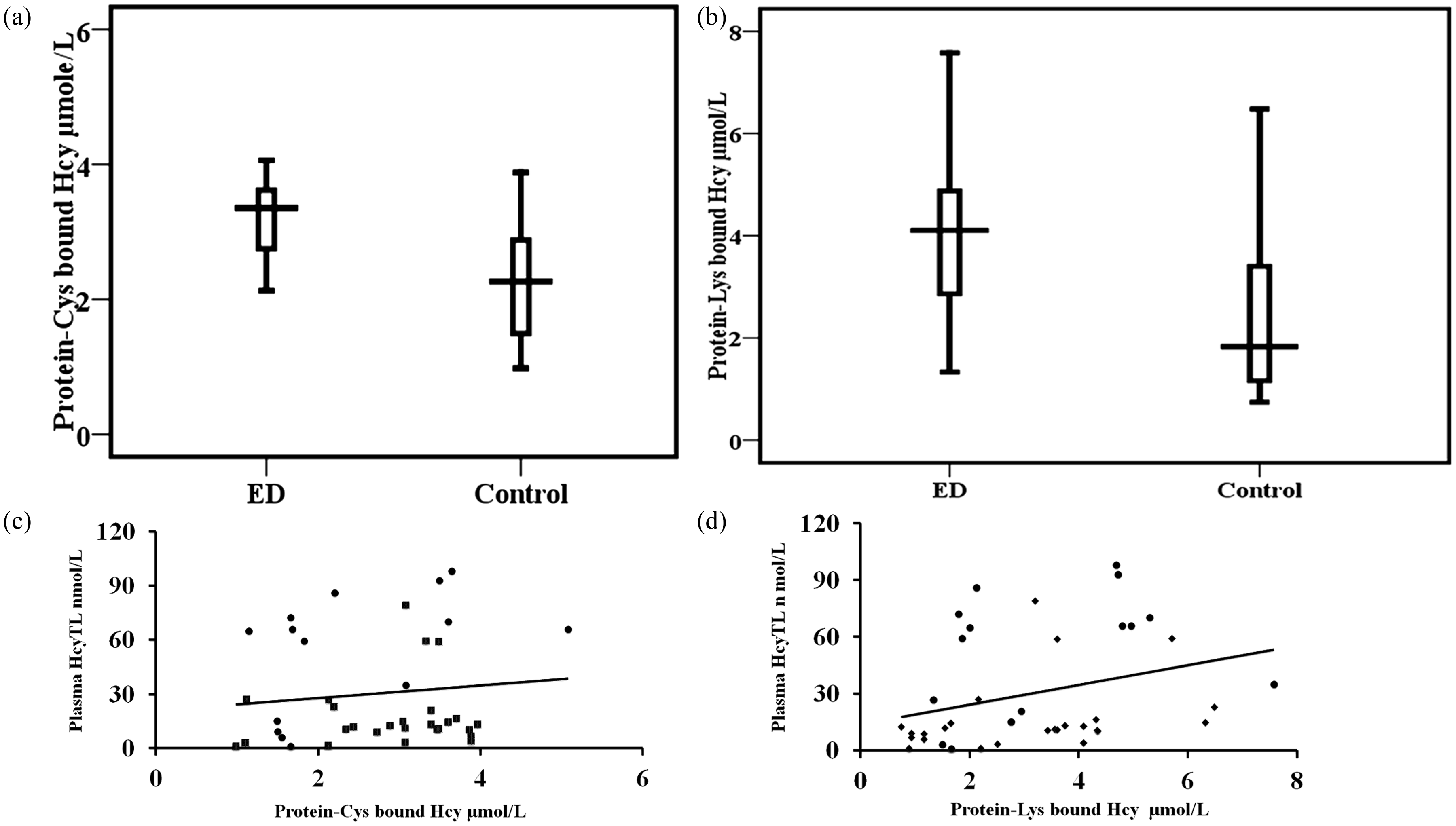
Increased Hcy and its metabolite HcyTL negatively correlated with trace element – copper
Increase of Hcy causes a deficiency of copper.
29
We conducted AAS analysis of plasma copper to establish whether any change in copper is seen in ED (Table 1). Increase of Hcy and HcyTL decreases the plasma copper concentration in ED patients compared to controls, and there was a negative correlation between plasma copper and plasma Hcy and plasma HcyTL in circulation (Figure 4).
