Abstract
Background
Urinary 18-hydroxycortisol has been investigated as a marker of aldosterone-producing adenoma (APA). The aim of this study was to develop and validate a method for the measurement of 18-hydroxycortisol using liquid chromatography-tandem mass spectrometry (LC-MS/MS).
Methods
Urine was collected over a 24-hour period in patients with APA (n = 11), idiopathic hyperaldosteronism (IHA, n = 9), and essential hypertension (EH, n = 6). 18-Hydroxycortisol was extracted in solid-phase, and measured by LC-MS/MS based on selected reaction monitoring.
Results
The method allowed quantification of 18-hydroxycortisol with a lower quantification limit of 0.26 nmol/L, intra- and inter-assay coefficients of variation of <3.4% and a range of analytical recovery of 98.0–103.7%. Urinary 18-hydroxycortisol excretion for APA, IHA and EH were determined as 725 (SD 451), 102 (SD 68) and 88 (SD 76) nmol/day, respectively.
Conclusions
The proposed method met the basic analytical requirements and was considered to be useful in the screening and differential diagnosis of APA.
Introduction
Primary aldosteronism (PA) is a syndrome characterized by arterial hypertension caused by aldosterone excessively secreted from the adrenal gland. A portion of patients are complicated with hypokalaemia due to renal potassium loss. Recent studies reported the incidence of PA as high as 3.2–21.7%. 1 Several guidelines and algorithms for the diagnosis of PA were proposed in recent years.2–6 As screening testing, the measurements of plasma aldosterone concentration, plasma renin activity (PRA) and the aldosterone–renin ratio are common in use. After positive screening, confirmatory testing is done using captopril challenge test, furosemide upright test and/or saline infusion test. 7 Finally, laterality and subtype are determined by adrenal venous sampling (AVS) with or without adrenocorticotropine (ACTH) stimulation and by radiological imaging.
PA is usually caused by aldosterone-producing adenoma (APA) or bilateral hyperplasia of the zone glomerulosa (idiopathic hyperaldosteronism, IHA), although it can be caused with much less incidence by other subtypes of PA, such as unilateral adrenal hyperplasia, unilateral multiple adrenocortical nodules, bilateral APAs and hereditary glucocorticoid-remediable aldosteronism (GRA). 8 Screening and differential diagnosis of APA are crucially important, since adrenalectomy is the first choice of therapy for APA, whereas drug therapy is chosen for IHA. The AVS is known to be most useful for differential diagnosis between APA and IHA. 9 However, the AVS is difficult to carry out in many hospitals and clinics because it requires skills and special facilities. A simple and easy testing that can substitute at least partly for AVS seems potentially useful.
18-Hydroxycortisol is known as a ‘hybrid steroid’ characterized by the co-presence of 17α- and 18-oxygenated carbons. 10 This steroid has been recognized as a marker for differentiation of APA and rare GRA from other subtypes of PA.11–13 Hence, 18-hydroxycortisol might be useful not only to detect APA but also to differentiate APA from IHA. Various methods have been reported for the measurement of urinary 18-hydroxycortisol, including radioimmunoassay (RIA),13–15 gas chromatography-mass spectrometry (GC-MS),15,16 high-performance liquid chromatography (HPLC),17,18 enzyme immunoassay (EIA)19,20 and time-resolved fluorometric assay.15,21 Although GC/MS has been regarded as the ‘gold standard’, it involves derivatization prior to GC-MS analysis. 16 On the other hand, derivatization is not necessary in liquid chromatography-tandem mass spectrometry (LC-MS/MS), in which a sample extract can be directly applied to LC-MS. For this reason, LC-MS is getting widespread through clinical laboratories. Here, we describe a simple and rapid method for measuring urinary 18-hydroxycortisol using LC-MS/MS and its usefulness for differential diagnosis of APA.
