Abstract
2′-Deoxy-2′-flouro-5-methyl-1-β-
Introduction
The rapid progress in molecular genetics has given scientists the ability to sequence, characterize, and manipulate genes [1]. Recent development of reporter gene technology [2–5] has complemented or, in some instances, replaced the conventional methods of genetic analysis such as hybridization techniques [1]. In the last decade, the herpes simplex virus thymidine kinase (HSV-tk) gene has gained much interest as a suicide gene in gene therapy of malignant tumors [6–9]. In animal models, malignant tumors were successfully treated with suicide gene therapy using HSV-tk gene and ganciclovir [6], [7]. However, clinical success with this method has been limited for the treatment of human cancers [8], [9]. Therefore, an in vivo method to assess the HSV-tk enzyme activity after gene transfer is required for the optimization of gene delivery in order to achieve clinically effective gene therapy protocols. Nuclear medicine, such as positron emission tomography (PET), can provide repeated, noninvasive, and quantitative assessment of the expression of genes in tissues and organs [2,4,10–12]. In vivo imaging of gene expression was first reported by Tjuvajev et al. [13] in experimental animal models using HSV-tk as a marker gene and 2′-deoxy-2′-flouro-5-iodo-1-β-
In contrast to the mammalian kinase, which phosphorylates thymidine preferentially, HSV-tk can phosphorylate a wide range of nucleoside analogues such as acycloguanosine and uracil derivatives that are not phosphorylated efficiently by cellular kinase [1,13–18]. The HSV-tk reporter gene along with various reporter probes has been studied and is of current interest [2], [11], [14], [15]. Reporter genes can be used to assess vector targeting and the level of suicide gene (HSV-tk) expression and to quantitatively monitor the level of enzyme activity associated with gene therapy with the help of in vivo imaging probes [2], [3], [5], [10].
Radiolabeled probes for PET imaging of gene expression using HSV-tk are being developed in our laboratory [14,15,19–21]. [18F]-Labeled acycloguanosine derivatives, 9-[(3-18F-fluoro-1-hydroxy-2-propoxy)methyl]-guanine ([18F]-FHPG), and 9-(4-[18F]-fluoro-3-hydroxy-methylbutyl)guanine ([18F]-FHBG) were first synthesized and reported by us [19], [20]. Biological evaluation of these compounds revealed that FHBG is superior to FHPG as a PET imaging agent for HSV-tk gene expression [15], [21], [22]. [18F]-Labeled at the C-8 position of acyclovir (FACV), ganciclovir (FGCV), and pencyclovir (FPCV) have also been reported by others [16], [17], [23], [24]. Among these acycloguanosine analogues, FPCV has been found to be better reporter probe than FGCV [17]. In addition to the acyclonucleoside analogues, cyclic pyrimidine nucleoside derivatives such as FIAU and 2′-deoxy-2′-flouro-5-methyl-1-β-D-arabinofuranosyluracil (FMAU) have also been reported as markers for PET imaging of HSV-tk gene [4], [5], [13], [25]. Among these probes, FIAU has been shown to be superior to the acycloguanosine derivative FHPG and FHBG in one type of cell line [26]. A few other cyclic pyrimidine nucleoside analogues have also been reported as potential gene imaging agents [27–31].
The noncatabolized thymidine analogue [11C]-FMAU was first synthesized in our laboratory for development as a marker for cell proliferation by PET [32]. In preclinical studies, [11C]-FMAU was shown to be a substrate for host-tk, although the rate of monophosphorylation was less compared to thymidine [33], [34]. Most recently, we have developed a synthesis of [18F]-FMAU in high yields [35]. [18F]-FMAU (t1/2 = 110 min) has an advantage over the [11C]-labeled product (t1/2 = 20 min) in that longer imaging intervals are possible, and that the F-18 analogue can be distributed to other facilities in the local region. A recent report on [11C]-FMAU as a potential gene imaging agent [25] prompted us to investigate further the role of radiolabeled FMAU in gene imaging and compare results with FHPG and FHBG in HT-29 human colon carcinoma wild-type and transduced cells expressing the HSV-tk gene.
