Abstract
To examine the ascending projections from the headache-related trigeminocer-vical complex in rats, biotinylated dextran amine (BDA) was injected into the ventrolateral dorsal horn of segments C1 and C2, a region previously demonstrated to receive input from sensory nerves in cranial blood vessels. Following injections into laminae I–II, BDA-labelled terminations were found bilaterally in several nuclei in the pons and the midbrain, including the pontine reticular nucleus, the parabrachial nuclei, the cuneiform nucleus and the periaqueductal grey. In the diencephalon, terminations were confined to the contralateral side and evident foremost in the posterior nuclear group, especially its triangular part, and in the ventral posteromedial nucleus. Following injections extending through laminae I-IV, anterograde labelling was more extensive. Some of the above regions are likely to be involved in the central processing of noxious signals of craniovascular origin and therefore putatively involved in mechanisms associated with primary headaches.
Introduction
The expression of pain in primary headaches is associated with activity in intracranial perivascular sensory nerve fibres, which originate in the trigeminal ganglion and project to the trigeminocervical complex in the brainstem. To understand the mechanisms of head pain in the pathogenesis of headaches, it is important to identify the central nervous system (CNS) regions that process nociceptive information from the trigeminovascular system (1). Accumulating evidence demonstrates that a major central target of some of the cranial perivascular nerves is located in a ventrolateral region of the dorsal horn extending from segment C3 rostrally into the caudal part of the spinal trigeminal nucleus (Sp5C) (2–9). This region, referred to as the ‘trigeminocervical complex’, is likely to be involved in headaches (6). We have previously demonstrated, using transganglionic tract tracing, that sensory nerve fibres in the superficial temporal artery, the superior sagittal sinus and the middle meningeal artery (MMA), all terminate in this region (7–9) (see also Table 1). Thin calibre primary afferent fibres [labelled with wheat germ agglutinin–horseradish peroxidase conjugate (WGA-HRP)] were found to terminate in laminae I–II, whereas larger calibre fibres [labelled with cholera toxin subunit B (CTb)] terminated in laminae III–IV.
CTb, cholera toxin subunit B; WGA–HRP, wheat germ agglutinin–horseradish peroxidase conjugate; Sp5C, caudal part of the spinal trigeminal nucleus.
Evidence from animal experiments indicates that many regions in the brainstem and diencephalon may be involved in the pathophysiology of migraine (10–13). Using positron emission tomography and functional magnetic resonance imaging (fMRI), several regions in the human brainstem, thalamus, hypothalamus and cerebral cortex have been observed to increase their activities during somatic nociceptive stimuli and in migraine attacks (14–17). It is unclear to what extent such activities depend on direct input from trigeminocervical neurons relaying nociceptive signals from cranial blood vessels. Therefore, to understand further the neural mechanisms behind primary headaches, it is important to know where neurons in this headache-related trigeminocervical complex project in the brainstem and in the thalamus. Such knowledge may also be of value in developing novel strategies to relieve headaches.
Ascending projections from the spinal dorsal horn have been extensively examined in different species using the high-resolution anterograde tracers Phaseolus vulgaris leucoagglutinin (PHA-L) and/or biotinylated dextran amine (BDA) (e.g. (18–24)). However, none of these studies has specifically addressed the ascending projections from the cranial blood vessel-receptive area in the rostral cervical spinal dorsal horn. Furthermore, our previous studies have demonstrated that the ventrolateral part of the C1 and C2 spinal dorsal horns contains the common termination sites of the sensory nerves from the superficial temporal artery, the superior sagittal sinus and the MMA (Table 1). We therefore in this study traced the brainstem and thalamic projections from this area, using BDA as anterograde tracer.
