Abstract
2′-Deoxy-2′-fluoro-5-methyl-1-β-
Introduction
Herpes simplex virus type-1 thymidine kinase (HSV1-tk) is being used as a suicide gene for gene therapy of cancer [1–9]. In animal models, malignant tumors have been successfully treated with suicide gene therapy using HSV1-tk gene and gancyclovir [6,7]. However, clinical results with this method suggest that gene delivery to the tumor cell in human is not sufficient to demonstrate efficacy [8,9]. Therefore, an in vivo method to assess the HSV1-tk enzyme activity after gene transfer may be useful for the assessment of gene delivery and establishment of treatment efficacy. Positron emission tomography (PET) can provide repeated, noninvasive, and quantitative assessment of the expression of genes in tissues and organs [2,4,10–12]. In vivo imaging of reporter gene expression was first reported in experimental animal models using HSV1-tk as a marker gene and 2′-deoxy-2′-fluoro-5-iodo-1-β-
In contrast to the mammalian kinase, which phosphorylates thymidine preferentially, HSV-tk can phosphorylate a wide range of nucleoside analogues such as acycloguanosine and 2′-deoxynucleoside of uracil derivatives that are not phosphorylated efficiently by cellular kinase [1,13–18]. We developed the radiolabeled probes 9-(4-fluoro-3-hydroxy-methyl-butyl)guanine ([18F]FHBG) and 9-[(3-fluoro-1-hydroxy-2-propoxy)-methyl]-guanine ([18F]FHPG) for PET imaging of HSV1-tk gene expression [14,15,19–21], and demonstrated that [18F]FHBG is more potent than [18F]FHPG for this purpose [20,21]. [18F]-Labeled acyclovir (FACV), gancyclovir (FGCV), and pencyclovir (FPCV) at the C-8 position have also been reported by others [17,22,23], where FPCV was found to be better reporter probe than FGCV [17].
Compared to the acycloguanosine analogues, [18F] FHPG and [18F]FHBG, pyrimidine nucleoside derivatives such as FIAU and 2′-deoxy-2′-fluoro-5-methyl-1-β-
Materials and Methods
[14C]FMAU, specific activity 5 mCi/mmol and radiochemical purity > 99% was purchased from Moravek Biochemicals (Brea, CA). [18F]FMAU, [18F]FHPG, and [18F]FHBG were synthesized following the methods developed in our laboratory [19,20,27]. Human breast cancer cells were obtainedfrom ATCC (Rockville, MD), and transduced with the retroviral vector G1Tk1SvNa with and without HSV1-tk gene following literature methods [29,30]. The transduced cells were periodically tested for HSV-tk expression and found to stable during the studies. Non-transduced (wild type) and transduced without HSV1-tk (tk-negative) cells were used as controls in this study.
In Vitro Studies
Cells transduced with HSV1-tk gene (tk-positive), without HSV1-tk gene (tk-negative) and wild-type cells (1–2 million) were plated in duplicate to a Petri dish (100 mm) and cultured in 10 mL media for 72 hr. Logarithmically growing cells were incubated with about 37 KBq (1 μCi) of [14C]FMAU or 111–148 KBq (3–4 μCi) of either [18F]FMAU, [18F]FHPG, or [18F]FHBG (sp. activity ~500 mCi/μmol) in separate experiments for various time points. Following incubation and removal of media, the cells were washed with phosphate-buffered saline (PBS, 3 × 10 mL) and treated with 0.3N perchloric (PCA, 2 mL). The acidic mixtures were cooled to 4°C for 10 min, centrifuged for 1 min at 14,000 rpm and the supernatant was separated. The cell pellets were washed twice with PCA (1 mL), centrifuged, and the acid soluble fractions, washings, and cell pellets were treated with complete counting scintillation cocktail (RIA-SOLVE-II, Research Products Intl., Mount Prospect, IL). Radioactivity was counted in a liquid scintillation counter (Beckman LS 9000, Fullerton, CA) using the external standard method of quench correction. Fluorine-18 activity was counted in a Cobra II auto gamma counter (Packard Inst., Meriden, CT). The number of cells was counted using a hemocytometer from an identical Petri dish with the same number of cells grown. Radioactivity in the acid-soluble fraction, washings, and cell pellets were combined and normalized to activity (cpm) per million cells and the mean was calculated for three experiments with standard deviation. For [14C]FMAU, the activity (cpm) was converted to dpm per million cells. Activity in the cell pellets was considered as uptake in macromolecules such as DNA and RNA. The average ratios of activity uptake between tk-positive and tk-negative cells, and tk-positive and wild-type cells were calculated with standard deviation. To determine molecular distribution of radio-label, the acid-soluble fraction was neutralized with 2.5N KOH, and analyzed by high-performance liquid chromatography (HPLC), and the acid-insoluble fractions were analyzed for radioactivity in DNA and RNA as reported earlier [24].