Materials and Methods
[14C]-FMAU, specific activity 18 mCi/mmol and radiochemical purity >99% was synthesized by Merck Research Laboratory (West Point, NY) following the method developed in our laboratory [31]. HT-29 human colon cancer cells were obtained from ATCC (Rockville, MD), transduced with the retroviral vector G1Tk1SvNa following literature methods [36], [37], and used in earlier studies [15], [21]. Nontransduced (wild type) cells were used as controls for this study.
In Vitro Studies
Cells transduced with HSV-tk gene and wild-type cells (1–2 million) were plated in triplicate to a petri dish (100 mm) and cultured in 10 ml media for 24 hr. Logarithmically growing cells were incubated with about 1 μCi of [14C]-FMAU for 1, 3, and 5 hr. Following incubation and removal of media, the cells were washed with ice-cold phosphate-buffered saline (PBS, 3 × 10 ml) and trypsinized. After neutralization with 10% fetal bovine serum (FBS) in DMEM medium (1.5 ml), cells were centrifuged for 5 min at 1000 rpm, and supernatants were discarded. Cells were resuspended in cold PBS, and an aliquot was removed for cell counting. The remaining cells were centrifuged, and the cell pellets were treated with 0.3 N perchloric acid (PCA, 2 ml). The acidic mixtures were cooled to 4°C for 10 min, centrifuged for 1 min at 14,000 rpm, and the supernatant was separated. The cell pellets were washed twice with PCA (1 ml) and centrifuged, and radioactivity in the acid-soluble fractions, washings, and cell pellets were counted in a liquid scintillation counter (Beckman LS 9000, Fullerton, CA) using the external standard method of quench correction. The number of cells was counted using a hemocytometer in the fractions of cell suspension removed from each experiment. Radioactivity in acid-soluble fraction, washings, and cell pellets were combined and normalized to activity (cpm) per million cells and averaged for three experiments. The ratio of activity uptake in transduced and wild-type cells was obtained for each experiment per time point, and an average ratio was calculated with a standard deviation.
To determine molecular distribution of radiolabel, the acid-soluble fraction was neutralized with 2.5 N KOH and analyzed by high-performance liquid chromatography (HPLC) after removing the perchlorate salt by centrifugation. HPLC was performed using a Waters Associates system (Milford, MA) equipped with Model 510 pumps, a variable wavelength UV detector operated at 254 nm, and a Partisil 10 SAX column (Whatman, NY). Elution was performed at a flow rate of 1 ml/min with a linear gradient from 100% A (10 mM potassium dihydrogen phosphate in water, pH 3.7) and 0% B (1 M potassium dihydrogen phosphate in water, pH 3.7) to 0% A and 100% B over a 15-min period. Fractions were collected at 1-min intervals up to 30 min. Radioactivity in each fraction was measured in a liquid scintillation counter and was plotted.
The acid-insoluble fractions were further analyzed for radioactivity in DNA, RNA, and protein as follows. To the cell pellet, potassium hydroxide (1 ml, 0.12 M) was added, followed by vortexing and incubation overnight at 37°C. On the next day, the suspension was cooled on ice, PCA (1 ml, 0.3 N) was added, and the suspension was kept for 10 min at cold temperature. The suspension was centrifuged at 3000 rpm for 5 min at 4°C. The supernatant containing RNA was counted on a scintillation counter. The precipitate was suspended in 1.4 N PCA (1 ml) and was heated in a water bath for 15 min at 70°C. The mixture was cooled in ice and centrifuged as above. The supernatant containing DNA and the precipitate containing protein were counted on a scintillation counter.
In Vivo Studies
In vivo biodistribution studies were conducted on tumor-bearing nude mice at 1, 2, and 5 hr postinjection. Tumors were grown in 6-week-old athymic nude mice (Harlan, Indianapolis, IN) by inoculation of 10 million cells (transduced with HSV-tk or wild type) under the skin in the left thigh region. When the tumor was about 1 cm in size, animals were used for the experiment as described below. Animal studies were performed under an approved Institutional Animal Care and Use Committee (IACUC) protocol.
