Abstract
The therapeutic efficacy of adipose mesenchymal stem cells (Ad-MSCs) for acute kidney injury (AKI) has been investigated extensively, and the anti-apoptotic, anti-inflammatory, and proangiogenic effects of heme oxygenase-1 (HO-1) reportedly ameliorate AKI. We hypothesized that the therapeutic efficacy of Ad-MSCs is dependent on their expression level of HO-1. The viability and migration ability of cisplatin-treated human renal proximal tubular epithelial cells were assessed. Sprague–Dawley rats were divided into control, cisplatin (10 mg/kg), and cisplatin plus Ad MSCs (with high and low HO-1 expression) groups. The HO-1 expression level in hAd-MSCs increased with increasing passage number, peaking at passage 4 and decreasing thereafter. The viability and migratory ability of hAd-MSCs with high HO-1 expression were greater than those of hAd-MSCs with low HO-1 expression. Renal tubular toxicity in cisplatin-treated rats was ameliorated by administration of hAd-MSCs with high HO-1 expression, although the levels of blood urea nitrogen and serum creatinine did not differ according to the level of HO-1 expression. The magnitude of reactive oxygen species induced DNA damage was lower in hAd-MSCs with high HO-1 expression than in those with low HO-1 expression. Administration of hAd-MSCs significantly suppressed cisplatin induced apoptosis. Also, hAd-MSCs with high HO-1 expression were more resistant to cisplatin-induced apoptosis than were those with low HO-1 expression. hAd MSCs with high HO-1 expression have therapeutic potential for cisplatin induced nephrotoxicity, based on our in vitro and in vivo results. These findings will facilitate the development of novel therapeutic strategies for cisplatin-induced AKI.
Introduction
Heme oxygenase-1 (HO-1) catalyzes the breakdown of heme, releasing iron, carbon monoxide, and biliverdin; biliverdin is subsequently converted into bilirubin. Induction of HO-1 expression is associated with protection against acute kidney injury (AKI) due to its anti-apoptotic, anti-inflammatory, and pro-angiogenic effects; in contrast, global deficiency or inhibition of HO-1 worsens renal function.1–3 Both induction of HO-1 expression and its overexpression protect against ischemia-reperfusion injury, nephrotoxin-induced renal injury, and rhabdomyolysis.4–8 However, other studies have reported that up- or down-regulation of HO-1 results in exacerbation of, or protection against, AKI.9–11 Moreover, the use of chemical inhibitors of HO-1 expression may have hampered evaluation of the role of HO-1 in AKI. 12
Mesenchymal stem cells (MSCs) are undifferentiated adult cells that can be isolated from a variety of tissues. The therapeutic effects of MSCs on AKI have been investigated. Various in vitro and in vivo studies showed that MSCs ameliorated renal injury and accelerated its repair.13–18 Adipose tissue is a readily accessible reservoir of mesenchymal progenitor cells.19–21 Adipose tissue-derived MSCs (Ad-MSCs) have characteristics similar to those of their bone-marrow counterparts, including in terms of their morphology, capacity for proliferation, and multilineage differentiation.22,23 Indeed, Ad-MSCs can differentiate into osteogenic, chondrogenic, myogenic, neurogenic, or endothelial cells.24–28 Furthermore, Ad-MSCs, including those harvested from adults, have a low senescence rate and do not trigger immune rejection. 29 The treatment of AKI using Ad-MSCs has been reported.30,31 According to our previous study, 31 infusion of Ad-MSCs resulted in a broad therapeutic effect, including reductions in apoptotic cell death, renal signaling, inflammation, and the release of toxic cytokines, improved renal function and tissue morphology, and increased survival. However, no study has assessed the impact of the expression level of HO-1 on the therapeutic efficacy of Ad-MSCs. We reported previously that Ad-MSCs ameliorate cisplatin-induced nephrotoxicity in rats through a variety of mechanisms. 31 In this study, we investigated the effect of the level of HO-1 expression on the protective efficacy of Ad-MSCs against cisplatin-induced nephrotoxicity in rats.
Materials and methods
Ethics approval
The Gyeongsang National University Institutional Animal Care & Ethics Committee approved this study (GLA-120120-R0002).
