Abstract
Background
Clinical heterogeneity is a peculiarity of vestibular migraine, in contrast to other vestibular disorders that have a more stereotypical expression. Migraine presents a range of variability in symptoms depending on the age of the patient. Supposing that migraine headache and vestibular migraine share the same pathogenetic mechanisms, a multiparametric analysis was performed to verify the hypotheses of an age-related influence on the clinical features of vestibular migraine at the onset.
Methods
In this retrospective study, we analysed the clinical records of 72 consecutive patients affected by vestibular migraine from June 2012 to November 2018: 64 females and eight males; mean age 38.2 ± 9.6. We considered only patients that reported onset of vestibular symptoms within 12 months preceding inclusion into the study.
Results
Statistical analysis shows a significant increase in the diagnosis of probable vestibular migraine with increasing age and a decrease in vestibular migraine diagnosis (p = 0.034). The incidence of spontaneous dizziness increases with age (p = 0.012); by contrast, external spontaneous vertigo, and visually induced vertigo decrease after 40 years of age (p = 0.018), clinically characterising the onset of juvenile forms. Spontaneous vertigo, head motion-induced vertigo/dizziness, and positional vertigo did not show significant variations with age.
Conclusion
Our data show that the type of vestibular symptoms in vestibular migraine varies according to the age of onset.
Introduction
The link between migraine and vertigo has long been known, and it is based on epidemiological data, pathogenetic hypothesis, and clinical evidence (1–3). In the last few decades, several authors have proposed different approaches to classification (4–6), and some of them have been structured on the International Headache Society (IHS) definition of migraine headache. Nowadays, the classification criteria are those included in the consensus paper edited by the Bárány Society and IHS in 2012 and define two entities: Vestibular migraine (VM) and probable vestibular migraine (PVM) (7).
Clinical heterogeneity is a peculiarity of VM (5), in contrast to other common vestibular disorders such as benign paroxysmal positional vertigo (BPPV) and vestibular neuritis (VN), which have a more stereotypical clinical expression. In addition to the variability regarding symptoms and characteristics of nystagmus (8), extreme variability in temporal characteristics of VM is described: Attacks lasting seconds, minutes, hours, or days are reported (3,4,9–12).
The natural history of head pain in patients with migraine presents age-related characteristics. This is particularly true for the period from pre-school age to puberty (13) and elderly migraine patients (14). For example, several studies have shown that migraine attacks are shorter in children (<2 hours) than in adults (15,16). Unilateral head pain seems to be more frequent in adolescents than in children, in whom the pain is frequently bilateral, becoming unilateral with increasing age (17). Gastrointestinal symptoms (mostly nausea and vomiting) characterise childhood more than adult migraine (18). Photophobia and phonophobia do not seem to present age-related features (18). It is reported that migraine characteristics are different in older subjects and that the headache becomes less typical (14). The mean duration of the aura was shorter in patients with late onset of a typical aura than those with the usual onset of typical aura. The headache was found to be absent in more cases with late-onset age and headache, when present, was found to be less frequently of the migraine type (14,19).
To our knowledge, there are no data in the literature regarding natural history and age-related expression of VM. Supposing that migraine headache and VM share some common pathophysiology (20–24), it is reasonable to hypothesise that there maybe also an age-related variability in the clinical presentation of the latter. This study aimed to verify if the variability of some aspects regarding the clinical expression of VM could be influenced by age, justifying an age-related variability of VM. Notably, the target of the study was to evaluate, through a multiparametric analysis, the hypothesis of variability in clinical aspects of the VM onset depending on age. Thus, we included patients who reported the beginning of vestibular symptoms within the 12 months preceding their vestibular assessment.