In Vivo Studies
In vivo studies, including biodistribution, were conducted on tumor-bearing nude mice at 1 and 2 hr postinjection. Tumors were grown in 6-week-old athymic nude mice (Harlan, Indianapolis, IN) by inoculation of approximately 4 million cells (tk-positive, tk-negative, or wild type) under the skin in the thigh regions and shoulder. When the tumor was about 1 cm in size, animals were used for the experiment as described below. Animal studies were performed under an approved Institutional Animal Care and Use Committee (IACUC) protocol.
Two groups of mice (n = 6) with wild-type tumor on the shoulder, tk-positive tumor on the right thigh and tk-negative tumor on the left thigh were injected intravenously via a tail vein with a mixture the radiotracers [14C]FMAU (−137 KBq) and [18F]FHBG (370–444 KBq). Animals were anesthetized with Nembutal (40 mg/kg) and sacrificed at 1 and 2 hr postinjection. Organs and tumors were excised and weighed. Tissue radioactivity for [18F]FHBG was measured with the gamma counter and organ samples were stored in the freezer. After 2 days, tissue samples were homogenized by a sonicator (BRAUN-SONIC-1510), mixed with the scintillation cocktail and [14C]FMAU was measured on the scintillation counter using the external standard method of quench correction. For each mouse, [14C]FMAU and [18F]FHBG uptake was expressed both as percent injected dose per gram tissue (%ID/g), and tk-positive tumor-to-organ ratio. Mean uptake (%ID/g) for each tracer was calculated with standard deviation and compared between the control and experimental tumors. Statistical significance of comparison between the control and experimental groups was based on two-tailed t test.
For PET imaging, tumors were grown by subcutaneous injection of cells, wild type on the shoulder, tk-negative on the left flank, and tk-positive on the right flank. Animals (n = 2) were injected with [18F]FMAU (~7.4 MBq, 0.2 mCi) through the tail vein, and imaging was performed using a micro-PET scanner (Concorde Microsystems, Knoxville, TN, spatial resolution of 1.2 mm). Static scans were performed at 30 min, 1 hr, and 2 hr with 10-min acquisitions. Images were reconstructed using ordered subset expectation maximization (OSEM) algorithm. On the following day, animals were injected with [18F]FHBG (~7.4 MBq, 0.2 mCi) and imaging was performed as described above. Regional tumor radioactivity concentrations (KBq/cc) were estimated from the maximum pixels within regions of interest drawn around the tumor on trans-axial slices of the reconstructed image sets. The radioactivity uptake in tumor (KBq/cc, μCi/cc) was converted to %ID/g and compared with the biodistribution data using [14C]FMAU and [18F]FHBG.
Results
Figure 1 summarizes accumulation of [1 C]FMAU in vitro in HSV1-tk-expressing and control cells. Between 30 and 240 min, activity uptake in tk-positive cells was 14-to 16-fold (p < .001) higher than tk-negative cells. At the same time points uptake in tk-positive cells was 146- to 233-fold (p < .001) higher than wild-type cells, and that in tk-negative cells was 10- to 14-fold (p < .001) higher than in the wild-type cells.
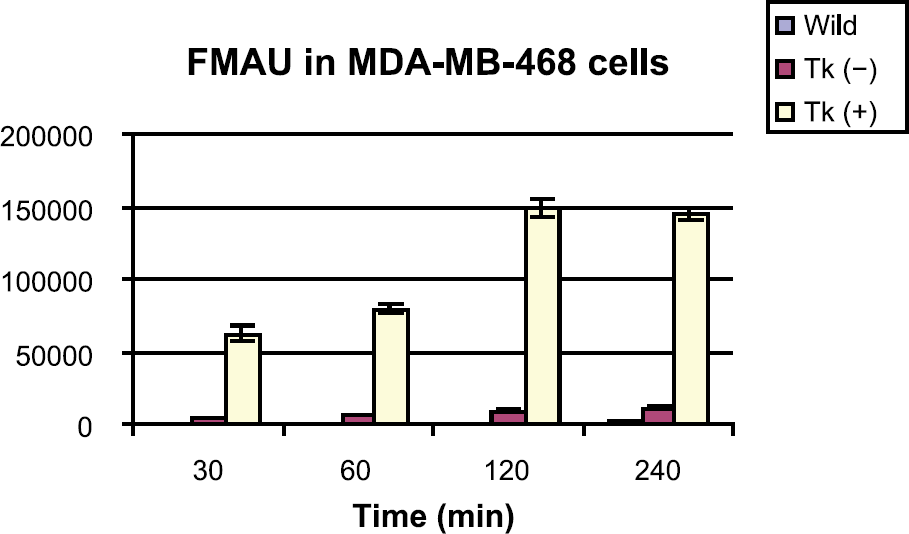
Accumulation of [14C]FMAU in MDA-MB-468 cells, wild-type, transduced with HSV1-tk and without HSV1-tk.

