Abstract
Our aim was to assess the 1-year prevalence of migraine headache in a rural population within the catchment area of the Haydom Lutheran Hospital in northern Tanzania. From December 2003 until June 2004 a community-based door-to-door survey was carried out, using a questionnaire based on the criteria of the International Headache Society, including 1192 households with 7412 individuals selected by multistage cluster-random sampling. The overall 1-year prevalence of migraine headache was found to be 4.3% [316/7412, 95% confidence interval (CI) 3.8, 4.7] with an age-adjusted rate of 6.0% and a male : female ratio of 1:2.94 (P < 0.001). Of these individuals, 132 did not fulfil all criteria for migraine headache, hence, these patients had to be classified as migrainous disorders with a crude prevalence rate of 1.8% (132/7412. 95% CI 1.5, 2.1). The remaining 184 patients met all criteria for migraine resulting in a 1-year prevalence of 2.5% (184/7412, 95% CI 2.1, 2.9) and a male : female ratio of 1:2.51 (P < 0.001). The present survey shows that migraine headache is not uncommon in northern Tanzania. The recorded prevalence of migraine headache is located within the median of previous African prevalence surveys, which confirms the trend of lower migraine frequencies in rural Africa compared with western countries.
Introduction
Prevalence of migraine in various African countries

AHC, Ad Hoc Committee Criteria; IHS, International Headache Society; comm., community-based.
With an increased knowledge of headache prevalence we can address a number of important questions about headache symptoms, which can help create preventive strategies for lowering the suffering from headache (15). Worldwide, recurrent headache is found to interfere with normal daily activities and productivity. It results in absence from work and constitutes a major health problem with significant deleterious personal, familial and social consequences (11). Headache disorders are still underestimated, under-recognized and undertreated in developed as well as developing countries. The World Health Organization (WHO) suggests that education of healthcare providers should ensure correct recognition, diagnosis and treatment of common headache disorders. Moreover, with increased knowledge of the burden of common headache disorders in the general population, the disability load can be estimated (16). Therefore, Tanzania and other developing countries require accurate headache prevalence studies with well-defined epidemiological methods to verify the existing data and to determine the impact of headache.
Several difficulties exist when comparing headache prevalence data acquired from SSA with data derived from western countries. Data concerning headache disorders from SSA are scant and, from a scientific point of view, often difficult to interpret. Differences in diagnostic criteria, variation in time periods used for prevalence estimates, methodology of data collection, population characteristics, patient selection, and the population surveyed add to difficulties in comparing the prevalence data from SSA with those from other parts of the world.
With the introduction of the International Headache Society (IHS) criteria two decades ago (17,18), the Headache Classification Committee provided new explicit diagnostic criteria for all headache disorders and new opportunities for valid epidemiological headache research. Application of the IHS criteria in recent headache surveys from SSA (2–4,12,19) has led to more reliable prevalence estimates and a more coherent picture of headaches has emerged. However, only few headache studies in SSA have used the IHS criteria and therefore little is still known about the current prevalence and distribution of headache in rural areas of the African continent.
As part of a comprehensive epidemiological investigation [see also Winkler et al. (19)], the primary objective of this study was to report the prevalence of migraine headache and its subtypes in a rural population from SSA. Further objectives were the analysis of prevalence rates by age and gender and to compare these findings with those obtained in other populations.
Subjects and methods
As the present survey is part of a comprehensive epidemiological study project, large parts of this Subjects and methods section have been previously published by our team (19). Beside headache disorders, we also investigated the prevalence of backache disorders and epilepsy.
Study site
The study area is situated in the southwest corner of the Mbulu district, in the northern highlands of Tanzania at an altitude of 1700 m (range 1300–2200 m) above sea level. The district is part of the Manyara region that is situated south of the famous Ngorongoro crater about 300 km southwest from Arusha (Figure 1). As study base we selected the local Haydom Lutheran Hospital (HLH). The total immediate catchment area of the hospital comprises 268 120 people and the total greater reference area is extrapolated to 1795 632 people (20). The nearer catchment area of HLH, comprising the two districts closest to the hospital, was chosen as study area. It has approximately 41 937 inhabitants and 6577 households (20).
