Abstract
Background
Supported liquid extraction (SLE) techniques are relatively new compared to other sample preparation approaches such as solid phase extraction (SPE), liquid–liquid extraction (LLE) and protein precipitation (PPE). We investigated the use of SLE as an alternative to SPE for the liquid chromatography tandem mass spectrometry (LC-MS/MS) measurement of aldosterone.
Methods
Samples (n = 83) were analysed by the routine method using SPE. The same samples were subsequently analysed using two different SLE 96-well plate devices (Thermo and Biotage) with methyl-tertiary butyl ether extraction. A direct comparison of the three extraction techniques on two different mass spectrometers was also performed.
Results
Both results using SLE plates gave excellent agreement with the results from the SPE analysis. The area counts obtained with the Biotage plates were considerably higher than those obtained using the Thermo plate.
Conclusions
SLE is an acceptable alternative to SPE for the LC-MS/MS analysis of aldosterone. Using SLE reduces the time required for sample preparation.
Introduction
Aldosterone analysis by liquid chromatography tandem mass spectrometry (LC-MS/MS) is technically challenging because of the low concentrations in plasma. Analysis also requires the use of negative ion mode which is generally not as efficient as positive ion mode used for other steroid analyses. Any attempt to improve the sample clean up step to enhance sensitivity will therefore be highly beneficial.
A wide variety of sample preparation techniques have been reported in the literature for the analysis of steroids by LC-MS/MS including protein precipitation (PPE), 1 liquid–liquid extraction (LLE)2,3 and solid phase extraction (SPE). 4 Supported liquid extraction (SLE) is a relatively novel approach for sample preparation.
In SLE, the aqueous sample is held onto a diatomaceous earth support and is subsequently treated with solvent for the extraction of hydrophobic molecules. The manufacturers propose that this method gives cleaner extracts than LLE.
We investigated the use of SLE as an alternative to SPE for the LC-MS/MS analysis of aldosterone.
Methods
Plasma samples (n = 83) were analysed by SPE using Oasis C18 96-well plates (Waters, Manchester, UK) as described in the method of Hinchliffe et al. 5 using a Waters Xevo TQS mass spectrometer (Waters). SLE (400 µL) plates were obtained from Thermo (Hemel Hempsted, UK) and Biotage (Hertfordshire, UK). The sample preparation for both of these plates was the same. Sample (250 µL), D7 aldosteone internal standard (10 µL, 60 µg/L or 166 nmol/L in methanol) and water (150 µL) were pre-mixed in 1.5 mL microcentifuge tubes. After a brief vortex (10 s), all the diluted sample mixture was transferred to a well of the SLE plate. The SLE plate was transferred to a vacuum manifold and vacuum was applied for approximately 5 s to encourage the sample mixture to descend into the diatomaceous earth layer. After 5 min, a collection plate was placed under the SLE plate (2 mL square well; Porvair Sciences, Leatherhead, UK) and methyl-tertiary butyl ether (MTBE, 900 µL), was added to the wells. The MTBE was allowed to pass through the plate by gravity and then the vacuum was re-applied for approximately 10 s. The collection plate was removed and MTBE was evaporated to dryness, then the extract was reconstituted using 80 µL of 35% methanol.
A serum sample (111 pmol/L) was extracted using the three methods described above and analysed on Waters Xevo TQ and TQS mass spectrometers to allow direct comparison of sensitivity.
Results
The results using the Biotage plate showed excellent agreement with the SPE results (Figure 1a) with an average bias of −0.2 pmol/L. The Passing-Bablok regression analysis gave the equation, Biotage = 1.02 × SPE−8.5 pmol/L The results using the Thermo plate also demonstrated good agreement with the SPE results (Figure 1b) with an average bias of −24 pmol/L. The Passing-Bablok regression analysis gave the equation, Thermo = 0.97 × SPE−1.5 pmol/L. The peak area counts were consistently lower using the Thermo plate compared to the Biotage plate. The Passing-Bablok regression analysis of the aldosterone area using each SLE plate gave the equation, Thermo = 0.31 × Biotage + 100 pmol/L. The % coefficient of variation (%CV) of the peak area counts for the internal standard in the serum samples were 45% for SPE, 28% for Biotage SLE and 14% for Thermo SLE. The difference in area counts between the non-matrix matched calibrators and the plasma samples was −45% for SPE, −44% for Biotage SLE and +6% for Thermo SLE, thus suggesting the Thermo plate exhibited a more reproducible extraction and/or less ion suppression.