Accumulation of [18F]FHBG in MDA-MB-468 cells, wild-type, transduced with HSV1-tk and without HSV1-tk.
Figure 2 represents in vitro accumulation of [18F]FHBG in HSV1-tk-expressing and control cells. Activity uptake in tk-positive cells was 9- to 14-fold (p < .001) higher than in the tk-negative cells between 30 and 240 min. At the same time points, uptake in tk-positive cells was 55-to 136-fold (p < .001) higher than wild-type cells. FHBG uptake in tk-negative cells was 6- to 10-fold higher (p < .001) than in the wild-type cells.
Figure 3 summarizes accumulation of [18F]FHPG in vitro in HSV1-tk-expressing and control cells. Between 30 and 240 min, uptake in tk-positive cells was 2- to 3-fold (p < .05) higher than tk-negative cells. During the same time activity uptake in tk-positive cells was 3- to 8-fold (p < .001) higher than wild-type cells. Uptake of FHPG in tk-negative cells was one to two times higher than wild-type cells.
Figure 4 represents DNA incorporation of FMAU in wild-type and transduced cells at 1, 2, and 4 hr. DNA incorporation in tk-positive cells was 2.7-, 6.7-, and 7.1-fold higher compared to the wild-type cells at 1, 2, and 3 hr, respectively, and that in tk-negative cells was 1.5-, 3.1-, and 3.0-fold higher than wild-type cells at the same time points. Approximately 95% of total activity in the cell pellets were in DNA for all three types of cells at these time points. HPLC analysis of the acid-soluble fraction at 4 hr in the wild-type cells showed three peaks corresponding to the free compound, FMAU, and its diphosphate and triphosphate in approximately 32%, 32%, and 36%, respectively. In transduced cells, both tk-negative and tk-positive, three peaks were also observed as reported earlier [24] corresponding to mono-, di-, and triphosphates in approximately 30%, 32%, and 38%, respectively, with no free compound detected. No significant DNA incorporation of FHPG or FHBG was observed as expected.

Accumulation of [18F]FHPG in MDA-MB-468 cells, wild-type, transduced with HSV1-tk and without HSV1-tk.

DNA incorporation of [14C]FMAU in MDA-MB-468 cells, wild-type, transduced with HSV1-tk and without HSV1-tk.
Tables 1 and 2 summarize the biodistribution of [14C]FMAU and [18F]FHBG in tumor-bearing nude mice at 1 and 2 hr postinjection, respectively. At 1 hr, uptake of FMAU (%ID/g) in tk-positive, tk-negative, and wild type tumors was 13.04 ± 3.45, 3.57 ± 0.64, and 2.81 ± 0.49, respectively (Table 1). The ratio between tk-positive and tk-negative tumors was 3.7 (p < .001), and that between tk-positive and blood was 4.3 (p < .001). Uptake ratio between tk-positive tumor with other organs was in the range of 3.2–7.8. Uptake in wild-type and tk(–) tumors was similar as the blood activity. At 2 hr, uptake of FMAU (%ID/g) in tk-positive, tk-negative, and wild-type tumors were 24.5 ± 4.18, 4.72 ± 2.81, and 4.21 ± 1.11, respectively (Table 2). At this time point, uptake of FMAU was significantly higher in the tk-negative and wild-type cells compared to blood activity, presumably due to phosphorylation by the host kinase. The ratio between tk-positive and tk-negative tumors was 5.5 (p < .001), and that between tk-positive and blood was 13.4 (p < .001). Uptake ratio between tk-positive tumor and other organs was in the range of 5.5–15.6.
