Abstract
Recently, there has been tremendous interest in developing techniques such as MRI, micro-CT, micro-PET, and SPECT to image function and processes in small animals. These technologies offer deep tissue penetration and high spatial resolution, but compared with noninvasive small animal optical imaging, these techniques are very costly and time consuming to implement. Optical imaging is cost-effective, rapid, easy to use, and can be readily applied to studying disease processes and biology in vivo.
In vivo optical imaging is the result of a coalescence of technologies from chemistry, physics, and biology. The development of highly sensitive light detection systems has allowed biologists to use imaging in studying physiological processes. Over the last few decades, biochemists have also worked to isolate and further develop optical reporters such as GFP, luciferase, and cyanine dyes. This article reviews the common types of fluorescent and bioluminescent optical imaging, the typical system platforms and configurations, and the applications in the investigation of cancer biology.
Technology of In Vivo Optical Imaging
For many years, molecular biologists have performed live cell assays and various biochemical assays on the cellular scale. For example, using fluorescent proteins, scientists are able to track cells and proteins on the micrometer scale with microscopy. In recent years, this technology has been adapted to perform imaging of optical reporters in living animals. Not only can whole-body measurements be made, but higher-resolution microscopic images in live animals can also be generated—as in the case of developments in intravital microscopy. The typical techniques of optical imaging widely performed today are fluorescence and bioluminescence imaging.
Optical Reporters
Fluorescent reporters. In fluorescence imaging, excitation light at a defined wavelength is required to excite a particular fluorophore, which in turn generates a signal in the form of light at a specific wavelength that can be detected. Although there are many fluorophores available, each fluorophore has unique optical properties, most notably, in excitation and emission wavelengths. The most common fluorophore currently used in optical imaging is green fluorescent protein (GFP). A typical fluorescence-based imaging system configuration is shown in Figure 1.
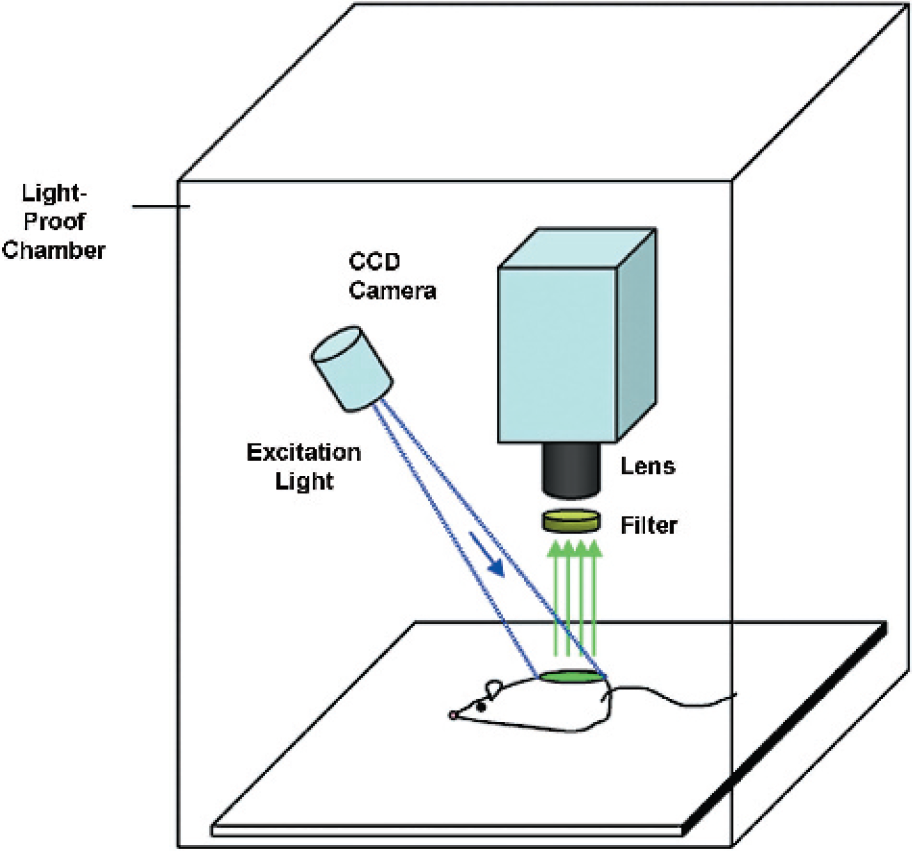
Shown here are the typical components and setup of a fluorescent imaging system. Fluorescent imaging systems require an excitation light source and the appropriate filters. The illustration is for demonstration of principles only. The light source depicted is regional and unidirectional. However, one may design an imaging system with multiple light sources (laser, halogen-based, etc.) and whole-body illumination. For optimal fluorescent imaging conditions, a whole-body and uniform illumination field may be important design considerations.