The study region within its geographical context (The United Republic of Tanzania).
The study area is unique in that it includes the four main language groups of East Africa. The Iraqw (Cushitic), Datoga (Nilotic), and Iramba (Bantu) are the largest groups, with a smaller group of the indigenous Hadzabe (Khoisan) also represented. Although the main spoken language is Kiswahili, many of those who have not had schooling speak only the local languages. More than half of the population stated that they were literate and nearly two-thirds were Protestant (40% Catholics, < 1% Islamic). The Iraqw are agro-pastoralists while the Datoga traditionally have been nomadic pastoralists, though many are now settling down to begin agriculture.
Survey
A door-to-door survey was carried out (December 2003 to June 2004) using a pre-tested questionnaire in order to identify people with a history of headache. The sample comprised 7412 people of all ages, corresponding to 17.7% of the population of the study area (20). The study was designed as a cross-sectional study using multistage cluster-random sampling, which has previously been shown to be the appropriate approach to obtain an acceptable sample size in our study area (19,21). In detail, at first a simple non-random sample of 12 evenly distributed villages within the study area was chosen according to the distance from the hospital. Subsequently, within the selected villages, a simple random sample of sub-villages and 10-cells was chosen (the latter representing the clusters). A 10-cell was composed of at least 10 households under the leadership of a 10-cell leader. All people within one 10-cell were interviewed. Using this methodology, the team investigated 1192 households corresponding to 7412 individuals.
During the survey, the procedure started with informed consent of the representative heads of the families or a substitute, informing them about the voluntary character of their participation. After collection of demographic details of the family, headache screening was conducted by posing the following questions to the head of household: ‘Have you or your family member (including anyone living in the household on a permanent basis) got a headache at present?’; ‘In your (his/her) life, have you or your family member ever had a headache?’; and ‘Did you (he/she) have a headache in the previous year?’. Receiving a positive headache history from any of the family members was followed by a personal interview of the identified headache sufferer by the team. In our study, we did not a priori decide to exclude children below a certain age, as age limits deemed appropriate in the White population may not be valid for the African population.
The team and the questionnaire
The data were obtained by a team of three well-trained last-year medical students from Austria (B. S., K. K. and M. M.), three secondary school graduates from Tanzania who severed as interpreters, a local general physician (J. K.) who was employed as an assistant medical officer at a local hospital and was already very skilled in the preparation and performance of population-based studies, and a neurologist (A. S. W.) who was employed as a medical doctor at the HLH and was the initiator and coordinator of the study. Members of the investigative team were selected based upon specific personal qualities to ensure maximum subject compliance in answering the questionnaire. The presence of the three locally recruited interpreters, respected by the villagers, as well as the presence of the 10-cell leader encouraged the villagers not only to participate but to give careful consideration to all questions. Prior to survey, the study team underwent intensive theoretical and practical training about the objectives and rationale of the study. During this training a 2-week hospital-based study was performed to test the research tools and obtain feedback from this medically selected cohort. Subsequent to this tool testing, the study team performed a separate pilot study outside the study area in a village, consisting of 85 households with 387 people. Thus, the team tested the overall methodology and logistical challenges of the survey under actual field conditions and took the chance to train and improve cooperation within the team. After this extensive preliminary training the full-scale survey began.
During the study, the interpreters had to translate the interviews from English to Kiswahili. Therefore, to minimize inaccuracy in translation, misinterpretation and observer bias, the interpreters received basic training in medical terms, the conduct of field surveys and the application of the questionnaire. For this purpose, the questionnaire was translated from English into Kiswahili and the interpreters were directed to follow the exact wording of the form. During the training (hospital-based and pilot study) the interpreters were guided by a general physician experienced in field study (J. K.) who could greatly improve the interpreters' basic knowledge of the investigated disorders and medical terminology. Moreover, as the investigators were last-year medical students, misunderstood questions and terms were continuously explained in the field.
