Abstract
Introduction:
Using functional magnetic resonance imaging (fMRI), we aimed to explore the habituation behaviour to trigemino-nociceptive as well as olfactory stimuli in migraine patients. We exclusively focussed on intrasessional behavioural rating patterns and the related blood oxygen level dependent (BOLD) signal changes.
Findings:
We observed that groups significantly differ in the time course of pain intensity ratings during the stimulation session: whereas interictal migraineurs sensitized (increasing pain ratings), control subjects habituated (decreasing pain ratings). Pain ratings of ictal patients remained unchanged. This behaviour is accompanied by a similar time course of neuronal activity in the bilateral anterior insula, in the middle cingulate cortex and in the thalamus. In these areas, the brain activity increased in migraineurs but decreased in the control group during the session. In contrast to these findings, the rating patterns for the olfactory stimuli (rose odour) did not differ between patients and controls and a gradual decrease of perceived stimulus intensity was found in all three groups. This stimulus specific response may occur because the olfactory system is the only sensory system not passing the thalamus.
Conclusion:
Our data suggest that impaired habituation in functional brain systems in migraine is fundamental only to specific modalities including the trigemino-nociceptive, but, at least, excluding the olfactory system. Our findings further suggest that there is no single neuronal modulator responsible for the altered rating pattern in migraineurs.
Keywords
Introduction
There is strong evidence that an altered cortical excitability level is fundamentally involved in the pathogenesis of migraine (1). A large number of studies have investigated the habituation behaviour and neuronal excitability in migraine patients, mainly using neurophysiological techniques including evoked- and event-related potentials and non-invasive brain stimulation techniques such as transcranial magnetic stimulation (2–4). By contrast, no imaging study has been published so far focussing on this topic.
The most consistent finding is that migraine patients with and without aura exhibit a lack of habituation to repetitive stimulation in pain-free periods (2,5,6). During acute headache attacks migraineurs display similar habituation behaviour to healthy subjects by showing a gradual decrease of perceived pain intensity during repetitive sensory stimulation (2,5,6). Deficient habituation in migraineurs during the pain-free interval has been confirmed across several sensory modalities, particularly in the visual and auditory domain (2,7,8). An excellent review regarding this consistent intrasession behaviour in migraine patients was recently published by Copolla et al. (1). In addition, a lack of habituation also has been observed in back pain patients as well as in tinnitus patients (4). However, cycling behaviour of habituation and non-habituation over a relatively short time interval (between two migraine attacks) seems to be highly specific for migraine patients (1,2,5).
A loss of habituation in interictal migraineurs during repeated sensory input has been suggested to be a key mechanism predisposing the brain to generate a migraine attack (2,5,6). The neuronal mechanisms responsible for this inability to habituate are, however, not fully understood. Dysfunctions of a neuronal inhibitory network have been extensively discussed in the past but there is an ongoing controversial debate about whether the migraineurs’ brain is hyper-excitable or hypo-responsive (2,5,6).
Evidence for impaired inhibitory mechanisms has been revealed in a previous study focussing on repetitive nociceptive stimulation over several days (9). We found that migraineurs and control subjects did not differ regarding pain ratings over several stimulation days (both groups habituated over time), but the brain activity in structures known to be involved in the endogenous pain control system increased in migraineurs and decreased in control subjects over the stimulation days (9). However, neuronal mechanisms responsible for intrasessional habituation may be completely different to those underlying between-session habituation. Thus, the present imaging study is the first one shedding light on intrasessional associations between sensory rating patterns and corresponding brain activity in migraine patients during and outside attacks. Data using the same stimulation approach but focussing on main effect differences between groups have been published elsewhere (10,11). Here, we related the intensity ratings of the two sensory modalities (pain and odour) during a single stimulation session to the cortical blood oxygen level dependent (BOLD) signal measured with means of fMRI. Based on the current literature (2,5,6), we expected that rating values would keep unchanged or even increased during the stimulation session in migraine patients between attacks (lack of habituation/ sensitization) and that ratings would decrease in patients during acute headache attacks as well as in healthy control subjects (habituation). We were further interested in possible changes of the brain activity during the session which directly correspond to the behavioural rating patterns in migraineurs and controls.