Green fluorescent protein. GFP was originally isolated from the jellyfish, Aequoria Victoria. GFP excites at a wavelength of ˜470 nm and emits at a wavelength of ˜510 nm. GFP has been used extensively to study subcellular processes such as gene expression and protein localization [1,2]. There are ongoing efforts in developing more GFP variants that have altered emission and excitation wavelengths, enhanced brightness, and improved pH resistance [3]-Derivatives of the original Aequoria proteins such as cyan (CFP) and yellow (YFP) variants have been introduced [3].
DsRed. DsRed is a red fluorescent protein that was recently cloned from Discosoma coral [3–5]. DsRed demonstrates homology to GFP, but distinctively exhibits an excitation wavelength of ˜560 nm and an emission wavelength of ˜580 nm. As emission wavelength increases, absorption by tissues concurrently decreases, thereby the light signal becomes less attenuated. As a result, red-shifted proteins allow for better depth penetration of optical signals. Thus far, few published studies in whole-body live mouse imaging have taken advantage of DsRed. However, several groups have begun to use DsRed in conjunction with other fluorescent proteins to perform multicolor imaging in cellular assays [6,7]. The stability and high intensity of brightness of DsRed make this novel fluorescent protein a viable option for both cellular assays and animal work. We can probably expect more reports using DsRed to be published in the near future. Recent and continuing efforts in engineering and optimizing both DsRed and GFP has been summarized in a comprehensive review by Zhang et al. [8].
Near-infrared fluorophores. An even higher wavelength, in the near-infrared (NIR) region, allows for further depth penetration. Compared to GFP, fluorophores based on NIR dyes offer better visualization of signals deeper within the tissue. Most commonly reported in recent literature has been the use of cyanine (Cy5 and higher), probably due to wide availability and relative nontoxic nature. Photon penetration into and out of tissue is high for NIR fluorophores that have emission spectrums of 700–900 nm [9–11]. As will be discussed below, signals at wavelengths <600 nm are highly attenuated via absorption by hemoglobin. Tissue absorption, scattering, and autofluorescence are also lower in NIR fluorophores [9,10]. For more information regarding the latest in NIR light, please refer to a review article by Ntziachristos et al. [12].
Bioluminescent. The firefly luciferase protein is an enzyme that was originally isolated from the American firefly, Photinus pyralis. Upon reaction with a substrate, luciferin, in the presence of ATP and oxygen, light is produced [13,14]. Luciferase-encoding genes have been extensively used by molecular biologists as reporter genes for protein expression. This technology has since been applied in vivo, namely, in small animal bioluminescence imaging.
One primary difference between fluorescence imaging and bioluminescence imaging in animals is the required substrate injection. Luciferin is a water-soluble substrate that penetrates cell membranes easily. However, luciferin appears to carry minimal toxicity as reported in some studies [13,14].
One primary advantage of bioluminescent imaging when compared with fluorescence imaging is the high sensitivity due to low background noise. As a result, the detection of very small amounts of light signal in bioluminescence imaging is possible. When high sensitivity is required, bioluminescence may therefore be the technique of choice.
Optical Imaging in Tissue
Whether the emitted light is from a fluorophore (GFP, DsRed, or a NIR dye, or a luciferase reaction), it is important to understand the principles of tissue optics in interpreting images from and implementing fluorescent and bioluminescent optical imaging. Mammalian tissue is a semi-transparent medium through which photons are absorbed and scattered [15,16].
Absorption and scatter depends significantly on the cell and tissue type and the presence of proteins, especially hemoglobin. Absorption in tissue attenuates any optical signal transmitted through to the surface. For example, substances in tissues of an animal, such as hemoglobin, absorb light in the blue-green region of the spectrum but is relatively transparent in the red spectrum (>600 nm). Scatter is the reflection, refraction, or diffraction of light due to cells, lipids, and other components of tissue, which changes linear path of propagating light within tissue. In case of emitted light through a medium such as tissue, significant scatter will result in a diffuse signal at the surface. Because depth of the signal is an important consideration in animal imaging, the deeper the signal within a medium, the more the signal will be subject to further absorption or scatter.
In fluorescent imaging, the understanding of incident light is also important. An excitation light source is required to generate a signal from a fluorophore within cells in fluorescent imaging. In fluorescence, following the absorption of photons, excited molecules return to a more stable state by the emission of a lower energy photon—the signal. When a beam of light enters the tissue, part of it will be reflected by the skin surface, while the rest will be transmitted into the tissue and be absorbed and scattered. As a result, the deeper the fluorophore, the less excitation light is available to generate a signal.
Overcoming the effects of absorption and scatter in tissues are clear challenges to researchers currently developing optical reporters for use in biomedical applications. Various dyes and proteins used in optical imaging have different optical characteristics, such as emission wavelengths and signal intensity. For example, higher wavelength dyes have the ability to pass through more tissue with less attenuation by absorption. Also, compared with bioluminescent proteins, fluorescent proteins tend to have a higher signal intensity.