Biodistribution of [14C]FMAU and [18F]FHBG at 1 hr in Nude Mice Bearing Wild-Type, tk-Negative, and tk-Positive Tumors

Results are average of 5 animals ± SD.
Biodistribution of [14C]FMAU and [18F]FHBG at 2 hr in Nude Mice Bearing Wild-Type, tk-Negative, and tk-Positive Tumors
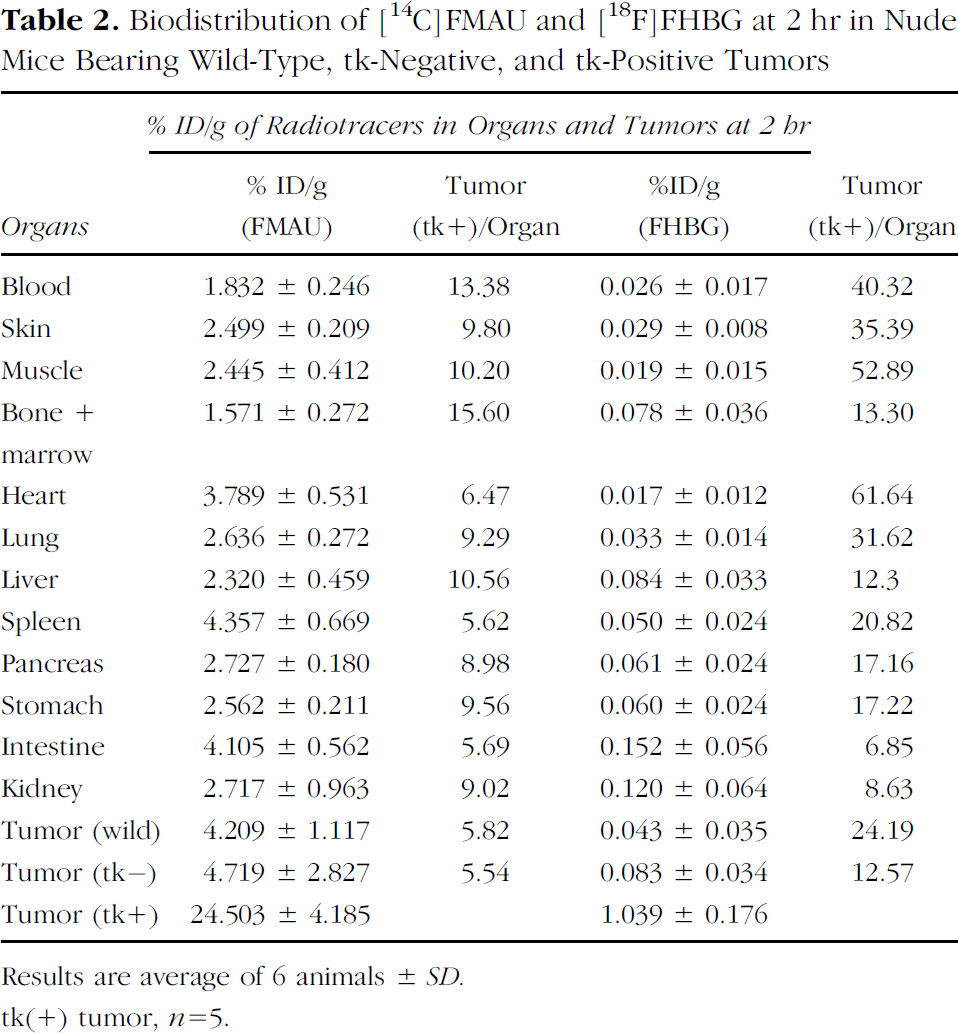
Results are average of 6 animals ± SD.
tk(+) tumor, n = 5.
Incorporation of FHBG (%ID/g) at 1 hr in tk-positive, tk-negative, and wild-type tumors were 0.78 ± 0.19, 0.18 ± 0.03, and 0.151 ± 0.03, respectively (Table 1). The ratio between tk-positive and tk-negative tumors was 4.2 (p < .001), and that between tk-positive and blood was 9.2 (p < .001). Uptake ratio between tk-positive tumors with other organs was in the range of 2.5–11.7. Uptake of FHBG (%ID/g) at 2 hr in tk-positive, tk-negative, and wild-type tumors were 1.039 ± 0.176, 0.083 ± 0.034, and 0.043 ± 0.035, respectively (Table 2). The ratio between tk-positive and tk-negative tumors was 12.6 (p < .001) and that between tk-positive and wild-type tumor was 24.2 (p < .001). Uptake ratio between tk-positive and blood was 40.3 (p < .001), and that between tk-positive and other organs was in the range of 12.3–61.6.
