Abstract
The objective of this study was to evaluate, in patients with migraine and healthy volunteers, with and without a history of motion sickness, the degree of discomfort elicited by drifting striped patterns. Eighteen healthy volunteers (HV) and 30 migraine patients participated in the study. Discomfort was greater in migraine patients than in HV, and in individuals with a history of motion sickness than in those without, but the effect of history of migraine was independent of history of motion sickness. Generalized Estimating Equations models for binary correlated data revealed that these differences did not depend on levels of duty cycle, spatial and temporal frequencies. Visual discomfort in migraine patients was associated with worse performance. There was a significant correlation between median degree of discomfort across conditions and number of migraine attacks in the past month. Discomfort to drifting striped patterns may be related to central sensitization in migraine patients.
Introduction
Visual processing differs in migraineurs compared with non-migraineurs (1–4). Sensitivity to static striped patterns occurs in 82% of migraine patients with or without aura, and correlates with pain frequency (5,6). In this regard, spatial frequencies of 2–8 cycles/degree (cpd) of visual angle are reported to be more uncomfortable (6). Since cortical visual neurons have orientation tuning, greater sensitivity to spatially repetitive patterns such as stripes suggests abnormal cortical visual processing (7).
Even between attacks, migraine patients report greater discomfort when observing striped or flickering patterns compared with non-migraineurs (3–6) and perform differently from non-migraineurs in motion perception tasks (8–12). One of the theories of migraine pathogenesis correlates pain and emesis to abnormal activity in brainstem nuclei modulating nociception and vascular control (13). These nuclei are believed to be involved in activation of the trigeminovascular reflex, in central sensitization (reflected, for example, in reduced habituation of event-related potentials) (14) and peripheral sensitization (evidenced by hyperalgesia of the skin of the face and scalp) (15). Additional mechanisms, such as baseline occipital hyperexcitability and decreased threshold for activation (13), or reduced sensory cortex preactivation (16), leading to decreased habituation to repetitive stimuli such as drifting stripes, may underlie greater unpleasantness reported by migraine patients compared with healthy volunteers (HV).
Migraine patients also have increased susceptibility to movement-induced and visually induced motion sickness (17). There is evidence that motion sickness and migraine are reciprocally connected. A history of motion sickness in childhood is considered a risk factor for developing migraine (18). Mechanisms underlying these phenomena are still unclear, but several hypotheses have been raised: peripheral and central vestibular abnormalities, dysfunction in multisensory integration in the brainstem, decrease in brain serotonin, or disinhibition of brainstem and gastrointestinal neurons (19,20).
Considering that migraine patients have increased sensitivity to striped patterns and to moving stimuli, we hypothesized that their responses to striped, moving stimuli would be abnormal, and would correlate with clinical features. The goals of this study were to investigate, in migraine patients and HV: (i) discomfort elicited by observing drifting striped patterns; (ii) effects of temporal frequency, spatial frequency and duty cycle on discomfort; (iii) association between degree of visual discomfort and number of migraine attacks, intensity of the last attack and duration of migraine; and (iv) performance in detection of drift direction.
Methods
Forty-eight subjects participated in the study: 18 HV and 30 migraine patients. HV 18–80 years old were included if they had no personal history of migraine, neurological or psychiatric disorders, and had no history of any headache in the past month. In the migraine patient group, those with a diagnosis of migraine with or without aura according to International Headache Society criteria (21) were included, if they had at least one migraine episode in the past month, had no history of other neurological disorders and had not used antimigraine prophylactic drugs, or psychoactive drugs, in the past month. HV were also required to be drug free. In both groups, visual acuity was at least 20/40 in both eyes and visual fields were normal. Subjects were asked whether or not they had experienced motion sickness while on a boat, car or bus; answers were recorded as ‘yes’ or ‘no’. Migraine patients were recruited in the outpatient Headache clinic and from hospital faculty members/workers. HV were either colleagues/friends of patients with migraine, or faculty members/workers from the hospital. All subjects provided written informed consent. The experimental protocol was approved by the Ethics Committee and conformed to ethical standards described in the Declaration of Helsinki.
There were 12 women in the HV group and 25 in the migraine patient group. Mean age was 33.6 ± 10.1 years in HV, and 38.7 ± 11.3 years in the migraine patient group. Of migraine patients, 33% (10/30) had migraine with aura and 67% (20/30) without aura. Median number of attacks in the past month was three (1–20) and pain intensity in the last attack, on a scale of 0–10, was 8 (3–10). Time since the last attack was 7.4 ± 1.5 days and duration of disease was 14.4 ± 2.2 years (mean ± standard error).
