Abstract
Background
Measurement of red blood cell thiopurine S-methyl transferase (TPMT) enzyme activity before commencing thiopurine therapy is recommended to avoid severe bone marrow suppression in TPMT-deficient patients. Patient's samples go through preanalytical transportation and storage steps before measurement. We studied patient's TPMT activity sample data to assess the effect of preanalytical variables including transportation time.
Methods
A total of 8524 TPMT enzyme activity analyses were conducted from 2002 to 2010 in a single laboratory, with samples sent from seven centres throughout New Zealand. TPMT activity was correlated with time of arrival at the reference laboratory, patient gender and age and centre of sample collection.
Results
The 6348 (74%) selected TPMT measurements that fulfilled selection criteria ranged from 0 to 25.8 IU/mL. Median delay to sample analysis was 42 h. Median TPMT activity was significantly lower for all centres compared with the reference centre (P < 0.001). Delay in sample arrival was significantly and independently correlated with TPMT enzyme activity (ANCOVA; P < 0.001), which showed a 0.011 (95% CI, 0.008–0.014) IU/mL decrease per extra hour of delay. After correcting for these data, one centre still had a significantly lower TPMT enzyme activity compared with the reference centre.
Conclusions
There was a significant negative correlation between TPMT enzyme activity and delay from sample collection to analysis. Transportation time is therefore an important preanalytical variable influencing TPMT activity. Samples from one centre had a lower TPMT activity after correcting for transportation delay, suggesting that other factors related to sample processing may also be relevant.
Keywords
Introduction
The thiopurine drugs azathioprine and 6-mercaptopurine (6-MP) have a wide variety of treatment indications, including haematological malignancies, autoimmune diseases such as inflammatory bowel disease and rheumatoid arthritis and organ transplantation.
Both drugs are subject to complex metabolism in which the genetically polymorphic thiopurine S-methyl transferase (TPMT) enzyme is the most clinically relevant in the pathway. Individuals with low or absent TPMT enzyme activity are at increased risk for developing myelotoxicity during treatment, primarily as a result of overproduction of 6-TGN. 1 The importance of TPMT enzyme activity measurement lies in its use to detect these individuals. 2
The clinical importance of the TPMT enzyme has led to development of laboratory assays to accurately measure TPMT enzyme activity. Commonly, TPMT activity is measured in red blood cells (RBCs) and the first description of a method to do this was published by Weinshilboum et al., 3 using a radio-chemical assay. It has been shown that measurement of TPMT enzyme activity in RBCs reflects TPMT enzyme activity in leukaemic blasts. 4
Although the analytical procedure of TPMT enzyme activity measurements has been extensively developed and studied to reduce laboratory errors, evaluations of the contribution of preanalytical variables such as the stability of RBC TPMT enzyme activity samples before measurement, are sparse. Moreover, the stability of TPMT enzyme activity has most often been described in a controlled laboratory setting.5,6 The effects of preanalytical variables in a relevant operational setting, 7 have not been reported previously.
We therefore collated all patient TPMT enzyme activity results from a large national laboratory database, which included samples from different centres throughout New Zealand, and assessed the effect on TPMT enzyme activity of variables including time delay between sample collection and arrival in the Christchurch reference laboratory, gender, age and collection centre.
Materials and methods
Samples and study design
Canterbury Health Laboratories (CHL) in Christchurch provides testing for TPMT enzyme activity, 6-TGN and 6-MMP concentrations in New Zealand. All blood samples in New Zealand taken for analysis of TPMT or thiopurine metabolite concentrations are transported to CHL for analysis.
We identified retrospectively TPMT enzyme activity results from the CHL database from all patients in New Zealand. We used these data to describe the distribution of TPMT enzyme activity in our population.
Samples were included if date and time of sample collection and date and time of sample arrival in CHL were recorded and if demographic data (gender, age and centre at which the sample was taken) were available. Also, we only collated data from the centres that send samples directly to the reference laboratory rather than indirectly via an intermediary centre. To expedite this we only studied data from the seven largest centres in New Zealand (Figure 1). Samples with a reported delay longer than one week (168 h) were excluded from analysis (n = 85) as it was considered that there was an error in the recorded time.