Methods
Subjects and clinical data
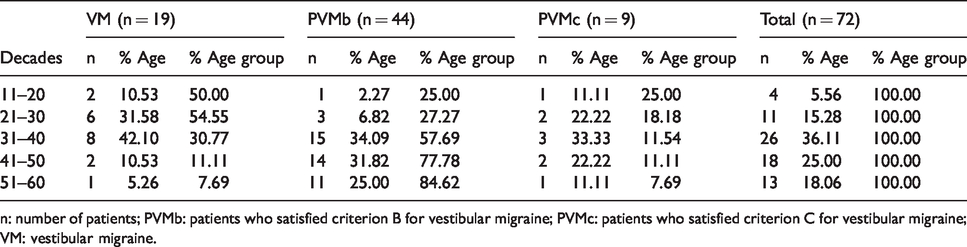
In a retrospective study, we analysed the clinical records of 72 consecutive patients (64 females and eight males; mean age 38.2 ± 9.6) affected by VM who came to our attention during the period from June 2012 to November 2018. The diagnosis has been based on diagnostic criteria established in the consensus paper edited by the Bárány Society and IHS in 2012 (7). The classification considers two entities: VM and PVM. PVM patients were divided into two groups: PVMb included patients who satisfied criterion B for VM, while PVMc had patients who satisfied criterion C (Table 1).
Diagnostic criteria for vestibular migraine (VM) and probable vestibular migraine (PVM).

In all patients, we evaluated the temporal relationship between vestibular symptoms and other migraine symptoms. Headache, visual aura, and photo-phonophobia were considered as migraine symptoms. Vestibular symptoms encompassed spontaneous vertigo (SV), including internal SV (ISV), a false sensation of self-motion, and external SV (ESV), a false impression that the visual surround is spinning or flowing (25). Additional vestibular symptoms included positional vertigo (PV), visually induced vertigo (VIV), and head motion-induced vertigo/dizziness (HMIV/D). Patients in VM + PVMc groups had coexisting vestibular symptoms and migraine symptoms in at least 50% of attacks. Cases where migraine and vestibular symptoms did not coexist, alternated, or coexisted in less than 50% of the attacks, made the PVMb group. We considered the quality of vestibular symptoms, defined following the international classification of vestibular disorders of the Bárány Society (25).
In this study, we also considered spontaneous dizziness (SD). Among patients with SD, we also studied those who presented at least one episode lasting more than 72 hours. Although SD is included in the International Classification of vestibular disorders, it may be due to other possible causes and is not in keeping with the criteria for VM.
We reviewed clinical records of patients that reported the onset of vestibular symptoms within 12 months. We excluded patients with history, referred to the same time frame, of single and/or recurrent episodes of BPPV. In the same way, we excluded patients with a history of VN during their lifetime, with Menière’s disease and/or other peripheral vestibular disorders. All patients were screened with cerebral imaging (MRI with/without gadolinium, MR-angiography) to exclude tumour, vascular, or degenerative pathologies, as possible causes of headache and vertigo/dizziness.
In our study, we planned an age-related distribution of patients based on five decades, from 11 to 60 years old (11–20, 21–30, 31–40, 41–50, 51–60). This distribution permitted a multiparametric analysis regarding VM and PVM diagnosis and the onset of vestibular symptoms, based on decades of life.
Standard protocol approvals, registrations, and patient consents
The study was conducted at the Department of Otolaryngology of the University of Perugia, Italy. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional guidelines on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. The local ethics committee of the University of Perugia approved the conducting of this study. Informed consent was obtained from all participants in the study.
Statistical analysis
The distribution of patients and study variables were described using frequencies and percentages frequency tables for categorical variables, evaluated with Chi-square analysis (Stata Version 4.2, Stata Corp., Houston, TX, USA). A two-tailed p-value < 0.05 was considered statistically significant.
Results
Nineteen (26%) of the 72 patients were affected by VM, 18 females (95%) and one male (5%). Fifty-three (74%) patients were affected by PVM, 46 females (87%) and seven males (13%). The distribution of the diagnosis of both VM and PVM, based on 5 decades, is shown in Table 2. The statistical analysis showed a significant increase in the diagnosis of PVM with increasing age and a decrease in VM diagnosis (p = 0.034) (Figure 1). Equally statistically significant is the percentage increase in PVMb diagnosis compared to PVMc + VM (p = 0.013).