Much work has been done to better understanding tissue optics. Physics of light behavior in mammalian tissue can be explained by a diffusion model [15]. Optical spectroscopy is a technology that takes advantage of photon absorption and scatter within the tissue to noninvasively differentiate between normal and pathologic tissue [16]. As a result, much progress has been made in better understanding principles of scatter and absorption—essential in the development of optical spectroscopy (for a more detailed mathematical explanation, as well as further details regarding optical properties of tissues, and spectroscopy, refer to Rice et al. [15] and Tromberg et al. [16].
Examples of In Vivo Optical Imaging Configurations
In the arena of bioluminescence and fluorescence body imaging, advances have been accomplished that allow two-dimensional and three-dimensional images to be obtained. For example, planar or reflectance imaging is used to measure the total or composite signal emitted from an array of depths in the tissue through the surface of the animal, allowing the acquisition of a semi-quantitative projected planar image. However, tomographic imaging allows more precise and quantitative resolution of signal originating deep within tissues. The technology of optical tomographical techniques has been recently validated in small animal models [12,17–19].
Fluorescent-mediated molecular tomography enables the resolution and quantification of fluorophores located within the tissue. Through mathematical modeling of light propagation in tissue and detection technology, tomography is now possible in optical imaging. Through the use of multiple measures and acquisition of projections around the illuminated source, mathematical calculations allow the reconstruction of the image. FMT has recently been used by Ntziachristos et al. [12,18] to image cathepsin B activity within mice (for more information regarding the emergence of optical tomography, the reader may refer to Refs. [12,17–19]).
Currently, a typical equipment arrangement includes a charge-coupled device (CCD) camera, a light-proof enclosure, imaging software and hardware, excitation light source(s), emission and excitation filters, camera lens and/or microscope, and animal support devices.
CCD. The hallmark of an optical imaging system is the CCD camera. Although there are other detection systems such as photo-multiplier tube detectors, CCD cameras are widely available and cost-effective. CCD cameras are very sensitive devices based on the principle that charge corresponds to variable shades of light. CCD spectral response, quantum efficiency (QE; over desired wavelengths of detection), sensitivity, image quality/geometric stability, dynamic range, resolution, chip resolution, signal-to-noise ratio, and baseline noise are important specifications of the camera that need to be considered. In order to detect sources up to a few centimeters deep, the camera must have the lowest noise possible, highest sensitivity, or QE [15,20,21]. Baseline noise should be minimized in applications where maximal sensitivity is required. Depending on the type of fluorophore or type of imaging required for a particular experimental goal, a camera equipped with a spectral response needs to also be properly selected. Chip size is also another issue where the higher the chip size, the higher will be the resolution in most cases.
Two basic types of CCD cameras are commonly available on the market. Image-intensified photon counting CCDs are a popular choice for bioluminescent applications. However, a second, better choice for in vivo applications is a cooled back-thinned and back-illuminated integrating CCD as suggested by Rice et al. [15].
Photon counting CCDs consist of a high-gain image intensifier that counts individual photons while maintaining a low noise level. However, a disadvantage of these types of CCDs are low QEs of 10–15% due to the use of bi-alkali photocathodes [15]. QEs drop to <1% at wavelengths above 600 nm in some camera types [15]. These cameras also exhibit limited dynamic range for bright signals.
On the other hand, cooled back-thinned and back-illuminated integrating CCD cameras offer a very high QE, low noise levels, and high spatial resolution. QE approaches 85% in some cameras and remains high in wavelengths >600 nm, in the red and NIR spectral ranges. Dark current is a source of noise in integrating CCDs but is reduced by cooling. Cooling level depends on the manufacturer of the CCD but cooling CCD to –105°C reduces dark current to insignificant levels [15]. Higher spatial resolution is also another advantage of integrating CCDs.
For more information, a good review by Rice et al. [15] and Hooper et al. [20,21] discuss CCD selection and imaging system design in more detail.
Lens and filter selection. In vivo imaging can be performed at varying magnification. As a result, CCD cameras can be either attached to a macroscopic zoom lens or to a microscope. Although a macroscopic video-camera lens can offer magnifications that allow whole-body level animal imaging, a microscope offers higher magnification to visualize individual cells in vivo. In choosing a lens or adapter for a microscope, one must insure that the CCD chip size (common sizes range from 1/2 to 2/3 in.) is accommodated. Lenses with a wide aperture is also desired in order to maximize the transmission of light to the CCD chip. The focal length of the lens is also an important attribute that needs to accommodate the desired proximity of the source to the detector.
In the case of intravital microscopy, one may desire to attach a microscope (confocal, stereodissecting scope, etc.) to a CCD camera. Intravital microscopy is a technique that allows real-time imaging of molecular and cellular processes in intact living animals with high resolution (1–15 μm) using molecular probes (fluorescent usually). For example, in an animal model for intravital microscopy, chronic-transparent windows have been implanted that allow the visualization of angiogenesis and vessel dynamics with systemically injected FITC-dextran [22] (for more information about intravital microscopy, please refer to a report by Jain et al. [22]).