Methods
Participants
Demographical features for patients and control subjects, with clinical characteristics listed separately for the interictal and ictal migraine patients.

Attack severity: the intensity of head pain was recorded from each patient scanned during acute headache attacks by using a numerical rating scale (from ‘0’ (no pain) to ‘10’ (highest imaginable pain); mean value and range are given.
SD: standard deviation; SEM: standard error of the mean.
Migraine diagnosis was based on the classification of the International Headache Society (12). None of the patients took any migraine prophylaxis, and patients and control subjects were further instructed to take no medication up to 24 hours before and after the experiment. The interictal patients were defined as not experiencing a migraine attack at least 72 hours before and 72 hours after the experiment (10). None of the participants had a history of internal, psychiatric or neurological disease, and in particular they had no history of other headache or pain syndromes.
The participants gave written informed consent in accordance with the declaration of Helsinki and approved by the local Ethics Committee. All were informed about the purpose of the study.
Study design
The stimulation equipment and design used in the present study is described in detail elsewhere (9,13). Briefly, we used three substances which were conveyed through a thin tube placed in the subject’s right nostril: ammonia was used in order to stimulate the trigeminal nerve (=pain), rose odour was chosen in order to excite the olfactory nerve (=smell) and odourless air puffs were administered as the neutral condition to the subjects using a custom-built olfactometer. The subjects were instructed to breathe orally during the experiment to avoid fluctuations in stimulus concentrations due to respiratory airflow.
At the beginning of each trial, subjects underwent a reaction time task. Following this, each condition (ammonia, rose odour and air puffs) was presented 15 times per session in a randomized order, but the same stimulus modality could not be received more than three times in succession. The inter-stimulus interval varied between 30 and 40 seconds between two stimuli. The intensity of each stimulus was then rated on a numerical rating scale (NRS), ranging from ‘0’ (no perception) to ‘10’ (strongest imaginable pain/ odour intensity). Prior to the fMRI experiment, the subjects were trained with this paradigm outside the scanner.
Image acquisition
Echo-planar images were collected on a 3-Tesla scanner (Siemens-Trio, Erlangen, Germany) using a 12-channel head coil. Functional scans used the following parameters: 42 axial slices were acquired; voxel size = 3 mm3; gap = 1 mm, time to echo (TE) = 30 ms; repetition time (TR) = 2620 ms, flip angle 80°, field of view (FOV) 192 mm2. Additionally, high-resolution T1-weighted structural images (voxel size = 1 mm3) were acquired, using a MPRAGE sequence.
Analysis of behavioural data
Behavioural data analysis was performed using the statistic software ‘R’.
(1) Characteristics of intensity ratings (pain and odour stimuli)
Mean values and standard errors (mean ± SEM) of averaged pain and odour ratings during the stimulation session (15 nociceptive and 15 olfactory stimuli) were calculated. A normal distribution of stimulus ratings was tested using the non-parametric Kolmogorov-Smirnov test.
To assess the characteristics of stimulus perceptions for all groups we applied linear mixed effects models (LMM) to the data. Separately for each group, we determined (a) the intensity of perceptions as well as (b) the changes in ratings over time (sensitization or habituation). The estimated group-specific fixed effect parameters indicate the intra-individual dependence of pain and odour ratings on stimulation order (time point 1 to 15).
(2) Time course of intensity ratings
To test whether stimulus intensity and temporal changes of stimulus ratings differ between control subjects and migraineurs in their interictal as well as ictal state, two further LMM were computed (factor group). Similarly, to test whether migraineurs in their interictal state exhibit a different level of stimulus perception or a different temporal characteristic of stimulus ratings compared to their ictal state, we computed an additional LMM (factor state). The main effects of group or state represent different absolute levels of stimulus perception. The interaction between time point and group, as well as between time point and state, on stimulus ratings indicates differences in habituation or sensitization.