Culture of human adipose tissue–derived MSCs
Human adipose tissue–derived (hAd)-MSCs were isolated from the adipose tissue of patients who had undergone general abdominal surgery at Gyeongsang National University Hospital, South Korea. All eligible patients (4 males and 1 female, age range 45–79 years, mean age 62.3 years) or their parents provided written informed consent, and permission to isolate MSCs from adipose tissues was granted by the Institutional Review Board of Gyeongsang National University Hospital (MR7404-130). The culture methods were based on those used in a previous study. 31
Phenotypic analysis
The phenotypic analysis was based on those used in a previous study. 31 Flow cytometric analysis for MSCs phenotype was doing using FC500 (Bekman). Cells were harvested and washed in flow cytometry buffer and incubated for 20 min in flow cytometry buffer containing fluorescein-conjugated monoclonal antibodies directed against differentiation of MSCs antigens (CD49d, Chemicon) and against hematopoetic antigens (CD34, Miltenyl biotech). Cells at passage 3–5 were used for in vivo experiments.
Animals
Male Sprague–Dawley rats (200–230 g) were maintained under a 12/12 h light/dark cycle in a temperature- and humidity-controlled facility and were provided standard rat chow and water ad libitum. All procedures involving rats were conducted according to the Gyeongsang National University Guide for the Care and Use of Laboratory Animals (GNU-150428-R0024). The rats were divided into four groups. The rats in the control group received a single intraperitoneal injection of saline (n = 5), and those in the cisplatin group were administered a single intraperitoneal injection of cisplatin (10 mg/kg body weight; n = 10). The rats in the hAd-MSC groups with high (n = 10) and low HO-1 expression (n = 10) received cisplatin and hAd-MSC (5 × 105 cells/head) injections into the tail vein 1 day after cisplatin injection. The rats were euthanized at day 3 after cisplatin injection, and blood and tissue samples were collected for analysis of renal function and tissue damage.
Immunoblot analysis
Cells from each passage and individual were lysed in RIPA buffer (#89900, Thermo Scientific. Waltham, MA, USA). Amounts of protein were measured by BCA assay kit (Pierce, Rockford, IL, USA). Proteins (30 µg) were loaded and electroblotted. The blots were probed with primary antibodies against polyclonal anti-HO-1 (Enzo Biochem, New York, NY, USA) at 4°C overnight. The primary antibody was visualized by a secondary antibody and an ECL kit (Amersham Pharmacia Biotech, Piscataway, NJ, USA).
Cell viability assay
Human renal proximal tubular epithelial (HK-2) cells (American Type Culture Collection, Manassas, VA, USA) were used in this study. HK-2 cells were seeded into a 24-well plate at 40,000 cells per well, were treated with 8 µM cisplatin for 6 h, and then HK-2 cells were washed and cultured with hAd-MSCs at 50,000 cells per insert, with high and low HO-1 expression for 24 or 72 h. The viability was assessed by WST-1 assay using a Cell Counting Kit-8 (Dojindo, Rockville, MD, USA). The absorbance was measured at 450 nm with a microplate reader (Bio-Rad). Cell viability is presented as a percentage of the control.
Cell migration assay
The migration ability of hAd-MSCs was assessed by Transwell™ assay. HK-2 cells (4 × 104/well) were exposed to 8 µM cisplatin in α-minimal essential medium (α-MEM) containing 5% fetal bovine serum (FBS) for 6 h in 24-well plates. Subsequently, cisplatin was removed by washing twice with phosphate-buffered saline. hAd-MSCs (5 × 104/insert) were loaded into Transwell inserts and cocultured with HK cells in α-MEM containing 5% FBS. After incubating 24 or 72 h, the medium was removed, cells were fixed in 4% paraformaldehyde for 20 min, washed three times with phosphate buffered saline (PBS), and stained with hematoxylin for 20 min. The cells adhering to the upper surface of the membrane were removed using a cotton applicator. The migrated cells on the lower side of the membrane were counted per high-power field (×400 magnifications).
Assessment of renal function
The levels of blood urea nitrogen (BUN) and serum creatinine (Cr) were assayed using standard diagnostic kits in an ADIVA 1650 Autoanalyzer (Bayer, Tokyo, Japan).