Map of New Zealand showing geographic positioning of centres used for analysis.
TPMT enzyme activity essay
TPMT enzyme activity in CHL was measured by a radio-chemical method based on the conversion of 6-MP to radio-labelled 6-MMP, using radio-labelled S-adenosyl-
For analysis, an aliquot of the lysate with 6-MP in dimethylsulphoxide (DMSO) and another aliquot with DMSO only were incubated with S-adenosyl-
Statistical analysis
All the data retrieved from the CHL database were entered in MS Excel spreadsheets. Statistical analysis was performed using GraphPad Prism for Windows (version 4.01) and SPSS Statistics for Windows (version 17.0).
In order to describe the characteristics of the TPMT enzyme activity, a frequency distribution of all collected TPMT measurements was used, regardless of any missing demographic data. When the patients had repeat TPMT enzyme activity measurements, only the first measurement was used.
Comparison of the relative times of transportation from each centre was conducted using the Kruskal–Wallis and Mann-Whitney U non-parametric tests. The effect of transportation delay on TPMT enzyme activity was analysed using a simple linear regression. To determine the independent contribution of transportation delay, age, gender and centre on TPMT enzyme activity, an analysis of covariance (ANCOVA) was undertaken. Estimates of means and pairwise comparisons among centres were generated with this model. Christchurch, as the referral laboratory with the least delay, was used as the reference for these comparisons. Two-sided tests were used and P values <0.05 were considered as statistically significant.
Results
Population distribution
Out of a total of 8524 TPMT enzyme activity measurements identified from the laboratory database, 7383 (86.6%) measurements were from individual patients. Analysis of the data from these individual patients showed an expected tri-modal distribution with 0.4% low activity, 15.7% intermediate activity, 81.1% normal/high activity and 2.8% ultra-high activity (Figure 2).
Distribution of thiopurine S-methyl transferase enzyme activity recorded from New Zealand-wide samples at Canterbury Health Laboratories.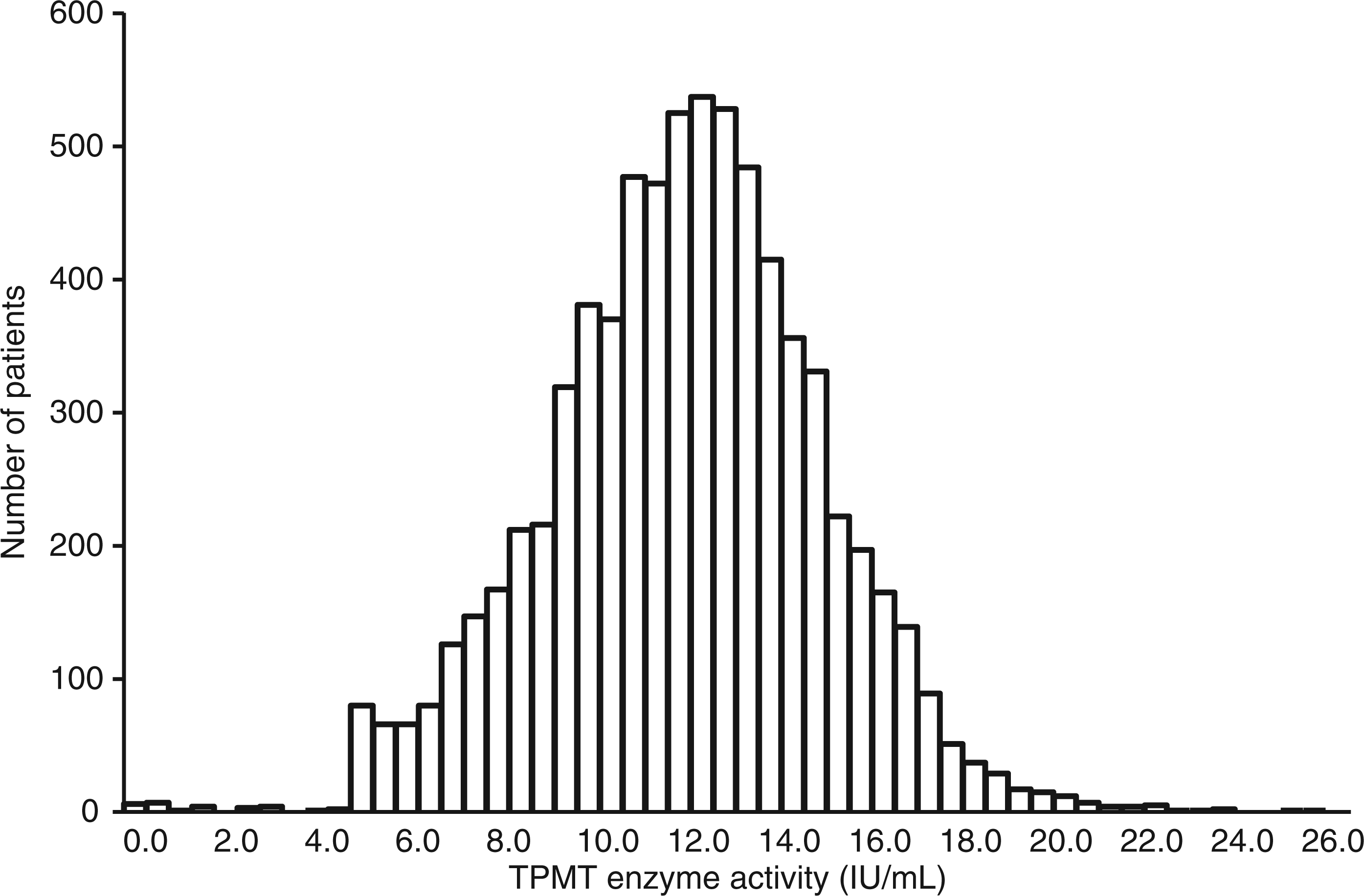
Demographic characteristics
Descriptive report of characteristics of patient RBC TPMT samples.
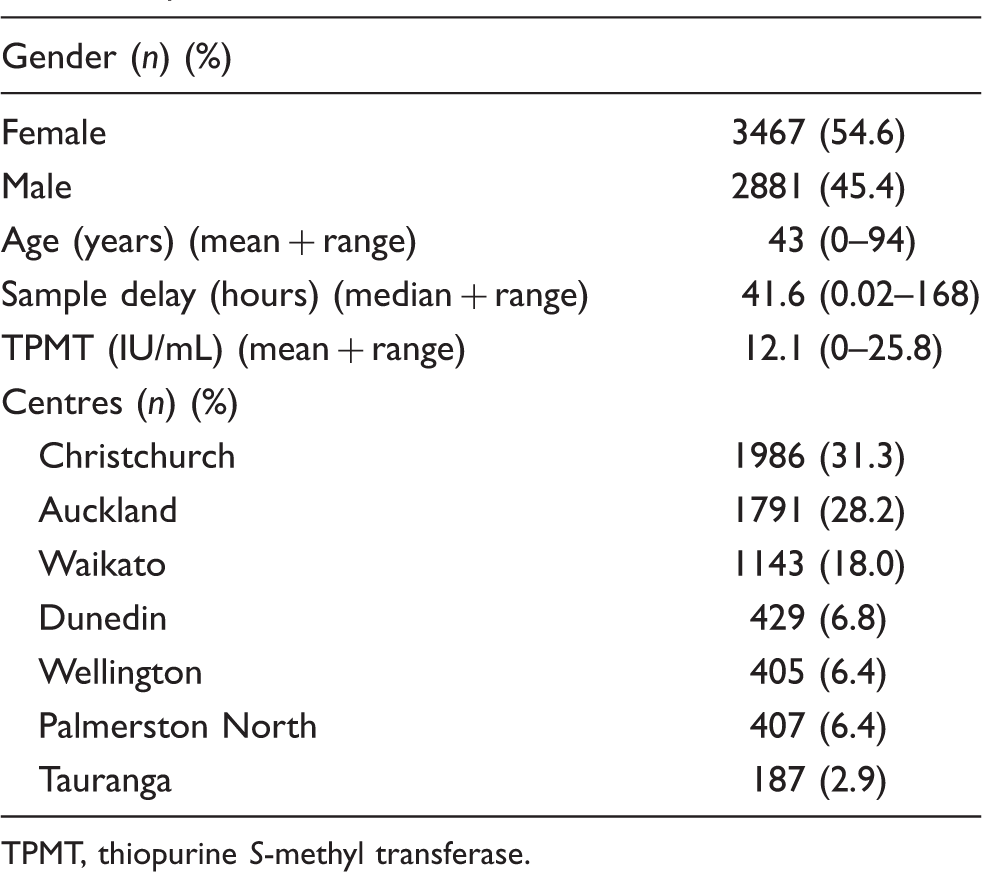
TPMT, thiopurine S-methyl transferase.