Filter selection depends on the type of imaging to be performed, bioluminescent or fluorescent imaging. Typically, bioluminescent imaging with luciferase does not require a filter. Fluorescent imaging requires careful selection of filters that block out undesired wavelengths of light while maximizing detected signal from the fluorophore of interest. Important aspects to consider when choosing a filter includes wavelength, shape and width of passband, long-term stability, and compatibility with lens or microscope.
Excitation light source (fluorescent imaging). Another important consideration includes selection of the excitation light sources in the case of fluorescence imaging. Intensities of fluorescent signals depend on the extent and quality of the delivered light source. Widely available and affordable lasers or filtered white-light sources can typically be used. Typical white-light sources include high-intensity halogen light sources attached to fiber-optic cables. However, lasers deliver excitation light of higher intensity and narrower wavelength bandwidths, which may offer advantages where significant au to fluorescence occurs, and excitation and emission spectra overlap. However, lasers require more time for scanning the subject.
An important consideration in the design of the excitation light source is the uniformity of illumination. Although lasers offer greater uniformity in excitation, fiber-optic-based light sources require greater care in design. In order to have effective uniformity, multiple light sources maybe needed as well as careful positioning of light sources to minimize variance of the light intensity.
Light-tight imaging box. The CCD camera, lens, and excitation light source (in fluorescent imaging) are mounted to a light-tight chamber where the animal is placed. Important considerations for the imaging box is the proximity of the detector to source and animal support devices. The desired distance from the detector to source is an important factor in lens selection. Closer proximity of the source to the detector can usually enable higher-resolution imaging and enhanced detection of light. The light-tight imaging box must also be built such that the excitation light sources provide maximal uniformity in the excitation illumination for fluorescence.
One may also desire to build in animal support devices. For example, commercial imaging systems such as the Xenogen IVIS system currently can be equipped with a nose-cone anesthesia system and a temperature-regulated platform [15].
Detection sensitivity. As in other imaging modalities, maximizing sensitivity of detection is of high priority. Steps must be taken on both hardware and biology side to insure optimal sensitivity.
In choosing the hardware, optimizing the CCD camera, lens, and excitation light source (for fluorescence) are most important. When choosing a camera, the highest QE for the desired spectral response should be chosen. The spectral responses must also match the wavelength specifications for fluorophore or bioluminescence reaction in the experiment. A high-performance and reliable cooling system should also be installed, thereby minimizing the noise (dark current) in the CCD camera. Binning features of the CCD camera can also be used to increase sensitivity and the signal-to-noise ratio, while sacrificing spatial resolution. Choosing the appropriate lens can also facilitate maximal light passing to the CCD camera, namely, by having a wide aperture. In the case of fluorescent imaging, an excitation light source that provides uniform and high-intensity illumination would also enhance the maximal excitation of the source fluorophore.
With regard to the biology and animal model, one can also take steps here to increase sensitivity. Whether it is fluorophore, such as GFP or cyanine, or a bioluminescent protein, such as luciferase, an abundance will result in brighter signals. Through increasing gene expression or increasing the amount of dye present in the source, a brighter signal will mean increased sensitivity. Because dark fur or dark skin absorbs light very well, the strain of mouse matters significantly in small animal imaging. For example, minimizing dark fur by shaving or the use of nude mice can increase the light emitted.
The design of the appropriate animal model with the optimal hardware configuration may therefore lead to the highest signal-to-noise ratios, the best quality images, and significant insights into molecular and cellular processes via noninvasive means.
Applications of Optical Imaging in Cancer Research
Imaging Tumor Kinetics, Localization, and Treatment
Optical imaging certainly holds utility in assessing tumor growth in xenograft, allograft, spontaneous-transgenic, and orthotopic tumor mouse models. In tumor-mouse models, the practical and simple quantitation of tumor burden is often desired. Tumor cells that express luciferase or GFP or any other reporter gene can be essentially marked and tracked by optical imaging. The tracking of labeled-tumor cells in vivo has numerous applications in monitoring tumor growth kinetics, treatment, and extent of metastatic disease. Review articles by Contag et al. [23] and Hoffman [24] may provide further information on fluorescent and bioluminescent imaging of tumor models.
Growth kinetics. Although subcutaneous and surface tumors can be measured by calipers, assessment of tumors implanted orthotopically, intraperitoneally, and intracranially requires sacrifice of the animal. If tumor burden over time needs to be assessed at regular time points, large numbers of animals will be needed in order to conduct long time-course experiments. However, optical imaging can offer a number of advantages in tracking tumor growth in individual animals due to its noninvasive, objective, and quantitative features. Numerous studies have demonstrated the capability to noninvasively monitor tumor cell proliferation in various animal models [25–28]. For example, imaging of spontaneously developing pituitary tumors, difficult to quantify with traditional measurements, has been noninvasively assessed over time (Figure 2). Validation studies have been conducted in our laboratory and many others where the in vivo growth of tumor cells engineered to express luciferase and GFP have been shown to correlate linearly with both tumor volume and mass [25–29].