Materials and methods
Chemicals
18-Hydroxycortisol was chemically synthesized as described previously. 22 6α-Methylprednisolone, the internal standard (IS), was purchased from Sigma–Aldrich Corp. (St. Louis, MO, USA). Formic acid and all the other solvents used were of HPLC grade or better, and were acquired from Wako Pure Chemical Industries (Osaka, Japan).
Sample collection and ethical procedures
Urine samples were collected over a 24-hour period from 26 hypertensive patients in Sapporo City General Hospital, and were stored at −30℃ until use. The patients were classified into three etiological groups: APA (n = 11), IHA (n = 9) and essential hypertension (EH, n = 6). The patients were screened in accordance with the guidelines provided by the Japan Endocrine Society. 8 Finally, APA was diagnosed by pathological findings after adrenalectomy. Diagnosis of IHA was based on confirmation of bilateral aldosterone hypersecretion in ACTH-stimulated AVS. The study was approved by the ethics review board at the Faculty of Health Sciences, Hokkaido University (approval number 11-08), and informed consent was obtained from each patient.
Sample preparation
An aliquot of urine (200 µL) was mixed with 100 µL of IS solution (2.65 µmol/L) to which 700 µL of distilled water was added. After vortex mixing, the mixture was applied to a HyperSep C18 SPE column (50 mg of octadecyl silica, ODS, Thermo–Fisher Scientific, Waltham, MA, USA) pre-treated sequentially with 2 mL of methanol and 2 mL of distilled water. The column was rinsed sequentially with 1 mL of distilled water and 2 mL of 5% aqueous methanol solution. The analytes were eluted with 2 mL of methanol. After evaporation to dryness, the residue was dissolved in 1 mL of 50% aqueous methanol solution, and a 10 -µL aliquot of the solution was subjected to LC-MS/MS analysis.
LC-MS/MS
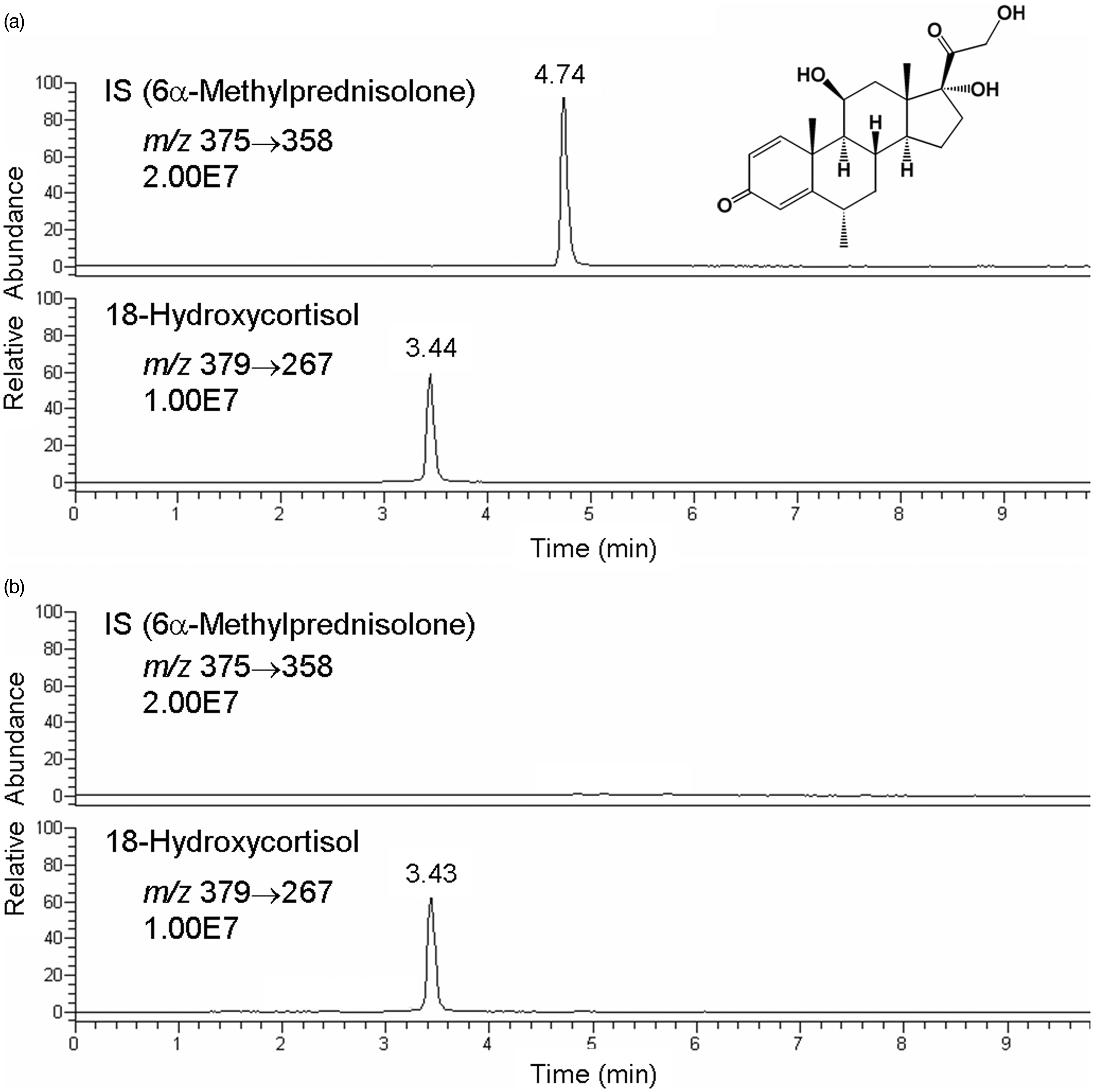