Median delay in hours of TPMT samples from different centres.
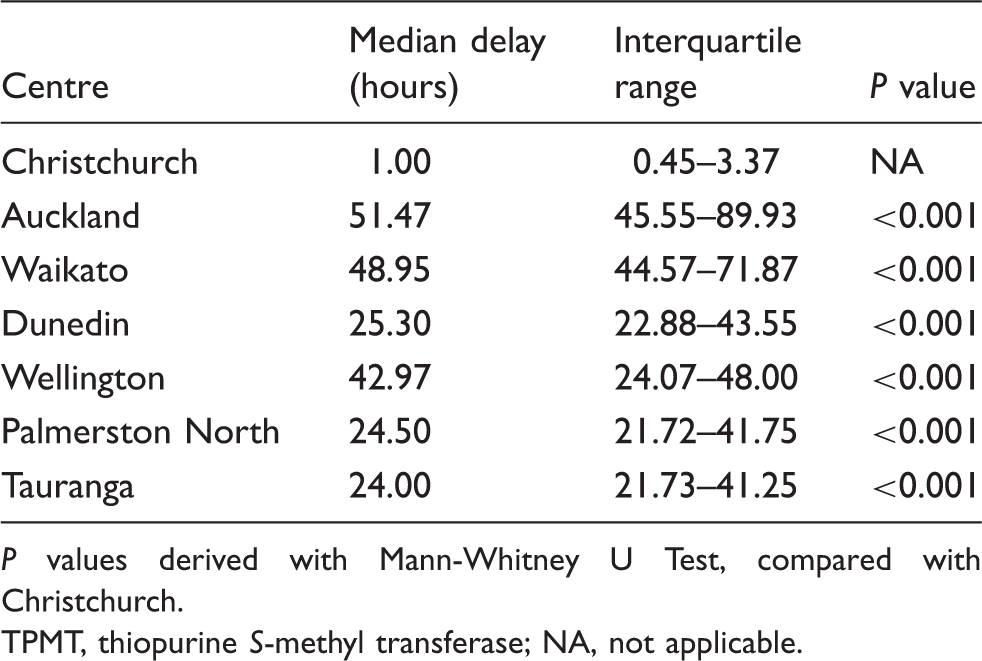
P values derived with Mann-Whitney U Test, compared with Christchurch.
TPMT, thiopurine S-methyl transferase; NA, not applicable.
Effect of preanalytic variables on TPMT enzyme activity
There was a significant negative association between TPMT enzyme activity and sample delay, with the slope of the linear regression analysis indicating a 0.016 (95% CI, 0.014–0.018, P < 0.001) IU/mL decrease in TPMT enzyme activity for every extra hour of sample delay from collection to arrival. This equates to a 0.4 IU/mL decrease in TPMT enzyme activity for every additional day of delay.
Mean TPMT enzyme activity levels (expressed in IU/mL) in patient's samples from different centres, adjusted for age, gender and transportation delay.