The questionnaire itself was semistructured, consisting of both open-ended and closed questions. It was tested intensively during the hospital-based study and, as Dent et al. had already employed the questionnaire in southern Tanzania (12), the present version was adopted with helpful theoretical and practical suggestions. As there were almost no patients diagnosed with migraine during the hospital-based study, resulting in an inappropriately small cohort, calculations to evaluate the sensitivity and specificity of the questionnaire were unsuccessful.
Diagnostic criteria and case definition
Analogous to the tension-type headache survey (19), subjects were diagnosed in accordance with the most frequently experienced type of headache in the previous year. Migraine diagnosis was based on IHS criteria of 1988 (17). According to these criteria, migraine patients were divided into migraine without aura, migraine with aura or migrainous disorders not fulfilling the above criteria (IHS criteria 1.7: patients fulfil all but one of the IHS criteria for migraine, and headache does not fulfil criteria for tension-type headache) (17).
After a positive headache screening, the headache questionnaire was applied to all affected patients and, at the end, medical students quoted their favoured diagnosis in accordance with the patients' symptoms. During the survey, after a day or two, all student-based established diagnoses were discussed in the team in the presence of A. S. W. Thus, all cases where there was doubt about the accuracy of the established diagnosis were personally re-evaluated by A. S. W. Cases with IHS diagnosis 1.7 were re-evaluated with special care, and therefore could be distinguished from tension-type headache with a high level of certainty.
To exclude secondary headache disorders, at the beginning of the headache questionnaire patients were asked if their ‘headache was related to any other disease?’ (e.g. malaria, diarrhoea, coughing, hypertension, poor vision, mental stress, abuse of alcohol or other drugs, malnutrition, head injury, toothache, eye/ear/sinus problems, common cold, . . . ). In a suspected case of malaria we asked for any accompanying symptoms such as fever, chills, sweats, and the course of the disease. Based on this additional information, differentiation between malaria and other primary or secondary headaches was feasible with a high degree of probability. Moreover, because of the proximity of HLH, more than a quarter of those diagnosed with malaria-related headaches had a positive blood slide.
Ethical considerations
After approval by the ethics committee of the National Institute of Medical Research and permission from the Tanzania Commission for Science and Technology in Dar es Salaam, research permits from the regional administrations and district councils were obtained. Written informed consent was obtained from the community leaders as well as from the village, sub-village and 10-cell leaders. Verbal informed consent of each head of household and all eligible individuals was obtained and information about the objective of the study and its voluntary character was given.
Statistical analysis
Patients' data was processed in Excel and for statistical analysis transferred into
Results
Study population
The investigated study population involved 7412 people living in 1192 households (for a detailed age distribution see Figure 2). This corresponds to 18.1% of all households (approximately 6577) and 17.7% of all inhabitants in the study area (approximately 41 937) (12). The initially proposed study population would have encompassed 1219 households. However, because of the absence of 27 households at three announced interview appointments, these households had to be suspended from the survey. The eligible study population was highly compliant.