LC-MS/MS was performed using an Accela high-speed liquid chromatography system (Thermo–Fisher Scientific) and a TSQ Quantum Access triple-stage quadrupole mass spectrometer with a heated-electrospray ionisation (H-ESI) probe (Thermo–Fisher Scientific). LC was carried out on a Hypersil GOLD analytical column (5 µm, 50 mm × 2.1 mm i.d., Thermo–Fisher Scientific). The mobile phase consisted of (A) 0.05% formic acid aqueous solution with (B) acetonitrile at a flow rate of 0.2 mL/min. Separation was conducted at 35℃ according to the following gradient program: 0–5 min, 15% (B) to 65% (B); 5–10 min, 65% (B). Under these conditions, 18-hydroxycortisol and the IS were eluted at 3.4 and 4.7 min, respectively.
To evaluate the performance of the method, the limit of detection (LOD), 23 the lower limit of quantification (LLOQ), 23 the linearity, the recovery, the carryover and the ion suppression were assessed. Linearity was evaluated in six runs of 18-hydroxycortisol standard samples of eight concentrations between 5.3 and 1322.8 nmol/L, equivalent to 26.4–6613.8 nmol/L in urine. The extraction recovery was evaluated by analysing nine samples from patients with APA (n = 3), IHA (n = 3) and EH (n = 3) by adding IS before and after extraction. The analytical recovery was evaluated by analysing samples from four patients with various concentrations of 18-hydroxycortisol, spiked with various amounts of 18-hydroxycortisol standard before extraction. Ion suppression was evaluated by postcolumn continuous infusion. 24
Preparation of standard solutions
Standard 18-hydroxycortisol and the IS were stored at −30℃ as stock solutions of 2.67 µmoll/L containing 5% aqueous methanol. Before use, the 18-hydroxycortisol solution was thawed and diluted with methanol and distilled water to prepare working standard solutions in 50% aqueous methanol at 18-hydroxycortisol concentrations of 5.3, 13.2, 26.5, 52.9, 132.3, 264.6, 529.1 and 1322.8 nmol/L. Each standard solution also contained 267.4 nmol/L of the IS.
Statistical analysis
Statistical analyses for the daily urinary 18-hydroxycortisol excretion were performed among the three subject groups with the non-parametric Kruskal-Wallis test with Scheffe’s multiple post hoc test, using Statcel (OMS, Saitama, Japan).