All participants were given full instructions before the experimental session. During the session, they were comfortably seated in a dark room and were instructed to look at the centre of a computer monitor (resolution 1024 × 768 pixels at 60 Hz) at a visual angle of 7°. Visual stimuli consisted of horizontal square wave gratings drifting up or down, within an overall circular shape presented in front of an isoluminant, black background. Images were on for 2 s at randomized intervals, alternating with a fixation cross. We used a variable interstimulus interval according to a Poisson distribution peaking at 5 s. The interval between runs was the time required to set the computer for the presentation of the next run (approximately 1 min). Stimuli were based on the static stimulus that elicited discomfort in the seminal work by Wilkins et al. (6).
We aimed at spatial and temporal frequencies in the range of previously published parameters associated with either visual discomfort, stronger Blood Oxygenation Level Dependent (BOLD) activity in functional magnetic resonance images (fMRI) effects, or both (1 cpd, 2.2 cpd, 9 Hz), and also at frequencies not previously reported to produce discomfort (10 cpd, 1 Hz, 20 Hz). When exposed to static striped patterns, migraine patients report greater distress at spatial frequencies of 1.2–8 cpd (6,22). Peak BOLD coincides with spatial frequencies (numbers of cycles of the pattern per degree of visual angle) associated with greater discomfort in migraine patients with aura (22). In HV, BOLD activation in V1 peaks at spatial frequencies of 1–4 cpd, and in V5/MT at 0.4 cpd, and is greater for moving, compared with static gratings. Activation by exposure to a unidirectional drift in a grating pattern is greatest at low spatial frequencies in V1, V2, V3 and V3AB, and at 7 cpd in V4 and V5 (23). In addition, a temporal frequency dependence of fMRI activation in cortical visual areas has been described after exposition to checkboard patterns, peaking at 6–9 Hz, compared with lower or greater frequencies (24,25). Static gratings with a duty cycle (ratio of bar width to space width) of 0.5 were reported to be more unpleasant than those with a duty cycle of 0.7 (Young, personal communication).
Number of subjects with visual discomfort in each condition—comparison between healthy volunteers (HV) and migraine patients (MP)

Number of subjects (in parentheses, %) who reported moderate to severe discomfort in each of the 10 conditions. Spatial frequency (cpd), duty cycle and temporal frequency (Hz) for each condition are shown.
χ2 or Fisher's exact tests.
One run consisted of eight stimuli drifting up and eight drifting down in a given condition. The sequence of runs was randomized across subjects. After each run, subjects were asked to grade the discomfort produced by the image as: 0, no discomfort; 1, mild discomfort; 2, moderate discomfort; 3, severe discomfort; 4, very severe discomfort, not able to look at the image at all. Grades 0 to 1 were classified as absent/mild discomfort, and 2–4 as moderate to severe.
Each experimental session lasted about 60 min. During each session, the left index was positioned on the letter ‘A’ of the computer keyboard and the right index on the letter ‘L’. Subjects were instructed to press ‘A’ when the stripes moved upwards, to press ‘L’ when they moved downwards, and to keep fingers at rest when the fixation cross was presented. Visual Studio 6.0 (Microsoft, Seattle, WA, USA) was used for presentation of visual stimuli and recording of key press responses. ‘Hit and miss’ accuracy in each run was determined.
The following end-points were evaluated: (i) differences in visual discomfort across the conditions, between HV and migraine patients, with and without a history of motion sickness; (ii) effects of duty cycle, temporal frequency, spatial frequency on visual discomfort; (iii) association between degree of visual aversion in migraine patients and migraine characteristics (number of attacks in the past month, intensity of the last attack, time from last attack, or disease duration); and (iv) differences in accuracy in key press responses (drift upwards, downwards, or rest) between HV and migraine patients.
χ2 or Fisher's exact tests, when appropriate, were
used to compare proportions of aversion (absent/mild; moderate to severe) between
groups (HV and migraine patients). Generalized Estimating Equations (GEE) models for
binary correlated data (26) were performed to evaluate effects of age, gender, history of
movement-induced motion sickness, group, temporal frequency, spatial frequency and
duty cycle on aversion. GEE is used to analyse correlated data, especially when they
are binary or in form of counts, and allows non-linear logistic regression with
repeated measures. GEE analysis was chosen to take into account the correlations
between responses in the same subject (26). Given the parameters estimates
(β) and values of independent variables (x), the probabilities of aversion
were estimated by
Spearman's ρ was used to evaluate associations between median degree of aversion across the conditions in migraine patients and: intensity of the last migraine attack; time from last attack; number of attacks in the past month; disease duration. A level of significance of 5% was chosen in all statistical analyses.