Distribution of study population by gender and age.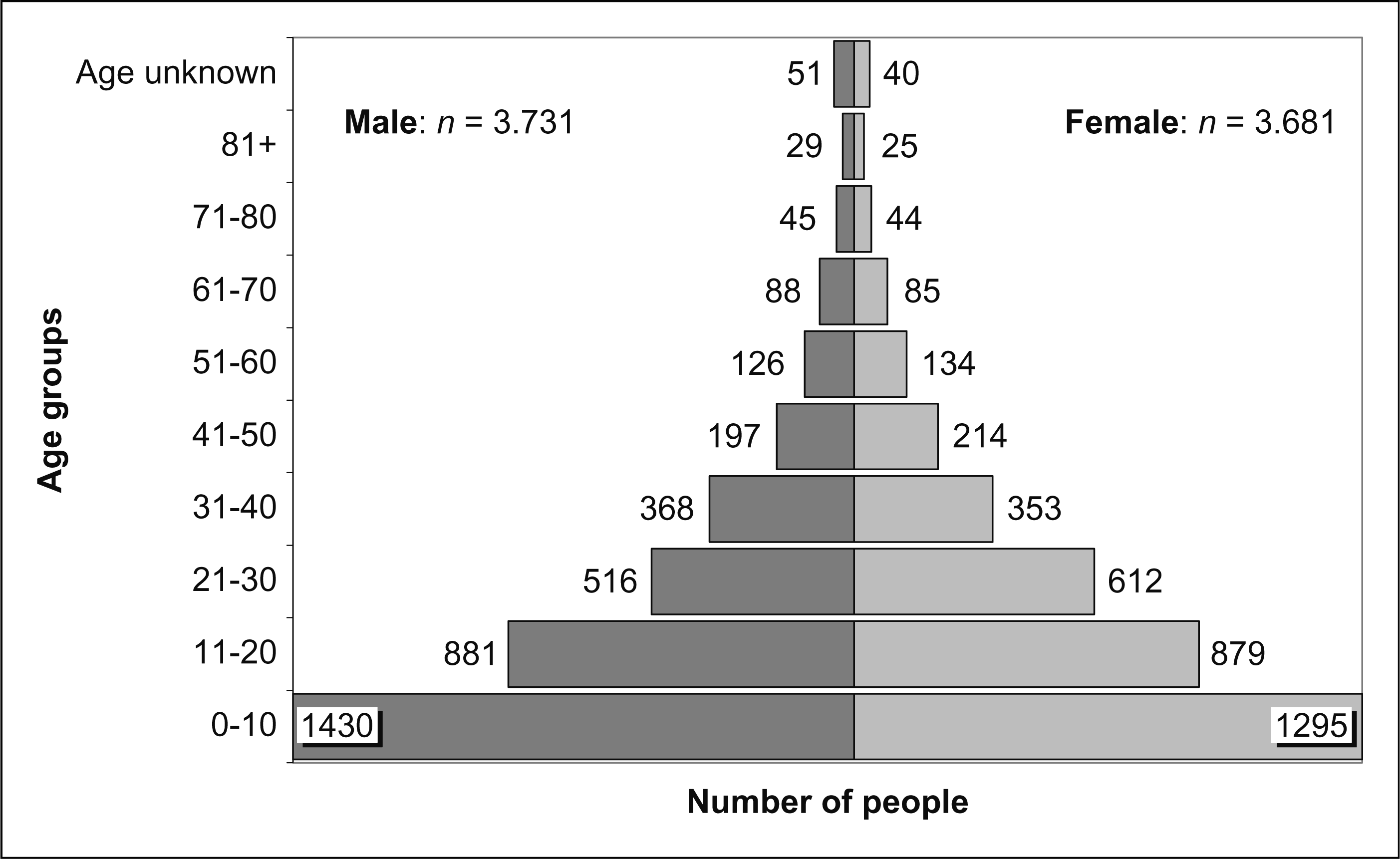
Overall migraine headache
Of the 895 people who had complained about headaches during the last year, a total of 316 respondents met the criteria of overall migraine headache, including chronic daily migraine headache and migrainous disorders. The calculated 1-year prevalence was 4.3% (316/7412, 95% CI 3.8, 4.7). The exclusion of children 0–5 years old, in whom we were unable to establish a diagnosis of headache, led to a 1-year prevalence of 5.4% (316/5858, 95% CI 4.9, 5.9). Eighty-one of the patients with migraine headache were men (2.2%, 95% CI 1.7, 2.7) and 235 women (6.4%, 95% CI 5.6, 7.2), with a male : female ratio of 1:2.94 (P < 0.001). After exclusion of seven people of unknown age, the 1-year prevalence rate of overall migraine headache was 4.2% (309/7405, 95% CI 3.7, 4.7) with an age-adjusted rate of 6.0%. As many western studies have dealt with adult populations starting between 12 and 20 years (23–30), we also calculated the crude prevalence rate for our study participants aged ≥ 21 years, which was 8.5% (250/2927, 95% CI 7.5, 9.6).