Results
Optimization of LC-MS/MS
LC-MS was performed in the positive-ion mode with H-ESI. The measurement parameters were optimized to obtain the best detection sensitivity for both 18-hydroxycortisol and the IS. On the full-scan spectrum of 18-hydroxycortisol, the base peak was observed at m/z 379 ([M + H]+) (Figure 1(a)). The product-ion spectrum of 18-hydroxycortisol (m/z 379) showed various ions generated at a collision energy of 19 V (Figure 1(b)). The ion signal at m/z 267 was chosen for the selected reaction monitoring (SRM) measurement because of its abundance. Similarly, the detection condition for the IS was optimized as m/z 375 → 358 at a collision energy of 9 V. Other parameters and conditions were as follows: electrospray voltage, 3.0 kV; vaporizer temperature, 250℃; ion transfer tube (capillary) temperature, 300℃; sheath gas (nitrogen) pressure, 50 p.s.i.; auxiliary gas flow, 40 arbitrary units; tube lens offset voltage, 105 V and collision gas (argon) pressure, 1.5 mTorr. After optimization of LC conditions, good separation of 18-hydroxycortisol and the IS was obtained by a linear gradient with a 0.05% formic acid aqueous solution and acetonitrile (Figure 2).
H-ESI mass spectra (positive-ion mode) of 18-hydroxycortisol in (a) full-scan mode and (b) product-ion scan mode by dissociation of [M + H]+ at m/z 397. SRM of (a) standard 18-hydroxycortisol and IS, and (b) a urine extract from a patient with APA. SRM conditions: m/z 375 → 358 for IS (a and b; upper) and m/z 379 → 267 for 18-hydroxycortisol (a and b; lower).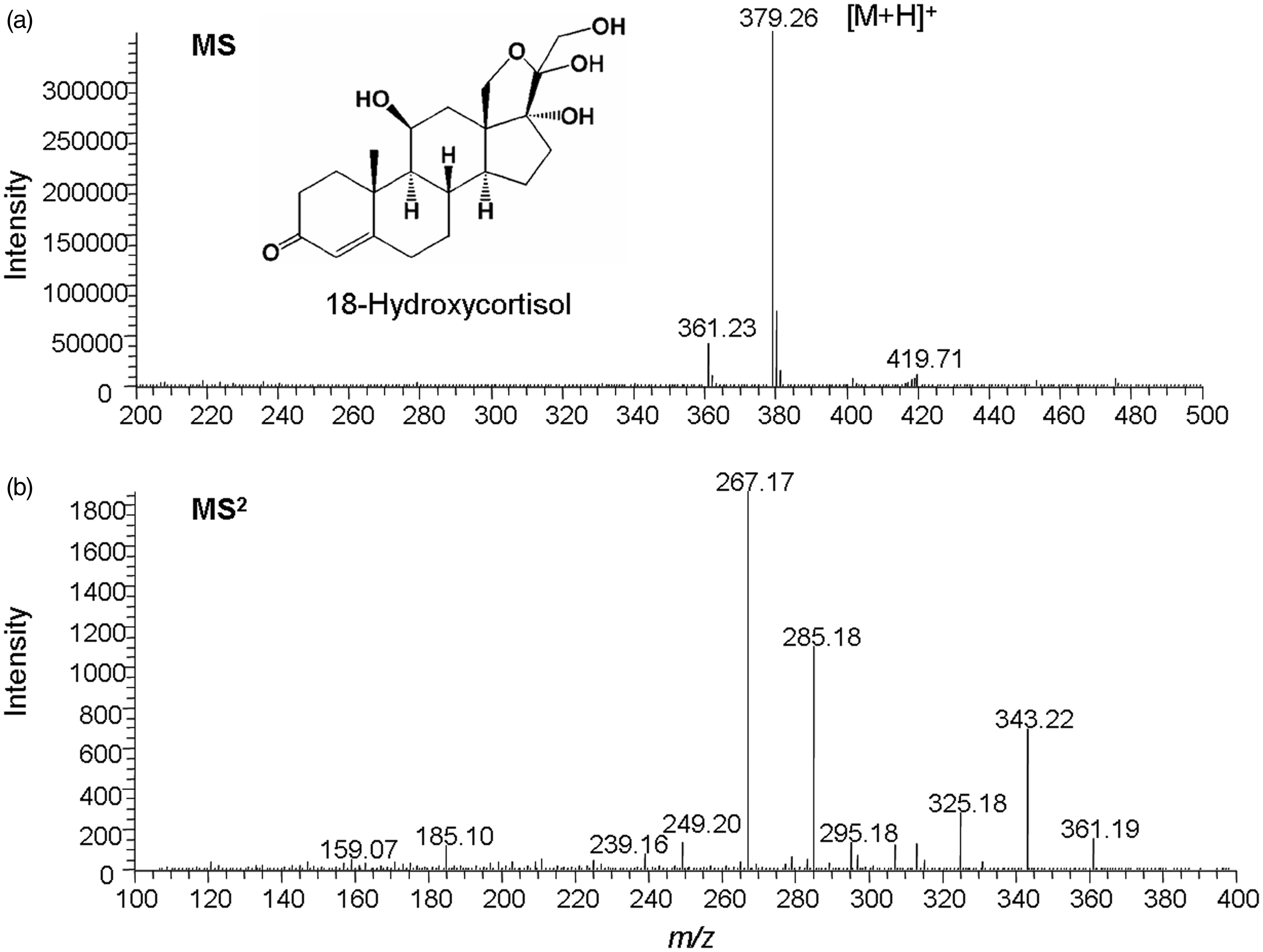