Results
There were no significant differences in age (P = 0.126) or sex (P = 0.288) between HV and migraine patients. History of motion sickness was more common in migraine patients (21/30) than in HV (7/18) (P = 0.034). Migraine attacks were not triggered by the experimental procedure in any of the subjects.
Results of binomial Generalized Estimating Equations model for visual discomfort
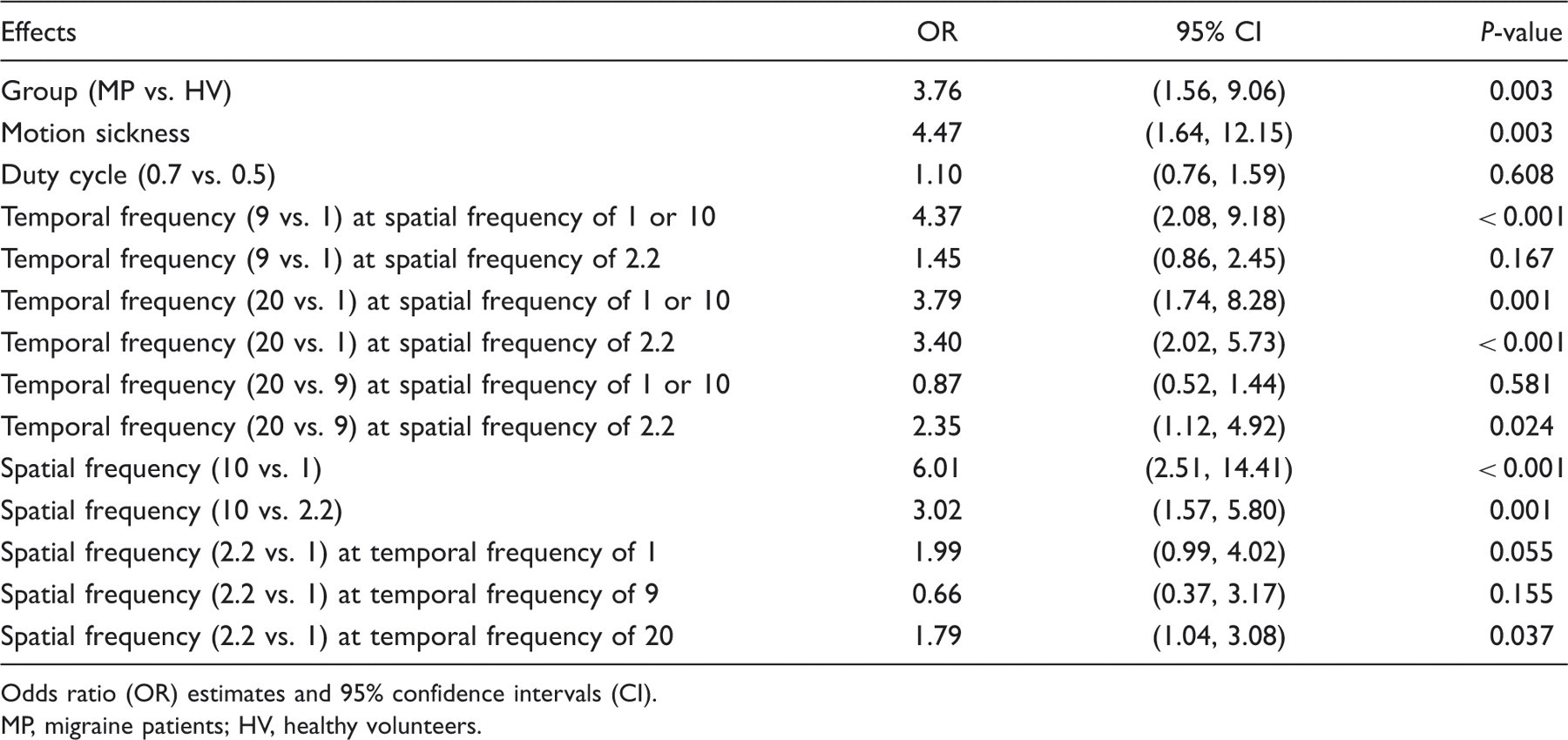
Odds ratio (OR) estimates and 95% confidence intervals (CI).
MP, migraine patients; HV, healthy volunteers.