Six groups of mice (n = 5 or 6 per group), three with HSV-tk--expressing tumors and three with control tumors, were injected intravenously via a tail vein with the radiotracer [14C]-FMAU (~1 μCi, 200 μl). Blood samples were collected from mice bearing HSV-tk-expressing tumor (n = 2) in capillary tubes (1–2 μl) from the contralateral tail vein after rupture with a needle. These samples were obtained at 1, 2, 5, 10, 20, 60, and 120 min postinjection. Activity in each blood sample was measured with the scintillation counter, and percent injected dose per gram was calculated and plotted. Animals were anesthetized with Nembutal (40 mg/kg) and were sacrificed at 1, 2, or 5 hr postinjection. Organs and tumors were excised and weighed. Tissue radioactivity was measured with the scintillation counter. For each mouse, [14C]-FMAU uptake was expressed both as percent injected dose per gram tissue and as tumor to organ ratios. Mean activity uptake was calculated for each time point and was compared between the control and experimental groups of animals. The statistical significance of comparison between the control and experimental groups was calculated based on a two-tailed t test.
Results
Figure 1 summarizes accumulation of [14C]-FMAU in vitro in HSV-tk-expressing and control cells. At 1, 3, and 5 hr, activity uptake in transduced cells was 2.4-fold (p < .02), 4.0-fold (p < .001), and 5.3-fold (p < .05) higher, respectively, than in the control cells. Over time, there was increasing uptake of FMAU in transduced and nontransduced cells with less accumulation observed in nontransduced cells.
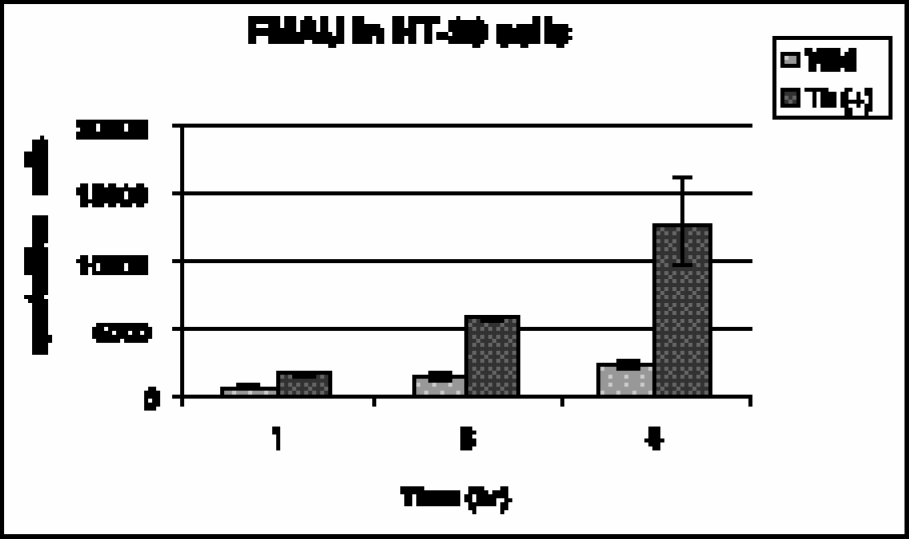
Accumulation of [14C]-FMAU in HT-29 cells, wild type and transduced with HSV-tk.
FMAU uptake in macromolecules (DNA, RNA, and protein) was approximately 88% of the total uptake in wild-type cells, and 46% in transduced cells at 3 hr. At 5 hr, these values were 93% and 60% in wild-type and transduced cells, respectively. Further analysis of this macromolecular species revealed that the [14C]-FMAU preferentially accumulated into DNA (>95%). DNA accumulation was about 2-fold higher in transduced cells compared to the wild-type cells (Figure 2).