Micro-PET images on tumor-bearing mice using [18F]FMAU is shown in Figure 5 (A = 1 hr coronal, B = 1 hr projection image). Tumors were grown with MDA-MB-468 cells, wild type on the shoulder, tk-negative on the left flank, and transduced tk-positive cells on the right flank. As the images show, [18F]FMAU accumulates primarily in tk-positive tumor on the right flank, consistent with the biodistribution results. All other organs demonstrated low accumulation except the bladder. Uptake in wild-type tumor and tk-negative tumor was as low as the background activity by 1 hr. A high-level activity in the projection image taken at 1 hr (Figure 5B) as opposed to the single frame at the same time point (Figure 5A) is due to urine in the bladder.
Micro-PET images on tumor-bearing mice (same group) using [18F]FHBG is shown in Figure 6 (A = 1 hr coronal, B = 1 hr projection image). As the coronal image shows, [18F]FHBG accumulates in tk-positive tumors on the right flank, consistent with the biodistribution results. Activity in the organs including intestine was extremely high, which obscure the visibility of the tk-expressing tumor on the right flank (Figure 6B). However, from the coronal image, tumor-expressing HSV-tk was visible.
A direct comparison between [14C]FMAU and [18F]FHBG in terms of sensitivity and specificity is represented in Figure 7 by plotting the %ID/g (A) and uptake ratio between tk-positive and tk-negative tumors (B). Total incorporation (%ID/g) of FMAU in tk-positive tumor was 18-fold and 24-fold higher than FHBG at 1 and 2 hr, respectively. At 1 hr, the uptake ratio between tk-positive and tk-negative tumors for FMAU was 3.7 ± 0.3, and that for FHBG was 4.3 ± 1.1. At 2 hr, these values were 5.5 ± 1.3 and 12.6 ± 2.9, respectively.
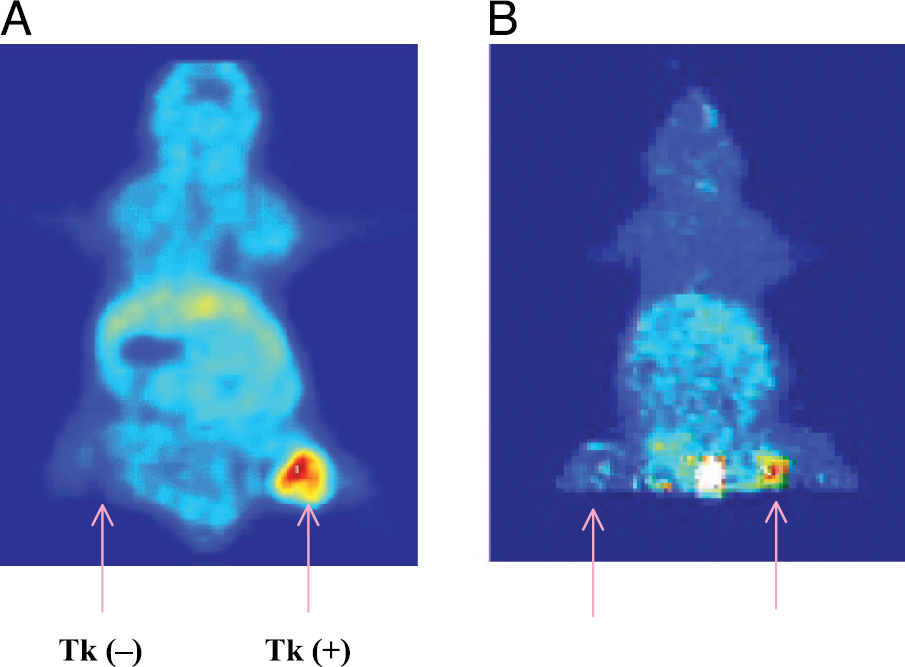
Micro-PET images of animals using [18F]FMAU. A = 1 hr coronal image; B = 1 hr projection image.