Migraine headache [according to IHS 1.1 and 1.2 (17)]
A total of 184 respondents met the case definitions for migraine headache including migraine headache with and without aura, resulting in a 1-year prevalence of 2.5% (184/7412, 95% CI 2.1, 2.9) with a male : female ratio of 1:2.51 (P < 0.001).
The age-specific prevalence peak was between 31 and 40 years with a prevalence rate of 5.7% (41/721, 95% CI 4.1, 7.6). Just as in the overall migraine headache patients, prevalence rates increased steeply from the first decade of life to the mentioned peak, with a somewhat shallower decline thereafter (Table 2). In both genders, the age-specific prevalence peak was in the fourth decade, with 8.2% (29/353, 95% CI 5.6, 11.6) in female and 3.3% (12/368, 95% CI 1.7, 5.6) in male patients (Table 2).
Number of people with migraine headache with and without aura [according to IHS 1.1 & 1.2 (7)]

Migrainous disorders [according to IHS 1.7 (17)]
Of the total 316 sufferers from migraine headache, 132 met the diagnostic criteria for ‘migrainous disorders’, which resulted in a 1-year prevalence rate of 1.8% (132/7412, 95% CI 1.5, 2.1). Twenty-eight were men (0.8%, 95% CI 0.5, 1.1) and 104 women (2.8%, 95% CI 2.3, 3.4), with a male : female ratio of 1:3.76 (P < 0.001). The peak age-specific prevalence rates irrespective of gender occurred in the fourth decade with 4.6% (33/721, 95% CI 3.2, 6.4). In women the peak was found in the fifth decade with 7.5% (16/214, 95% CI 4.3, 11.9), whereas in men it was recorded in fourth decade with 2.7% (10/368, 95% CI 1.3, 4.9) (Table 2).
Discussion
This is the second ‘community-based’ headache prevalence survey in rural Tanzania, with a special focus on migraine headache, and the first in a population of northern Tanzania. The isolated nature of the investigated area with its semi-urban and rural population structures, the low level of population fluctuation and the unadulterated origin of its residents made this region eminently suitable for the implementation of such an epidemiological survey.
Limitations of the study
Designing studies on headache in a different socio-cultural context with relatively little knowledge on the distribution of the disorder within the population may be difficult. Communication about pain and the concept of pain may not be the same as in the investigators' cultures. In this study, secondary school graduates were employed as interpreters, translating the questions from English into Kiswahili. Sometimes, especially in interviews with the elderly, questions had to be translated into tribal languages like Kiiraqw and Kidatoga, which made translation even more prone to mistakes, particularly when medical terms had to be translated. Thus, high demands were put on the interpreters. To reduce this source of mistake and to minimize possible observer bias, interpreters received specific and intensive training. Furthermore, the questionnaire was translated into Kiswahili and the translators were instructed to follow the exact wording. However, a lack in interpreters' background knowledge might even have had a positive effect, because it can be assumed that the interpreters did not influence the interviewees subjectively. Still, some bias caused by the interviewees themselves cannot be avoided.
A second element of uncertainty could be the employed sampling system. Some people became suspicious that only selected families were chosen for participation. This problem was reduced by screening the whole 10-cell and by explaining the structure of door-to-door surveys. Furthermore, the 10-cell leader, who was a person of trust for the villagers, explained the voluntary nature of this study. Therefore, after a detailed briefing, no potential study attendant refused to participate. Also, questioning about epilepsy, a stigma-associated disorder, in the same interview as for headache may have led some people to deny their headache symptoms and therefore potentially to underreporting of migraine headache. Underreporting may have also been caused by the fact that only the head of the household was interviewed with the screening questions. However, in the close confines of African life it is difficult to conceal physical illness and it can be assumed that the head of household is a reliable source of information. In the African context, putting the screening questions to each member of the family separately may even have resulted in biased information, as women, especially, are not used to discussing their health problems in public. On the other hand, with the knowledge that migraine headache also represents a familial and possibly genetic disorder, one may hypothesize that the employed sampling system with inclusion of biologically related family members may have led to an overrepresentation of affected family members. However, because of the large number of study attendants this factor can probably be ignored.