Liver metastases shown in a model where intrasplenic injections of tumor cells were given. Metastases were tracked by GFP fluorescence imaging. These are pseudocolor images.
Sensitive detection and metastases. Comparing the two most commonly used reporter genes to track tumor proliferation, luciferase is significantly more sensitive than GFP in detecting small numbers of tumor cells. Because the signal detected by GFP-based imaging is affected by autofluorescence in skin, tumors need to be a sufficient size or express enough fluorophore before they can emit a detectable signal overcoming background autofluorescence. However, in bioluminescent imaging, there is a high signal-to-noise ratio. Background noise is very low because mammalian tissues do not emit light. Bioluminescent imaging has been demonstrated to allow the sensitive detection for small numbers of cells. Studies have demonstrated the detection of as few as 1000 cells in the peritoneal cavity [8]. The reported work in the literature has been done using mostly CCD cameras with macroscopic and microscopic lenses, but also luminometers, photon-counting devices, and photomultiplier tubes [15,30–32]. Yang et al. [33] demonstrated that GFP-fluorescence based in vivo imaging can detect as few as 50 cells. It is important to emphasize that the work was conducted using a microscope attached to a CCD camera. In some cases, the skin was removed and windows were created. A study that determines the minimum number of cells detectable on a macroscopic scale using fluorescence imaging has yet to be performed.
Bioluminescence imaging is certainly more sensitive than fluorescence imaging. However, one strength of fluorescent fluorophores is the ability to perform high-resolution microscopy of GFP-expressing cells. As a result, if tracking of small numbers of cells at high magnification and perhaps in real time is desired, fluorescence-based methods can be considered.
The sensitivity of the technique can be very important depending on the application, especially where few numbers of cells need to be detected. For example, noninvasive imaging can be applied to identifying mice that bear minimal tumor burden for studies evaluating early intervention and treatment regimens. Micrometastases consisting of few numbers of cells may also be monitored [33–36]. Figure 3 shows the presence of liver metastases (Libutti SK, unpublished data) as a result of intrasplenic implantation of tumor cells as detected by GFP imaging. Similar work has been done by Yang et al. [33,34], Li et al. [35], and Wetterwald et al. [36]. Again, significant background from the skin is associated with GFP imaging. Expression levels of GFP may also limit the use of fluorescence. As a result, skin flaps and skin windows may be required to aid in detection of GFP-labeled metastases. Achieving a high enough tumor signal-to-background ratio is essential to detect small groups of cells. Literature thus far suggests that more sensitive methods such as bioluminescence imaging may be a better tool to use in the case of monitoring metastasis [25,36].

Noninvasive quantification of pituitary tumor growth over time. This is an analysis of tumor development in POMCcre–POMCluc;RbF19/F19 mice. Animals were measured in 2-week intervals from 4 weeks onward. Pseudocolor images of longitudinal tumor growth in POMCcre–POMClucF19/F19 animal are shown on the left. Quantification of emitted photons shows an approximately exponential tumor growth between 12 and 16 weeks. (Adapted from Vooijs et al. [28].)
Monitoring cancer treatment. Noninvasive detection of tumor cells would also certainly aid in the testing and evaluation of potential anti-cancer therapies. The ability of noninvasive optical imaging to monitor tumor growth kinetics and metastases can be applied in the assessment of new therapeutic agents. The power of optical imaging allows not only the characterization of subcutaneously implanted tumors, but also that of tumor models where cells are implanted intraperitoneally, intracranially, and orthotopically. The monitoring of the efficacy of chemo therapeutics, cytokine, or immunotherapy has been illustrated in numerous studies [26–29]. These studies implement both bioluminescence and fluorescence techniques and show the potential to monitor early effects of treatment, and also tumors that are not otherwise easily measured due to implantation within the body cavity.
Imaging Molecular Events with Optical Sensors
Optical imaging has not only been applied to tracking endogenously labeled tumor cells by reporter genes, but several research groups have also designed agents that can be detected by imaging. Specifically, labeled peptides that can act as optical tracers have been engineered to noninvasively elucidate physiological and molecular processes. Currently, the most commonly used organic dyes are a class of cyanine dyes, specifically those with emission spectrums in the NIR range (700–1000 nm). Analogous to radiolabeling peptides, cyanine conjugates can be tracked. This potentially is a safer alternative because cyanine conjugates do not rely on ionizing radiation. Cyanine-peptide conjugates have also been shown to exhibit stability in vivo and produce no toxicity at detectable doses in the small animal [37–39].