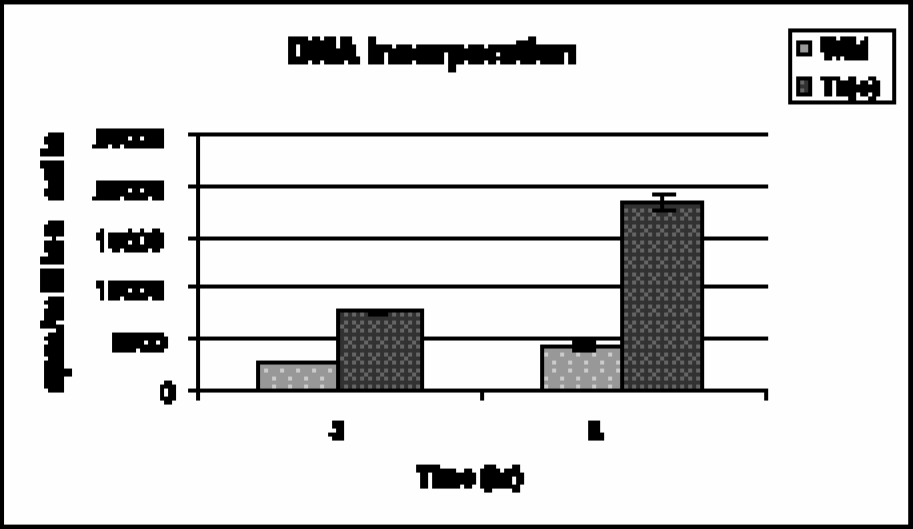
DNA incorporation of [14C]-FMAU in HT-29 cells, wild type and transduced with HSV-tk.
Figure 3 shows high-performance liquid chromatography (HPLC) chromatograms of the acid-soluble fraction from transduced and wild-type cells at 3 hr of incubation, along with a standard mixture of FMAU and thymidine phosphates. In transduced cells, four peaks were obtained at 5, 8, 11 plus 12, and 19 min corresponding to FMAU and its mono-, di-, and triphosphates, estimated as 26%, 13%, 13%, and 48%, respectively (Figure 3A). In wild-type cells, on the other hand, three peaks were obtained at 5, 11 plus 12, and 19 min corresponding to FMAU and its di- and triphosphates estimated as 19%, 26% and 55%, respectively (Figure 3B). A standard mixture of FMAU, thymidine mono-, di-, and triphosphates co-eluted with [14C]-FMAU and its mono-, di-, and triphosphates, respectively, as detected by UV (Figure 3C).

HPLC chromatograms of [14C]-FMAU metabolites and standard mixtures: (A) Radiochromatogram of acid-soluble fraction from transduced cells after 3 hr of incubation. FMAU (a), FMAU monophosphate (b), FMAU diphosphate (c), and FMAU triphosphate (d); (B) radiochromatogram of acid-soluble fraction from wild-type cells after 3 hr of incubation. FMAU (a), FMAU diphosphate (c), and FMAU triphosphate (d); (C) UV trace, mixture of cold FMAU (a), thymidine monophosphate (b), thymidine diphosphate (c), and thymidine triphosphate (d) coeluted with acid-soluble fractions of cell extract.
The blood clearance curve for [14C]-FMAU in nude mice (n = 2), uncorrected for metabolites, is shown in Figure 4. Radioactivity cleared from the blood rapidly in an exponential manner. The peak radioactivity was seen at 1 min (~40% ID/g) following tail vein injection, after which a rapid decrease was observed, followed by a very slow clearance after 20 min. The plasma half-life (t1/2) of the tracer in nude mice was calculated from the clearance curve to be less than 10 min. About 4% of the injected dose remained in the blood at 1 hr, which subsequently cleared very slowly.

Blood clearance curve from tumor-bearing nude mice carrying HSV-tk gene, average of 2 animals ± SD.
Tables 1, 2, and 3 summarize the biodistribution of [14C]-FMAU in tumor-bearing nude mice at 1, 2, and 5 hr postinjection, respectively. At 1 hr, activity uptake in transduced tumors was 8.86 ± 1.16, more than 2-fold higher than the control tumors (p < .001). Activity in the blood and other organs was comparable in both control and experimental animals, and was quite high. The only difference in the radiotracer accumulation was observed in tumors, indicating that HSV-tk–expressing cells have 2-fold higher uptake than wild-type tumors lacking HSV-tk gene. From the blood clearance curve, it has been observed that approximately 4% of the circulating activity remains in the blood at 1 hr postinjection. Tumor/blood ratio at this time was less than 1 in control animals; however, it was two times higher in experimental animals.
Biodistribution of [14C]-FMAU at 1 hr in Nude Mice Bearing Wild-Type and Transduced Tumors

Results are average of 5 animals ± SD
Biodistribution of [14C]-FMAU at 2 hr in Nude Mice Bearing Wild-Type and Transduced Tumors

Results are average of 6 animals ± SD.
