Abstract
Serum pregnancy-associated plasma protein-A (PAPP-A) is measured in Down’s syndrome screening, routinely offered to women in pregnancy. We present the case of an undetectable pregnancy-associated plasma protein-A concentration on the PerkinElmer AutoDELFIA system where immunoassay interference was suspected. Investigations performed, including dilution and recovery studies and antibody-blocking tube incubations, all yielded serum pregnancy-associated plasma protein-A concentrations of <25 mU/L. Pregnancy-associated plasma protein-A was also undetectable on two alternative pregnancy-associated plasma protein-A assays. An experimental manual Delfia procedure suggested the site of interference was between the secondary antibody and the pregnancy-associated plasma protein-A molecule. This case of negative interference in the PerkinElmer pregnancy-associated plasma protein-A assay produced a falsely high Down’s syndrome risk that might have led to an unnecessary invasive procedure with the potential for fetal loss. This highlights the need for Down’s syndrome screening laboratories to be vigilant to immunoassay interference due to the significant impact of the results on patient decision outcome.
Introduction
Screening for Down’s syndrome is offered to all females in England during pregnancy. The ‘combined test’, offered in the first trimester is the National Screening Committee’s test of choice 1 and consists of a fetal nuchal translucency (NT) measurement performed by ultrasound scan and the measurement of serum markers: free beta-human chorionic gonadotropin (free hCGβ) and pregnancy-associated plasma protein A (PAPP-A). The risk of the pregnancy being affected by Down’s syndrome is calculated by software that accounts for gestational age, weight, family origin and smoking status, which affect marker levels. The software produces a corrected multiple of the median (MoM), from which the risk result is generated. If the risk is 1 in 150 or greater, the woman is offered an invasive diagnostic procedure.
PAPP-A is an insulin-like growth factor-binding protein (IGF)-1BP cleaver involved in placental growth. 2 A low serum PAPP-A concentration in conjunction with raised serum-free hCGβ concentration and raised NT measurement is associated with Down’s syndrome.3,4 Low serum PAPP-A concentration in isolation is associated with intrauterine growth retardation (IUGR), preterm delivery and poor pregnancy outcome.5–9
Here, we present the case of a patient undergoing routine Down’s syndrome screening who had an undetectable serum PAPP-A concentration.
Case report
A sample was received at the regional Down’s syndrome screening laboratory from a 34-year-old female at 12 weeks and four days’ gestation.
Initial screening results using the PerkinElmer-Elmer AutoDELFIA (Turku, Finland) system gave a free hCGβ concentration of 50.4 IU/L (median level for gestation = 36.41 IU/L, corrected MoM of 1.47). NT measurement was 1.1 mm, (median for gestation = 1.67 mm, corrected MoM of 0.67). The PAPP-A result was less than 25 mU/L (median level for gestation 2292 IU/L, corrected MoM of 0.01). The calculated risk of Down’s syndrome was high at 1:110, incorporating a truncated low PAPP-A MoM at 0.2 MoM. Interference in the PAPP-A assay was suspected and further work undertaken.
Investigations
Contamination
EDTA, oxalate and sodium citrate contamination is a cause of undetectable serum analyte concentration using the AutoDELFIA system, as they chelate the Europium and Samarium conjugates employed in the assays. 10 Contamination was excluded by measurement of adjusted calcium, potassium and sodium (2.33 mmol/L, 4.3 mmol/L and 140 mmol/L, respectively). This was further supported by the measurable free hCGβ using the same assay system, which would also be affected by anticoagulant contamination, if present. Furthermore, repeat serum samples showed the same pattern of results.
Interference
Interference has previously been reported in the PerkinElmer AutoDELFIA, AFP assay used for second trimester Down’s syndrome screening in females with complement activation. 11 The sample was left for 7 days at 4℃ to deplete complement and then re-analysed which gave a serum PAPP-A concentration of <25 mU/L.
Given the PAPP-A method utilizes mouse monoclonal anti PAPP-A immunoglobulin G (IgG), the sample was incubated with 1:1 with mouse serum (Sigma-Aldrich, Gillingham UK) for 2 h to remove any antimouse antibodies (HAMA) and the PAPP-A concentration remained <25 mU/L.
Upon serial dilution with manufacturer’s diluent, the PAPP-A remained undetectable. A 1:2 dilution with a serum sample from a control patient with a PAPP-A concentration of 3090 mU/L, (corrected MoM, 1.35) the measured PAPP-A was 505 IU/L, 70% less than the expected recovery. This indicates the presence of a substance in the serum, which actively inhibits PAPP-A measurement.