Bland-Altman analysis of aldosterone results (n = 83) in pmol/L when comparing the SPE with the Biotage SLE plate (a) and the Thermo SLE plate (b).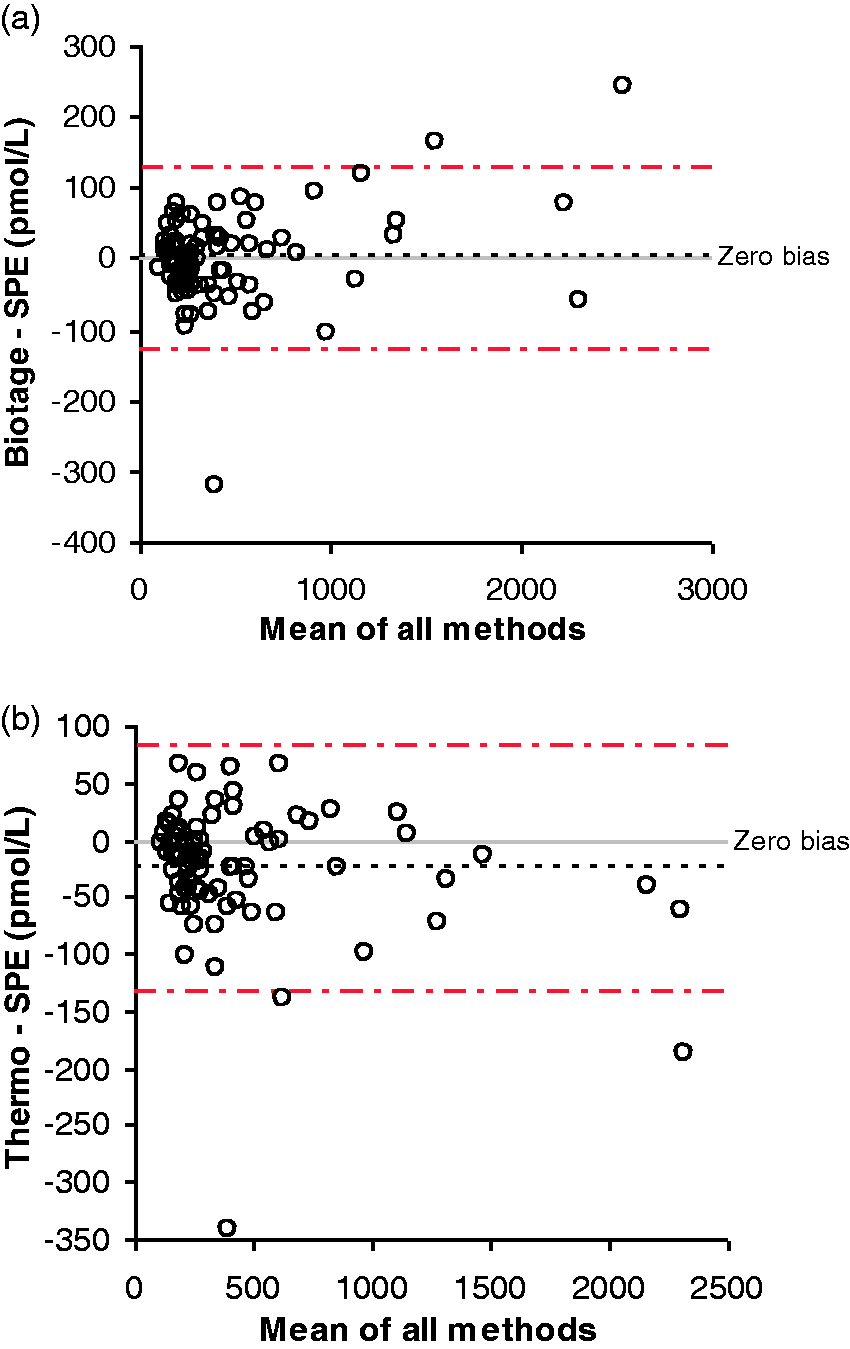
The signal to noise (peak to peak) ratio obtained for an extracted serum sample (111 pmol/L) was 5:1 using SPE, 11:1 using Biotage SLE and 2:1 using Thermo SLE when measured on a Waters Xevo TQ mass spectrometer. The signal to noise (peak to peak) ratio obtained for the same extracts was 34:1 using SPE, 56:1 using Biotage SLE and 12:1 using Thermo SLE when measured on a more sensitive instrument (Waters Xevo TQS).
Discussion
Both SLE plates demonstrated an excellent agreement with the SPE results. Furthermore, the sample preparation using the SLE plates contained fewer steps and was therefore simpler and less time consuming than SPE. During SPE sample preparation, the serum samples first undergo protein precipitation in microcentrifuge tubes followed by centrifugation. SLE does not require protein precipitation and therefore saves time during this stage. The dilution of the samples can be performed in a 96-well plate followed by the use of a multichannel pipette to transfer to the SLE plate further speeding up the process. The SLE plates also do not require wash steps as in SPE, therefore also minimizing the time spent performing the extraction. Alternative strong solvents, such as dichloromethane, may also be investigated to achieve optimum results but we achieved good results using MTBE and we found it safer to use. It was apparent that the Biotage SLE plates consistently gave the most sensitive results and this was confirmed when the signal to noise ratio of an extracted plasma sample was compared using all three methods.
The difference in area counts and signal to noise ratios obtained using the SLE and SPE plates highlights the need to investigate devices supplied by different manufacturers to establish which would be best suited to the equipment available. For example, all sample extraction techniques reported here were adequate when using a highly sensitive mass spectrometer. However, the Thermo SLE plate was not suitable for sample preparation when a less sensitive mass spectrometer was used, because the lower area counts and signal to noise ratio obtained may compromise the lower limit of the assay. Since changing our extraction technique from SPE to Biotage SLE plates, we have been able to perform routine aldosterone analysis on a less sensitive mass spectrometer (Waters Xevo TQ), whilst still achieving adequate sensitivity (<100 pmol/L) for the screening of Conn’s syndrome. This has obvious advantages for laboratories that cannot afford an expensive research grade instrument.
However, the use of the Thermo plate has advantages in other areas. It had the most stable counts suggesting least ion suppression and/or more reproducible extraction efficiencies. This may be as a result of the different composition of the Thermo SLE plate affecting better phospholipids removal as the SLE plates are recommended for this use by the manufacturer. The Thermo plate was also the most cost-effective of the three extraction techniques used, the list price for which is approximately half of the Biotage SLE plate and SPE plates although prices for these will vary depending on usage and local discounts given by the manufacturers.
In conclusion, SLE provides a quicker, more sensitive and potentially cheaper alternative to SPE for the LC-MS/MS analysis of aldosterone.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
BK.
Contributorship
BK and LO conceived the study. LO performed the sample and data analysis and wrote the manuscript. BK reviewed and edited the manuscript.