Age distribution according to diagnosis.
n: number of patients; PVMb: patients who satisfied criterion B for vestibular migraine; PVMc: patients who satisfied criterion C for vestibular migraine; VM: vestibular migraine.

VM vs. PVM diagnosis: Age distribution. Note the increase in diagnosis of PVM with increasing age and the decrease in VM diagnosis.
All patients presented spontaneous vestibular symptoms as vertigo and/or dizziness in their history (Table 3). Sixty-five patients (90%) reported SV: Among them, ISV was reported by 51 (79%) and ESV by 46 (71%). In detail, 32 (49%) reported both ISV and ESV in their history, 19 (29%) exclusively ISV, 14 (22%) exclusively ESV. The age distribution of SV was: Four (100%) patients from 11–20, 11 (100%) from 21–30, 23 (89%) from 31–40, 16 (89%) from 41–50, 11 (85%) from 51–60. The age distribution of the 14 patients affected by exclusively ESV was: Four (100%) from 11–20, five (46%) from 21–30, three (13%) from 31–40, one (6%) from 41–50, one (9%) from 51–60. The age distribution of the 19 patients with ISV exclusively was: None (0%) from 11–20, three (27%) from 21–30, six (26%) from 31–40, five (31%) from 41–50, five (46%) from 51–60. Last, the distribution of the 32 patients with both symptoms (ESV and ISV) in their history was: None (0%) from 11–20, three (27%) from 21–30, 14 (61%) from 31–40, 10 (63%) from 41–50, five (46%) from 51–60. Statistical analysis did not reveal significant age-related differences in the incidence between percentage values of the SV symptom globally considered (p > 0.05). However, a significant decrease of ESV from the first decade to the third decade was recorded (p = 0.018).
Vestibular symptoms reported by each patient.

ESV: external spontaneous vertigo; ISV: internal spontaneous vertigo; SD: spontaneous dizziness; SD > 72h: spontaneous dizziness lasting more than 72 hours; HMIV/D: head motion induced vertigo/dizziness; VIV: visual induced vertigo; PV: positional induced vertigo.
Forty-one patients reported SD, in association with SV in 34 of them, alone (exclusively SD) in seven patients. The age distribution of SD was: Age group 11–20: No cases (0%); age group 21–30: Two cases (18%); age group 31–40: 12 cases (46%); age group 41–50: 15 cases (83%); age group 51–60: 12 cases (92%) (Figure 2). A spontaneous dizziness lasting more than 72 hours affected, at least once, 17 (42%) of the 41 patients with SD in their history, showing this age distribution: None (0%) from 11–20, none (0%) from 21–30, two (17%) from 31–40, six (40%) from 41–50, nine (75%) from 51–60 (Figure 3). Statistical evaluation showed a significant percentage increase in incidence of SD with increasing age (p = 0.012) (Figure 2).

Statistical evaluation showed a significant increase of SD with increasing age (p = 0.012), while no significant difference emerges between the decades of life considered with regards to SV, which always reaches high values.

An SD lasting more than 72 hours (SD > 72 h) affected patients over the age of 30 with a progressive increase in the following decades.
Regarding triggering symptoms, patients reported a HMIV/D in 65 (90%) cases, PV in 35 (49%) and VIV in 37 (51%). The age distribution was: from 11–20 years old, four (100%) patients reported HMIV/D, two (50%) PV, four (100%) VIV; from 21–30, 11 (100%) HMIV/D, six (55%) PV, 11 (100%) VIV; from 31–40, 22 (85%) HMIV/D, 14 (54%) PV, 16 (62%) VIV; from 41–50, 17 (94%) HMIV/D, nine (50%) PV, four (22%) VIV; from 51–60, 11 (85%) HMIV/D, four (31%) PV, two (15%) VIV. The statistical evaluation showed a significant decrease of VIV with increasing age. HMIV/D and PV did not show significant variations in relation to age (Figure 4).