Calibration and sensitivity
An eight-point calibration curve ranging from 5.3 to 1322.8 nmol/L for 18-hydroxycortisol was evaluated. The calibration curve was constructed by plotting the 18-hydroxycortisol/IS peak area ratio (y) against the 18-hydroxycortisol concentration (x). The least-square regression line obtained from the mean of six analytical runs was y = 0.00292109 (SD 0.00006841)x + 0.00011812 (SD 0.00082455), with a squared correlation coefficient (r2) of 0.9999.
The LOD and LLOQ were 1.3 pmol (10 µL of 132.3 pmol/L) and 2.6 pmol (10 µL of 264.6 pmol/mL), which showed a signal-to-noise ratio more than 3:1 and 13:1, respectively, in three runs. The carryover peak that reached LOD was not observed until 18-hydroxycortisol and IS concentrations of 2.65 and 2.67 μmol/L, respectively, under the condition that the sampling needle of autosampler was washed with 3 mL of methanol.
Ion suppression
To rule out potential ion suppression or enhancement, the background signal concentrations in the chromatogram were determined with and without continuous infusion of standard 18-hydroxycortisol (Figure 3). The results showed no change in the background signal concentration except for the void volume (0.6–1.1 min), negating the likelihood of ion suppression or enhancement for 18-hydroxycortisol (Figure 3(a) and (b)). The same results were obtained for IS (data not shown).