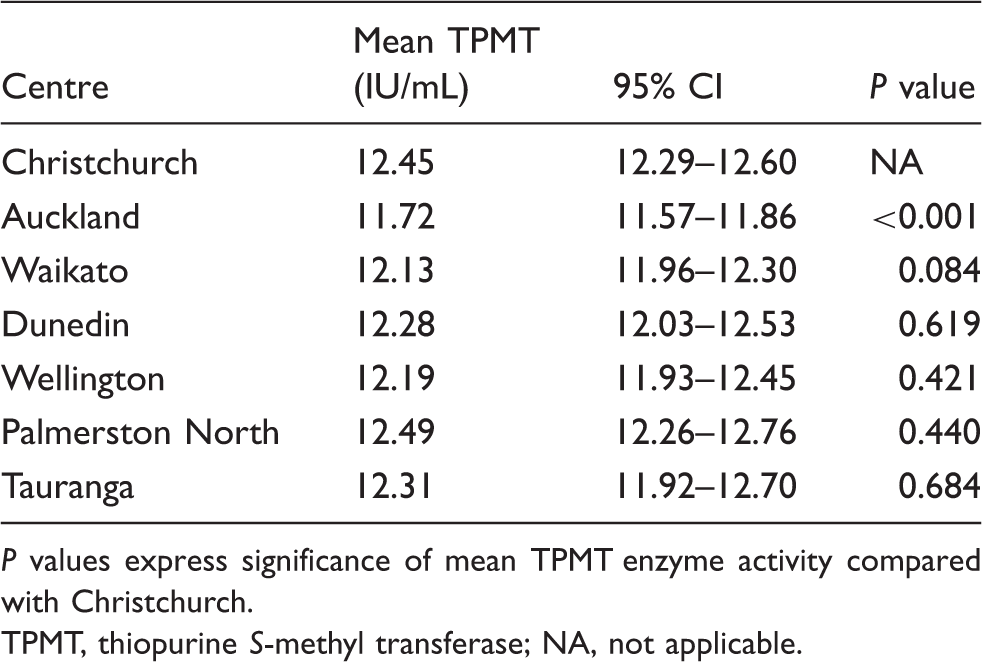
P values express significance of mean TPMT enzyme activity compared with Christchurch.
TPMT, thiopurine S-methyl transferase; NA, not applicable.
Discussion
Our study shows that delay in transport of blood samples has a small, but significant reduction on the resultant measured TPMT enzyme activity. The difference in TPMT activity of samples in this study was 0.26 IU/mL for every extra day of delay, independent of other contributing factors. As the delay for TPMT enzyme activity samples differed significantly between centres, samples that are sent from geographically distant centres may have altered measurement outcomes. Lower TPMT enzyme activity as a result of a delay in sample analysis, although unlikely to be clinically significant per se, may potentially lead to incorrect phenotypic classification and treatment decisions with regard to adjustments made to thiopurine dose, since patients with intermediate TPMT enzyme activity require an up to three-fold lower dose. 2
Few studies have provided data on the stability of TPMT enzyme activity over time. It seems that samples from healthy individuals are relatively stable for several days, especially at storage temperatures equal to or below 4℃. 5 However, there is evidence that samples may not be as stable in some disease populations and that TPMT activity can decrease significantly at room temperature. 6 Our samples are usually transported at a temperature of 4℃ and subsequently stored at 4℃ prior to analysis. The assay procedure is the same for all samples, thus other preanalytical variables such as transportation time and sample handling appear to contribute to changes in TPMT enzyme activity.
There are a number of possible explanations for our results. We analysed blood samples taken from patients and transported in an operational clinical setting, which may therefore be exposed to more confounding variables than in previously reported studies in a controlled laboratory setting. Although recommendations are made regarding sample handling (e.g. transportation with ice packs), we are unable to verify to what extent these are followed explicitly. Temperature during transportation can potentially increase up to 20℃. We postulate that this may lead to enzyme degradation, which in turn causes decreased measured TPMT enzyme activity with long sample delay. Suggested lower stability of TPMT enzyme activity in patients compared with healthy individuals could make samples more vulnerable to the effects of transportation time and sample handling in a clinical setting. Further research would be needed to investigate these possibilities.