Another limitation of the study may be the fact that our survey focused only on the most frequently experienced type of headache. This implies that migraine would not be diagnosed in anyone with tension-type headache occurring more frequently than migraine. The prevalence of tension-type headache was 7% in our cohort (19). Therefore, patients suffering from a combination of migraine and tension-type headache were allocated only to one headache entity, which might be a possible explanation of a lower estimate of prevalence of migraine headache in our cohort.
Inclusion of migraine in young children may represent another drawback of our study. Choosing to investigate children < 10 years old, the age group with most people in our study population, but diagnosing relatively few cases may have had some effect on the low prevalence of migraine headache in our cohort of patients. However, excluding a priori children < 10 years old may neglect important aspects of the disorder. As in our study we were not able to identify any cases < 5 years old, we have also calculated the prevalence of migraine headache excluding the population of < 5-year-olds, resulting in a 1.1% increase of prevalence, which however is still low.
Prevalence of migraine headache in other African countries
Migraine prevalence data from SSA are still scant and the available community-based prevalence rates differ from 3% in Ethiopia (4) to 7.2% in Dar es Salaam, Tanzania (11). To put our results in the context of other published prevalence data from Tanzania, the study carried out by Dent et al. in southern Tanzania offers the best basis for comparison, because of its similarity in study design, the application of the IHS criteria for classification and its recent date (12). Dent and co-workers found a crude 1-year prevalence of migraine headache of 5.0%, which is slightly higher than the 4.3% we found. In contrast to our study population, in southern Tanzania all children < 11 years old were excluded from statistical analysis. After adjustment of our study population to the age groups investigated by Dent et al., our prevalence rate would be 6.7%, which is even higher than the findings from southern Tanzania. Matuja et al. performed a community-based survey in urban workers and students of higher education aged 20–40 years in Dar es Salaam, with a migraine prevalence of 7.2% (11). After adjustment of our study population to these age groups, the crude prevalence rate of migraine headache would be 9.0%. Contrary to the bulk of studies from western countries, where a trend of higher migraine prevalence rates has been reported for urban areas, our results do not support this finding. In contrast, in our rural population migraine headache was more prevalent compared with Matuja et al. in Dar es Salaam (11). Comparison of our prevalence data (4.3%) with other population-based studies from African countries shows slightly lower numbers in Ethiopia (3.0%) and Zimbabwe (4.2%) (4, 14) and somewhat higher prevalence rates in consecutive studies from Nigeria (5.3% in 1992, 6.3% in 1988 and 6.9% in 1982) (5, 7, 8). However, because of the application of the Ad Hoc Committee Criteria in the Nigerian studies, comparison of these results with ours may be considered invalid. In summary, those prevalence surveys of migraine headache from African countries applying the IHS criteria reported prevalence rates at a low level ranging from 3.0% in Ethiopia (4) up to 4.3% in northern and 5.0% in southern Tanzania (12).
Dent et al. reported that 1.4% of their study population in South Tanzania suffered from migraine without aura and 3.6% from migraine with aura (12). In our study, we also found more patients reporting migraine headache with aura compared with those without, but the difference between the two groups was less pronounced (1.2% without aura symptoms, 1.3% with aura symptoms). However, results from other countries show migraine headache without aura to be more common (4, 7, 14, 23–25) which may point towards a different pathomechanism in genetically diverse populations.
Prevalence of migraine headache in European and North American countries
To compare our results with data from Europe and North America, we calculated the age-adjusted prevalence rate, which was 6.0% in our population (22). In addition, as many western studies dealt with adult populations starting between 12 and 20 years (23–30). we also generated the crude prevalence rate for our study participants aged ≥ 21 years, which was 8.5%. For comparison, we considered only those western studies investigating the 1-year prevalence of migraine headache and also applying the IHS criteria for headache classification. Migraine prevalence rates from Tanzania were lower than those from European and North American studies, where prevalence rates ranging from 9.5% in the USA to 17.0% in Sweden (23–30) were reported. Moreover, even after non-consideration of children and adolescents in our survey, the prevalence rate, although increased, was still below that of western studies.