Monoclonal antibodies. Earlier work was focused on conjugation of cyanine dyes to monoclonal antibodies that bind specifically to receptors on tumor cells [37–40]. Work by Ballou et al. demonstrated that Cy5.5 and Cy7 monoclonal antibody conjugates allowed for effective tumor visualization. Translation to humans remains a challenge because conjugated antibodies need long periods of time to accumulate in sufficient concentrations to be useful for the detection of lesions [38,41].
Proteases. More recently, cyanine-based tracers have evolved to image specific molecular events. Weissleder and colleagues designed novel compounds conjugated to cyanine dyes that would be activated by protease cleavage [39]. These probes were engineered such that they are dark in their initial (quenched) state but emit light only when cleaved by specific proteases. Taking advantage of a higher rate of endocytosis and overexpression of proteases in tumors, Weissleder et al. developed probes that would emit light upon intracellular cleavage by lysosomal protease activity in tumor cells. Using this technology, it was demonstrated that micrometer-sized tumors were noninvasively detected in vivo [39,42,43].
Of the probes that have been reported in the literature thus far, probes specific to cathepsin B/H and matrix metalloproteinases (MMPs) show promising results [44,45]. Not only may these probes be useful in tumor detection, but there also appears to be potential for these activatable probes to describe tumor aggressiveness and also evaluate the efficacy of anti-tumor agents. Using probes activated by cathepsin activity, Bremer et al. [44] demonstrated that higher grade and more aggressive tumors, specifically highly invasive metastatic human breast cancer compared with a well-differentiated human breast cancer, demonstrated significantly higher fluorescence as revealed by optical imaging. Bremer et al. [45] demonstrated that probes which were cleaved and activated by MMPs allowed for direct correlation of MMP activity to signal intensity. As a result, it was demonstrated that the inhibition of MMP activity could be detected within hours of administration of an MMP inhibitor, Prinomastat (Figure 4).
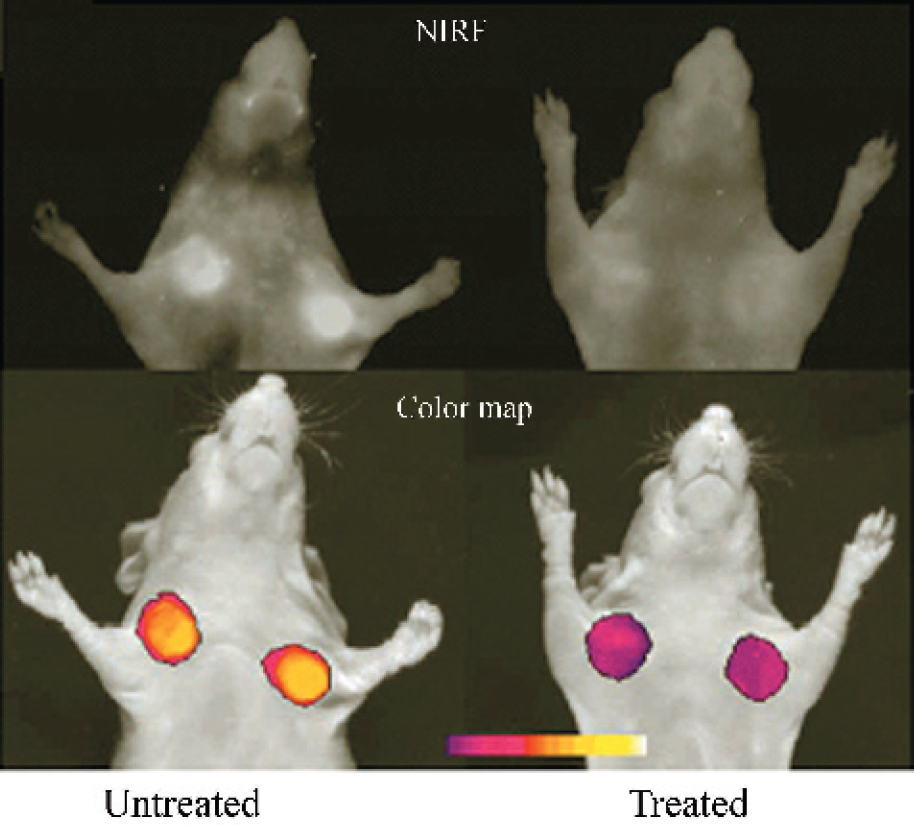
Imaging of MMP activity in vivo and response to MMP inhibitors. In vivo NIRF imaging of HT 1080 (human fibrosarcoma) tumor-bearing animals. Top row shows raw image acquisition obtained at 700-nm emission. Untreated animal is on the left. Prinomastat-treated (twice a day intraperitoneally for 2 days) animals are on the right. Bottom row shows color-coded maps of MMP-2 activity superimposed on gray-scale images. (Adapted from Hoffman [24].)