(3) Correlations between the attack distance and a) averaged pain ratings and b) the intrasessional time course of pain ratings
In order to explore whether the height of the pain intensity ratings was associated with the distance to the last or next migraine attack, we performed a correlation analyses testing for a significant correlation effect between the individual intensity ratings (=mean values from the interictal migraineurs) and the time to the last as well as to the next headache attack (=number of days).
Similarly, in order to investigate whether the time course of pain intensity ratings was associated with the distance to a migraine attack, we tested for a significant correlation between individual slopes (individual betas coefficients of the LMM) and the time to the last and next headache attack (=number of days).
The statistical threshold for significance for behavioural analyses was set to p < 0.05.
Preprocessing and statistical analysis of the functional imaging data
fMRI data were statistically analyzed using SPM 5 (statistical parametric mapping; Welcome Department for Imaging Neuroscience, London, UK). Pre-processing included slice time correction, realignment (to the first volume) and spatial normalization into the Montreal Neurological Institute (MNI) stereotactic space (14). Finally, these data were smoothed. We used a 10 mm3 full-width at half-maximum (FWHM) isotropic Gaussian kernel which might be optimal for cortical regions. Additionally, we used a 6 mm3 FWHM smoothing kernel in order to detect smaller regions, especially brainstem and midbrain structures.
Statistical data analyses were performed using the general linear model (GLM). The following types of events were modelled as delta functions convolved with a canonical haemodynamic response function with its time derivative as implemented in SPM5: (1) ‘trigemino-nociceptive stimuli’ (=ammonia), (2) ‘odour’ (=rose), (3) ‘odourless’ (=air puffs), (4) ‘reaction time task’ (onset of fixation-cross until button press after colour-changing), (5) ‘rating procedure’ (onset of presenting the NRS and duration until rating was done) and (6) movement parameters (translation and rotation in three axes).
As we were interested in correlation effects between the brain activity in response to pain stimuli and the height of the individual pain ratings, we inserted each rating value (n = 15 values corresponding to 15 pain stimuli) of all patients and controls at each time point and inserted these data into each first level model as a parametric regressor. Data were then analyzed for each subject individually and contrast images were inserted into a random effects second-level model in order to analyze group effects. Based on behavioural results demonstrating group differences (opposed rating behaviour over time) between the interictal migraine patients and the control group, we tested whether there are functional correlates underlying the rating patterns in both groups. Using a null conjunction analysis (15) including contrast images resulting from the parametric regressor, we aimed to explore whether there are brain areas in which the height of the activity level directly corresponds to the rating behaviour (=height of pain perception). As the behavioural data demonstrated that pain rating data increased in interictal migraine patients but decreased in controls, we have consequently focused on brain regions in which the BOLD response to the respective increased in the interictal migraineurs but decreased in the controls.
For the conjunction analysis we used a threshold of p < 0.005 uncorrected for whole brain comparisons.
Results
Behavioural data
(1) Mean values of intensity ratings
The non-parametric Kolmogorov-Smirnov test demonstrated that both pain and odour intensity ratings were normally distributed in each group (statistically not significant Z values). Analyses of variance (one-factorial design) showed that the three groups (controls, interictal and ictal patients) did not differ in averaged pain and odour intensity ratings. Mean values and standard errors of the mean are listed in Table 1.
(2) Time course of intensity ratings during the stimulation session and differences between the three groups
Pain
LMM showed that the pain ratings of the control group significantly decreased during the session (=habituation) (t = −2.78; p < 0.01). Interictal migraine patients demonstrated significantly increased pain ratings during the session (=sensitization) (t = 3.59; p < 0.001), whereas the ictal group showed no statistically significant changes in pain ratings over time (t = 1.26; p < 0.21).
The only group comparison that revealed a significant difference regarding pain ratings over time was the contrast between the interictal migraineurs and the control group (t = 4.37, p < 0.001) (Figure 1). No significant effects were observed between ictal patients and interictal patients, or between ictal patients and controls.
Behavioural intensity rating data (NRS) of the three groups during the stimulation session (15 stimuli of each condition). On the left side (a) mean values of pain intensity ratings (ammonia are shown, whereas odour intensity ratings (rose) are presented on the right side (b). Regression lines were overlaid for each of the three groups: the interictal migraineurs (red line), the control group (blue line) and the ictal migraineurs (green line).