Biodistribution of [14C]-FMAU at 5 hr in Nude Mice Bearing Wild-Type and Transduced Tumors
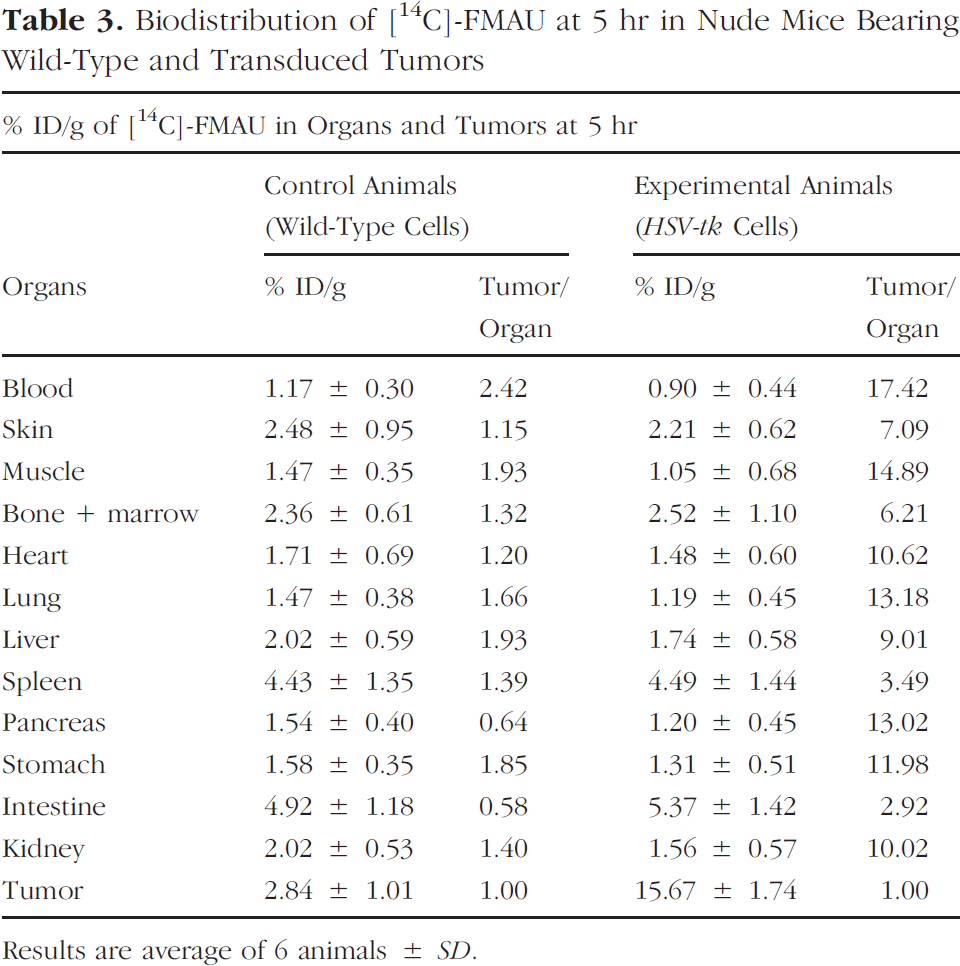
Results are average of 6 animals ± SD.
At 2 hr, activity uptake in transduced tumors was 11.23 ± 2.46, 3-fold higher than in wild-type tumors (p < .001) (Table 2). By 2 hr, the activity in the blood was much less than that at 1 hr similar to that observed in the clearance curve (Figure 4). Most organs had comparable uptake of the tracer in both control and experimental animals, with tumors being the exception. Tumor/blood ratio in control animals was 2.08, whereas in the experimental animals, it was 6.41. In the experimental animals, tumor/organ ratios at 2 hr were 2- to 6-fold higher (Table 2) with a high tumor/blood ratio (6.4).