Receptors. Optical imaging can also image the presence of receptors on tumors. Becker et al. developed a peptide–dye conjugate that allowed early detection of tumors that overexpressed receptors for somatostatin (SST), such as neuroendocrine gastroenteropancreatic tumors. Conventionally, radiolabeled SST analogs have been used clinically for receptor scintillography. In this study, Becker however showed that an NIR fluorescent dye-labeled peptide was a sensitive tracer for tumor detection [37] (Figure 5). Conjugates of vasoactive intestinal peptide (VIP) have also been developed and applied in a similar fashion for the detection of receptor-positive tumors [37,46,47].

In vivo fluorescence images demonstrate targeting of Cy5.5-peptide conjugate targeting, specifically SST analogs, targeting to tumors (RIN38 pancreatic tumor expressing the somatostatin receptor subtype 2). (A and B) These show images of mouse before injection of dye-peptide conjugate. The binding of the conjugate leads to elevated tumor signal (C), whereas a control conjugate does not generate signal (D). Nontumor sites also appear to demonstrate low levels of fluorescence. This may be due to scatter/reflection from the main tumor site or nonspecific accumulation of the probe in the dorsal skin of the animal. (Adapted from Ntziachristos et al. [17].)
Apoptosis. The development of “smart sensors” or “smart probes” has gone further than the assessment of enzyme activity and receptor density. Petrovsky et al. [48] have converted an in vivo radiation-based assay for apoptosis into an optical imaging-based technique. Instead of using radioactive annexin V, fluorophore Cy5.5 was conjugated to annexin V protein, and the resulting Cy–annexin probe was shown to bind to cells undergoing apoptosis. Upon cyclophosphamide treatment, treated tumors underwent apoptosis that was detected by optical imaging. This is another example of how conjugation of optically detectable fluorophores, here a Cy dye, to peptides can be used as probes to assay for specific biological phenomena in vivo.
Treatment kinetics and distribution. Camphausen and colleagues have also applied optical imaging in monitoring drug trafficking and kinetics (K. Camphausen, personal communication). To study in vivo behavior of the anti-angiogenic peptide endostatin, Cy5.5–endostatin conjugates were systemically injected into mice. Optical imaging was then used to serially monitor fluorescence signal over time in subcutaneously implanted murine tumors in the legs of mice. Imaging revealed early drug uptake and also a peak concentration localized in the tumor over time (Figure 6). As a result, using molecular imaging allows for investigation of peptide trafficking in vivo. Implementing optical imaging as a key strategy in tracking proteins in vivo will accelerate both drug and treatment protocol designs.
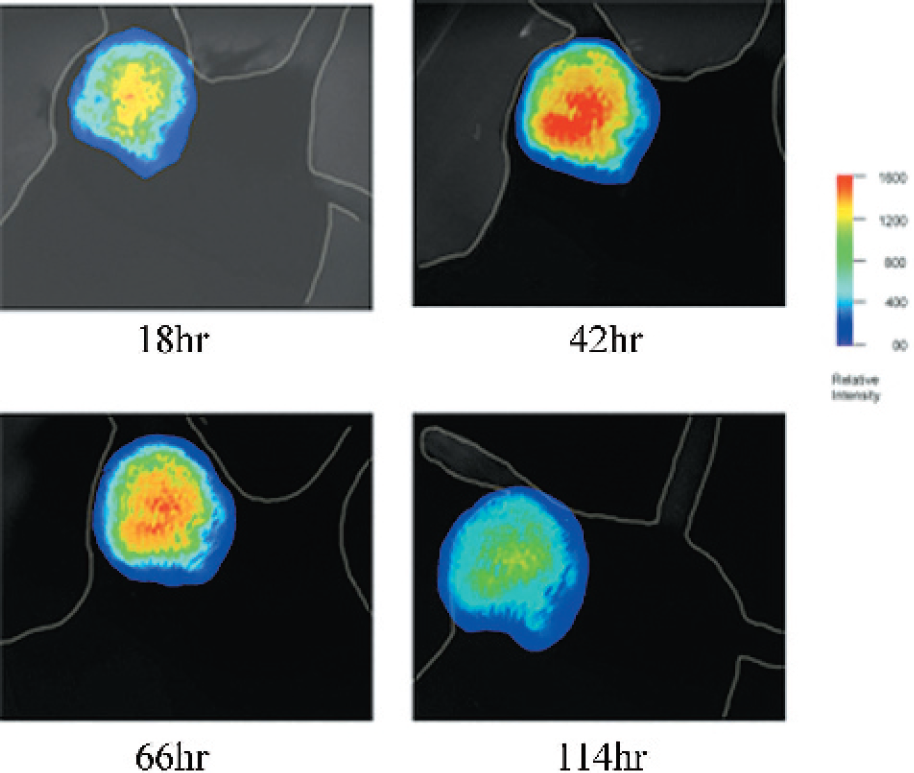
This figure shows the monitoring of conjugated Cy5.5-endostatin accumulation in suhcutaneously implanted tumor over time. In this experiment, a peak in signal and accumulation of conjugate is demonstrated at ˜42 hr (Courtesy of K Camphausen, NCI, personal communication.)