Cross-comparison with other analytical platforms
Summary of PAPP-A concentration and calculated corrected multiple of the medians (MoMs) in the sample on three different analytical platforms and following incubation with antibody blocking tubes on the PerkinElmer and Brahms system.
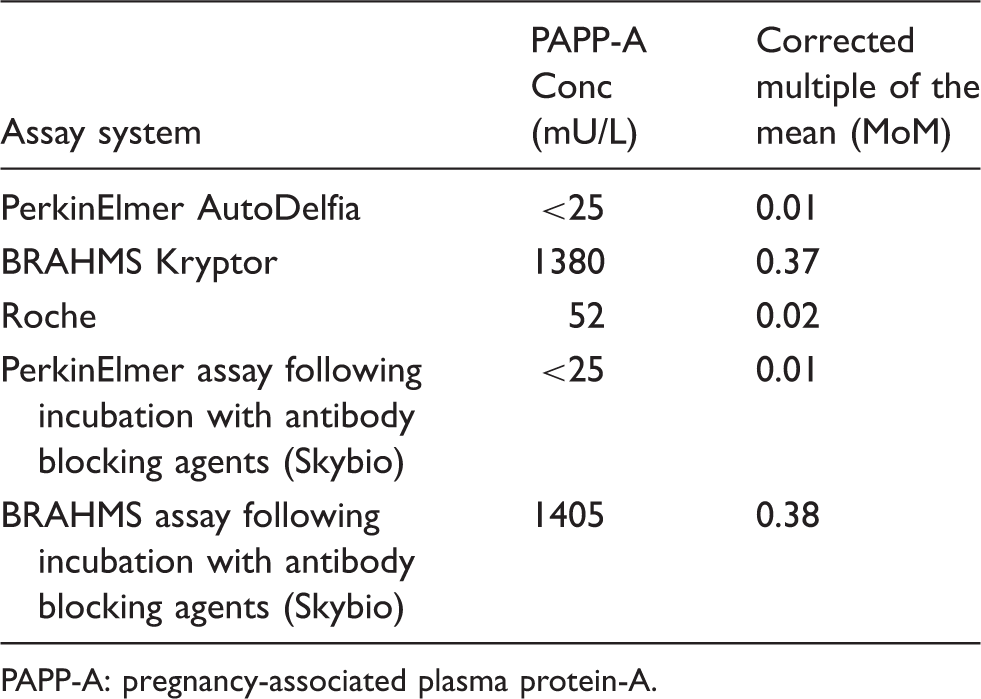
PAPP-A: pregnancy-associated plasma protein-A.
Experimental Delfia procedure
The PerkinElmer AutoDELFIA PAPP-A assay is a two-site immunoassay comprising a biotin-labelled mouse anti PAPP-A IgG, which binds to streptavidin on the wall of a microtitre well, to which PAPP-A binds and then a second mouse anti PAPP-A IgG antibody linked to a Europium conjugate is added which binds to the bound PAPP-A. The bound europium is detected through the addition of an enhancement solution and measured by the AutoDELFIA fluorimeter. The amount of fluorescence is directly proportional to the PAPP-A concentration in the sample.
To investigate the site of the interference, an experimental variation of a manual Delfia procedure (PerkinElmer kit insert) was carried out (Appendix 1).
In the procedure, the first antibody was added to the well and incubated with the serum, which was then removed before the addition of the second antibody and control maternal serum of a known PAPP-A concentration. Following incubation, washing and enhancement, the fluorescence was measured on the AutoDELFIA plate reader. The PAPP-A concentration in the control maternal serum was 4817 mU/L when analysed by the conventional AutoDELFIA procedure and 6520 mU/L (mean result of triplicate measures) when analysed in combination with the abnormal serum in the experimental manual method. The PAPP-A concentration measured in the serum containing interference remained at <25 mU/L in both the AutoDELFIA and the experimental manual Delfia assay.
The results suggest that the interferent was removed at the first wash step in the experimental procedure, thereby allowing the PAPP-A in both the control maternal serum and in the sample with interference to be detected. In the experimental procedure, the interference sample is removed before the addition of the secondary antibody, thereby allowing it to bind and generate a signal, whereas in the routine AutoDELFIA system, the patient sample and secondary antibody are added and removed at the same time.
