Abstract
To clarify if migraine-associated vertigo (MAV) and Meniere's disease (MD) share a common pathophysiology, vestibular-evoked myogenic potentials (VEMP) were measured in 11 patients with MAV, 11 with unilateral MD and eight healthy subjects. As acoustic stimuli, tone bursts (TB; 250, 500, 1000 and 2000 Hz) were presented. In healthy subjects, 500-Hz TB evoked the largest amplitude. To quantify this tendency, 500-1000 VEMP slope was calculated, and 500-1000 VEMP slope was the smallest on the affected side of MD patients. Among the 11 MD patients, five had significantly decreased 500-1000 VEMP asymmetry (shift of the tuning to 1000 Hz). Three of the 11 MAV patients also showed a significantly decreased 500-1000 VEMP slope. This finding suggests that MAV might share a common pathophysiology with MD. In addition to this finding, four of the other eight MAV patients showed prolonged p13 latencies. This suggests that MAV could consist of patients with different lesion sites.
Introduction
Patients with migraine often report vertigo/dizziness. The clinical association of migraine with vertigo/dizziness has been supported by several epidemiological studies (1, 2). Migraine-associated vertigo (MAV), migraine-associated dizziness or migrainous vertigo (MV) has been proposed as one of the main clinical entities of balance problems (1–5). However, the pathophysiology of MAV remains to be clarified.
Meniere's disease (MD) is also one of the most common diseases that cause recurrent vertigo attacks. In addition to recurrent vertigo, patients with MD have fluctuating hearing loss, tinnitus, and a sensation of aural fullness (6). Histopathological studies of the temporal bone have revealed endolymphatic hydrops in the affected ears (7–9). Differential diagnosis of MAV from MD is sometimes hard because patients with MAV could have tinnitus and/or aural fullness during vertigo attacks (10), and a controlled study has provided evidence of the association of MD with migraine (11). One might therefore assume that the two conditions, MAV and MD, could share common pathophysiological mechanisms (12).
Vestibular-evoked myogenic potential (VEMP) has been regarded as a vestibulo(sacculo)-collic reflex evoked by acoustical stimulation and applied as a clinical test of the vestibular system (13–15). On the basis of previous reports, the incidence of abnormal VEMP in patients with MD (absence of responses or decreased amplitudes) is considered to be around 40–50% (16, 17). Rauch et al. (18) reported the alteration of tuning characteristics of VEMP in MD patients. According to previous studies (18–20), the best frequency of VEMP is between 300 and 700 Hz. Rauch et al. (18) reported that, in healthy subjects, the largest amplitude and the lowest threshold were obtained to 500-Hz tone bursts (TB), whereas patients with MD showed less tuning at 500 Hz and shifts of the best frequency to 1000 Hz. Rauch et al. attributed this shift to the change of resonant frequency in the saccule. We assumed that VEMP in patients with MAV could show the same tuning characteristics as those in patients with MD if MAV and MD share common pathophysiological mechanisms. Of course, MAV patients might show other abnormal findings. Concerning VEMPs in patients with migraine or MAV, there are only a few reports (21, 22), which showed decreased amplitudes of VEMP in patients with migraine.
Here, we studied characteristics of VEMP responses in patients with MAV and compared those with patients with MD and healthy subjects
Materials and methods
Subjects
Eleven patients with MAV (two men, nine women, 18∼60 years of age, mean 41.3 years), eight healthy volunteers (four men, four women, 30∼48 years of age, mean 37.3 years) and 11 patients with unilateral definite MD (three men, eight women, 44∼78 years of age, mean 58.2 years) were enrolled. As the diagnostic criteria of definite MD, American Academy of Otolaryngology-Head and Neck Surgery guidelines (1995) (6) were adopted (Table 1). As the diagnostic criteria of migraine, we adopted International Headache Society criteria (23). As the diagnostic criteria of MAV, we used the criteria shown in Table 2, which were processed on the basis of Neuhauser's criteria (12) and Brantberg's criteria (3). Adopting our criteria, patients with associated unilateral hearing loss were excluded in order to differentiate MD clearly. This study was prospective. None of our subjects had conductive hearing loss on either side.
Diagnostic criteria definite Meniere's disease (6)