SRM (m/z 379 → 267) chromatograms of the postcolumn infusion experiments for evaluation of ion suppression: (a) blank solvent (50% aqueous methanol) injection with infusion of 18-hydroxycortisol standard (5 µL/min of 264.5 nmol/L), (b) urine ODS column extract injection with infusion (same as (a)) and (c) urine ODS column extract injection without infusion.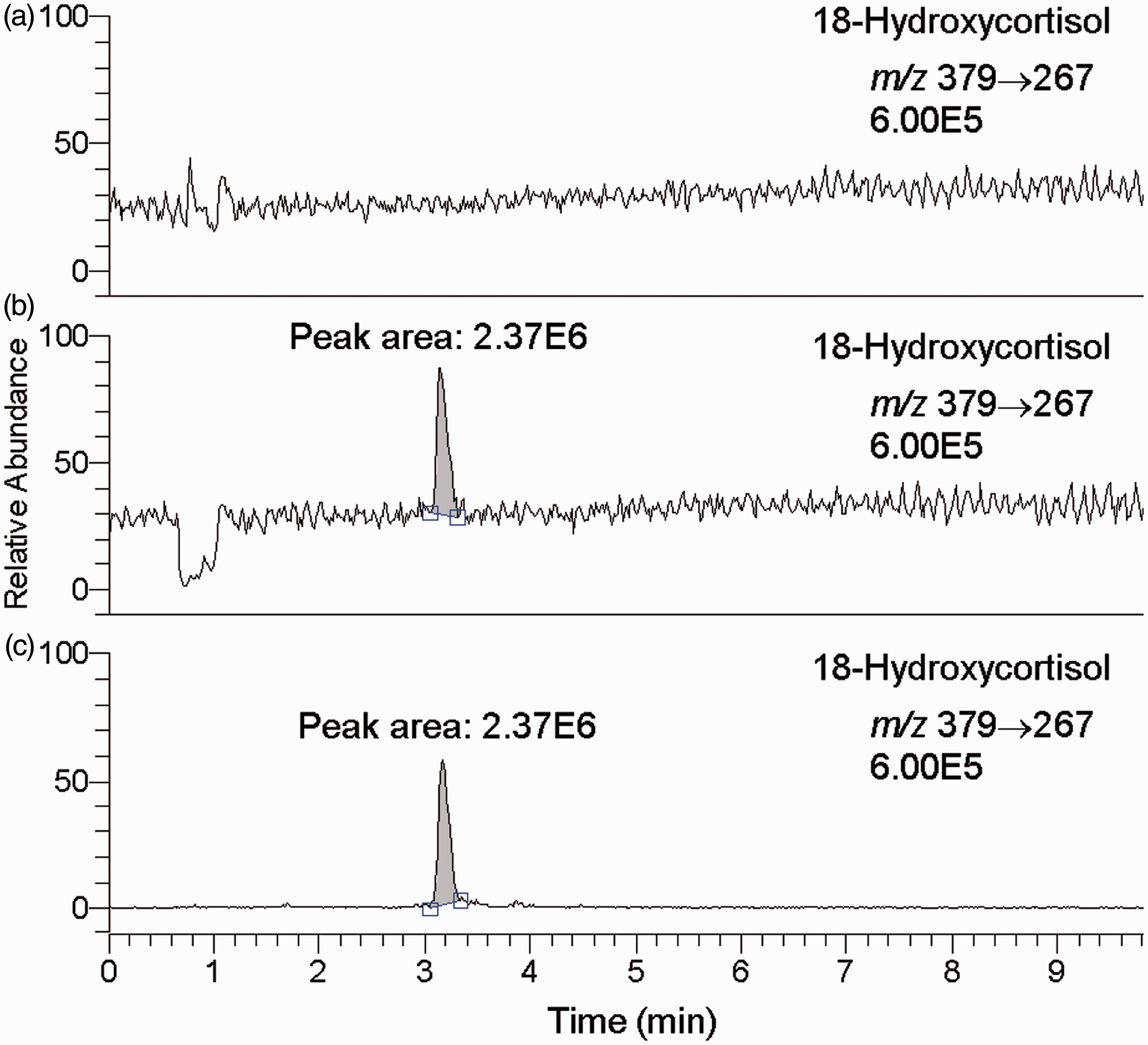
Recovery and reproducibility
Recovery of the solid-phase extraction with an ODS column was 99.7% (96.3–101.6%), and there was no significant difference between the APA, IHA and EH groups. The precision of the assay was examined using urine samples containing various concentrations of endogenous 18-hydroxycortisol obtained from six subjects within the above groups. The intra- and inter-assay coefficients of variation for all subjects were less than 3.4%. Analytical recovery rates ranged from 98.0% to 103.7%, and the grand mean of the recovery rates was 100.0%.