Furthermore, if the interference was with the primary antibody then this would remain blocked during the addition and removal of the control patient sample and the signal would be undetectable, assuming the interferent is in excess relative to the antibody titre within the well.
The average manual PAPP-A value of 6520 mU/L is comparable to the expected value if the PAPP-A in the interference sample (BRAHMS 1380 mU/L) and the normal serum (AutoDELFIA value 4817 mU/L) were both detected by this system. This assumes that there is no interference in the BRAHMS assay, which cannot be excluded (Figure 1).
Experimental manual Delfia assay. The sample with suspected interference ‘test sample’ (TS) is incubated with the primary antibody and then removed prior to the addition of the secondary antibody and the normal maternal control serum. The removal of the test sample and therefore the interfering substance before the addition of the secondary antibody allows binding and an increased signal due to the detection of the PAPP-A in both samples.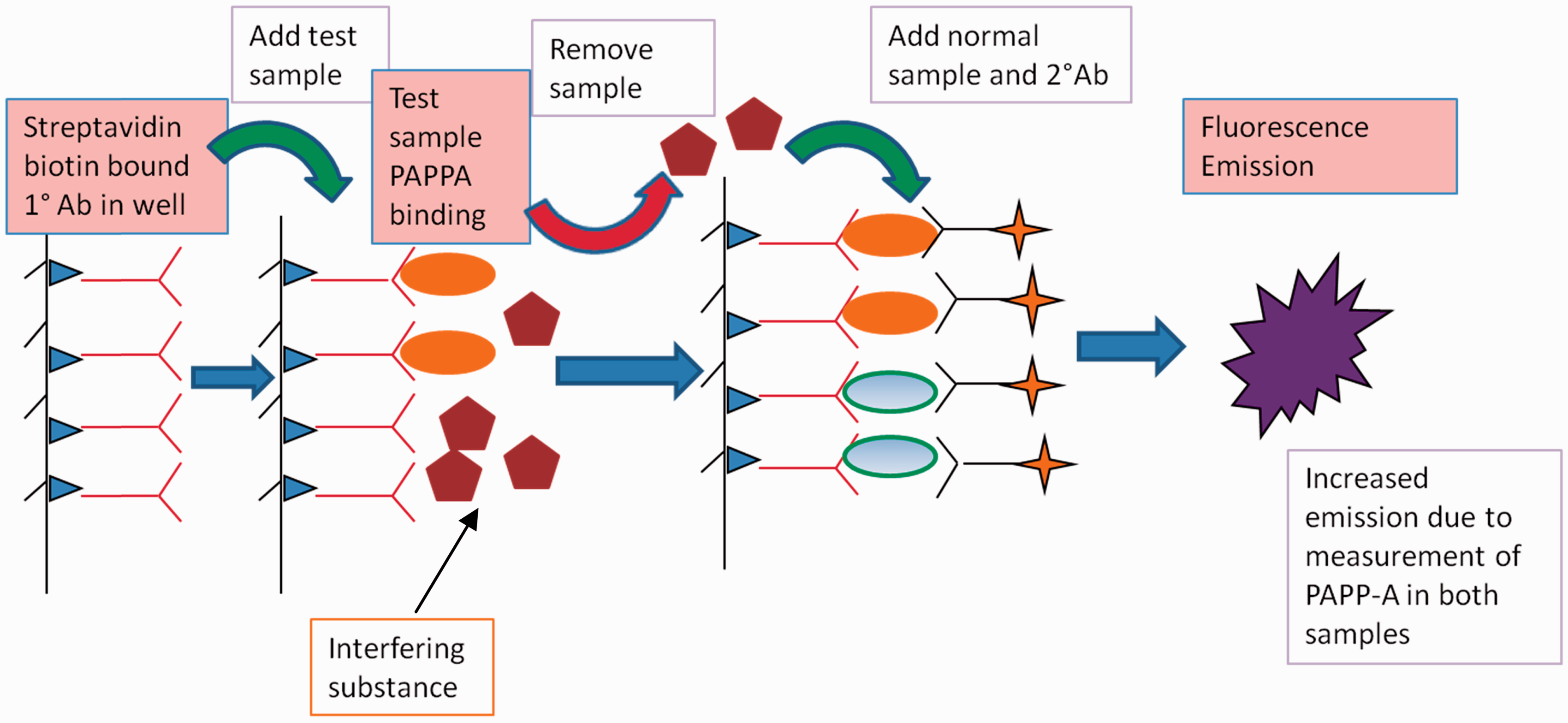
Therefore, it can be hypothesized that the site of interference in the PerkinElmer AutoDELFIA assay in this sample is with the secondary anti PAPP-A IgG antibody bound to the Europium conjugate, which is prevented from binding and thereby produces no signal.
