Abstract
To determine whether motion sickness induced by optokinetic stimulation would trigger migraine-like attacks, 27 migraine sufferers and 23 controls attended the laboratory up to three times at intervals of at least 3 weeks. On one occasion subjects experienced up to 15 min of optokinetic stimulation, followed by three 30-s applications of ice to the temple at 4-min intervals. On another occasion, the ice applications preceded and accompanied optokinetic stimulation. On a third occasion, one hand was immersed in ice water for 30 s, three times at 4-min intervals before and during optokinetic stimulation. Subjects recorded headache activity in a diary over the course of the study. None of the controls experienced a migraine-like attack at any stage of the experiment. In migraine sufferers, the incidence of migraine-like attacks was greater than the expected daily incidence of 8% after sessions that involved painful stimulation of the temple during or after optokinetic stimulation (44% and 28% of the group, respectively) (P < 0.001). In contrast, migraine-like attacks developed in only 13% of migraine sufferers after the session that involved immersing the hand in ice water during optokinetic stimulation (not significant). The development of nausea and headache during optokinetic stimulation increased the likelihood of migraine-like attacks afterwards. These findings indicate that motion sickness and head pain increase susceptibility to migrainous attacks in migraine sufferers, and suggest that the symptoms of migraine build upon each other in a vicious circle. Thus, targeting multiple symptoms should be more effective than targeting individual symptoms, both for preventing and treating attacks of migraine.
Introduction
Motion sickness resembles migraine in several ways – both are associated with vestibular disturbances, nausea, vomiting, pallor and headache (1–6). Indeed, heightened susceptibility to motion sickness in migraine sufferers (5, 7–9) suggests some overlap of mechanisms. Discharge of sensitized or disinhibited trigeminal nociceptive neurons may increase susceptibility to migraine (10–12) and augment headache during attacks (13–15). Similarly, excitability in brainstem emetic neurons might boost nausea in migraine sufferers both during motion sickness (9, 16) and during attacks of migraine.
If similar mechanisms are involved in motion sickness and migraine, then a bout of motion sickness could increase the likelihood of an attack of migraine. This possibility was investigated in sailors competing in a ‘round the world’ yacht race (17, 18). During the race, participants recorded the occurrence of motion sickness and characteristics of headache in a daily log. Although sailors who suffered from migraine during the race were more susceptible than other sailors to motion sickness, migraine and motion sickness apparently developed at different times. However, these findings are not definitive because the participants may have had difficulty distinguishing between symptoms of motion sickness and migraine in the presence of ongoing motion sickness provocation.
The aim of the present study was to determine whether migraine-like headaches would develop after brief exposure to motion sickness provocation. In particular, the incidence of prolonged headache and nausea in migraine sufferers and controls was investigated after exposure to the illusion of movement induced by watching black and white stripes move past (an optokinetic effect). As reported elsewhere (9), the main goal of this research was to determine whether painful stimulation of the head would enhance nausea in migraine sufferers during optokinetic stimulation. One of the sessions involved applying an ice block to the temple (thus inducing head pain) during optokinetic stimulation, whereas the subject's hand was immersed in painfully cold ice water during optokinetic stimulation in another session. In a third session ice was applied to the temple after optokinetic stimulation, to determine whether head pain would influence residual symptoms of motion sickness.
Subjects recorded details of their attacks in a headache diary over the course of the study. It was hypothesized that the incidence of migraine-like attacks would be greater after optokinetic stimulation than at other times during the recording period. Since painful stimulation of the head might excite the trigeminal nociceptive neurons that mediate headache, it was also hypothesized that the incidence of migraine-like attacks would be greater after optokinetic stimulation accompanied by painful stimulation of the head than after optokinetic stimulation accompanied by painful stimulation of the hand.
Subjects and methods
Subjects
The migraine sample consisted of 22 women and five men (mean age ± SD, 40.7 ± 11.2 years; range 20–59 years) who met International Headache Society criteria for migraine with aura (n = 3) or without aura (n = 24) (19). Participants did not take prophylactic medication for migraine and had no other major medical conditions. The control group consisted of 17 women and six men (mean age 39.7 ± 11.8 years; range 18–62 years) who reported less than 12 headaches per year that did not meet diagnostic criteria for migraine and that were relieved by analgesics. Each participant provided informed consent for the procedures, which were approved by the Murdoch University Human Research Ethics Committee.