Renal histology and damage scoring
Sections (5-µm-thick) of renal tissue were stained with hematoxylin and eosin. Tubular injury was defined as tubular epithelial necrosis, intratubular debris, and loss of the brush border, and was scored by grading the percentage of affected tubules per high-power field (×400 magnification), as follows: 0, 0%; 0.5, <10%; 1, 10%–25%; 2, 26%–50%; 3, 51%–75%; and 4, 75%–100%. To score tubular injury, the numbers of whole tubules per field were counted under ×400 magnification. The injury score was calculated in at least 10 randomly selected areas of the renal cortex, as follows: injury score (%) = (number of injured tubules ÷ number of whole tubules) × 100.
Terminal deoxynucleotidyl transferase dUTP nickend labeling assay
DNA fragmentation was assayed using a kit (Roche, Indianapolis, IN, USA). A semi-quantitative analysis was performed by enumerating terminal deoxynucleotidyl transferase dUTP nickend labeling (TUNEL)-positive cells in at least 10 randomly selected areas of the renal cortex at ×400 magnification.
Immunohistochemistry
Sections of rat renal tissue were deparaffinized and incubated with a polyclonal primary antibody against 8-OHdG (Abcam (Dawinbio Inc.), Hanam, South Korea) followed by a biotin-conjugated secondary IgG (diluted 1:200; Vector Laboratories, Burlingame, CA, USA), avidin–biotin–peroxidase complex (ABC Elite Kit; Vector Laboratories), and diaminobenzidine tetrahydrochloride. We visualized the tissue sections by light microscopy and captured and analyzed digital images (Nikon Eclipse, Tokyo, Japan).
Statistical analysis
Data are expressed as means ± standard error of the mean (SEM). Statistical analyses were conducted using Prism 8 software (GraphPad Software Inc, San Diego, CA, USA). A value of
Results
Phenotypic analysis of hAd-MSCs
Flow cytometric analysis showed that the Ad-MSCs were CD34−, CD45−, CD29+, CD49d+, and CD105+. 32 The expression of these phenotypic markers on the hAd-MSCs did not differ according to their level of HO-1 expression (Supplementary Figure 1).
HO-1 expression in hAd-MSCs according to passage number and source
The level of HO-1 expression in hAd-MSCs, isolated from one individual according to passage number, is shown in Figure 1(a). The HO-1 expression level increased with increasing passage number, peaking at passage 4 and decreasing thereafter. This pattern was similar in the hAd-MSCs isolated from the other individuals. The level of HO-1 expression in hAd-MSCs at passage 4 varied according to the individual from which they had been isolated (Figure 1(b)). Therefore, we used hAd-MSCs with high HO-1 (HHO-1) expression (e.g. passage 4 in Figure 1(a)) and low HO-1 (LHO-1) expression (e.g. passage 2 in Figure 1(a)) in subsequent experiments.

Heme oxygenase-1 (HO-1) expression levels in human adipose tissue-derived mesenchymal stem cells (hAd-MSCs). Cells were lysed in RIPA buffer and proteins (30 µg) were loaded and probed with polyclonal anti-HO-1: (a) HO-1 expression level according to passage number in hAd-MSCs isolated from “c” patient, (b) HO-1 expression in passage 4 hAd-MSCs isolated from different 5 individuals. β-actin was used as the internal control. p4, passage 4. a–e corresponds to different individuals.
hAd-MSCs with high HO-1 expression ameliorated cisplatin cytotoxicity in renal epithelial cells
To assess the effect of the HO-1 expression level in hAd-MSCs on cisplatin-induced cytotoxicity, HK-2 cells were exposed to cisplatin in the presence of hAd-MSCs with high and low HO-1 expression. The cytotoxicity of cisplatin was significantly enhanced at 24 and 72 h (Figure 2(a) and (b)). However, the cytotoxicity of cisplatin against HK-2 cells was reduced by co-culture with hAd-MSCs - with high or low expression of HO-1 for 24 and 72 h (Figure 2(a) and (b)). We next determined the effect of the HO-1 expression level on the migration of hAd-MSCs.