Methods
Surgery and tracer injections
The experimental procedures were reviewed and approved in advance by the Animal Care and Use Committee of Malmö/Lund. Thirty adult male Sprague–Dawley rats weighing 250–400 g were used. The rats were deeply anaesthetized with chloral hydrate (300 mg/kg, intraperitoneally) and mounted in a stereotaxic frame. A sagittal incision was made on the back of the neck and a laminectomy was performed to gain access to the C1 and C2 segments of the spinal cord. A glass micropipette (outside tip diameter 25–40 μm), filled with 10% BDA (Molecular Probes, Eugene, OR, USA), was positioned about 1.6 mm lateral to the midline of segments C1 or C2 and with an angle of 10–15° towards the sagittal plane. The tip of the micropipette was carefully inserted through a small opening of the dura mater into the ventrolateral part of dorsal horn, aiming at either the superficial (laminae I–II) or the deeper layers (laminae III–IV). The BDA was injected iontophoretically using an interrupted current (5 μA, 7 s on/7 s off) for about 20–25 min. Only one injection was made in each rat and all injections were placed on the right side. After a postoperative survival period of 1–3 weeks, the rats were deeply anaesthetized with sodium pentobarbital and terminated by transcardial perfusion. Following a rinse with 500 ml of 0.1 M phosphate-buffered saline (PBS), the rats were fixed by perfusion with 700 ml of 4% paraformaldehyde in PBS at room temperature for 30 min. The brain and the rostral part of the spinal cord were removed and placed in fixative overnight at 4°C. The tissues were then put into PBS containing 30% sucrose for 1–2 days.
Tissue processing
The brain and the rostral spinal cord containing the injection site were blocked and cut coronally into 40-μm-thick sections on a freezing microtome. Four sets of sections were collected. One set was mounted on slides and stained with thionin (Merck, Darmstadt, Germany). To visualize BDA labelling, one adjacent set of sections was rinsed in PBS with 0.3% Triton (PBS–T) and then incubated in Avidin–Biotin Complex Elite solution (Vector Laboratories, Burlingame, CA, USA; one drop of avidin plus one drop of biotin in 5 ml of PBS–T) for 4 h at room temperature or overnight at 4°C. After several rinses in 0.01 M PBS (pH 7.5), the sections were processed with the chromogen Vector SG (Vector Laboratories; three drops each of the chromogen and the H2O2 solution in 5 ml 0.01 M PBS) for 10–15 min until a Golgi-like black staining of fibres and terminals appeared. The sections were then rinsed and mounted on slides. The sections containing the injection sites were counterstained with neutral red to reveal the cytoarchitecture of the spinal cord. The sections were dried, cleared in xylene and cover-slipped with DPX Mountant (VWR International AB, Stockholm, Sweden).
Data analysis
Anterograde labelling in the brain and spinal cord sections was examined in a light microscope (Nikon Optiphot-2) and photographed with a Q-Imaging Micropublisher 5.0 digital camera. The photomicrographs were processed in Adobe Photoshop (version 9.0) to enhance the visibility of labelled profiles. Every third or fourth section containing BDA-labelled terminal-like varicosities was plotted using a computer-assisted plotting system (MD2 & MDplot system; Minnesota Datametrics Corp., Shoreview, MN, USA) connected to the microscope. The resulting plots were compared with the adjacent thionin-stained sections to define the cytoarchitectonic borders of different nuclei. The atlas of Paxinos and Watson (25) was used as a reference for terminology and delineation of nuclei.
Results
Injection sites
Of 30 rats that were subjected to BDA injections, nine displayed injections in the ventrolateral part of C1 (n = 2) or C2 (n = 7) dorsal horn (Fig. 1), a region that has been demonstrated to receive cranial blood vessel sensory nerve terminations in our previous work (7–9). Of these, seven displayed injections that were confined to either laminae I–II (n = 3) or laminae I–IV (n = 4) (Fig. 1A–C,E,F). Two additional rats had injections in laminae I–IV that also extended slightly into the adjacent lateral cervical nucleus (LCN; Fig. 1D). The remaining 23 rats had injections extending outside of these regions, and the ascending projections labelled in these rats were therefore not analysed in detail.