Diagnostic criteria of migraine-associated vertigo in this study

IHS, International Headache Society.
The study was performed according to the Declaration of Helsinki. Informed consent was obtained from each subject. Ethical approval was received from the Tokyo Postal Services Agency Hospital ethics committee.
Methods
For recording VEMPs, electromyographic (EMG) activity was recorded from a surface electrode placed on the upper half of each sternocleidomastoid muscle (SCM), with a reference electrode on the lateral end of the upper sternum and a ground electrode on the nasion (24). Recording was performed with Neuropack system (Nihon Kohden Co. Ltd, Tokyo, Japan). During the recording, subjects in the supine position were instructed to raise their heads from the pillow to contract the SCM.
All healthy subjects underwent VEMP testing using air-conducted TBs (rise/fall time 1 ms, plateau time 5 ms, 250, 500, 1000 and 2000 Hz), and patients with MD or MAV did VEMP testing using 500-Hz and 1000-Hz TB. Acoustical stimuli were presented through a headphone (Type DR-531; Elega Acous. Co. Ltd, Tokyo, Japan) (25). The sound intensity was 130 dB sound pressure level (SPL) for 500, 1000 and 2000 Hz, but 120 dB SPL for 250 Hz due to the limitation of the device. EMG activities were amplified and bandpass-filtered (20–2000 Hz). The stimulation rate was 5 Hz, and the time window for analysis was 100 ms (−20 to 80 ms). Responses to 25 TB stimuli for each run were averaged. Reproducibility of responses was confirmed by two runs for each frequency. We used the average of two runs as the amplitude of p13-n23 and latencies of p13 and n23 (Fig. 1).

Parameters measured in vestibular-evoked myogenic potentials.
We analysed the amplitude of the first biphasic responses (p13-n23) on the ipsilateral SCM to the stimulated ear (13) and the peak latencies of p13 and n23 (26) (Fig. 1). In order to eliminate the effect of variance of muscle activities, the mean background amplitude was calculated from the integral of rectified background activities during a pre-stimulus period of 20 ms (27, 28). Then corrected amplitudes (CA) were calculated as follows:

In order to evaluate interaural asymmetry of VEMP responses, interaural VEMP asymmetry was calculated as follows (24):
Interaural VEMP asymmetry = 100 × |CAr − CAl|/(CAr + CAl) in healthy subjects and patients with MAV, and, in MD patients:
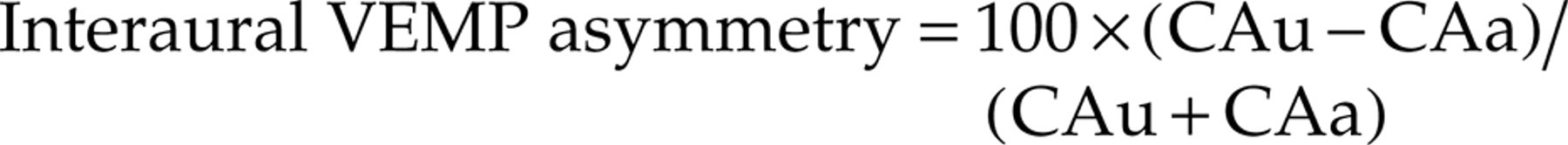
where CAr(l) is the corrected amplitude of p13-n23 on the right (left) SCM to the right (left) ear stimulation, CAu(a) the corrected amplitude of p13-n23 on the unaffected (affected) side to the unaffected (affected) side stimulation, and |CAr − CAl| is the absolute value of (CAr − CAl).
In order to compare CA to 500-Hz TB with CA to 1000-Hz TB, 500–1000 slope was calculated as follows:

where CA500 is the corrected amplitude of p13-n23 to 500-Hz TB, and CA1000 is the corrected amplitude of p13-n23 to 1000-Hz TB.
Therefore, the positive value of 500–1000 VEMP slope indicates the dominancy of VEMP responses in 500 Hz, whereas the negative value is the dominancy in 1000 Hz.
Results
VEMPs to TBs in healthy subjects
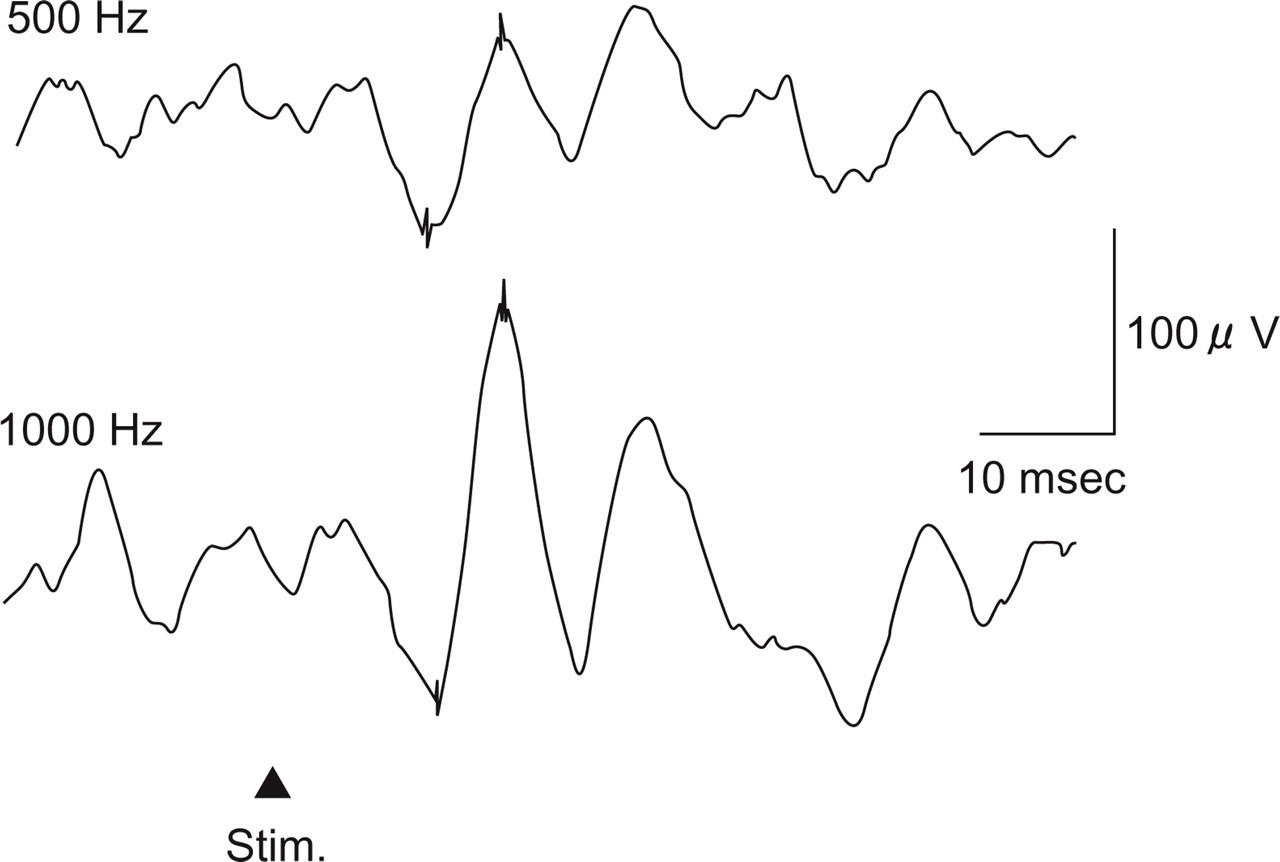
Both sides of all the healthy subjects showed clear VEMP responses to 500-Hz and 1000-Hz TB. On the other hand, responses to 2000 Hz and 250 Hz were observed in 87% (14/16) and 68% (11/16), respectively. The mean of CA was the largest to 500-Hz TB in this study (Figs 2 and 3 and Table 3).