Effect of hAd-MSCs with high and low HO-1 expression on cisplatin-treated human renal proximal tubular epithelial (HK) cells. After exposure to cisplatin (8 µM) for 6 h, HK-2 cells were washed and cultured with hAd-MSCs, with high and low HO-1 expression, for (a)24 or (b) 72 h. Con, cisplatin-untreated; Cis, cisplatin-treated; Cis + HHO-1, cisplatin plus hAd-MSCs with high HO-1 expression; Cis + LHO-1, cisplatin plus hAd-MSCs with low HO-1 expression. Viability was measured by WST-1 assay. Data are means ± SEM. Differences were significant at 24 and 72 h (*
Migration of hAd-MSCs increased with increasing HO-1 expression
MSCs localize to the damaged kidney and promote its morphological and functional recovery (Herrera et al. 13 and Morigi et al. 17 ). Based on this, we examined the effect of HO-1 expression on the migration of hAd-MSCs - toward cisplatin-treated HK-2 cells using a Transwell system. After 24 h, the hAd-MSCs with high HO-1 expression (Right in Figure 3(a) and (b)) exhibited significantly greater migration than those with low HO-1 expression (Left in Figure 3(a) and (b)). Thus, we next evaluated the effect of the level of HO-1 expression on the ability of hAd-MSCs - to protect against cisplatin-induced AKI.

Effect of HO-1 expression level on the migration of hAd-MSCs. Representative image (a) and quantitative data (means ± SEM) (b) of Transwell migration assays. The cells adhering to the upper surface of the membrane were removed using a cotton applicator. The migrated cells on the lower side of the membrane were fixed in 4% paraformaldehyde, washed, stained with hematoxylin, and were counted. Differences were significant at 24 h (*
hAd-MSCs with high HO-1 expression ameliorated cisplatin-induced AKI
Cisplatin resulted in increased levels of BUN and serum Cr, and those changes were significantly attenuated in the hAd-MSC–injected rats. However, the levels of BUN and serum Cr did not differ according to the expression level of HO-1 in the hAd-MSCs (Figure 4). Cisplatin-treated kidneys exhibited extensive tubular damage, including tubular epithelial necrosis (asterisk), intratubular debris (arrowhead), and loss of the brush border (asterisk) (Cis in Figure 5(a) and (b)). hAd-MSCs with high or low expression of HO-1 suffered less severe damage (Cis + HHO-1, Cis + LHO-1 in Figure 5(a) and (b)). Finally, hAd-MSCs with high HO-1 expression ameliorated cisplatin-induced renal tubular damage more effectively than did those with low HO-1 expression.