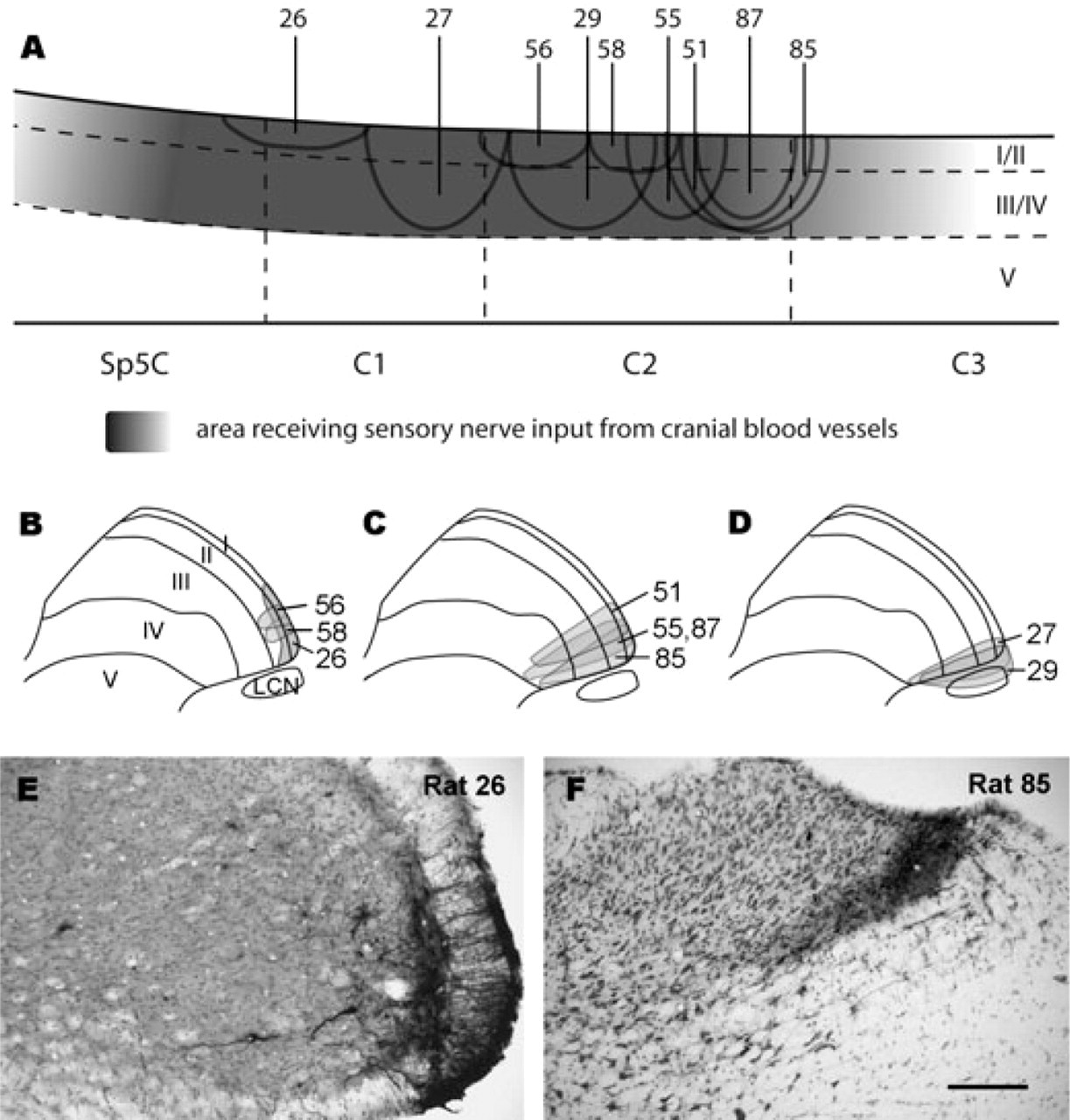
Drawings and photomicrographs depicting the biotinylated dextran amine (BDA) injection sites in the different cases. (A) Schematic drawing of a parasagittal section showing the longitudinal distribution of BDA injection sites in all nine rats. The shaded background in laminae I–IV indicates the area in which terminations from cranial vessel sensory nerves were previously detected (7–9). (B–D) Schematic drawings of the injection sites on transverse plane in laminae I–II (B; rats 26, 56 and 58), laminae I–IV (C; rats 51, 55, 85 and 87) and laminae I–IV with extension into the lateral cervical nucleus (D; rats 27 and 29). (E,F) Examples of injection sites in the ventrolateral dorsal horn, either limited to laminae I–II in rostral C1 (E; rat 26) or laminae I–IV in rostral C2 (F; rat 85). Scale bar = 200 μm in (F) (valid for E and F).