Vestibular-evoked myogenic potentials of a 43-year-old healthy woman (responses on the left side). She showed the largest response to 500-Hz tone bursts among the four frequencies.

Corrected amplitudes in healthy subjects and patients with Meniere's disease (MD) and migraine-associated vertigo (MAV). Dots and bars represent means and standard deviations, respectively. See also Table 7.
Detection rates and corrected amplitudes (CAs) of vestibular-evoked myogenic potentials in healthy subjects

We calculated interaural VEMP asymmetries to 500-Hz and 1000-Hz TBs because the detection rates of responses to these two frequencies were 100% in healthy subjects. They were 15.4 ± 6.7 (mean ±
We also calculated 500–1000 VEMP slope. In healthy subjects, 500–1000 VEMP slope ranged from −16.6 to 30.0 [mean (
500–1000 vestibular-evoked myogenic potential slope

Bonferroni test indicated that significant difference was seen between the affected side and the unaffected side of MD patients (P < 0.0001).
MD, Meniere's disease; MAV, migraine-associated vertigo.
The means (
Peak latencies in healthy subjects (ms)

VEMPs in patients with MAV
For patients, 500-Hz and 1000-Hz TBs were applied. All patients with MAV had VEMP responses to 500-Hz TB, whereas all but one side of one patient showed responses to 1000-Hz TB (Table 6). The means and
Detection rates of vestibular-evoked myogenic potentials in healthy subjects and patients

MD, Meniere's disease; MAV, migraine-associated vertigo.
Mean +

Bonferroni test indicated that significant differences were seen between healthy subjects and the affected side of MD patients (P < 0.0001) and between MAV patients and the affected side of MD patients (P = 0.001).
Bonferroni test indicated that significant differences were seen between healthy subjects and the affected side of MD patients (P = 0.0012) and between healthy subjects and the unaffected side of MD patients (P = 0.0015).
MD, Meniere's disease; MAV, migraine-associated vertigo.
In this group, the mean (
Vestibular-evoked myogenic potentials of a 48-year-old woman with migraine-associated vertigo (MAV) (responses on the left side). She showed significantly greater responses to 1000-Hz tone bursts (TB) than to 500-Hz TB (500–1000 slope = −23.8).
Incidence of significantly decreased 500–1000 vestibular-evoked myogenic potential slope

MD, Meniere's disease; MAV, migraine-associated vertigo.
The peak latencies (mean +
Incidence of significantly prolonged peak latencies

MD, Meniere's disease; MAV, migraine-associated vertigo.
Peak latencies (mean +

MD, Meniere's disease; MAV, migraine-associated vertigo.
VEMPs in patients with MD
Detection rates of VEMP responses are summarized in Table 6. Apart from absence of responses, two patients had significantly large interaural VEMP asymmetries suggesting decreased amplitudes on the affected side to 500-Hz TB. The means and
In this group, the means (