The effects of headache diagnosis (group) and motion sickness were significant
(P < 0.01) and did not
depend on condition (levels of duty cycle, spatial and temporal frequencies) (Fig. 2). There was no
significant interaction between group and motion sickness
(P = 0.78). There were
significant interactions between spatial and temporal frequencies, i.e. the effect
of temporal frequency depended on the level of spatial frequency (Table 2). There
were no significant effects of duty cycle. Examples of visual stimuli. (A) Duty cycle, 0.7; temporal frequency,
1 Hz; spatial frequency, 1 cpd. (B) Duty cycle,
0.7; temporal frequency, 1 Hz; spatial frequency,
2.2 cpd. (C) Duty cycle, 0.7; temporal frequency,
9 Hz; spatial frequency, 2.2 cpd. The motion
version is online only (see Supporting
information). Estimated probabilities of discomfort (×100) in migraine
patients and healthy volunteers with different temporal and spatial
frequencies of the visual stimuli. Duty cycle was 0.7 in all presented
conditions. Estimates were based on the binomial Generalized Estimating
Equations model.

Figure 2 shows estimated probabilities of discomfort at a duty cycle of 0.7 for each temporal and spatial frequency. Regardless of condition, the probability of discomfort was higher in migraine patients with motion sickness and lower in HV without motion sickness. HV with motion sickness behaved similarly to migraine patients without motion sickness. At a spatial frequency of 2.2 cpd, discomfort increased with temporal frequency.
There was a significant correlation (ρ = 0.51; P = 0.004) between median degree of discomfort across conditions and number of migraine attacks in the past month. There were no significant correlations with intensity of the last migraine attack, time from last attack, or disease duration (P > 0.05). There were no significant differences between severity of aversion in patients with migraine with or without aura (P > 0.99). However, statistical power for these comparisons was limited by the number of subjects.
In two HV and in six migraine patients, responses were not registered by the computer
keyboard. Therefore, accuracy was analysed in 37 subjects. Table 3 shows the results of the
Poisson GEE model. The effect of group was significant only when associated with
moderate to severe discomfort: patients with migraine and moderate to severe visual
discomfort performed worse than those HV with discomfort
(P = 0.004). For individuals
with absent or mild discomfort, group effect was not significant
(P = 0.181). Spatial frequency
of 10 cpd and temporal frequency of 9 Hz and
20 Hz were associated with worse performance in all groups. GEE
estimates at the spatial frequency of 10 cpd are shown in Fig. 3. Estimated number of correct answers in drift direction in migraine
patients and healthy volunteers. Estimates were based on the Poisson
Generalized Estimating Equations model. Results of Poisson Generalized Estimating Equations model for the number
of correct responses Estimated effect in the mean of correct answers. Spatial frequency of 1 cpd and temporal frequency of
1 Hz were considered as reference. CI, confidence interval; HV, healthy volunteers; MP, migraine
patients.

Discussion
History of movement-induced motion sickness was more common in migraine patients, but the lack of significant interaction between group and history of movement-induced motion sickness indicates that migraine and motion sickness independently contributed to visual discomfort. Motion sickness is more common in migraine patients than in individuals with tension headache (19), but motion-induced and visually induced motion sickness are not associated in migraine patients (17), suggesting that different mechanisms underlie discomfort evoked by environmental movement and by movement of visual stimuli. Ischaemia of peripheral or central structures related to vestibular processing due to repeated migraine attacks has been hypothesized to increase susceptibility to movement-induced motion sickness in migraine patients (17) as well as abnormal gains of vestibular-ocular reflexes, abnormal central processing of vestibular information (27) and increased excitability of central gastrointestinal neurons (17).
Even though different mechanisms seem to be responsible for movement-induced and visual-induced motion sickness, increased susceptibility to these phenomena in migraine patients may share predisposing conditions. Decreased serotonergic activity has been implicated in migraine pathogenesis and is a candidate explanation for increased likelihood of movement-induced and visual-induced motion sickness in migraine patients (20). Serotonergic drugs improve emesis during motion sickness and tryptophan depletion (leading to decreased serotonin synthesis in the brain) enhances visual-induced motion sickness in HV and migraine patients (20,28). Decreased serotonin activity in the brain may make migraine patients more prone to motion sickness and to visual-induced motion sickness or discomfort, but the presented results indicate that differential processing of visual stimuli with specific psychophysical properties in migraine patients and HV plays an important, independent role on visual aversion to moving stripes.
