Abstract
Background: Vestibular migraine (VM) has gained recognition as a distinct clinical entity in recent years. The pathophysiology is unclear. Vestibular evoked myogenic potential (VEMP) is a validated method to test the vestibulocollic reflex and peripheral vestibular hypersensitivity to noise. The aim of our study was to evaluate the vestibular function and sensitivity in a series of VM patients in comparison to ordinary migraine (M) patient and healthy controls.
Methods: Thirty-seven patients diagnosed with VM according to the Neuhauser criteria, 32 migraineurs, and 30 healthy persons underwent VEMP testing (response and sound intensity threshold) and a motion sickness susceptibility questionnaire.
Results: We found absence of unilateral or bilateral VEMP response at 90 dB normal hearing level (nHL) in 44% of the VM patients, in 25% of the migraineurs, as compared to in 3% of the healthy controls (p = 0.001). The sound intensity threshold and latencies were similar in all the groups. Migraineurs (VM > M) reported more motion sickness than healthy controls (p = 0.006).
Conclusion: The results indicate more pathology in the VEMP circuitry in migraineurs than in healthy controls. We did not find support for peripheral vestibular hypersensitivity in terms of lower VEMP threshold among VM patients, but they are more sensitive for motion triggers than other migraineurs.
Introduction
Neuhauser criteria for ‘definite migrainous vertigo’

Vestibular symptoms are rotational vertigo, another illusory self or object motion, positional vertigo, head motion intolerance, i.e. sensation of imbalance or illusory self or object motion that is provoked by head motion. The symptoms may be spontaneous or positional. Vestibular symptoms are ‘moderate’ if they interfere with, but do not prohibit, daily activities and ‘severe’ if the patients can not continue daily activities.
In general, migraineurs seem to have a global hypersensitivity to different sensory stimuli (motion, visual, auditory, and olfactory) and to vestibular testing. VM patients are even more sensitive to vestibular activation than those with other migraine forms, demonstrated by a strong emetic response during caloric testing (3) and a higher frequency of migraine attacks after vestibular testing (4).
The pathophysiology in VM is unknown. The pathology could be localized anywhere in the vestibular system; from the sensory receptor organs in the inner ear, the eight nerve and the central pathways to vestibulo-cerebellum, vestibular and oculomotor nuclei in the brainstem, thalamus, and vestibular cortex. Previous studies of VM describe a variety of abnormal vestibular findings, some indicating dysfunction in the central vestibular system and others in the peripheral vestibular system (3,5−9). The sensation of vertigo in VM patients may arise from a decreased or an increased activation of the vestibular system, either peripheral or central, generating a tonic imbalance between these structures.
The click stimulus of vestibular evoked myogenic potentials (VEMPs) elicits an inhibitory oligosynaptic brainstem reflex, the vestibulocollic reflex. Testing of VEMP can reveal functional abnormalities of the vestibular system, in particular the saccular function, the inferior vestibular nerve, the vestibular nuclei, and its pathway through the lower brainstem to the motor neurons in the sternocleidomastoid muscle (SCM). VEMP has also been useful in documenting peripheral vestibular hypersensitivity to noise in the sense of abnormally low sound intensity thresholds in persons with the Tullio phenomenon. This occurs in persons with fistula or superior semicircular canal disorders (10−12).
The aim of our study was to investigate if there were any differences in vestibular sensitivity and function in VM patients compared to ordinary migraine patients and healthy controls, and thereby to contribute to increased understanding of the pathophysiology of VM. To achieve this we assessed the saccular otolith pathway (VEMP) and measured the motion sickness susceptibility.