Vestibular-evoked myogenic potentials of a 70-year-old woman with left Meniere's disease (MD) (responses on the affected side). She showed significantly greater responses to 1000-Hz tone bursts (TB) than to 500-Hz TB (500–1000 slope = −20.5).
The peak latencies (mean +
Comparison of the groups
Detection rates of VEMP responses were significantly lower on the affected side of MD to 500-Hz TB (P = 0.0003, χ2 test), while there was no significant differences concerning 1000-Hz TB (P = 0.19, χ2 test) (Table 6).
Concerning 500–1000 VEMP slope, there were significant differences among the four groups (P = 0.0012, one-way
Concerning CA, there were significant differences among the four groups (P = 0.0007 to 500-Hz TB and P = 0.001 to 1000-Hz TB, one-way
There was no significant difference in the peak latencies among the four groups (P > 0.05, one-way
Discussion
Rauch et al. (18) reported that in healthy subjects, the largest amplitude and the lowest threshold of VEMP were obtained to 500-Hz TB, whereas patients with MD showed less tuning at 500 Hz and shifts of the best frequency to 1000 Hz. Rauch et al. attributed this shift to the change of resonant frequency in the saccule. Node et al. (29) showed the same tendency and also showed that this shift of the best frequency was normalized by furosemide administration, suggesting that this shift could be caused by endolymphatic hydrops. To quantify this tendency concerning frequency tuning, we adopted 500–1000 VEMP slope as a new parameter. Using this parameter, we confirmed that the affected side of MD patients shows a shift of VEMP response tuning to 1000 Hz (Table 4). We adopted this new parameter, 500–1000 VEMP slope, for MAV patients because we hypothesized that MAV and MD might share some common pathophysiological mechanisms. The comparison of the MAV group with healthy subjects did not show significant difference of 500–1000 VEMP slope. However, this result was unsurprising, because MAV may be heterogeneous. In other words, perhaps, MAV has diverse aetiologies (12). The fact that the rate of significantly decreased 500–1000 VEMP slope, indicating the shift of VEMP response tuning to 1000 Hz in MAV patients, was relatively high (3/11, 27%) (Table 8) supports our hypothesis that MAV and MD might share some common pathophysiological mechanisms.
What are the common pathophysiological mechanisms between MAV and MD? Node et al. showed that this shift of the best frequency was normalized by furosemide administration (29), suggesting that the shift could be caused by endolymphatic hydrops. Therefore, the shared common condition could be endolymphatic hydrops. Ishiyama et al. (30) have reported that patients with MAV could have vestibular drop attack, Tumarkin's otolithic crisis, or sudden fall without loss of consciousness, which is usually seen in patients with MD (31–33).
How does migraine cause endolymphatic hydrops? Dieterich et al. (34) and Iwasaki et al. (10) reported that patients with MAV had canal paresis on the caloric test, which implies peripheral vestibular dysfunction. Patients with MAV could have inner ear damage. Trigeminal nerve endings have been found in the vessels in the inner ear (35). Neurogenic inflammation could cause change of the blood flow in the inner ear, which might lead to change of the inner ear condition. Furthermore, expression of transient receptor potential channel vanilloid subfamily1 (TRPV1), which is a nociceptive receptor associated with migraine, has been found in the human endolymphatic sac (36). Therefore, migraine might affect the absorption of the endolymph in the endolymphatic sac, resulting in endolymphatic hydrops.
Concerning VEMPs in patients with migraine or MAV, Allena et al. and Roceanu et al. have shown decreased amplitudes of VEMP in patients with migraine (21, 22). Although our study did not confirm this tendency, it might be due to the difference of stimulation (tone bursts vs. click) or the smallness of the size of our study.
Finally, we would like to point out another interesting finding in this study. We found that significantly prolonged p13 latencies were observed in four ears of four patients (4/11, 36%). The four patients with prolonged p13 were different from the three patients with significantly decreased 500–1000 VEMP slope. Prolonged peak latencies suggest retro-labyrinthine lesions, including brainstem lesions (26). Therefore, the four patients with prolonged latencies might have vestibular nerve lesions or brainstem lesions, whereas the three patients with frequency shift might have labyrinthine lesions. Different types of abnormal findings on VEMP in patients with MAV might reflect differences in aetiology. In other words, VEMP might contribute to classification of MAV by difference of aetiology.