Possible causes of low migraine prevalence in countries of sub-Saharan Africa
It has not yet been clarified why prevalence rates of migraine headache in studies from SSA are consistently lower than those of western countries. Age adjustment to take into account the different population structure of African countries with a large proportion of very young people is unable to remove this difference. Differences in study design and classification systems may play an important role (31). However, even in studies conducted in Europe and the USA, higher prevalence rates of migraine headache have been found among Whites compared with Afro-Americans (23, 29, 32). There have been some attempts to explain this observation.
Headaches may be perceived differently in other cultures. Haimanot et al. assumed that villagers in an African rural setting could not easily recall vital information and events leading to a disease (33). This could be the consequence of higher pain thresholds or the fact that headache is less noticed compared with the threat of starvation and malaria, among others. In addition, unacceptability in the family and community may be a cause of migraine underreporting. Thus, awareness of migraine may be low. Also, sociodemographic and socioeconomic risk factors such as education, income, marriage, etc. have been discussed as mechanisms for the lower prevalence of migraine headache in African communities as well as in black Africans compared with Whites in western countries (29, 32, 34–37). Furthermore, several inherited factors may influence susceptibility to migraine such as variation in neurotransmitter metabolism and receptors (32). Stewart et al. suggested that the higher platelet level of the tyramine conjugating enzyme phenolsulfotransferase in African-Americans compared with Whites may protect against migraine by metabolizing suspected dietary triggers (29). In summary, environmental and dietary (38) factors, genetic differences or cultural diversity in the reporting of pain may explain the apparent difference in migraine susceptibility in African and western countries and should be topics of future research (23).
Gender-specific prevalence rates
In accordance with studies worldwide, we found higher prevalence rates of migraine headache in women (4, 5, 7, 8, 11, 12, 23–25, 27–30, 32, 34). Age-specific prevalence rates in our study in northern Tanzania were 2.2% in men and 6.4% in women, which corresponds to a male : female ratio of 1:2.94. An almost identical proportion has been found in southern Tanzania with a male : female ratio of 1:2.8 (12). This is in agreement with prevalence figures from studies worldwide, which have reported similar male : female ratios (23, 24, 27–30, 32). Reasons for female preponderance in migraine headache remain unclear. However, Rasmussen has suggested an influence of hormones as possible underlying constitutional and predisposing factors and a relationship to menarche and pregnancy (39).
Age-specific prevalence rates
Prevalence rates of migraine are usually described to increase rapidly after puberty with a gender-specific peak between 30 and 60 years old in women and 25 and 40 years old in men. After this peak, migraine prevalence decreases with age. A minimum of migraine sufferers has been reported in children and in the elderly (4, 7, 12, 23, 24, 27–29, 33). In our study, the peak prevalence rate of migraine headache was found in the fourth decade (10.3%) followed by the fifth decade (9.2%). In women the peak prevalence rate was in the fifth decade (15.0%), whereas in men it was in the fourth decade (6.0%). These results concur with international findings.
In summary, the present survey has shown that migraine headache is not uncommon in this typical east-African population. The recorded prevalence rate of migraine headache is within the range of previous findings, confirming the trend of lower prevalence rates in rural SSA compared with western countries. Because migraine headache is a major health problem constituting a significant burden to the individual and society in the affected population, efforts should be undertaken to help all affected patients. In this context, we are aware that there is a clear need to define the impact of migraine on the relevant societies—an analysis of our study in northern Tanzania in that direction is underway.
Footnotes
Acknowledgements
The authors are greatly indebted to all their local helpers, without whom this study would not have been possible, and to all participants in this study. A. S. W. was supported by The Centre for International Migration, Frankfurt, Germany.