Subjects and methods
Subjects
The study population consisted of three groups: ‘definite’ vestibular migraine (VM) patients, ordinary migraine (M) patients and healthy controls (HC). Subjects were recruited from the outpatients clinic, Department of Neurology, Sørlandet Hospital HF, Kristiansand, Norway and Spesialistsenteret, Kristiansand, Norway, general practitioners (GPs) in the area (migraine patients without vertigo), and colleagues at the hospital (healthy controls). Recruiting period was from May 2007 to May 2009. The VM patients fulfilled the Neuhauser criteria for ‘definite’ migrainous vertigo (1,2) and were selected by a validated diagnostic interview for determination of vestibular migraine by Furman et al. (13,14). The group ‘ordinary migraine’ consisted of migraine patients who fulfilled the IHS criteria for migraine with and without aura (15). Other subtypes were not included. We chose to recruit them from GPs in order to get comparable samples with respect to migraine attack rate. In Norway, most migraine patients are treated by their GPs and are only referred in case of difficult treatable migraine or when having a complicated subtype of migraine (as vestibular type, hemiplegic type, etc.). VM patients do not necessarily have frequent or complicated migraine headache attacks, but are usually referred to the hospital because of the vertigo complaints. Triptans, antiepileptic drugs, drug prophylaxis for motion sickness, and other medication that could influence the vestibular testing results were cessated 1 week before testing. The VM and migraine patients were comparable for age, sex, and migraine type (with or without aura) and migraine attack rate. The control group consisted of 30 healthy volunteers, comparable for age and gender with the study groups. Exclusion criteria for all groups were abnormal hearing defined by audiometric testing, age ≥60 years, a history of other vestibular diseases, and chronic ear, ophthalmological or neurological disorders.
The study received prior approval from the regional ethical committee of South Norway and each subject gave informed consent to participate in accordance with the declaration of Helsinki.
Testing
VEMP recording was carried out as described by Colebatch et al. (16,17), except for local differences in the equipment used. We used a system for recording brainstem evoked responses and could therefore give air conducted click stimuli with an upper peak value at 90 dB normal hearing level (nHL), equivalent to approximately 130 dB sound pressure level (range 125–132 SPL). The clicks were monaurally delivered via earphones. Stimulus repetition rate was 5 Hz in two series of 150 rarefaction clicks. Offsets for 0 dB nHL for clicks (0.1 ms) are typically between 35–45 dB SPL. In order to test vestibular hypersensitivity to sounds, we reduced the sound intensity level in steps of 5 dB from 90 dB nHL until the VEMP response disappeared. Surface electromyogram activity was recorded using disc electrodes. The active electrode was placed at the midportion of the SCM, the reference electrode near the midportion of each clavicule and the ground electrode over sternum. The subjects lay in the supine position and were instructed to raise their heads and press it against the resistance of an assistant’s hand during testing. They got continuous mental feedback from the assistant during recording to assure adequate muscle activation of the SCMs. The testing was done by one trained investigator (MIB) in the interictal period, defined as absence of migraine headache and vertigo attacks for the last 2 days.
All patients answered a small, not validated, questionnaire about motion sickness and were asked to range how often they get sick or feel nauseated (never, rarely, sometimes, frequently, always) when exposed to different type of motion triggers (car, boat, plane, train).
Analysis of VEMP
Unrectified electromyogram responses were averaged and analogue filtered (pass band: 0.02–2 kHz). Presence of VEMP response was defined as a clearly visible, reproducible, ipsilateral positive 13−negative 23 waveform (Figure 1). Later responses were not reported, as they often are considered to be of cochlear origin. The threshold was defined as the lowest stimulus intensity capable of evoking a reproducible VEMP response on both ears. The evaluation of the VEMP results was not done blinded. The VEMP responses were fairly easy to interpret and were evaluated by two investigators (MIB and PM). Rectification of the electromyogram has mainly showed to be useful in reducing the bias for the degree of muscle contraction of the SCM. Since the rectification method is needed to do analyses on amplitude-associated parameters, we did not analyse the amplitudes and the habituation responses.
This is an example of a normal VEMP response from one of our patients.
Statistics
For comparison of unpaired categorical data, we used Pearson’s χ2 test. For continuous outcome variables, we used Student’s paired and unpaired t-tests and one-way ANOVA. When there was a significant difference between the groups, we performed post-hoc pairwise comparisons of proportions (Pearson’s χ2 test) and means (Bonferroni’s test) to investigate which groups that differed. P-values <0.05 were considered significant.
Statistical calculations were carried out using Statistical Package for Social Sciences version 16.0 (SPSS, Chicago, IL, USA).