Serum PAPP-A concentration rises physiologically in pregnancy and therefore it was of interest to see if the interference persisted during pregnancy. The patient donated a blood sample at 26 weeks gestation and PAPP-A was measurable on the PerkinElmer assay (30,000 mU/L) and the BRAHMS assay (120,000 mU/L). MoMs cannot be calculated for these as median values for PAPP-A are not established at this gestation as PAPP-A is not usually measured outside of the first trimester. However, the discrepancy between the AutoDELFIA and the BRAHMS assay results is still apparent.
Pregnancy outcome
The patient went on to have a second trimester quadruple test for Down’s syndrome, which returned apparently normal results (AFP = 36.9 U/mL, corrected MoM 1.29; free hCGβ = 12.5 U/L, corrected MoM 0.97; uE3 = 3.9 nmol/L, corrected MoM 1.21; Inhibin A = 263.1 pg/mL corrected MoM 1.80) with a Down’s syndrome risk of 1:8400. She delivered a normal healthy baby at full term.
Discussion
This is the first reported case of negative interference in the PerkinElmer AutoDELFIA PAPP-A assay that is not related to contamination from anticoagulants and is suggestive of interference in the Roche assay also. Both use a streptavidin/biotin sandwich assay system with fluorescence and chemiluminesence signal systems, respectively. The BRAHMS Kryptor assay appears to be unaffected or affected to a lesser extent by the interferent and employs a measurement system of Time Resolved Amplified Cryptate Emission, whereby proximity of molecules in an immunocomplex modifies the levels of emission. All three assays use two mouse monoclonal antibodies directed against the PAPP-A molecule. The PerkinElmer and Roche assays are heterogeneous, with a physical separation of free from bound antibody, compared to the BRAHMS which has a homogenous measurement system where there is no separation of free from bound prior to detection.
The nature of the interfering substance is unknown. Given that PAPP-A was undetectable at 12 + 4 weeks but detectable at 26 weeks’ gestation on the AutoDELFIA suggests that either the interferent concentration had decreased in the intervening weeks or that its concentration was not sufficient to fully suppress the higher PAPP-A level typically found at 26 weeks’ gestation indicating a competitive nature.
This also further supports interference rather than an altered form of PAPP-A found in this particular patient that is not detected by the antibody. Interference in immunoassays is widely reported with a quoted incidence of 0.4% to 4%.10,12,13 Antiruthenium antibodies have been described previously in some Roche assays to cause interference;14,15 however, the PerkinElmer system uses a Europium conjugate and therefore unlikely to be affected. In addition, the free hCGβ concentrations on all three assays correlate well and normal second trimester results were produced using the same assay systems. Therefore, this suggests a specific interference between the epitope of one of the antibodies in the assay and the PAPP-A molecule.
This negative interference in the PerkinElmer PAPP-A assay produced a higher risk result (1:110) which, if issued, may have led to an unnecessary invasive diagnostic procedure that carries a 1–2% chance of miscarriage. Interestingly, the PAPP-A result produced by the BRAHMS system is low for a normal pregnancy (corrected MoM 0.38) and therefore also produced a high-risk result (1 in 129), which is in contrast to the low risk seen in the second trimester sample (1 in 8400) and it is impossible to exclude partial interference in this assay also.
Conclusion
This case serves as a reminder to Down’s syndrome screening laboratories to be vigilant to grossly abnormal MoMs which may indicate analytical interference. The implications of issuing a Down’s screening risk incorporating such a result are considerable. There is the potential for fetal loss due to an unnecessary invasive diagnostic procedure, with associated maternal stress and anxiety and further intensive pregnancy monitoring. Although this may be an isolated case it is important to highlight the potential for such interference and the consequences for the patient result and outcome.
Footnotes
Acknowledgements
We would like to thank the Biochemistry Department at Central Manchester Foundation Trust for their gift of Skybio antibody blocking tubes.
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Ethical approval
Not applicable. The patient has given informed written consent to publication.
Guarantor
CW.
Contributorship
CW, KH and JM conceived the experimental interference investigations. CW performed the analytical work, MP and KS provided analysis of the samples at their site and contributed to the analysis of the results. CW wrote the manuscript. All co-authors reviewed and edited the manuscript and approved the final version.