Odour
LMM demonstrated that all three groups habituated in response to odour during the stimulation session (=decreased odour ratings over time) (interictal patients: t = −5.93, p < 0.001; ictal patients: t = −10.38, p < 0.001; controls: t = −13.38, p < 0.001). Groups did not differ significantly regarding time courses of odour ratings over time.
(3) Associations between the attack distance and a) averaged pain ratings and b) the intrasessional time course of pain ratings
Correlation analyses did not reveal any significant effect. Thus, there is neither a statistically significant association between (a) the attack distance and mean values of pain ratings nor between (b) the attack distance and the intrasessional time course of pain ratings.
Imaging data
In previous studies performed by our group, we published data demonstrating that several cortical and brainstem regions known from the pain-literature were active during trigemino-nociceptive stimulation in both migraine patients and healthy control subjects (13).
In the present study, we were specifically interested in which brain areas the BOLD activity directly correspond to behavioural rating values. As described above, we found significantly differences in rating patterns only for the condition pain (not for odour) and exclusively between two groups: interictal migraine patients sensitized, whereas healthy subjects habituated during the session. Consequently we focussed on neuronal correlates associated with this opposed pain rating patterns in interictal migraineurs and controls.
A null conjunction (resulting from the 10 mm smoothed data) which included the parametric contrasts from each individual (interictal patients and controls) revealed statistically significant BOLD signal changes in the middle cingular cortex (x = 9, y = 6, z = 39; t(38) = 3.02; p = 0.002 and x = −9, y = 18, z = 36; t(38) = 4.14; p < 0.001), and bilaterally in the anterior insular cortex (x = 39, y = 12, z = −15; t(38) = 2.88; p = 0.003 and x = −30, y = 18, z = −15; t(38) = 2.96; p = 0.003).
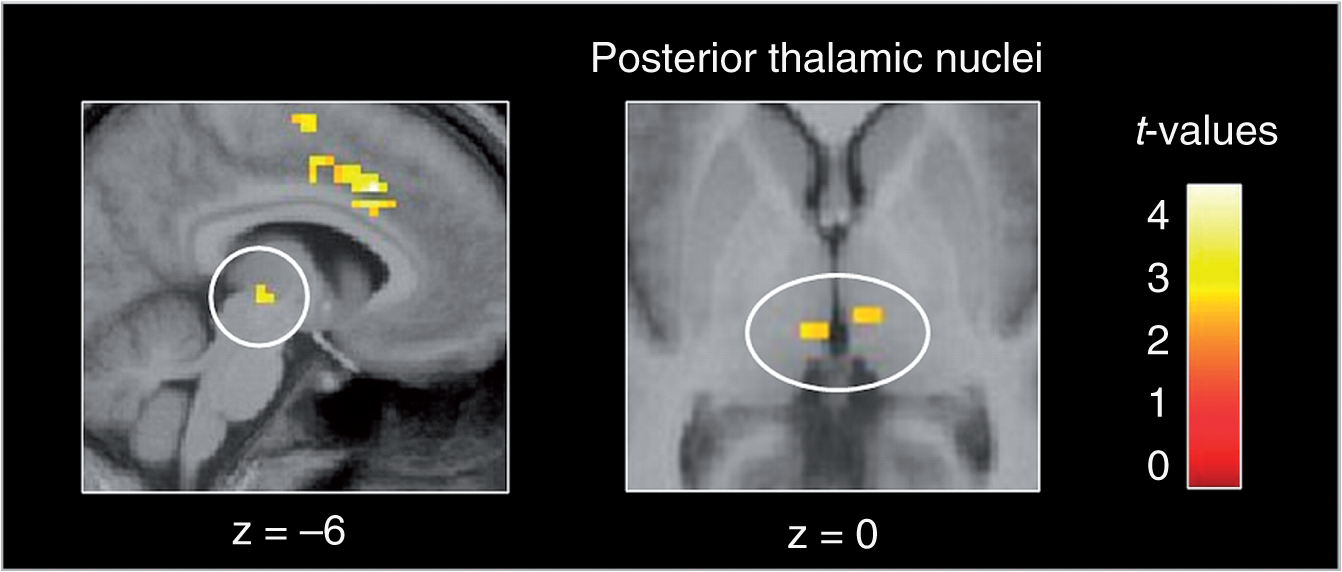
The same conjunction analysis but including the data smoothed with a smaller (6 mm) kernel replicated the cortical findings regarding the cingulate (x = 9, y = 3, z = 39; t(38) = 3.51; p = 0.001 and x = −9, y = 18, z = 36; t(38) = 4.83; p < 0.001) and the insular cortices (x = 42, y = 12, z = −15; t(38) = 2.83; p = 0.004 and x = −30, y = 18, z = −18; t(38) = 4.13; p < 0.001). Furthermore, in this analysis BOLD signal changes reached the significance threshold bilaterally in the posterior part of the thalamus (x = 6, y = −15, z = −3; t(38) = 2.95; p = 0.003 and x = −6, y = −21, z = −3; t(38) = 3.24; p = 0.001) as well as in the precentral gyrus (x = 54, y = −9, z = 42; t(38) = 2.91; p = 0.003 and x = −45, y = −15, z = 36; t(38) = 3.23; p = 0.001).
Activation patterns resulting from the 10 mm analysis are presented in Figure 2. Thalamic activity (resulting from the analysis including the data smoothed with the smaller 6 mm kernel) is shown in Figure 3. The opposite analyses (conjunctions including the contrasts testing for negative correlations) did not reveal significant results.