Results
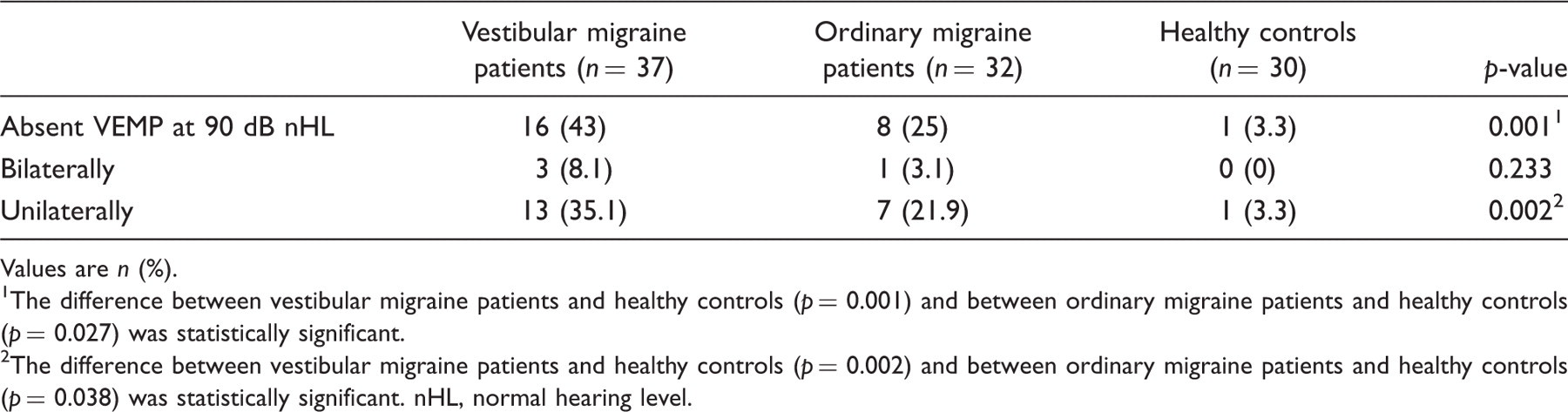
We identified 37 ‘definite’ VM patients, 32 ordinary migraine patients and 30 healthy controls (Table 2). The vertigo complaints of the VM patients are described in Table 3. Six of the VM patients used antiepileptic drugs (two valproate and four topiramate), seven used beta-blockers, and two patients other migraine prophylaxis. Among the ordinary migraineurs, four patients used beta blockers and three patients other prophylactic drug. Ordinary migraineurs used more attack medication (triptans) than the vestibular migraine patients did. Subgroup analyses revealed no differences in VEMP response pattern between the patients who used medication (either attack or prophylactic medication) and those who did not. Results from VEMP testing (absence of response, either unilaterally or bilaterally, threshold, and latencies) and from the questionnaire about motion sickness are shown in Tables 4 and 5 and Figure 2, respectively. We found no relationship between an absent VEMP response and motion sickness susceptibility (p = 0.240).
(A) Percentage of motion sickness susceptibility among vestibular migraine patients, ordinary migraine patients, and healthy controls. The degree of motion sickness was classified as ‘not susceptible’ when they answered never−rarely or sometimes and as ‘susceptible’ when they answered frequently and always. 1There was a difference between vestibular migraine patients and healthy controls (p = 0.006) and between vestibular migraine and ordinary migraine patients (p = 0.026). (B) Susceptibility to different triggers of motion (mean). The ratings were never (1), rarely (2), sometimes (3), frequently (4), and always (5) analysed as continuous data.
2
Post-hoc test (Bonferroni) showed a difference between vestibular migraine patients and healthy controls for boat, car, and plane.
3
Post-hoc test (Bonferroni) showed a difference between ordinary migraine and vestibular migraine patients for car. Patient characteristics Values are n (%) or mean ± SD. Description of the vertigo complaints of the vestibular migraine patients Values are n (%).
1Definitions for experienced vertigo symptoms are adapted from International classification of vestibular disorders (ICVD-1).
2Definition for migrainous features are adapted from IHS classification 2004.