Experiments were carried out when subjects were free from headache for at least 4 days, and women were tested between menstrual periods. All but four migraine sufferers and one control subject attended three sessions separated by at least 3 weeks. Two migraine sufferers began prophylactic medication after participating in one session and were not tested further, and the other two withdrew from the experiment after the first session because of prolonged headache and nausea. The control subject withdrew because of time constraints.
Procedures
The procedures have been described in detail elsewhere (9, 20). In brief, one session consisted of up to 15 min of optokinetic stimulation, followed about 20 min later by three 30-s applications of ice to the temple at 4-min intervals (Fig. 1). On another occasion, three ice applications preceded optokinetic stimulation and another three applications accompanied optokinetic stimulation at 4-min intervals. On a third occasion, one hand was immersed in ice water for 30 s, three times at 4-min intervals before and during optokinetic stimulation. The session order was counterbalanced across subjects.
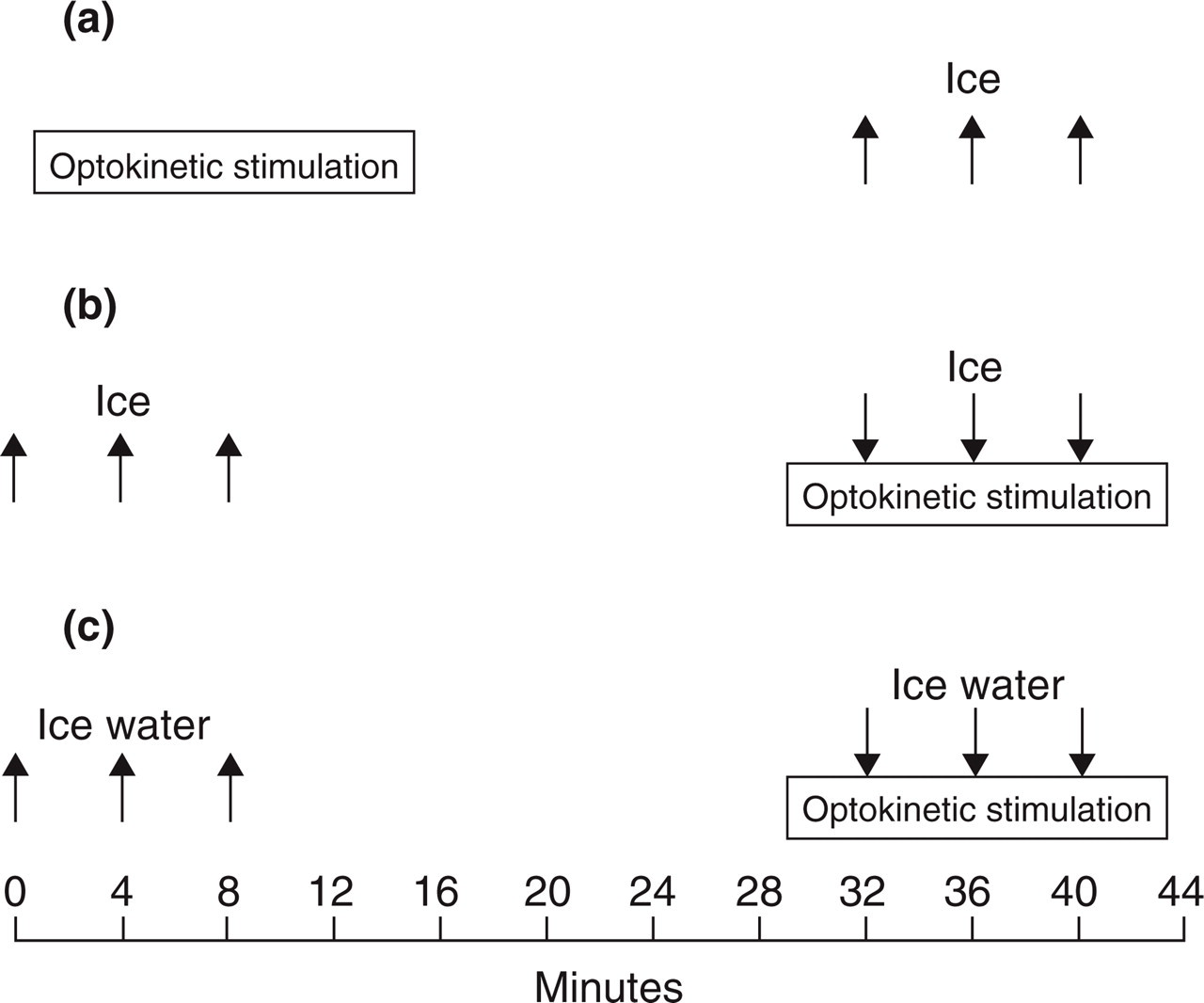
The sequence of procedures in the three sessions (a–c). In each session, the optokinetic stimulus lasted up to 15 min. Ice was applied to the temple, or the hand was immersed in ice water at intervals shown by the arrows. (a) Optokinetic stimulation followed by ice on the temple. (b) Ice on the temple before and during optokinetic stimulation. (c) Hand in ice water before and during optokinetic stimulation.