Measurement of urinary 18-hydroxycortisol
Urine samples collected over a 24-hour period from 26 subjects were measured for 18-hydroxycortisol by LC-MS/MS. The daily urinary 18-hydroxycortisol excretion for the three groups are shown in Figure 4. The mean 18-hydroxycortisol excretion in the patients with APA, IHA and EH were 725 (SD 451), 102 (SD 68) and 88 (SD 76) nmol/day, respectively. The patients with APA showed statistically significant elevation in comparison with APA (P < 0.01) and EH (P < 0.01). No significant difference was observed between APA and EH.
Urinary 18-hydroxycortisol excretion (nmol/day) by the proposed method in patients with aldosterone-producing adenoma (n = 11), idiopathic hyperaldosteronism (n = 9) and essential hypertension (n = 6). Statistical analysis was done using the non-parametric Kruskal-Wallis test with Scheffe’s multiple post hoc test.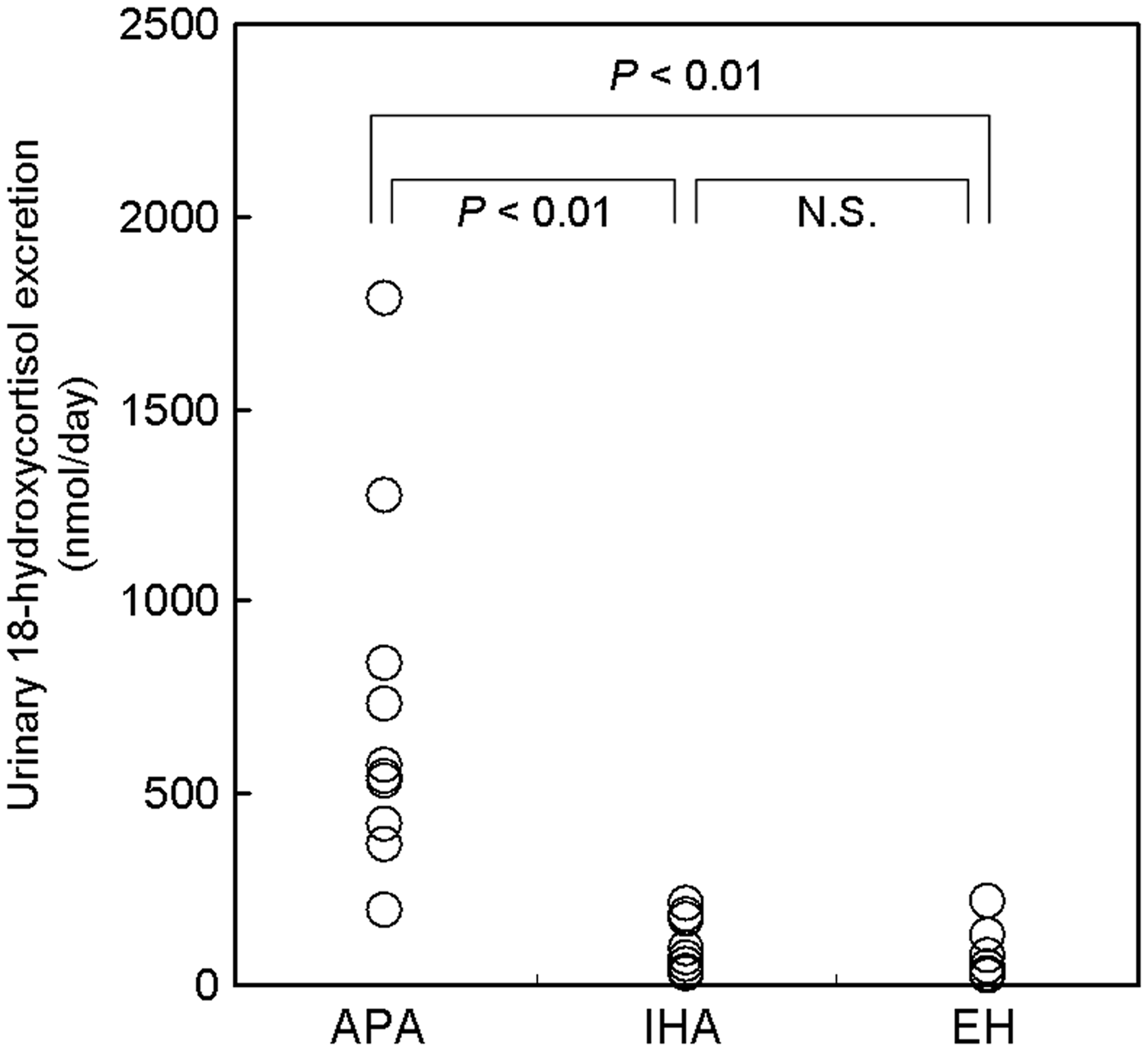
Discussion
The proposed LC-MS/MS method allows linear measurement of urinary 18-hydroxycortisol in the range 26.5–6613.8 nmol/L. In our previous study using monoclonal enzyme-linked immunosorbent assay, the urinary 18-hydroxycortisol concentration ranged from 27.9 to 485.2 nmol/L in normal individuals and from 809.8 to 4264.0 nmol/L in APA. 20 Therefore, the linearity of the proposed method seems sufficient for clinical use. The sensitivity of this method, 132.3 pmol/L, is much higher than those of monoclonal EIA (20 nmol/L), 20 RIA (8.5 nmol/L), 13 and HPLC (25 nmol/L). 18 The results in the recovery and reproducibility tests were fine.
Urine samples contain high concentrations of salts. This can cause ion suppression or enhancement in LC-MS. Such effect was not observed in the proposed method (Figure 3), presumably owing to the involvement of solid-phase extraction in this method.
Additional strength of this method is inexpensiveness and quickness. Consumables needed are the ODS column and a small volume of solvents. The lack of derivatization step makes this method simple, quick (total operation time <1–2 h), and feasible for the use in clinical laboratories.