3Multiple answers possible. Absent vestibular evoked myogenic potential response, either bilaterally or unilaterally Values are n (%). The difference between vestibular migraine patients and healthy controls (p = 0.001) and between ordinary migraine patients and healthy controls (p = 0.027) was statistically significant. The difference between vestibular migraine patients and healthy controls (p = 0.002) and between ordinary migraine patients and healthy controls (p = 0.038) was statistically significant. nHL, normal hearing level. Latencies and threshold of those who had a normal vestibular evoked myogenic potential response (VEMP) on both ears Values are mean ± SD. There were no individual differences in the mean latency time of left and right ear, thus the latencies are presented as the mean latencies (ms) of both ears. nHL, normal hearing level.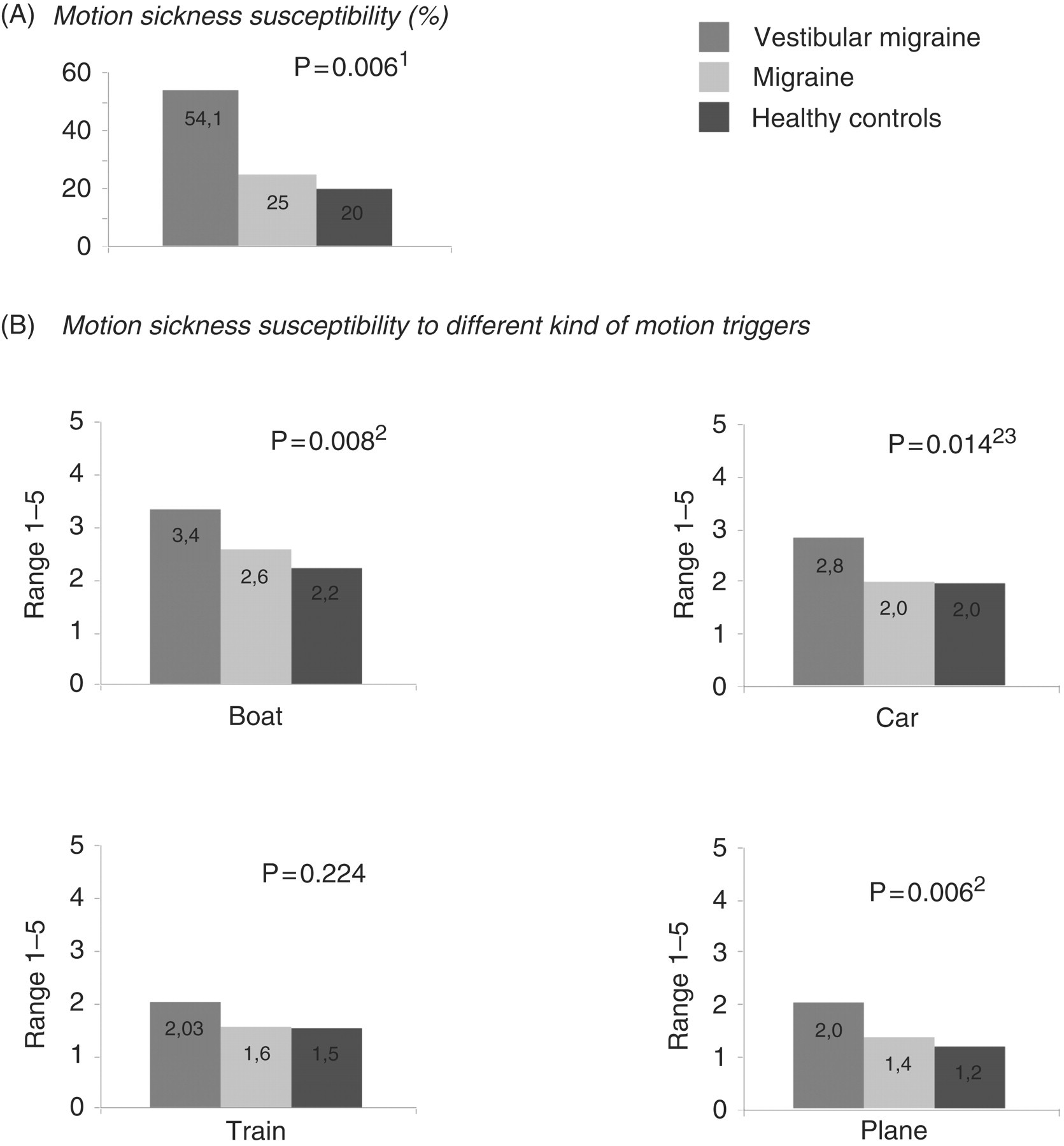



Discussion
We found that absence of VEMP response was more frequent among VM patients (44%) and patients suffering from ordinary migraine (25%) than in healthy controls (3%), but the latencies were normal in all groups. These results indicate a vestibulocollic dysfunction in migraine and especially VM, but cannot address the dysfunction to any special site as the entire saccular pathway, peripheral, central, or both could be involved. Our results are in line with a recent report by Vitokovic et al. (3) and an older study of basilar-type migraine (18) which found abnormal VEMP responses in 19.2% and 35%, respectively. Two other studies (19,20) found presence of VEMP response in almost all VM patients, but abnormalities in the sense of a reduced amplitude size (19,20) and a deficit of the habituation pattern of the amplitude. A lack of habituation of cortical- and subcortical-evoked potential has earlier been described in migraine patients as well as VM patients, indicating a underlying central pathophysiology (21). In our study and the study by Vitokovic et al, the VEMPs were recorded in definite VM patients following the Neuhauser criteria strictly. In the other studies (22) were the subjects a combination of definite and probable VM patient and tertiary referred migraine patients as controls (20). This might explain some of the differences between the findings of absent VEMP responses in some studies and not in others. Further on, also the heterogeneity of the VM patients and the time from last migraine and/or vertigo attack might affect the test results.