Subjects were asked to record details of their headaches in a diary over the course of the study. The number of days recorded depended on when the subject returned for the final session, and averaged 77 ± 27 days (mean ± SD) in migraine sufferers and 67 ± 15 days in controls. If the headache was rated as more than mild (i.e. > 3 on a 0–10 scale of intensity), subjects were asked to record headache details (the location of pain, treatment, and the presence of symptoms such as photophobia, phonophobia, nausea, vomiting, dizziness, sweating, drowsiness, and aura) every 8 h until the headache subsided. The accuracy of the diary recordings was checked each time the subject attended the laboratory.
Data reduction
Since headache details generally were not recorded consistently enough for diagnostic criteria to be applied for migraine or probable migraine (19), headaches were grouped into categories with or without nausea and those with nausea are referred to as migraine-like attacks. A rating of peak intensity was obtained from the headache diary and the duration of attacks was estimated to the nearest half day. Headaches on the day of testing either subsided without drug treatment shortly after optokinetic stimulation, or persisted for at least several hours despite drug treatment. Since the aim of this study was to determine whether optokinetic stimulation would trigger migraine-like attacks, headaches that subsided shortly after optokinetic stimulation were not considered further. To estimate general headache activity over the course of the study, the proportion of days during the recording period with headaches, with or without nausea, was calculated for each subject (excluding headaches that started during or shortly after optokinetic stimulation).
Results
None of the control subjects experienced prolonged headache or nausea after optokinetic stimulation. One control subject experienced headache with nausea at another time during the recording period, but this attack was unlikely to be migraine because it was associated with symptoms of upper respiratory tract infection. Control subjects reported headaches on only 1 ± 1% (mean ± SD) of days throughout the recording period, compared with 25 ± 20% of days in migraine sufferers [t(42) = 5.57, P < 0.001]. Since headache activity was minimal in control subjects, their headache details were not included in the analyses reported below.
A high incidence of headaches in a few migraine sufferers disproportionately boosted the mean number of days of headache (deviation of the distribution from normal, Kolmogorov–Smirnov Z = 1.886, P < 0.01 for headaches with nausea; and Kolmogorov–Smirnov Z = 1.923, P < 0.001 for headaches without nausea). Therefore, the expected daily incidence of headaches was based on the median rather than the mean number of days with headache throughout the recording period (excluding attacks that started during or shortly after optokinetic stimulation).
As shown in Table 1, 44% of migraine sufferers experienced a migraine-like attack (prolonged moderate or severe headache with nausea) after the session involving application of ice to the temple during optokinetic stimulation, compared with an expected daily incidence of only 8%[χ2(1) = 42.6, P < 0.001]. Similarly, the incidence of headache with nausea exceeded the expected incidence after the session involving application of ice to the temple after optokinetic stimulation [28% vs. 8%, χ2(1) = 13.0, P < 0.001]. Immersing the hand in ice water during optokinetic stimulation boosted the rate of headache without nausea [26% of cases vs. an expected incidence of 11%, χ2(1) = 4.85, P < 0.05] but not the rate of migraine-like attacks. In fact, headaches with nausea were less likely to develop after the hand was immersed in ice water during optokinetic stimulation than after ice was applied to the temple during optokinetic stimulation (13% vs. 44%; McNemar test, P < 0.05).
Percentage of migraine sufferers expected to experience headache on the day of the test, and the percentage who actually experienced a headache after each session

Actual incidence greater than the expected incidence
(∗P < 0.05;
P < 0.001).
Significantly less than the 44% of migraine sufferers who developed headache with nausea after ice was applied to the temple during optokinetic stimulation (McNemar test, P < 0.05).