Anterograde labelling in the brainstem and thalamus
The focus of the present study was to search for putative vascular sensory centres in the brainstem and thalamus. In the medulla, terminal labelling was evident only in the ipsilateral trigeminal nuclear complex. Because we cannot exclude that at least a proportion of this labelling resulted from uptake of the tracer in trigeminal tract fibres passing or terminating within the injection site, this labelling was not analysed in detail.
Table 2 lists the distribution and relative abundance of BDA-labelled varicosities in the pons, the midbrain and the thalamus for all examined cases. The density and distribution of terminal labelling differed between the different cases. The cases with an injection including laminae I–IV generally showed more abundant labelling than those with an injection limited to laminae I–II (see also Fig. 2).
Locations and density of biotinylated dextran amine-labelled varicosities in the pons, the midbrain and thalamus from all analysed rats

+, contralateral labelling; ¤, ipsilateral labelling.
Density of terminal fibres: +/¤, sparse; ++/¤¤, moderate; +++/¤¤¤, dense.
SubC, subcoeruleus nucleus; PnC, caudal part of the pontine reticular nucleus; LPB, lateral parabrachial nucleus; MPB, medial parabrachial nucleus; CnF, cuneiform nucleus; DpMe, deep mesencephalic nucleus; PAG, periaqueductal grey; CIC, central nucleus of the inferior colliculus; ECIC, external cortex of the inferior colliculus; InG, intermediate grey layer of the superior colliculus; DpG, deep grey layer of the superior colliculus; APT, anterior pretectal nucleus; SG, suprageniculate nucleus; PoT, triangular part of the posterior nuclear group; Po, posterior nuclear group; PIL, posterior intralaminar thalamic nucleus; VPM, ventral posteromedial nucleus; VPL, ventral posterolateral nucleus; VM, ventromedial nucleus.

Plots of the distributions of labelled terminations in the pons, midbrain and thalamus in two individual cases after biotinylated dextran amine (BDA) injection into laminae I–II (A; rat 26) or laminae I–IV (B; rat 85). BDA was injected into the right side of the spinal dorsal horn. In the pons and midbrain, anterograde labelling is observed both ipsilaterally and contralaterally, whereas in the thalamus terminal labelling is observed only contralaterally. Note the more extensive labelling (including, for example, the suprageniculate nucleus and the ventral posterolateral nucleus) in rat 85 than in rat 26. Scale bar = 1 mm.
BDA injections in laminae I–II
The group with injections confined to laminae I–II includes three rats (rats 26, 56 and 58; Fig. 1A,B). In rat 26, the BDA injection was located in the C1 segment, included the lateral part of lamina I–II and had a rostrocaudal extension of about 1000 μm (Fig. 1A,B,E). Anterograde labelling in this case was relatively abundant and BDA-labelled terminal-like varicosities were evident in several nuclei in the pons, midbrain and thalamus (Fig. 2A). In the pons, relatively dense terminal labelling was detected in the lateral parabrachial nucleus (LPB), whereas labelling was sparser in its medial part (MPB) (Fig. 3A). Terminal labelling was also detected in the caudal part of the pontine reticular nucleus (PnC) and in the subcoeruleus nucleus (SubC). In the midbrain, moderate labelling was detected in the cuneiform nucleus (CnF), in the lateral and ventrolateral parts of the periaqueductal grey (PAG), in the intermediate grey layer of the superior colliculus (InG) (Fig. 3B–D) and in the deep mesencephalic nucleus (DpMe). Sparse labelling was evident in the anterior pretectal nucleus (APT). In the thalamus, dense labelling was detected in the triangular part of the posterior nuclear group (PoT) and in the ventral posteromedial nucleus (VPM) (Fig. 4A,B). Moderate labelling was seen in the posterior nuclear group (Po) and in the ventromedial nucleus (VM). Except for in the DpMe, labelling in the pons and in the midbrain was bilateral, whereas labelling in the DpMe and thalamus was confined to the side contralateral to the BDA injection.