Part (a) shows the activation pattern resulting from a conjunction analysis, including parametric contrast images from the interictal migraine patients and the control subjects. Results are mapped onto an averaged template from the structural images of these patients and controls. An uncorrected statistical threshold of p < 0.05 was used. Part (b) demonstrates the signal intensities (y axes) during the scan session (x axes): signals were extracted out of the peak voxel of the middle cingulated cortex (MCC) at the MNI coordinate x = −9, y = −18, z = −36 from the underlying beta images of each individual at each time point (=15 pain stimuli). We then calculated mean values for the groups and plotted these data in the diagram for the interictal migraineurs (red line) and the control group (blue line). Result of the conjunction analysis including the 6 mm smoothed data: bilateral activations were observed in the posterior part of the thalamus corresponding to the pulvinar. In this brain region, the BOLD signal decreased in controls but increased in interictal migraine patients during the stimulation session (over the 15 pain stimuli). Results are mapped onto an averaged template from the structural images of these patients and controls. An uncorrected statistical threshold of p < 0.005 was used.
Discussion
The present study is the first one exploring the underpinnings of the intrasessional psychophysical behaviour during repeated trigemino-nociceptive and odour stimulation in migraine patients in and outside acute migraine attacks compared to healthy subjects.
We therefore recorded behavioural rating data as well as BOLD signal changes during the stimulation session. Our findings can be divided into three domains, namely perception of nociceptive stimulation, perception of olfactory stimulation and neuronal correlates related to these findings.
Nociceptive stimulation
As expected, we found that the interictal migraine patients and the control group significantly differ in pain ratings during the session: decreased pain intensity ratings were observed in the control group (habituation), whereas the interictal migraine patients suffered from a lack of habituation, i.e. they even demonstrated increased pain ratings during the painful stimulation session (sensitization). This is in line with earlier studies in trigeminal pain ratings (16) and several electrophysiological studies investigating contingent negative variation (CNV) (17–19) visual evoked potentials (VEP) (20,21), somatosensory evoked potentials (SEP) (22,23) and auditory evoked potentials (AEP) (7,24,25). By contrast, during acute migraine attacks patients showed unchanged pain ratings over time but did not differ to interictal migraine patients. However, contrary to the interictal patients, ictal patients also did not differ to control subjects. It is known from electrophysiological studies that cortical responses normalized just before and during attacks (19) might reflect an increase in the cortical pre-activation level due to enhanced activity in raphe-cortical serotonergic pathways (24). However, the present result of the ictal group has to be seen with caution because of the small group size. It may be that investigating more patients during head pain may have allowed identifying habituation effects over time. Increasing the number of stimuli during the stimulation session may also result in more robust statistical effects over time, but for ethical and practical reasons it is not feasible to use much more than 15 nociceptive stimuli per session.