Triggered symptoms reported by patients on VM onset. The statistical evaluation showed a significant decrease of VIV with increasing age. HMIV/D and PV did not show significant variations in relation to age.
None of our patients reported sound-induced vertigo/dizziness, Valsalva-induced vertigo/dizziness, orthostatic induced vertigo/dizziness or other triggered vertigo/dizziness.
Discussion
Our data confirm the extreme clinical heterogeneity of VM, which includes vestibular symptoms with various expression, by the international classification (25). There are spontaneous symptoms such as SV (I and/or E) and SD. These manifestations can occur singularly or, more often, both be present at different times in the patient history, showing an inter/intra-individual variability in symptomatology. Also triggered types of vertigo/dizziness, such as HMIV/D, PV, and VIV, are widely represented in the association. The different migraine pathogenetic mechanisms may contribute to the variability of VM (24). Neuroanatomic studies in animals point to a connection between the vestibular system and nociceptive brainstem structures, such as the noradrenergic locus coeruleus and the serotoninergic dorsal raphe nuclei (21–23). There are several neurotransmitters and neuropeptides that are involved in the pathophysiology of migraine, and which at the same time modulate the activity of central and peripheral vestibular neurons (5-hydroxytryptamine, noradrenaline, dopamine, calcitonin gene-related peptide) (20). The potential structures dysfunctioning in VM range from the inner ear to the brain stem, cerebellum, to the vestibular cortex, and this could explain why the clinical presentation may vary so much and has no specific eye movement abnormalities.
In our study, we conducted a detailed history of symptoms that led to VM diagnosis. This survey aimed to show if there is any difference in the clinical presentation of VM related to age. By revising literature, our study seems to be the first one with this purpose. The following considerations justify the temporal exclusion criteria adopted in our study. The age distribution based on life decades of new diagnosis time and related vestibular symptoms involved in particular the thriving phase of migraine, when pathogenetic mechanisms are considered more vivid and efficient (26).
For this reason, we considered a temporal cut-off of 60 years old, also based on the evidence of a radical decrease in the onset of migraine attacks from the age of 50, in an examined population of patients (27–29). Considering the prevalence of VM in the female gender, it appears appropriate to evaluate, for this study, the temporal time frame that goes from menarche to the period immediately after menopause. Another reason for exclusion is the fact that the appearance of polymorphous vestibular symptomatology in advanced age must be firstly considered as an expression of more relevant clinical issues (such as transient ischemic attack in the posterior circulation), even with verified migraine in the history of the patient (7). Also, due to cognitive deficits, taking a reliable medical history in this patient group is unpredictable.
Results from our study show that some clinical aspects in the onset of VM are influenced by age. First of all, the diagnosis of PVM significantly increases with age. Simultaneously, the percentage of VM diagnosis decreases just as significantly. Regarding PVM diagnosis, the majority concerned forms in which criterion B of VM was fulfilled. Therefore, our study shows that the temporal association of vestibular and migraine symptoms is not the most likely expression at the onset when VM occurs late. This evidence may raise some doubts regarding the correct interpretation of patients’ vestibular history.
It should be noted that patients were never aware of having a headache qualifying for the diagnosis, so it is often necessary to dig in detail in the past headache history, and then it is often possible to retrospectively establish past migraines. However, the result of this study confirms the fact that vestibular symptoms connecting with VM more often occur in early age concomitantly with migraine symptoms. We support the idea that, in younger age, the simultaneous migrainous activation of pain and vestibular networks is more common, whereas in more advanced age dissociated activation prevails. This variability in VM presentation is not surprising because migraine symptoms, such as visual aura, also may change over a lifetime. In particular, the mean duration of the aura was less in patients with late onset of a typical aura than those with the usual onset of typical aura, and headache was found to be absent in more cases (14,19).