We did not find support for a lowered VEMP threshold for sound stimuli in migraine patients with or without vertigo. Hence, our findings did not support the theory that hyperacusis or vertigo in migraine is caused by a peripheral sensitization in vestibular organs as in the syndrome of dehiscence of the superior semicircular canal. A lowered VEMP threshold in this clinical syndrome is typically about 20 dB lower than normal (70 dB nHL compared to 90 dB nHL) (11,23,24). Our mean VEMP threshold was 87 dB nHL.
As expected, VM patients regard themselves as more susceptible to all kinds of motion triggers than those with ordinary migraine and healthy controls. Seasickness was the most frequent experienced form of motion sickness reported (Figure 2).
Previous studies report that 50–67% of migraineurs suffer from motion sickness, a prevalence two−five times higher than in the general population (25−29). So far, to our knowledge, only one study has compared the severity and incidence of proneness to motion between migraine patients with and without vertigo (30). They found the same motion sickness pattern between the groups as we did (VM > migraine dizziness > classic migraine > controls). In this study the effects of motion sickness were tested by usage of the rotatory chair test and the authors hypothesize that an impaired cerebellar inhibition over the VOR may underlie the vestibular hypersensitivity in VM patients.
The pathophysiological mechanisms in migraine are areas for intense research. The prevailing explanation model suggests that patients with migraine suffer from an abnormality of the brain to control or modulate incoming signals (31), probably on a genetic basis (31). The model also involves changes in neurotransmitters, which cause neurovascular reactions in the trigemino-vascular reflex arc and protein extravasations into the dura and vessels (a sterile inflammatory response). Activation of the trigemino-vascular reflex arc is thought to be responsible for the pain in migraine and can be activated subcortically, cortically, or directly as a consequence of peripheral stimulation. There are reciprocal connections between the trigeminal reflex arc and the vestibular system (33). Imaging studies show activation of these areas during a migraine attack (34). There is a debate of what causes the vertigo symptoms in migraine. Some explain it with vasospasm within the vessels of the vestibular nerve and/or labyrinth (35). Others suggest a linkage between Meniere’s disease and VM, and thus vertigo caused by endolymphatic hydrops (36). It has also been speculated what role the neurotransmitters and neuromodulators play; several are involved in the inner ear (glutamate, acetylcholine, or calcitonine gene-related peptide). Furman et al. (13) suggested a simultaneous action on the vestibular nuclei, trigeminal structures, and the thalamocortical pathways as explanation for the various vestibular symptoms described in VM.
The clinical syndrome of motion sickness is traditionally explained by the sensory mismatch theory. The brain’s estimate of motion is based on vestibular, visual, and somatosensory information (velocity storage process). If prevailing inputs from these sensory organs are incongruent with earlier stored patterns derived from previous experiences about locomotion, a sensory mismatch is generated, thereby leading to the symptoms of motion sickness. The peripheral vestibular apparatus is necessary to develop motion sickness and subjects with a deficit peripheral vestibular system do not develop the symptoms of motion sickness, despite adequate stimuli (37,38). Researchers have proposed that asymmetries in the otolith function between the two labyrinths can lead to motion sickness (29,39). The theory is that, under self-propelled locomotion, central mechanisms will compensate for the discrepancy between the otolith organs and any sensory channel, but that this central compensation fails when further incompatible sensory input are added as in different types of travelling. The saccule is considered to be the main sensory organ for motion (40,41). Studies done in sea-sick susceptible persons reveal a higher VEMP threshold, but no asymmetries which could support the asymmetry hypothesis of the otolith function (42). We could not find any relationship between motion sickness susceptibility and absent VEMP response in migraineurs and therefore no support for the hypothesis that motion sickness susceptibility in migraineurs are attributed to canal-otolith asymmetry.