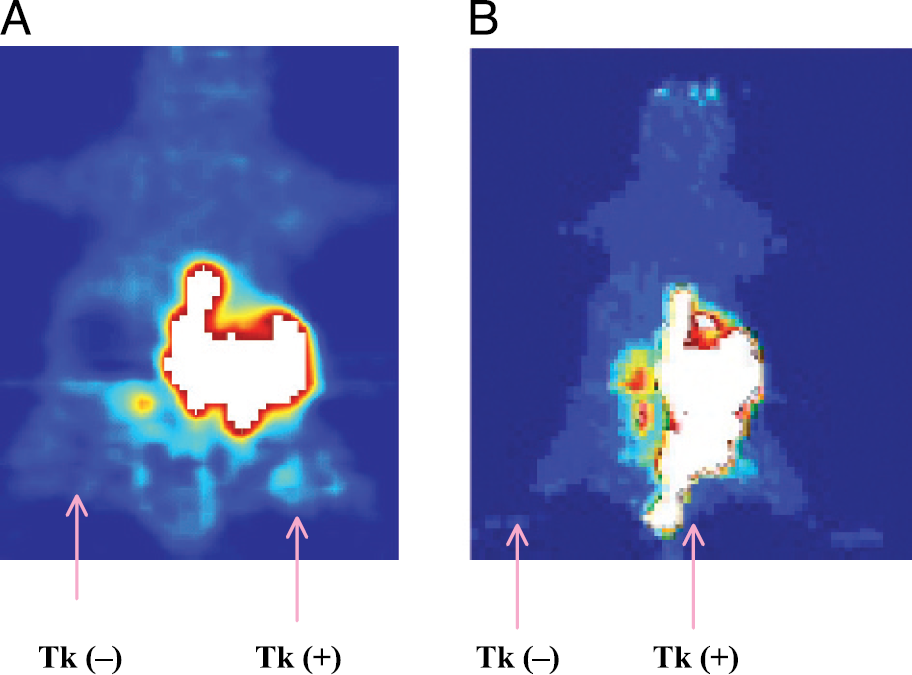
Micro-PET images of animals using [18F]FHBG. A = 1 hr coronal image; B = 1 hr projection image.
Discussion
The objective of this work was to compare representative arabinofuranosyl pyrimidine and acyclic guanosine nucleoside derivatives as potential radiotracers for in vivo imaging of HSV1-tk gene expression in human breast cancer, including an initial evaluation of the effect of cell transduction with retroviral vector on their uptake. FMAU was chosen due to the fact that radiolabeled uracil derivative FIAU has been shown to be superior to acycloguanosine derivatives FHPG and FHBG in certain cell lines [26], and it is not subject to deiodination in vivo. In earlier studies, accumulation of the tracers was compared between transduced cells and wild-type cells [2,4,17,24,25] without considering effects of transduction with the viral vectors. Identification of the most suitable radiotracer for evaluating a given cell line requires consideration of many parameters, including, but not limited to, cellular transport of the tracer, monophosphorylation by HSV1-tk, influence of nonspecific transduction with the viral vector, intracellular macromolecular distribution, as well as an understanding of the in vivo biodistribution of the tracer. In this study, we have used human breast cancer cells MDA-MB-468 transduced with HSV1-tk and without HSV1-tk, as well as wild-type cells. The in vitro and in vivo experiments were designed to provide a direct comparison between the arabinofuranosyluracil derivative FMAU and acycloguanosine derivatives FHPG and FHBG in MDA-MB-468 cells. The in vitro results obtained agree with earlier studies that demonstrate that FHPG is inferior to FHBG [20,24,25], therefore, animal studies were performed with only FMAU and FHBG for direct comparison. In addition, based on the in vitro observation that FMAU uptake in tk-positive cells was very fast, and plateaus after 2 hr, we chose to conduct the animal biodistribution studies at 1 and 2 hr postinjection, and micro-PET imaging within that time frame.

Comparison between FMAU and FHBG. Results are average ratio of tumor uptake (tk-positive/tk-negative), average of 5 ± SD.