Comparison of plasma copper among ED and healthy controls. Statistical significance between control (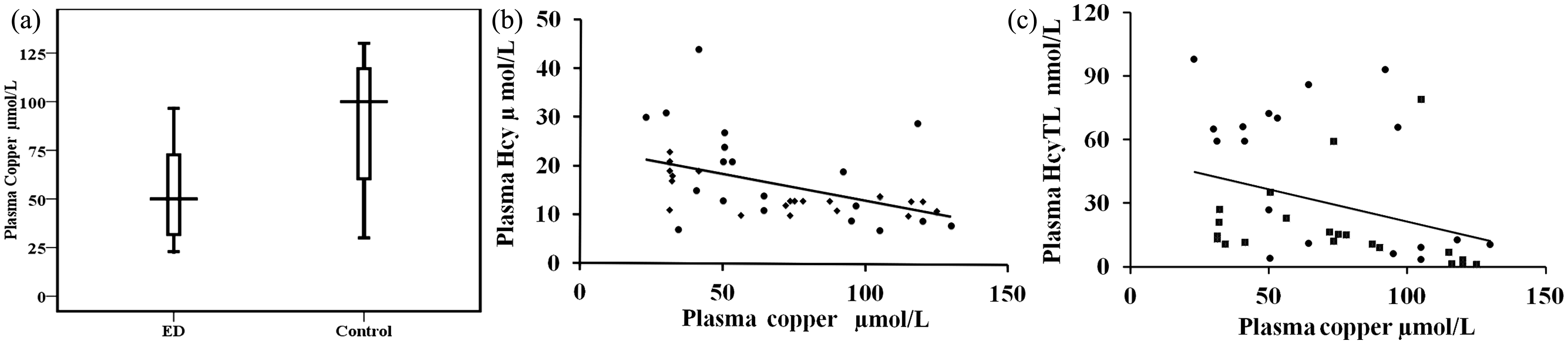
Discussion
Oxidative stress has been shown to be involved in the pathophysiology of intraocular inflammatory diseases. 30 Chronic intraocular inflammation is a major cause of blindness. The loss of vision is the result of damage inflicted by the infiltration of inflammatory cells into the retina, which is facilitated by the disruption of the blood-retinal barrier. 30 The retina is considered to be the most susceptible tissue to oxidative stress. This may be due to the following reasons: the presence of abundant mitochondria, 31 which consume large amounts of oxygen and the presence of photosensitive retinal pigments. The latter presents a high degree for photodynamic oxidation in the retina. 32
Protein peroxidation can occur by various mechanisms, such as an increase in the production of reactive oxygen species (ROS) and decrease in the rate of removal of oxidised species. 33 This study established the diminished GSH content and impairment of copper homeostasis. This could be the possible reason for the elevated HcyTL and Hcy which impairs the protein function in patients with ED. The decrease in plasma GSH in ED may be an outcome of greater GSH consumption by ROS generated by phagocytes during the process of inflammation. 34 Our earlier work has also shown diminished GSH content in erythrocytes and vitreous of patients with ED. 35
Clinical manifestations of ED occur due to three basic pathological changes 3,5 : inflammation (peripheral retinal perivasculitis), ischaemic changes (peripheral retinal capillary non-perfusion) and neovascularization of the retina or disc which often leads to vitreous haemorrhages. Most of these pathological changes can be brought about by increased Hcy leading to damage directly or indirectly. Hcy reacts with proteins through a process known as homocysteinylation 36 which denatures and impairs protein function causing vascular changes. In addition to the above, Hcy also causes the formation of ROS, chiefly hydrogen peroxide (H2O2) which further aggravates the extent of vascular pathology. 37 Besides the damage through the ROS, the proteins are further modified through cysteine and lysine residues conjugated to Hcy and HcyTL.