Effects of hAd-MSCs with high and low levels of HO-1 expression on cisplatin-induced renal dysfunction. Rats were divided into four groups; those in the control group (Con) were injected with saline and those in the cisplatin group (Cis) with a single dose of cisplatin (10 mg/kg, i.p.). In the other two groups, hAd-MSCs (5 × 105) with a high (Cis + HHO-1) or low (Cis + LHO-1) level of HO-1 expression were injected into the tail vein 1 day after cisplatin administration (Cis + HHO-1). Blood samples were collected for measurement of the BUN and serum creatinine (Cr) levels at Day 3 after cisplatin injection. Data are means ± SEM (n = 10) (*
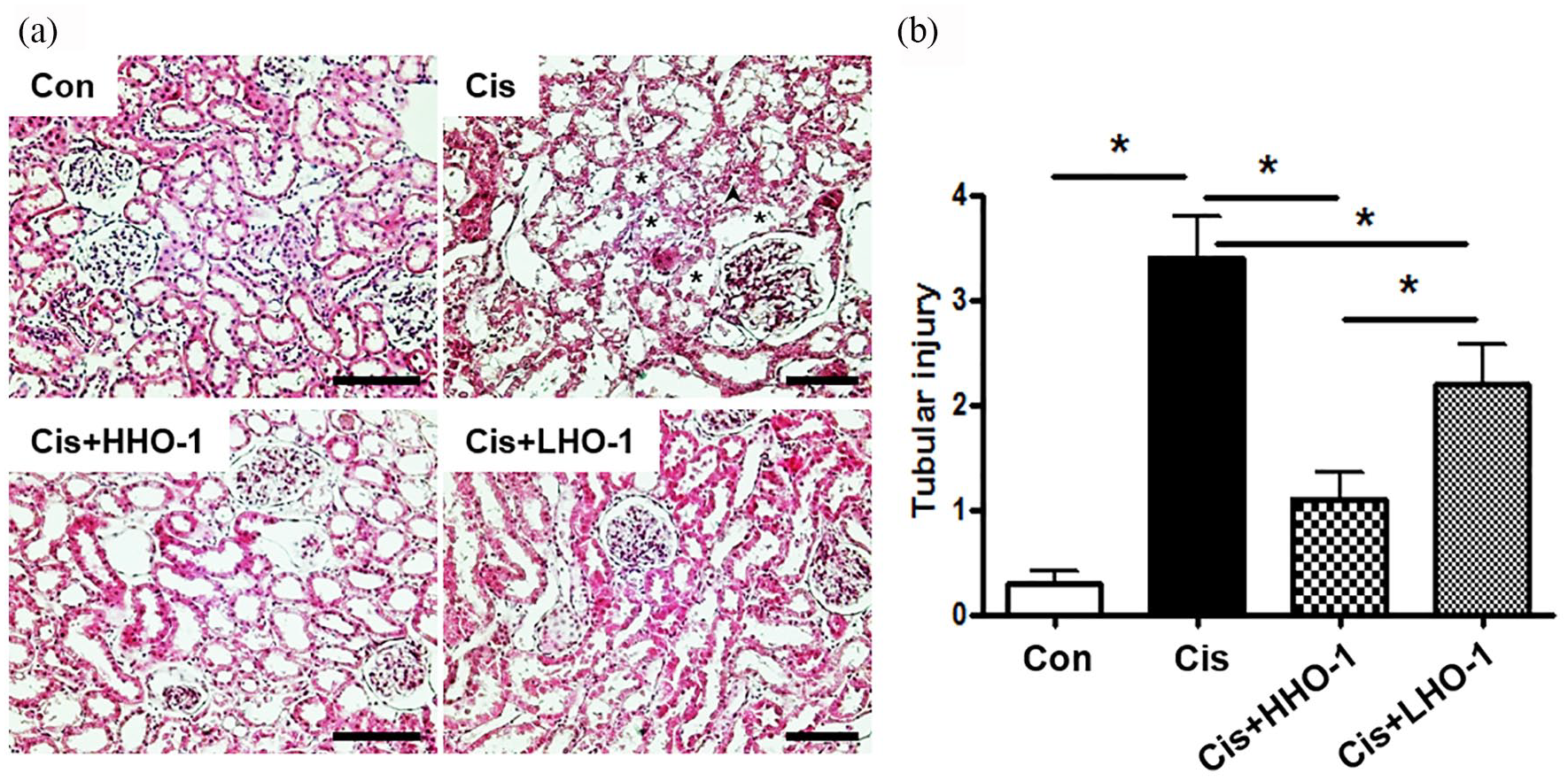
Protective effect of hAd-MSCs on renal morphology after administration of cisplatin. Hematoxylin and eosin staining was performed on sections of kidney tissue 3 days after cisplatin injection. The kidney sections of cisplatin-treated rats (Con) showed severe tubular damage. The kidney sections of Cis + hAd-MSCs rats (Cis + HHO-1, high level of HO-1 expression; Cis + LHO-1, low level of HO-1 expression) showed mild casts and tubular necrosis (a). Tubular injury was defined as tubular epithelial necrosis, loss of brush border, and intratubular debris. Tubules with tubular epithelial necrosis and loss of brush border were represented as asterisk and with tubular epithelial necrosis, loss of brush border, and intratubular debris were as arrowhead. The severity of injury was scored was based on the percentage of affected tubules. Cisplatin-treated kidneys had severe tubular injury (high scores in all categories), whereas tubular injury in hAd-MSCs-injected cisplatin-treated kidneys was significantly ameliorated (b). Data are means ± SEM (n = 10) (*
hAd-MSCs with high HO-1 expression were resistant to cisplatin-induced oxidative stress and apoptosis
Immunohistochemical staining for 8-OHdG, a marker of reactive oxygen species (ROS)-induced DNA damage, was performed to investigate the effect of the expression level of HO-1 on oxidative stress (Figure 6(a)-(d) and (i)). 8-OHdG-positive signals were detected in the nuclei of renal proximal tubular epithelial cells in sections of kidney tissue from cisplatin-treated rats (Figure 6(b)) and were markedly more intense than in the kidney sections from Cis + hAd-MSCs rats, irrespective of their HO-1 expression level (Figure 6(c) and (d)). Also, the signals in the sections of kidney tissue from Cis + HHO-1 rats (Figure 6(c)) were much less than those in the sections from Cis + LHO-1 rats (Figure 6(d)). A proportion of the renal proximal tubular epithelial cells was TUNEL-positive (Figure 6(e)–(h) and (j)) indicating active apoptosis (Figure 6(f)). Administration of hAd-MSCs significantly suppressed apoptosis in the kidney (Figure 6(g) and (h)) compared to that in cisplatin-treated rats (Figure 6(f)), in a manner dependent on the expression level of HO-1 (Figure 6(g) and (h)).