Other variables that showed an independent significant influence on TPMT activity were gender, age and sample collection centre. The gender-based difference in TPMT enzyme activity is not surprising, since it has previously been shown that men have a higher TPMT enzyme activity compared with women. 10 Age had a significant, but very small effect on TPMT enzyme activity. The increase of 0.006 IU/mL per additional year of age is not considered to be clinically relevant. Interestingly, there was one centre (Auckland) with significantly lower mean TPMT enzyme activity compared with Christchurch. This suggests a systematic issue, such as a higher frequency of samples not being sent with ice packs to maintain low temperatures during transportation. Although we cannot confirm this without more evidence, this might explain a lower TPMT enzyme activity, which is consistent with the view that other preanalytical variables in addition to transportation time are important factors to address. A possible factor that we did not address was the distribution of ethnic groups. New Zealand has a mix of predominantly Caucasian, Asian and Polynesian inhabitants and the distribution of these groups differs among the centres we studied. Furthermore, we did not assess the distribution of thiopurine indications, co-medications and co-morbidities for each centre.
Improvements in rates of analytical errors in clinical laboratories in the last decade are a result of improvement of the steps in the total testing process (TTP). Evidence is accumulating, however, that pre- and postanalytical steps are more error-prone than the analytical steps involved in the TTP and may cause potential adverse outcomes for patients. 7 There is therefore an imperative to investigate any potentially confounding variables in the TTP. Transportation time alone does not fully explain the observed variance in TPMT enzyme activity and we postulate that factors related to sample handling may also be important and that further research is needed to elucidate this. More meticulous attention to preanalytical variables in the TTP of TPMT enzyme activity could prevent potential errors of misclassification and improve clinical management.
Our observation of TPMT distribution among the study population showed an expected tri-modal distribution. This is in line with results of other studies on the distribution of TPMT enzyme activity 11 13 and previously reported data on the distribution of TPMT in New Zealand. 8 The proportion of patients with an intermediate phenotype was larger than described in previous studies,8,11--13 although our cut-offs were carefully validated 9 and in operational practice, all samples with activities less than 10.5 IU/mL are routinely submitted for genotyping. Preanalytical variables, including sample delay and variations in sample handling, may have contributed to underestimation of TPMT enzyme activity and phenotypic ‘misclassification’. Furthermore, a very small number of samples had TPMT activity between 1.0 and 4.5 IU/mL, which is not usually expected. These results could represent patients with low TPMT activity that have been transfused, an effect of sample analysis delay, or a rare TPMT genotype. However, we did not have sufficient information about these patients to explore this further.
Conclusion
We conclude that sample delay has a small but significant effect on TPMT enzyme activity, together with variations in sample handling. Samples sent from geographically distant places are prone to falsely low TPMT enzyme activity, which should be taken into account in the interpretation of the result. Further studies are required to investigate the effect of preanalytical variables involved in the TTP in order to assess what further improvements can be made.
Footnotes
Acknowledgements
We acknowledge Linda Pike and the staff of Specialist Biochemistry, Canterbury Health Laboratories for the TPMT assays and Associate Professor Chris Frampton for statistical advice.
Declaration of Conflicting Interests
None.
Funding
RvE received unrestricted educational grants for this research from Abbott, Schering-Plough (MSD) and Tramedico.
Ethical approval
No specific ethical approval was required as the research resulted from laboratory audit analysis of non-identified patient sample results.
Guarantor
MB.
Contributorship
RvE researched literature, assessed the patient results database, analysed the data and wrote the first draft of the manuscript. PC assisted with preparing the results database and data analysis. CS supervised the TPMT assay and prepared the results database. MB conceived the study, supervised RvE, and assisted with data analysis and manuscript drafting. All the authors reviewed and edited the manuscript and approved the final version of the manuscript.