At 5 hr, activity uptake in transduced tumors was 15.67 ± 1.74, more than 5-fold higher (p < .001) than in control tumors (Table 3). By 5 hr, activity in the blood was much less than that at 1 and 2 hr. At 5 hr, most of the nonspecifically bound activity presumably is washed out leaving the phosphorylated products in tumors in addition to some free activity in the circulation. Over time, continuing phosphorylation and washout of background activity enhance tumor/background ratio. Besides the uptake in transduced tumors, spleen and intestine uptake was slightly high in both control and experimental group of animals. Tumor/blood ratio in control animals was 2.42, similar to values at 2 hr, but in the experimental animals, it was much higher (17.42).
A direct comparison of biodistribution and tumor uptake between transduced and wild-type tumors revealed that [14C]-FMAU is incorporated preferentially into transduced cells (Figure 5A and B). By 2 hr, the difference between transduced and wild-type tumors was quite significant (3:1) (Figure 5A), with an even larger difference observed at 5 hr (6:1) (Figure 5B). The low-level uptake by proliferating tissues such as spleen and intestine are recognizable at 5 hr; however, bone marrow uptake was not significantly high compared to other organs.
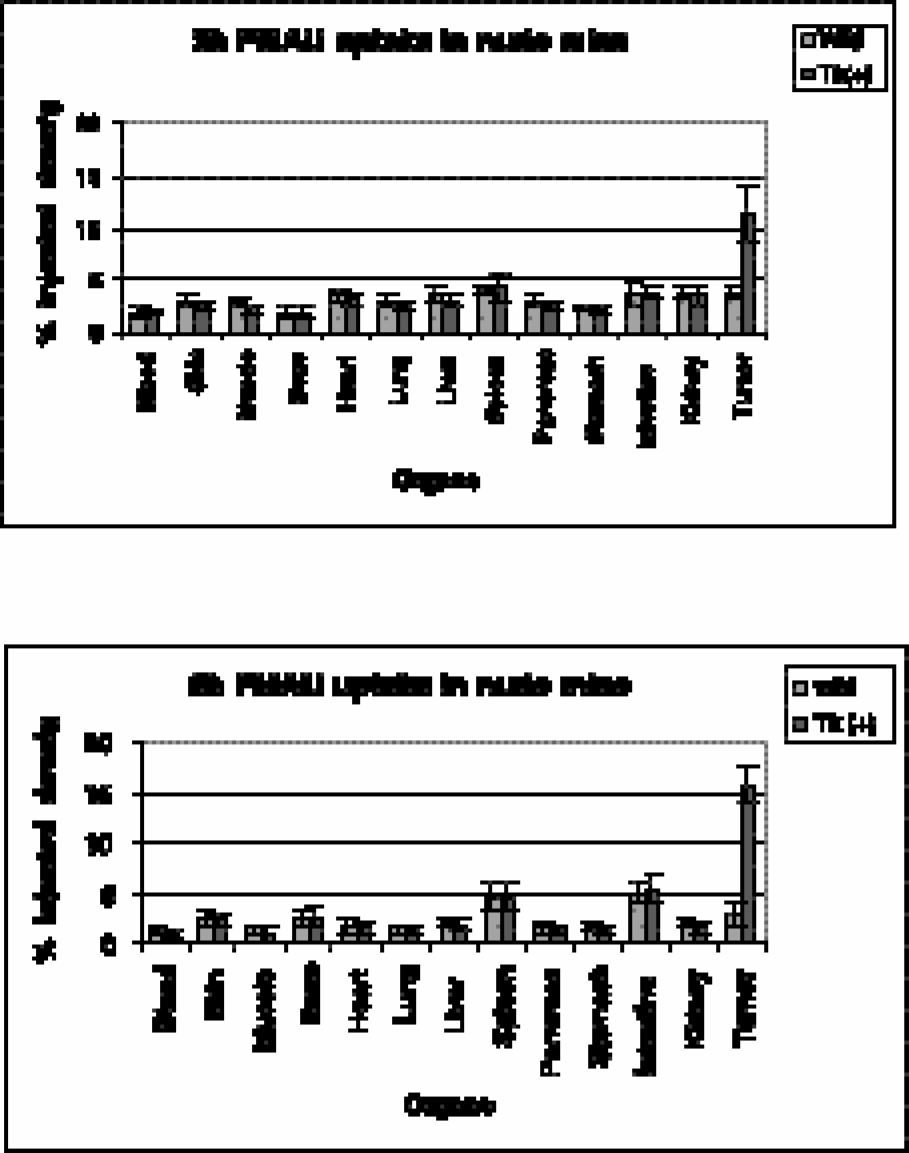
Biodistribution of [14C]-FMAU in tumor-bearing nude mice: (A) 2 hr; (B) 5 hr. Results are average of 6 animals ± SD.