Increased Hcy and HcyTL leads to protein alterations and diminishes the antioxidant capacity observed in patients with ED. The increase in thiolation is a consequence of increased Hcy. 36 An important factor affecting the proteins and thereby producing vascular changes is through the angiotoxic and atherogenic effects of HcyTL. 19 The amount of HcyTL formed is decided by the concentrations of Hcy; the higher the Hcy higher the HcyTL. 26,36 There are reports that some other factors could cause increased formation of HcyTL. Moreover, the biochemical half-life of serum or plasma HcyTL is also quite low, i.e., 0.5 h and 1.5 h, respectively. 26 However, in its short survival period and in increased concentration, the HcyTL reacts with proteins and causes homocysteinylation. Homocysteinylation results in severe damage to the protein structure resulting in changes in their function, leading to vascular pathology. 36 Hcy, as well as its metabolite, the thioester HcyTL, play an important role in atherothrombosis in the following ways. HcyTL modifies LDL 38,39 and inhibits lysyl oxidase 38 as well as insulin signalling. 40 Of special significance is the degeneration of multiple retinal cells and photoreceptors by HcyTL in HHcy, which was observed in the present study. Hence, it is suggested that the retinal proteins get homocysteinylated to a significant extent in ED, and this could contribute to ocular pathology.
It is interesting to note that the metabolism of Hcy and copper are intimately interlinked. Increased Hcy causes a deficiency of copper which would also mean weakened oxidant defence as copper is a component of the two enzymes superoxide dismutase (SOD) as Cu-Zn-SOD and caeruloplasmin, 41,42 a ferro-oxidase, which are antioxidants. Therefore, decreased copper concentration would aggravate the formation of ROS and thereby TBARS through a decrease of these two enzymes. 42 Being a ferro-oxidase, caeruloplasmin scavenges Fe2+ and mitigates the Fenton’s and Haber-Weiss reaction. Our earlier work has also shown diminished SOD and increased oxidative stress in erythrocytes of patients with ED. 43 Earlier, we have reported, an increase in copper in monocytes of ED patients when compared to healthy controls, 44 in contrast to our present study wherein there is a decrease in its concentration in circulation. At this point, the reason for this difference in the concentrations of copper intracellularly and extracellularly is not known, but it warrants further research related to copper transporter. In our work, we find that the copper deficiency and Hcy inhibit lysyl oxidase, 25,45 a copper-dependent enzyme needed for the formation of crosslinks in collagen and elastin. It has been shown that in our earlier study decreased lysyl oxidase activity in the vitreous 25 could result in inadequate cross-linking of collagen and elastin weakening the extracellular matrix integrity. Dietary supplementation of therapeutic amounts of copper is reported to reduce the concentrations of Hcy. 46
It is known that ED has an unknown aetiology. However, from our present work, it appears that increased Hcy might be one of the biochemical alterations for the ocular pathology. We observed this in our earlier study in CRVO. 47 Thus, Hcy and its metabolite, HcyTL, affect protein function through homocysteinylation. 17 There is oxidative stress and poor oxidant defence in ED. 5 This could be brought about by Hcy, and other factors like deficiency of antioxidant vitamins which could exacerbate it. ED affects young adult men but not women. 5 The concentration of Hcy is shown to be greater in men than in women and in the young more than the elderly. 6 ED is widely prevalent in the Indian subcontinent. 2 It is reported that Hcy concentrations of Indians are at least 6% higher than those of Caucasians, indicating that nutrition plays a role in the pathology of ED. 48 To the best of our knowledge, this is the first report on the concentrations of Hcy and its metabolite HcyTL in patients with ED.
From a therapeutic point of view, as copper supplementation has been reported to correct the increase of Hcy, copper should find an important place in the treatment of ED. Vitamin B12 and folic acid needed for the conversion of Hcy to methionine, and antioxidant vitamins E, A and C can also be considered for the treatment of ED. Besides, protein diet rich in glutamic acid, glycine and cysteine may help in the synthesis of GSH. It has to be administered to the patient to quench the oxidant stress and normalise the Hcy concentrations. In summary, normalizing the concentrations of Hcy appears to be the front line therapeutic management of ED.
Footnotes
Acknowledgements
The authors thank the Indian Council of Medical research (ICMR) for their funding. The authors would also like to thank Dr S. Bharathidevi, Mrs R. Punitham and Mr R. Swaminathan for their constant encouragement and help.
Declaration of conflicting interests
None.
Funding
Grant from (ICMR) Indian council of Medical research (ICMR/52/16/2007-BMS).
Ethical approval
All protocols were approved by the Institution’s Ethics Board (REC number 40-2006-P, 41-2006-P).
Guarantor
KNS.
Contributorship
MB processed the sample and analysed the data collected. SR and KNS conceived the work, analysed the data and wrote the paper. KC, AN and RS helped in manuscript correction and discussion. JB the ophthalmic consultant who treated the patients, obtained their consent, recruited and in filling the clinical proforma in this study.