We confirmed the larger urinary 18-hydroxycortisol excretion in the patients with APA than those with IHA and EH (Figure 4), as previously reported by many investigators.12,13,15–21,25,26 Although the ranges for APA and IHA are overlapping, the measurement of urinary 18-hydroxycortisol by the proposed LC-MS/MS method might be useful for quick detection of the patients with APA out of hypertensive subjects and leading them to AVS and surgical operation. A larger number of patients with hypertension are to be included in our next study.
In conclusion, the proposed simple and quick LC-MS/MS method for the measurement of urinary 18-hydroxycortisol satisfied the basic requirements for detection and differentiation of APA from IHA and EH, and seems feasible for the use in clinical laboratories.
Footnotes
Acknowledgements
We are grateful to the Central Research Laboratory, Faculty of Health Sciences, Hokkaido University, which kindly provided the work space and equipment. Toshihiro Sakurai is a Research Fellow of the Japan Society for the Promotion of Science.
Declaration of conflicting interests
None.
Funding
This research was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science and also by the Regional Innovation Strategy Support Program of the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Ethical approval
The study was approved by the ethics review board at the Faculty of Health Sciences, Hokkaido University (approval number 11-08).
Guarantor
Hitoshi Chiba.
Contributorship
Shigeki Jin and Hitoshi Chiba researched the literature and conceived the study. Yuji Takahashi, Toshihiro Sakurai, Hirotoshi Fuda and Seiji Takeda were involved in sample preparations and analyte measurements. Hitoshi Chiba, Shu-Ping Hui, Shigeo Ikegawa and Takao Kurosawa were involved in synthesizing the standard compounds. Norio Wada, Masato Fujikawa and Katsuyuki Yanagisawa were involved in providing samples and patient recruitment. Shigeki Jin prepared the first draft of the manuscript and Hitoshi Chiba reviewed and modified the paper. All authors reviewed and approved the final version of the manuscript.