In order to compare the efficacy of FMAU, FHPG, and FHBG in this cell line (HT-29), the ratios of tumor uptake in transduced versus nontransduced cells were calculated and plotted (Figure 6). At 2 hr, these ratios were 3.0, 3.3, and 4.4 for FMAU, FHPG, and FHBG, respectively. At 5 hr, these ratios were 6.1, 6.2, and 12.4 for FMAU, FHPG, and FHBG, respectively.

Comparison between FMAU, FHPG, and FHBG at 2 and 5 hr. Results are ratios of tumor uptake (transduced/wild type), average of 5 or 6 animals ± SD.
Discussion
The objective of this work was to develop a radiotracer suitable for in vivo imaging of HSV-tk expression in tumor cells using PET. FMAU was chosen due to the fact that radiolabeled uracil derivatives such as FIAU have been shown to be superior than acycloguanosine derivatives FHPG and FHBG in certain cell lines [26]. The in vitro and in vivo experiments were designed to provide a comparison between the thymidine analogue FMAU and acycloguanosine derivatives FHPG and FHBG studied earlier in the same cell lines [15], [21] in our laboratory. The results presented herein suggest that FMAU is a substrate for HSV-tk enzyme, in addition to the native host thymidine kinase, in agreement with the earlier report [39].
In vitro studies revealed the gradual increase in accumulation of [14C]-FMAU in HSV-tk-expressing cells with time indicating continuing monophosphorylation by HSV-tk enzyme (Figure 1). There is also an increasing accumulation of the tracer in nontransduced cells, suggesting that FMAU is phosphorylated by the host kinase albeit in low levels. However, accumulation of FMAU in HSV-tk-expressing cells is significantly higher (2- to 5-fold) compared to the wild-type cells.
It has been reported that the rate of phosphorylation for FMAU by mammalian thymidine kinase is approximately 10% that of thymidine in prostate tumor cells [38], therefore, rising DNA uptake in both wild-type and transduced cells was expected. In the wild-type cells, 88–93% of the activity accumulated into the macromolecules, most of which (95%) was found in the DNA pool over time (3–5 hr). Approximately 7–12% of the activity remained as free nucleotide with a small amount of the nonphosphorylated compound. In transduced cells, 46–60% of the activity accumulated in the macromolecules, approximately 95% of which enter the DNA pool. The remaining activity was found to be intracellular free nucleotide with a small amount of nonphosphorylated compound. The additional DNA incorporation seen in transduced cells (Figure 2) is due likely to augmentation of monophosphorylation by HSV-tk, which increases the concentration of nucleotide. A significant portion of activity (54% at 3 hr and 40% at 5 hr) in transduced cells appears to be the nonmacromolecular species, namely, FMAU and its mono-, di-, and triphosphates.
HPLC analysis of the acid-soluble fractions (Figure 3) revealed that wild-type cells had only the di- and triphosphate metabolites as opposed to the transduced cells, which had mono-, as well as the di- and triphosphates. All of these species co-eluted with a standard mixture of the respective thymidine phosphates. The absence of detectable levels of monophosphate metabolites in the wild-type cell supports the presumption that monophosphorylation is the rate-limiting step in the DNA synthetic pathway for FMAU. FMAU and thymidine have similar structures except for a fluorine atom at the 2′-position instead of hydrogen. In the ion-exchange column, phosphate counter parts of the molecule likely dominate the elution rather than the small difference between hydrogen and fluorine. Therefore, no significant difference in retention times was expected between the presumed FMAU phosphates and thymidine phosphate standards.