In vitro results revealed that accumulation of all three compounds in HSV1-tk-expressing cells with time was much higher compared to the transduced tk-negative cells and wild-type cells (Figures 1–3), suggesting a continuing monophosphorylation of the tracers by HSV1-tk over time except FMAU (Figure 1). Interestingly, uptake of these compounds in tk-negative cells was also much higher than in wild-type cells. For example, the average ratio of FMAU uptake between tk-positive and tk-negative cells was 14 ± 2, and that between tk-negative and wild-type cells was in the range of 10–14. Similarly, the average ratio of FHBG uptake in tk-positive and tk-negative cells was 11 ± 2, and that in tk-negative to wild-type cells was in the range of 6–10. Uptake of FHPG in tk-negative cells was one to two times higher than wild-type cells. It has been reported that viral infection of cells induces thymidine kinase activity [32]. Therefore, it is possible that uptake of these compounds in transduced tk-negative cells may be due to induction of host thymidine kinase by the retroviral vector itself. A recent study also showed similar results that adenoviral infection of cells enhances uptake of FHBG and FIAU [16]. Higher uptake of these radiotracers in transduced tk-negative cells compared to the wild type also may be due in part to altered nucleoside transport system kinetics as a result of the transduction process. Further studies will be necessary to better understand the general effects of cell transduction with retroviral vector on transport kinetics, and monophosphorylation and cellular trapping of these nucleoside analogues.
There was a gradual increase in uptake into DNA in all three types of cells over time (Figure 4). Analysis of the macromolecular species of FMAU in cell pellets revealed that 95% of total activity in the cell pellets was in DNA at all the time points sampled. Our earlier observations suggest that monophosphorylation of FMAU by mammalian thymidine kinase does occur in proliferating cells [24,31]. Therefore, baseline DNA uptake in both wild and transduced cells was expected due to cell division. The marked additional DNA incorporation seen in tk-positive cells is likely a result of augmentation of monophosphorylation by HSV1-tk, and expansion of the radiolabeled monophosphate pool available for subsequent action by host kinases. However, the major portion of activity (98% at 1 hr and 94% at 4 hr) in tk-positive cells appears to still be the nonmacromolecular species (i.e., mono-, di-, and triphosphates), as observed by HPLC, whereas in the wild-type cells at the same time points, these values are 77% and 42%, respectively. This suggests that although DNA incorporation is increased secondary to enhanced monophosphorylation, the process for further anabolism (i.e., to the diphosphate and beyond) may be saturable in the presence of HSV-tk. Furthermore, because, in the tk-negative cells these values are also 98% and 92%, respectively, the nonspecific transduction process itself leads to augmentation of host kinase monophosphorylation activity, and influences DNA incorporation. Presumably, when cells are saturated with nucleotides, further incorporation of the compound may be reduced and eventually stopped presumably by concerted feedback inhibition mechanisms sensitive to nucleotide pool size [32]. These results suggest that further investigation is necessary to better understand the kinetics of arabinofuranosyl nucleoside incorporation in DNA in the presence of transduction. On the other hand, no significant DNA incorporation of FHBG or FHPG was observed, as was expected.
In vivo tumor uptake of FMAU (%ID/g) in tk-positive cells at 1 and 2 hr was 13.04 ± 3.45 and 24.5 ± 4.18 (Tables 1 and 2), respectively. At 1 hr, uptake in other organs including control tumors was much lower and comparable with blood except in kidney. However, spleen, intestine, kidney, and control tumors (tk-negative and wild-type) had more than 2-fold higher activity than blood at 2 hr. The ratio between tk-positive and tk-negative tumor was 3.7 and 5.5 at 1 and 2 hr, respectively, and that between tk-positive and wild-type tumor was 4.6 and 5.8. Tumor uptake in tk-negative cells was approximately 24% and 12% higher than in wild-type cells at these time points. As opposed to in vitro results, however, which showed uptake in tk-negative cells was very high compared to wild type cells, only a small difference was observed in vivo between tk-negative and wild-type cells. Incorporation of FMAU in spleen, intestine, and wild-type tumor indicates monophosphorylation of FMAU by the host kinase, consistent with the in vitro result and earlier reports [24,26].
Uptake of FHBG (%ID/g) in tk-positive tumors was 0.776 ± 0.191 and 1.034 ± 0.17 (Tables 1 and 2), and in other organs was comparable with blood except higher uptake in the intestine and kidneys. The ratio between tk-positive and tk-negative tumor was 4.2 and 12.6 at 1 and 2 hr, respectively, and that between tk-positive and wild-type tumor was 5.1 and 24.2. The tk-positive tumor/blood ratio was very high, 9.2 and 40.3 at 1 and 2 hr, respectively. There was approximately 22% higher tumor uptake in tk-negative cells compared to wild-type cells, similar as FMAU. Interestingly, the uptake of FHBG in tk-negative tumors was about 90% higher than wild-type tumors at 2 hr. Incorporation of these tracers into tk-negative cells again suggests that transduction has some influence on monophosphorylation of these compounds, and must be considered in the estimation of gene expression. Likewise, because viral transduction augments uptake of FMAU, it may be important also to consider the presence of both transduction and viral infection when using FMAU as exogenous tracer to assess cell proliferation in cancer patients.