Olfactory stimulation
The three groups (migraine patients during the attack, migraine patients in the pain-free interval and controls) did not differ in response to repeated olfactory input. Each of the three groups demonstrated a significant habituation effect, i.e. showed decreased odour intensity ratings over the 15 odour stimuli during the session (see Figure 3). We included migraine patients without any specific inclusion or exclusion criteria regarding sensitivity to odours or trigger factors. Seven out of 20 patients from the interictal group and three out of nine patients from the ictal group reported odour sensitivity during attacks, which is in line with epidemiological data regarding the incidence of odour sensitivity in migraineurs (26). It may be that specifically investigating migraineurs suffering from hypersensitivity to odours and osmophobia outside as well as during headache attacks would yield different results. However, we aimed to study patients representative for the clinical picture and not focus on specific sub-groups.
Modality-specific differences
Whereas repeated trigemino-nociceptive input resulted in a loss of habituation in interictal migraineurs, olfactory stimulation did not. Moreover, we observed differential effects: significant group differences for nociceptive perception were found between the control group and the interictal patients, but not between the controls and the ictal patients. These results are similar to those described in neurophysiological studies. The fact that all three groups did not differ in response to olfactory stimulation suggests that the normal behaviour to repetitive sensory input, that is habituation, is not altered per se in migraineurs. Of note, the olfactory system is the only sensory system not passing the thalamus. However, we have only assessed intensity ratings, it may well be that asking for the valence of the odour stimulus would have yielded other/significant results. The appendix of the IHS classification defines osmophobia (aversion against smells and odours) during attacks as a diagnostic criterion for migraine, and about 25% of the migraine patients report themselves to be hyper-sensitive to odours during acute attacks (26). Thus, the pleasant, respectively aversive quality of the received odour might be significantly different between patients during and outside headache attacks.
Our data challenge the view that a lack of habituation is indeed the general nature or biology of the central nervous system in migraineurs. However, given findings of impaired habituation behaviour not involving pain, such as the visual system in migraineurs, it is not exclusively altered with respect to nociceptive or trigemino-nociceptive input. Future studies are certainly needed but it is probably fair to say that migraine specific impaired habituation in functional brain systems is fundamental only to specific modalities including the trigemino-nociceptive, but at least excluding the olfactory system and possibly the peripheral nociceptive system (16). It is important to note that our experimental design significantly differs to those used in electrophysiological studies. Whereas neurophysiology studies (i.e. event-related and evoked potentials) use a large number of repetitive stimuli with a rather short inter-stimulus interval (milliseconds to seconds), we applied, because of a design fitted to cover BOLD requirements, 15 stimuli with a relatively long time interval that varied between 30 and 40 seconds between two stimuli. Furthermore, contrary to electrophysiological studies, we used two different sensory modalities applied in a randomized order during one session. Hence, study design and approach significantly differ to those of EEG studies. It is not clear whether the neuronal mechanisms underlying sensitization/habituation in response to short stimuli (as used in EEG studies) are comparable with those to relatively long stimuli (as used in our design) or whether these mechanisms are completely different. Furthermore, it is not known whether the application order and the use of different sensory stimuli during one session may have significant impact on the outcome. However, it is remarkable that migraine patients (and also healthy controls) demonstrate similar rating patterns for painful stimuli independently of study design and approach.
Imaging findings
The neuronal mechanisms underlying altered habituation behaviour in migraine have been mainly studied using evoked potentials and EEG, whereas functional imaging has not been used so far. We performed the stimulation program in the MRI scanner in order to shed light on neuronal mechanisms which directly correspond to behavioural rating patterns.