In vivo results at 2 and 5 hr revealed that tumor uptake in transduced cells are much higher than nontransduced tumor cells (Tables 2 and 3). Tumor/blood ratios in the experimental animals were 6.41 and 17.42 at these two time points, respectively. The higher uptake in transduced cells indicates continuous accumulation of FMAU in HSV-tk-expressing cells presumably due to the monophosphorylation by HSV-tk enzyme and substrate trapping. A significant accumulation of [14C]-FMAU was observed in spleen (4.49%) and intestine (5.37%), and smaller amounts in nontransduced tumors (2.84%), indicating that the radiotracer is monophosphorylated by the host enzyme, consistent with in vitro results and earlier reports [39].
Total uptake of [14C]-FMAU both in vitro (cpm/million) and in vivo (% ID/g) was much higher at 2 and 5 hr compared to results reported earlier for [18F]-FHPG and [18F]-FHBG [15], [21]. For example, [14C]-FMAU uptake at 2 hr in transduced tumors was 11.28 ± 2.46% ID/g compared to 0.12 ± 0.08% ID/g and 0.51 ± 0.25% ID/g of FHPG and FHBG, respectively. At 5 hr, FMAU uptake was 15.67 ± 1.74% ID/g compared to 0.10 ± 0.01% ID/g and 0.37 ± 0.07% ID/g for FHPG and FHBG, respectively. These results are in agreement with the in vitro data, which also show high uptake of FMAU compared to FHPG and FHBG, suggesting that FMAU is much more sensitive than the acycloguanosine derivatives in cells transduced with HSV-tk. At 2 hr, the total accumulation (% ID/g) of FMAU is approximately 94 and 22 times higher than FHPG and FHBG, respectively, in HT-29 cells. At this time point, accumulation (% ID/g) of FIAU is approximately 41 and 15 times higher than FHPG and FHBG, respectively, in RG2 tk(+) cells [26]. While this suggests that FMAU is more sensitive than FIAU, accumulation of [14C]-FMAU in nontransduced tumor was also higher as opposed to FIAU [26], FHPG, and FHBG [15], [21]. It is important to realize, however, that comparison of results from different radiotracers across cell lines is of limited value given the variability of gene expression. Ideally, paired, multitracer studies would afford more useful data.
The efficacy of FMAU, FHPG, and FHBG in this cell line (HT-29) was evaluated by plotting the ratios of tumor uptake in transduced versus nontransduced cells. Figure 6 shows that the uptake ratios between transduced and wild-type cells at 2 hr for FMAU, FHPG, and FHBG are comparable, or have insignificant differences. At 5 hr, while the uptake ratios of FMAU and FHPG are similar, uptake of FHBG is significantly higher (p < .001). Although the sensitivity of FMAU at 5 hr is approximately 100 and 25 times higher than FHPG and FHBG, respectively; its specificity remains comparable with FHPG and less than FHBG. High accumulation of FMAU has an advantage over the low uptake of the acycloguanosines, that is, the tumor should be visualized much clearly at an earlier time point. Interestingly, in vitro data, at least in the cell lines studied, appear to correlate more closely in magnitude with results obtained in in vivo studies, compared to either FHBG or FHPG [15], [21].
Although there is low-level uptake in nontransduced tumors and some proliferating tissues, FMAU labeled with a radioisotope such as 18F may be useful in imaging tumors expressing HSV-tk gene by PET. Phosphorylation of FMAU by host kinase appears higher compared to other tracers studied, thereby decreasing specificity for gene therapy imaging. This potentially could limit the role of FMAU in differentiating transduced from wild-type tumor, despite the observed higher uptake in the transduced line. At low levels of transduction, this problem could be accentuated. Therefore, additional studies in other cell lines are required to ascertain the role of FMAU in gene therapy imaging.
Conclusion
In vitro and in vivo studies indicated that [14C]-FMAU is preferentially phosphorylated by the HSV-tk enzyme and accumulates in a much higher quantity in HSV-tk-expressing cells compared to the wild-type HT-29 cells. The compound is also phosphorylated by the host kinase to a very small extent. In comparison with [18F]-FHPG and [18F]-FHBG, the sensitivity of [14C]-FMAU for identifying transduced tumors appears high, while the specificity is low compared to FHBG. These results suggest that radiolabeled FMAU, especially [18F]-FMAU, may be a suitable radiotracer for imaging HSV-tk expression, although further studies in additional tumor lines are required.
Footnotes
Abbreviations:
Acknowledgments
This work was supported in part by National Cancer Institute Grant CA 72896.