Effect of hAd-MSCs on oxidative stress and apoptosis in renal tubules after administration of cisplatin. Immunohistochemical staining for 8-OHdG (a–d). The tubular epithelial cells of the kidney of cisplatin-treated rats were positive for 8-OHdG (b), but the signal intensity of 8-OHdG was lower in those from hAd-MSCs-injected rats (c and d). Analysis of apoptosis by terminal deoxynucleotidyl transferase dUTP nickend labeling (TUNEL) assay (e–h). Micrograph showing that the proportion of apoptotic tubular epithelial cells was greater in the cisplatin group (f) compared to the control (e) and hAd-MSCs-injected groups (g and h). Tubules with each positive signal were represented as arrow (b and f). Signal density of 8-OHdG and TUNEL level was measured in all groups (I and J). *
Discussion
We report that hAd-MSCs ameliorated the nephrotoxic effect of cisplatin according to their level of HO-1 expression. The viability and migration ability of cisplatin-treated hAd-MSCs increased with increasing HO-1 expression. Renal tubular toxicity in cisplatin-treated rats was significantly ameliorated by administration of hAd-MSCs with high HO-1 expression, although the BUN and serum Cr levels did not differ according to the level of HO-1 expression. These beneficial effects of hAd-MSCs - may be mediated by the antioxidant and anti-apoptotic activities of HO-1, as evidenced by decreased 8-OHdG staining and apoptosis.
The beneficial effect of MSCs on cisplatin-induced renal injury is associated with HO-1, as HO-1−/− MSCs demonstrated reduced expression and secretion of several important growth and pro-angiogenic factors compared to HO-1+/+ MSCs. 32 Moreover, conditioned medium of HO-1+/+ MSCs reversed the functional and morphological changes associated with cisplatin-induced AKI, while conditioned medium of HO-1−/− MSCs did not. These findings suggest that HO-1 plays a major role in MSC-mediated protection against AKI. There were two major differences between the previous study and our study. First, we used hAd-MSCs instead of mouse bone marrow–derived MSCs; second, we administered hAd-MSCs without conditioned medium. Therefore, our findings indicate that hAd-MSCs exert a direct, rather than a paracrine, effect on cisplatin-induced nephrotoxicity.
HO-1 plays an indirect role in the effect of hAd-MSCs on cisplatin-induced nephrotoxicity.33,34 Wang et al. showed that hypoxia-inducible factor-1α (HIF-1α)-transfected hAd-MSCs accelerate recovery from cisplatin-induced nephrotoxicity in vitro and ameliorate cisplatin-induced AKI in vivo. They also reported that the level of HO-1 expression in HIF-1
We reported previously that hAd-MSCs and their conditioned medium protect the kidney from cisplatin-induced nephrotoxicity in vitro and in vivo, by suppressing apoptosis and the expression of p53, JNK, ERK, and pro-inflammatory factors (e.g. IκB-α and COX-2). 31 In that study, we also detected hAd-MSCs - in the kidney. The beneficial effect of hAd-MSCs with high HO-1 expression may be mediated by the aforementioned mechanisms. In addition, cisplatin enhances the production of ROS, which induce oxidative damage in the kidney.6,32–34 In this study, the level of 8-OHdG (a marker of oxidative damage to DNA) was lower in hAd-MSCs with high compared to low HO-1 expression. ROS-mediated oxidative stress activates the mitogen-activated protein kinase (MAPK) signaling pathway such as JNK and ERK.35–38 These kinases have proapoptotic properties. 39 Based on our previous work and this study, the decreased ROS production due to HO-1 overexpression in hAd-MSCs might reduce apoptosis via less MAPK activation.