Functional neuroimaging studies suggest that enhanced neuronal activation in the visual cortex and discomfort elicited by visual stimuli are interrelated. Peak BOLD activity in fMRI images coincides with spatial frequencies associated with greater discomfort in migraine patients with aura (22). In the present study, discomfort was influenced by the interaction of temporal and spatial frequencies. fMRI studies in healthy volunteers have shown that spatial frequencies of grating patterns and temporal frequencies of checkboard patterns influence peak BOLD activation in primary and secondary visual areas (23–25). Several authors have proposed that the temporal frequency dependence may reflect resonance of the stimulus frequency and neuronal activation with a predominant rhythm in the visual cortex (α rhythm, 8–10 Hz), leading to a maximal response (23,24,29,30). Enhanced visual activation by moving stripes may be related to greater discomfort in MP.
Previous studies have reported difference in performance in HV and migraine patients, predominantly in visual tasks related to magnocellular processing (optimal for low spatial and high temporal frequencies) (7,8,31). However, abnormalities in response to tasks that demanded either magnocellular or parvocellular processing have also been described (32), supporting non-selective visual dysfunction in migraine patients. Parvocellular processing is more related to colour and stimuli with higher spatial frequencies. Even though most of the conditions tested in the present study were expected to involve predominantly magnocellular activity, the interaction between temporal and spatial frequencies suggests that function in both pathways influenced visual discomfort.
In owl monkeys, massive glutamatergic projections to the primary visual cortex through magnocellular and parvocellular systems have been reported (33). Increased glutamatergic activity or decreased GABAergic inhibition could contribute to hyperexcitability (7,34), i.e. increased responsiveness to stimulation (7). Alternatively, decreased excitation and increased inhibition could lead to chronic hypoexcitability and less habituation to repetitive stimuli (such as moving stripes) (16) in migraine patients. A putative role of GABA/glutamate imbalance in migraine pathogenesis could be considered a potential target for therapy.
Individuals who reported moderate/severe visual discomfort and were in the migraine patient group made more mistakes than those with moderate/severe discomfort in the HV group, suggesting greater overload in processing in migraine patients, in the presence of visual discomfort. Increased discomfort may have disturbed concentration and led to lower efficiency in migraine patients. Previous studies described a link between visual aversion and performance in visual tasks. High visual aversion, in either migraine patients or HV, is associated with worse performance on visual tasks, such as those requiring discrimination among distractors in a visual array (35,36). Overload in the magnocellular system due to increased sensitivity to pattern and light, in the high visual discomfort group, may decrease efficiency in sustained attention to the target. Alternatively, deficient parvocellular processing could lead to weaker capacity to focus spatial attention (36). Temporal frequencies of 9 and 20 Hz, and spatial frequency of 10 cpd, increased task difficulty and were associated with worse performance in all groups in the present study.
A limitation of this study is lack of reaction time recording. However, it was previously demonstrated that migraine patient response times do not differ from those of HV in conscious attention tasks (36). Overall, error rate was low in both migraine patients and HV in the present study: the task was relatively simple and demanded conscious detection of drift direction of a striped pattern.
There was a significant association between number of migraine attacks in the past month and visual discomfort, similar to previous results with static striped patterns (6). In contrast, McKendrick and colleagues did not find associations between number of migraine attacks in the past year and performance in a motion direction detection task, in a different paradigm (9). Likewise, in another study, a lack of association between frequency of attacks and illusions evoked by static striped patterns was reported (37). Discomfort elicited by drifting striped patterns may be a better surrogate of clinical features, due to closer involvement of mechanisms related to migraine pathogenesis than pure motion detection tasks or visual illusions. Frequent attacks may contribute to enhance central sensitization to drifting stripes by increasing activity in brainstem nuclei projecting to the trigeminovascular reflex, and/or by changes in excitability of the visual cortex. Alternatively, drugs used for treatment of acute attacks could disrupt sensitivity to visual stimuli. Further studies should address the relation between visual discomfort and migraine chronification.
In conclusion, this study has provided novel evidence of independently increased discomfort to drifting striped patterns in migraine patients and persons with a history of motion sickness. The exact mechanisms underlying the influence of spatial and temporal frequencies are not known, but may involve abnormal sensitivity to moving stimuli by central sensitization.
Supporting information
Additional Supporting Information may be found in the online version of this article:
Please note: SAGE are not responsible for the content or functionality of any supporting materials supplied by the authors. Any queries (other than missing material) should be directed to the corresponding author for the article.
Footnotes
Acknowledgements
This study was funded by Grant 143-06, Instituto Israelita de Ensino e Pesquisa Albert Einstein/Arthur Goldlust. We thank Marta Oliveira Simões de Freitas and Marta Jardim Diniz for patient recruitment.