In vitro results demonstrate that total uptake of [14C]FMAU (cpm/million cells) in tk-positive cells was much higher compared to both [18F]FHPGand [18F]FHBG (Figures 1 Figures 2 Figures 3). In vivo comparison between FMAU and FHBG (Tables 1 and 2) revealed that FMAU uptake in all organs including blood and tumors was also much higher compared to FHBG. This large difference in vivo is presumably due to two factors: (a) slower blood clearance of FMAU, and (b) the higher rate of monophosphorylation of FMAU. FMAU has a longer circulation time [24], and therefore, more likely to be transported into cells and phosphorylated by the HSV1-tk, resulting in much higher net accumulation. The higher rate of monophosphorylation also provides a greater chance for DNA incorporation of FMAU. Therefore, combining the effects of both longer blood circulation and higher monophosphorylation rate results in a significant difference in radiotracer uptake and cellular distribution.
Micro-PET images on tumor-bearing mice using [18F]FMAU (Figure 5) and [18F]FHBG (Figure 6A) also show that both [18F]FMAU and [18F]FHBG accumulate in tk-positive tumor, consistent with the biodistribution results. However, images with [18F]FMAU showed high uptake in tk-positive tumors, without any significant uptake in the control tumors and low background uptake in normal organs. As the biodistribution results show, the total accumulation (%ID/g) of FMAU was 18.5, and 24 times higher than FHBG at 1 and 2 hr, respectively. The disadvantage with FHBG is the high uptake by the intestine and organs, which limits visualization of the HSV-tk-expressing tumor. Combining data from biodistribution and micro-PET images, FMAU appears to be superior to FHBG for imaging gene expression in vivo at early time points when insignificant nonspecific accumulation of FMAU occurs.
In order to compare the efficacy of FMAU and FHBG in this cell line (MDA-MB-468), the %ID/g in tk-positive tumor and ratios of tumor uptake between tk-positive and tk-negative cells were calculated and plotted (Figure 7). At 1 hr, the %ID/g for FMAU was 18-fold higher than FHBG (Figure 7A), and uptake ratios between tk-positive and tk-negative tumors for FMAU and FHBG were comparable, 3.7 and 4.2, respectively (Figure 7B). At 2 hr, however, these ratios are significantly different; FHBG had 2-fold higher (p < .001) specificity than FMAU, although %ID/g for FMAU was 24-fold higher. After 1 hr, specificity of FMAU decreases due to the fact that FMAU uptake in tk-negative cells continues to rise over time, while in tk-positive cells little or no incorporation takes place. As noted above, negative feedback inhibition of cell transport of FMAU may explain declining uptake into tk-positive cells at later time points. Thus, at the early time point, total uptake of FMAU is significantly higher than FHBG with comparable specificity (ratio of tk-positive/tk-negative tumors). By 2 hr, however, specificity is superior with FHBG.
Compared to our earlier studies with FMAU, FHBG, and FHPG in HT-29 cells [24], total uptake of these compounds in HSV1-tk-expressing MDA-MB-468 cells was 2-fold higher. The difference in uptake between these two types of cells may be due to cellular transport or level of enzyme expression. Further studies will be necessary to explain this difference in incorporation of these compounds between HT-29 and MDA-MB-468 cells.
Conclusion
In vitro and in vivo studies indicated that [14C]FMAU, [18F]FHBG and [18F]FHPG are all readily phosphorylated by the HSV1-tk and accumulate in a much higher quantity in HSV1-tk-expressing MDA-MB-468 cells compared to wild-type cells. Although significant baseline activity was anticipated with FMAU because of its known phosphorylation by host tk and incorporation into DNA of proliferating cells, this was overshadowed by the higher accumulation in vitro and in vivo in transfected cells due to the presence of phosphorylation by viral tk, and was corroborated on micro-PET imaging of wild-type and transfected tumors. The magnitude of FMAU uptake also was significantly higher compared to the acyclic nucleosides, although at later time points, specificity (ratio tk-positive/tk-negative) was superior with FHBG. Excellent images of HSV1-tk-expressing tumors can be obtained using [18F]FMAU or [18F]FHBG, although lower nontumor background may favor FMAU depending on the location of the region of interest.
Footnotes
Acknowledgments
This work was supported by the National Cancer Institute grant CA 72896.