Among spontaneous symptoms, SV is the most frequent symptom at the onset, being present in all decades of life that we examined, without significant differences between them. SV can occur either exclusively as ESV or ISV, or together as happened in half of the examined patients who reported the presence of both manifestations during the anamnestic period considered in our study. In particular, exclusive ESV shows a significant decrease in incidence rates with age, therefore representing a characteristic feature of the juvenile forms onset.
Another critical emerging data from our study, regarding spontaneous symptoms, is that there is an increase of SD in the latter decades of life, representing a characteristic feature of the more advanced age onset of VM. Results overall show a higher probability of variability in VM onset the later the disease occurs, probably indicating a different involvement of the vestibular pathway to the process. Central and peripheral vestibular structures could become shared in older age. In the same way, a lower intensity, with age, of the migraine pathogenetic mechanisms operating in the vestibular pathways could be responsible for the onset of late symptoms, especially for dizziness. In the current classification, VM is seen as an episodic disorder; therefore, SD is not included as a vestibular symptom to qualify the diagnosis (7). Although a chronic variant of VM has been reported (11,30), it is difficult to distinguish between this form and psychiatric comorbidity in the presence of SD (31,32). However, our results concern patients who satisfied the diagnostic criteria for VM based on episodic vestibular symptoms. Therefore, it seems to be possible that a higher probability of onset, with increasing age, of VM forms in which episodic symptoms such as vertigo, which fulfilled the diagnostic criteria, are accompanied by prolonged dizziness. As is known, dizziness is a common and vague term that can include a wide array of medical disorders, especially in advanced age. Another potential explanation is that other vestibular disorders also manifest differently in older age, with more dizziness than in younger patients. So further investigations are needed to establish if prolonged constant dizziness can also be considered an age-related form of VM and confirm our data.
The most important findings of triggered manifestations concern VIV. In contrast to the HMIV/D, which keeps high values and does not show changes in incidence through age, VIV is a dominant characteristic feature of the inception of the juvenile vestibular migraine form, with its almost complete absence in the symptomatology of later-onset VM forms. In 1995, Bronstein described the symptom of visual vertigo as a sensation of unsteadiness or dizziness on exposure to complex or moving visual stimuli (33). In a prospective study, Cousins et al. found that patients who had persistent dizziness for at least 6 months following bouts of acute VN had greater visual dependence than those who recovered without chronic symptoms (34). The disorder is supported by a trait-like tendency to rely on visual information for spatial orientation. A posturographic study showed the destabilising effects of optokinetic stimulation on postural control of migraines, and these alterations seem to persist longer in migraine patients affected by VM (35). Thus, the strategy involved in postural control appears to be present also in VM. The high incidence of VIV could be attributed to an increased susceptibility to a developed visual dependence in younger migraine patients. Probably, a greater juvenile integrity of the extra-vestibular sensorial systems, such as vision, can provide an efficient vicarious effect on the occasion of transitory and recurrent vestibular dysfunctions induced by VM. However, the assumption for such efficiency is guaranteed by the presence of stable and known visual cues. The failure of these assumptions, due to exposure to complex sensorial contexts, reveals a new sensorial mismatch that makes the migraine patients prone to develop VIV.
Conclusion
Our data show that VM has a wide variety of vestibular symptoms within individual patients and across the patient population. Additionally, the type of vestibular symptoms varies according to the age of onset of VM. Further studies could explore the impact of comorbidities of the inner ear, the brain and emotional aspects on the clinical presentation.
Clinical implications
The diagnosis of probable vestibular migraine (VM) significantly increases with age, whereas the diagnosis of definite vestibular migraine decreases. Regarding spontaneous symptoms, external spontaneous vertigo represents a characteristic feature of the juvenile onset forms of VM. On the other hand, spontaneous dizziness is more characteristic of later onset VM. Visually induced vertigo is a hallmark of juvenile onset VM and is rarely seen in older age onset VM.
Footnotes
Acknowledgements
We thank Dr. Deanna Anderlini and Dr. Mariella Chirulli for linguistic review.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