Overlapping pathomechanisms of motion sickness and migraine has been suggested: a hypersensitive ‘emetic centre’ (43), hyperexcitability of the vestibular receptors or nuclei (29,44,45) and/or a common final pathway, probably within the brainstem. Reduced brain serotonin may be an important factor. A double-blind placebo-controlled study of the effect of rizatriptan (a 5-HT agonist) on motion sickness in VM patients gives a body of evidence to this theory (46).
Based on the findings of an increased vestibular sensitivity in the sense of motion sickness, but no signs of peripheral sensitization in terms of a lowered VEMP threshold in our VM patients, we suggest that they have a central hyperexitability. We also suggest that vertigo in migraine is caused by a combination of asymmetrical stimuli from peripheral sensory organs combined with the inborn mechanism of a disturbed central nervous sensory process. Endolymphatic hydrops, vasospasm of the vessels of the inner ear, or asymmetric neuropeptide release may perhaps be the peripheral trigger of the vertigo attacks, reversible or irreversible.
Some aspects of our study design need to be addressed. First or all, the case ascertainment is good (high specifity) and the characteristics of the VM patients are comparable to other published descriptions of VM patients using the definite Neuhauser criteria (1,47,48). The ordinary migraine patients used triptans more frequent than the VM patients did, but otherwise the groups were comparable regarding baseline characteristics. The difference in use of attack medication did probably not influence the results, as we could not find a difference in VEMP response pattern between those who used triptans and those who did not. Furthermore the half time of triptans is short (about 2 hours) and the effect is probably eliminated after a week of discontinuation. Antiepileptic drugs are neuromodulators and reduce the neuronal hyperexcitability and could possibly influence the VEMP threshold testing (false-negative results), but no studies have evaluated this aspect. In our patients, the plasma concentration of antiepileptic drugs should be zero and such an influence is unlikely anyway. Another aspect regarding antiepileptic drugs is a theoretically withdrawal effect and, after discontintuation, cerebral hyperexcitability is presumed to be the mechanism of the withdrawal seizures. However, there was no indication of a lowered VEMP threshold in any of the groups, so we think such an effect is ignorable among our patients.
An obvious limitation in our study was the VEMP technique, as we used a system and headphones made for recording brainstem-evoked responses. Our laboratory had an upper limit for stimulus intensity at approximately 90 dB nHL/130 dB SPL and therefore lower than routinely used (95 dB). Low intensity clicks at 90 dB nHL has been documented to give a normal VEMP response in healthy individuals (11), but this limitation in our equipment might have given false-negative responses in some. Our VEMP system also did not allow rectification of the electromyogram signal and thereby prevented calculations of the amplitude and the habituation-response pattern on repeated stimuli. The use of an unrectified VEMP method is not assumed to affect the latency of the VEMP or the determination of the VEMP response itself, as rectification only can reduce the bias from the degree of interindividual muscle contraction. Testing the stimulus threshold based on decreasing decibel is a documented method (10) in the study of the Tullio phenomenon. Testing the VEMP threshold based on the amplitude is more reliable (11) and less time consuming, but we do not think it affected the results of the VEMP threshold in this study as there was no indication of a reduced VEMP threshold in any of the groups. Another limitation was that we not applied the most commonly used motion sickness susceptibility questionnaire (27). This prevented exact comparison with other studies.
It might be that future studies including better methodological technique could specify the lesion site of the VEMP abnormalities and further elucidate the mechanisms underlying vertigo in VM patients
In conclusion, our study demonstrated a higher frequency of loss of VEMP in VM patients and ordinary migraineurs as compared to healthy controls. Somehow, the sacculus and/or the saccular pathway are affected. The underlying vertigo mechanism and anatomical site of origin, however, cannot be explained by this finding. We found more motion sickness susceptibility among migraineurs than among controls, but no support for peripheral vestibular hypersensitivity to noise in vestibular migraine in terms of a lower VEMP threshold. This could indicate that the motion sickness in migraine is due to an abnormality in central sensory modulation mechanisms.
Footnotes
Acknowledgements
The authors gratefully thank Dr Siri Økstad for her help during data collection and analysis. The authors also acknowledge the statistical assistance of Dr Scient Are Hugo Pripp. The authors thank the patients who consented to participate in the study.
Funding
This work was supported by Medisinsk forskningsfond i Agder ‘Sykehuset i våre hender’, Sørlandet sykehus HF, Kristiansand, and the Research Department, Sørlandet sykehus HF Kristiansand, Norway. The study received no support from industrial sponsors.
Conflicts of interest
None.