The proportion of migraine sufferers who treated their postsession headache with medication did not differ significantly across sessions (Table 2). Headaches lasted longer after sessions involving application of ice to the temple than after the session involving immersion of the hand in ice water [2.6 ± 2.9 days vs. 1.0 ± 1.1 days, t(32) = 2.38, P < 0.05]. However, neither the timing of application of ice to the temple (i.e. during vs. after optokinetic stimulation) nor the site of painful stimulation (temple vs. hand) affected the intensity of postsession headaches (Table 3).
Percentage of migraine sufferers who treated their attack after each session

The incidence of headache treatment did not differ significantly across the three experimental conditions.
Intensity and duration of postsession headaches for migraine sufferers who experienced those headaches

As shown in Table 4, the presence of nausea and/or headache during optokinetic stimulation increased the likelihood of prolonged headache in migraine sufferers after the session. The number of symptoms during optokinetic stimulation was proportional to the number of symptoms afterwards for sessions that involved applying ice to the temple during optokinetic stimulation (Spearman's ρ= 0.47, P < 0.05) and immersing the hand in ice water during optokinetic stimulation (Spearman's ρ= 0.63, P < 0.001); a similar relationship for the session that involved applying ice to the temple after optokinetic stimulation did not achieve statistical significance (Spearman's ρ= 0.29).
Relationship between nausea and headache during optokinetic stimulation and the development of prolonged headache

Nausea and headache were considered to be present during optokinetic stimulation if the mean rating was > 1 on a scale where 0 corresponded to ‘not at all’, 1 to ‘stomach or head awareness but no discomfort’ and 10 to ‘extremely intense nausea or headache’.
Discussion
Painful stimulation of the temple during and after optokinetic stimulation boosted the incidence of migraine-like attacks in migraine sufferers, and painful stimulation of the hand during optokinetic stimulation boosted the incidence of headaches without nausea. In contrast, none of the control subjects experienced migraine-like attacks after optokinetic stimulation or at any other time over the course of the study.
We previously reported that the immediate effects of optokinetic stimulation and painful stimulation of the temple and hand differed between migraine sufferers and controls. In particular, nausea and headache were greater in migraine sufferers than controls during optokinetic stimulation (9, 16), and could be aggravated by painful stimulation of the temple but not the hand (9). In general, migraine sufferers are more susceptible than controls to gastrointestinal disturbances (5, 7–9, 21–24) and photophobia (25–29) during the headache-free period, suggesting that the migraine mechanism persists subclinically between attacks.
Migraine sufferers who experienced nausea and headache during optokinetic stimulation were more likely to experience migraine-like attacks afterwards than migraine sufferers who did not develop nausea or headache during optokinetic stimulation. This association was not perfect – symptoms of motion sickness invariably resolved quickly after optokinetic stimulation in controls and only sometimes developed into a migraine-like attack in migraine sufferers. Nevertheless, the incidence of these attacks far exceeded the median daily incidence of headaches, implying that a short period of motion sickness sometimes triggered a prolonged attack of migraine.
Painful stimulation of the temple during and after optokinetic stimulation boosted the incidence of migraine-like attacks, presumably because trigeminal nerve discharge enhanced the provocative effect of motion sickness on the migraine mechanism. In contrast, painful stimulation of the hand boosted the incidence of headaches without nausea but not of migraine-like attacks. It is interesting to note that nausea and headache increased during painful stimulation of the temple but not during painful stimulation of the hand (9, 20). These findings support the notion that trigeminal nerve discharge and nausea build upon each other in a positive loop, sometimes culminating in an attack of migraine. In contrast, painful stimulation of the hand before and during optokinetic stimulation boosted the incidence of non-migrainous attacks, possibly due to the stressful nature of the tasks. Painful stimulation of the hand during optokinetic stimulation might also have provoked a modulatory effect (e.g. stress-induced analgesia or diffuse noxious inhibitory controls) that inhibited the full expression of migrainous symptoms.
In conclusion, the findings indicate that motion sickness and head pain increase susceptibility to migraine-like attacks in migraine sufferers but not controls, presumably because the provocation threshold for migrainous symptoms is lower in migraine sufferers than in controls. The findings also support the notion that symptoms of migraine build upon each other in a vicious circle. If so, targeting multiple symptoms (e.g. headache, gastrointestinal complaints, and disturbances of the special senses) should be more effective than targeting individual symptoms, both for preventing and treating attacks of migraine.
Footnotes
Acknowledgements
This study was supported by grants from the National Health and Medical Research Council of Australia and the U.K. Migraine Trust. We thank Ms Shiree Hassard for technical assistance.