Lymphocyte Trafficking
Optical imaging has made a significant impact on the understanding of immune responses by revealing lymphocyte trafficking patterns in vivo. Lymphocytes are mobile and widely distributed throughout the body. Their small numbers and their significant mobility become challenges in monitoring the migration of lymphocytes. Although a number of tools, such as cryosectioning of sample tissue, allow study of the trafficking patterns of lymphocytes, most are invasive and require the sacrifice of animals. Optical imaging offers a new paradigm to study trafficking patterns in the same mouse over time and has been demonstrated to be very useful [49–52]. Interestingly, from literature, the use of bioluminescence imaging to monitor the trafficking of lymphocytes seems to be more popular as a method of choice for this purpose. Although other techniques can certainly be employed, bioluminescence offers the high sensitivity required to monitor such small numbers of lymphocyte cells.
Immunotherapy. In order to track lymphocytes directly, many engineered-labeled lymphocyte methods have been reported in the literature. The use of retroviral transduction to introduce stable expression of luciferase by the lymphocytes of interest is one such method [50]. Another technique describes the creation of transgenic mice where all cells constitutively express the reporter construct (such as luciferase) and may serve as sources of labeled immune cell populations [51]. An example of the creation of labeled cells for the purpose of monitoring lymphocyte trafficking in adoptive transfer in immunotherapy is shown in Figure 7 (Steven Finkelstein, NCI, personal communication).
Immune response can also be evaluated spatially and temporally by using labeled lymphocytes. For instance, optical imaging has been used to directly visualize in vivo lymphocyte localization in joint inflammation, arthritis, and autoimmune encephalitis [49,52]. Visualization of the migration patterns of lymphocytes will lead to a better understanding of the immune system's function in general and lymphocyte behavior in vivo for the treatment of cancer patients in particular. A comprehensive review of lymphocyte tracking using bioluminescence imaging is available for further information [51].
Gene Therapy of Cancer
Gene therapy as a possible treatment for cancer faces many obstacles. In vitro and ex vivo gene expression can be quantitatively assessed with various techniques such as RT-PCR, Northern blots, Western blots, and other molecular biology techniques. On the other hand, optical imaging can spatially and temporally characterize in vivo transgene expression patterns. Assessing successful and targeted transgene expression in mammals remains a major challenge. Tools that enable efficient, high throughput, and valuable noninvasive assessment would be particularly useful to study transgene expression and monitor vector-delivered therapeutics. Monitoring exogenous gene delivery has been demonstrated in numerous studies. For example, Hasegawa et al. [53] demonstrated that an adenoviral vector expressing eGFP delivered to various organs can be visualized by optical imaging.
Targeted gene delivery: nanoparticles. One novel approach in gene therapy utilizes nanoparticles coupled to a ligand which enables preferential delivery of genes to endothelial cells in tumors [54]. In developing and validating the targeting specificity of these particles, optical imaging was employed to monitor where genes were delivered and when expression was detected.
Conclusion
Despite major technical advances and numerous implementations of optical imaging in cancer research, this novel molecular imaging technology is still in its infancy. New developments in both fluorophores and detection technologies are principal determinants of the future of optical imaging.
Combining the development of new optical detectors with the development of novel optical probes will likely result in exciting revelations into normal physiologic and disease processes. Already, there are new technologies in optical imaging systems being developed. Multichannel (multicolor) imaging technology, optical tomography, and autofluorescence reduction technology are being introduced [12,55,56]. As previously discussed, researchers have developed fluorescence-mediated optical tomography that employs the use of image reconstruction of data acquired from an array of CCD detectors to provide three-dimensional localization of a fluorophore emitting from deep within a mouse [12].
Novel fluorophores that allow for multicolor imaging, deeper tissue penetration, and detection of specific molecular interactions are actively being studied and developed in laboratories [57]. The development of novel fluorophores is a field in itself that is advancing at a rapid rate among biochemists.
Optical imaging is certainly not limited to noninvasively assessing biological processes in cancer. Image-guided investigations can complement research in immunological, neurological, cardiovascular, infectious, and metabolic diseases. Numerous optical imaging studies have already been reported in these areas [9,49,52,58].
Clinical translation is extremely promising as optical imaging is noninvasive, cost-effective, portable, and safe from ionizing radiation. At the moment, NIR optical tracers hold the greatest promise in fluorescence-guided endoscopy due to their centimeter-magnitude tissue penetration capability. Some studies have started to investigate the use of fluorescence to aid in the detection and diagnosis of precancerous abnormalities during endoscopy and surgical procedures.
Advances in optical imaging in the clinic and laboratory are actively being pursued. With the development of new fluorophores and new detection technology, optical imaging will undoubtedly continue to contribute to a better understanding of molecular phenomena underlying physiologic and disease processes.