The BOLD responses are significantly correlated with the pain intensity ratings in migraine patients and healthy controls respectively bilateral in the middle cingulate cortex, in the anterior insula cortex and in the posterior portion of the thalamus most likely reflecting the pulvinar nuclei (see Figure 1). In these brain areas, the signal intensity decreased in healthy controls corresponding to decreased pain ratings during the scanning session (habituation), whereas the signal increased in interictal migraineurs corresponding to the increasing pain ratings over time (lack of habituation). These brain areas are known to be involved in pain processing. The cingulate cortex is one of the most frequently activated brain regions during nociceptive stimulation (27). The anterior part of the cingulum has repeatedly been shown to be included in the emotional evaluation of pain and the endogenous pain control system (28,29), whereas increased activity in its middle part probably reflects increased attention to the location of innocuous thermal stimulation (30), bodily awareness and preparation of defending action to the presented stimulus (31). The anterior part of the insular cortex is specifically linked to the affective component of pain processing (27). The inversely arranged activation levels in interictal migraine patients and controls detected in the middle cingulate and the anterior insular cortices most likely reflect the direct correlate of the height of pain perception of the individuals. The finding that pain intensity ratings additionally correlated with the activity bilaterally in the thalamus is of specific interest because the thalamus has been previously discussed to play an important role in the pathophysiology and clinical picture of migraine (22,32,33).
The thalamus is the key structure in transmitting sensory input from the brainstem to the cortex. Given that any information travelling towards the cortex must pass this gate, one could argue that whether the perception of such a signal habituates over time or not, depends on modulating effects of precisely this gate. Moreover, we have to take into account that migraine is more than just head pain (34). Patients are suffering from phobic symptoms to various sensory stimuli just before, during and shortly after headache attacks (35). Additionally, altered sensory thresholds have been discussed in interictal migraine patients (36–38). It is interesting that effective medications used as preventatives in migraine such as beta-blockers and gabapentin, seem to exhibit their effect in the thalamus (39,40). This is in line with the finding that interictal electrophysiological abnormalities normalize after treatment with propranolol and this may be associated with a decrease in headache frequency (41). Our data point towards the involvement of thalamic nuclei underlying sensitization behaviour observed in interictal migraine patients during the stimulation session. However, a very recent study pinpoints the trigemino-cervical complex, rather than the thalamus as a possible source of sensory adaptation (42). Our findings further suggest that there is no single neuronal modulator responsible for the altered rating pattern in migraineurs but rather a thalamo-cortical network including several pain transmitting cortical and subcortical areas.
In summary, the present study demonstrates that migraine patients exhibited a loss of habituation between attacks, whereas control subjects habituated in response to repeated painful stimulation but not to olfactory stimuli. Interestingly, pain ratings of migraineurs during acute headache attacks remained unchanged. Taking previous electrophysiological findings into account, the thalamus or the trigemino-cervical complex may act as possible modulators of habituation/ sensitization effects in migraine. However, because of the small sample size of ictal patients, the present results are only explorative and have to be seen with caution until replicated. Moreover, as our study is the first imaging study focussing on neuronal correlates of habituation/ sensitization behaviour, we report uncorrected findings resulting from whole brain analyses and, because of a lack of a priori hypotheses from previous imaging studies, consequently did not reduce our analyses to pre-defined regions of interest. Therefore, further studies including larger groups of patients during attacks as well as at different time points during the pain-free period are necessary for replicating the present findings and for characterizing the functional consequences of oscillating cross-talk between trigeminal and thalamic neurons in spino-thalamo-cortical control of trigemino-nociceptive input.
Clinical implications
Migraine patients sensitized (increasing pain ratings) and controls habituated (decreasing pain ratings) during the stimulation session. Correlated with this behaviour is the neuronal activity in the bilateral anterior insula, in the middle cingulate cortex and in the thalamus (increased in migraineurs but decreased in the control group during the session). Impaired habituation in functional brain systems in migraine may be fundamental only to specific modalities including the trigemino-nociceptive, but at least excluding the olfactory system. This stimulus specific response may be due to the fact that the olfactory system is the only sensory system not passing the thalamus. These data support a better understanding of neuronal correlates of dishabituation in migraine.
Footnotes
Funding
This work was supported by grants from the DFG (SFB 936-Teilprojekt A5).
Conflict of interest
None declared.