Recent experimental studies showed protective properties of HO-1 expressed in various MSCs.40–42 Bone marrow–derived mesenchymal stem cells (BMSCs) and hemin (HO-1 inducer) administration protected acute hepatic injury induced by D-galactosamine (D-Gal)/lipopolysaccharides (LPS)-induced acute liver failure rats whereas the protective effects of BMSCs was partially attenuated by zinc protoporphyrin (ZnPP, the HO-1 activity inhibitor) suggesting the key role of HO-1 in the process. Chen et al. also revealed that the transduction of HO-1 into BMSCs had an increased beneficial effect on lipopolysaccharide (LPS)-induced inflammatory and oxidative damage in human pulmonary microvascular endothelial cells (PVECs) by enhanced paracrine function of the stem cells. They proposed that further testing of beneficial effects of MSCs enhanced by HO-1 in acute lung injury models. Yin et al. demonstrated that protective properties of HO-1 expressed in umbilical cord mesenchymal stem cells (UCMSCs) help restore the ovarian function of premature ovarian failure mice with UCMSC transplantation, which is mediated by the activation of JNK/Bcl-2 signal pathway-regulated autophagy and upregulating the circulating of CD8+ CD28– T-cells. These experimental researches presented the preventive and therapeutic potentials of various MSCs enhanced HO-1 expression. Our study also revealed that hAd-MSCs highly expressing HO-1 had beneficial effects on cisplatin toxicity both in vivo and in vitro.
This study had some limitations. First, the biggest limitation might be that we have used ad-MSCs from patients whose diseases can affect. The degree of HO-1 expression at the same passage from five patients who performed the surgery was different according to cell passage although patterns were similar. It is believed that experiments with ad-MSCs from healthy people might affect the outcomes. Second, HO-1 activity that is thought to be more important was not measured. Further study is needed to explore the relationship between HO-1 enzyme activity and protective effects. Third, we would not present exact mechanisms of beneficial effect in in vivo study, although we put forward evidence to support the superiority of cellular viability and migration in higher expression of HO-1 through in vitro experiment.
Conclusion
In conclusion, hAd-MSCs with a high level of HO-1 expression ameliorated cisplatin-induced nephrotoxicity in vitro and in vivo, and so may have therapeutic potential for cisplatin-induced AKI. Further experimental and clinical studies are needed to develop this concept.
Supplemental Material
Supplementary_figure_1 – Supplemental material for The protective effect of human adiposederived mesenchymal stem cells on cisplatin-induced nephrotoxicity is dependent on their level of expression of heme oxygenase-1
Supplemental material, Supplementary_figure_1 for The protective effect of human adiposederived mesenchymal stem cells on cisplatin-induced nephrotoxicity is dependent on their level of expression of heme oxygenase-1 by Hyun Seop Cho, Ha Nee Jang, Myeong Hee Jung, Si Jung Jang, Sang-Ho Jeong, Tae Won Lee, Eunjin Bae, Se-Ho Chang, Dong Jun Park and Jin Hyun Kim in European Journal of Inflammation
Footnotes
Author contributions
H.S.C. mainly designed the research and wrote the manuscript. H.N.L., M.H.J., S.J., S.H.J., T.W.L., E.J.B., and S.C. performed the research and collected data. D.J.P. and J.H.K. analyzed the data and revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All eligible patients or their parents provided written informed consent, and permission to isolate MSCs from adipose tissues was granted by the Institutional Review Board of Gyeongsang National University Hospital (MR7404-130).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a National Research Foundation of Korea grant funded by the South Korean government (MIST; 2013R1A1A3004443) and by grants from the Special Clinical Fund of Gyeongsang National University Hospital (2009 and 2010) to K.J.H. and Biomedical Research Institute Fund
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