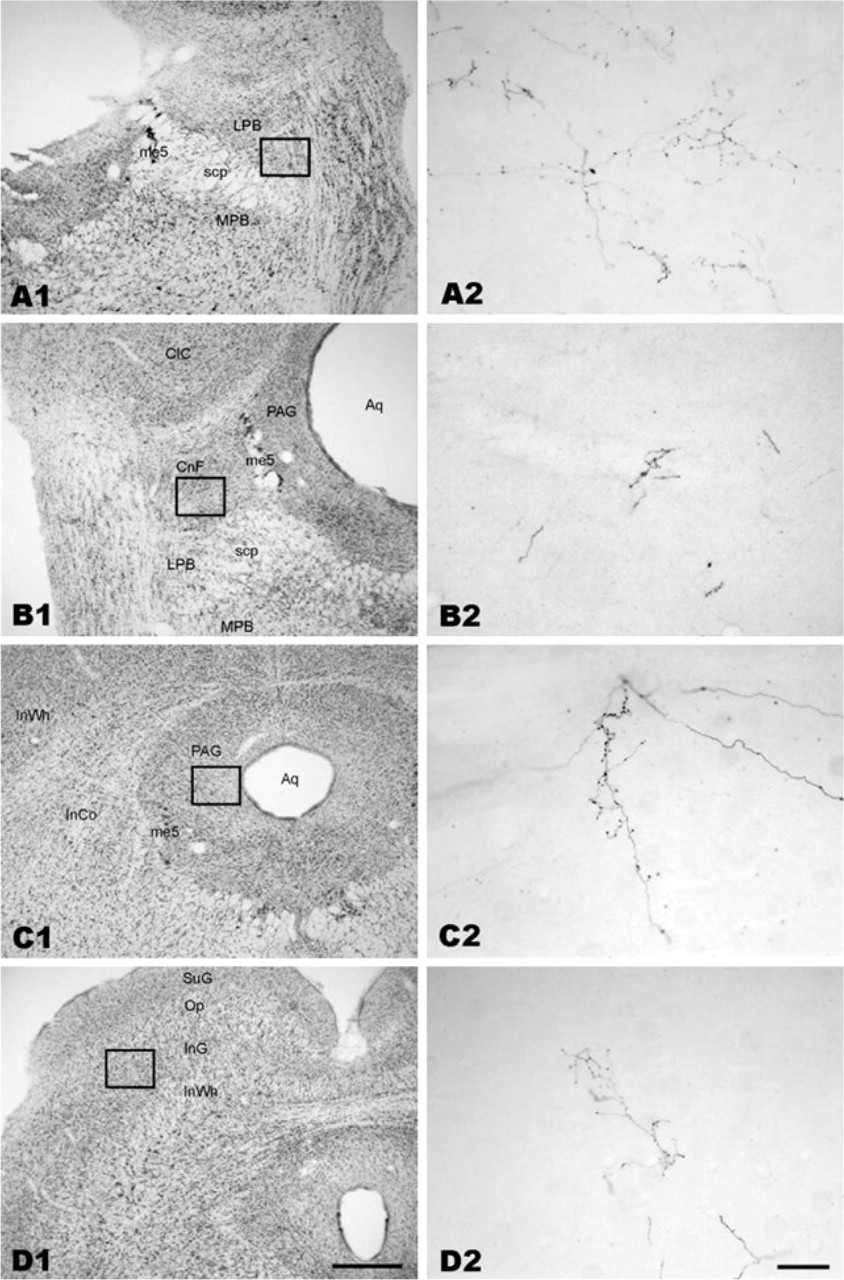
Photomicrographs showing anterograde biotinylated dextran amine (BDA) labelling in the pons and the midbrain after a BDA injection into laminae I–II of rat 26. (A1, B1, C1, D1) The cytoarchitecture of the selected brainstem sections (thionin staining). (A2, B2, C2, D2) are from the adjacent BDA-labelled sections and show the areas corresponding to those indicated by rectangles in (A1), (B1), (C1) and (D1). Scale bar = 500 μm in (D1) (valid for A1, B1, C1 and D1). Scale bar = 50 μm in (D2) (valid for A2, B2, C2 and D2).
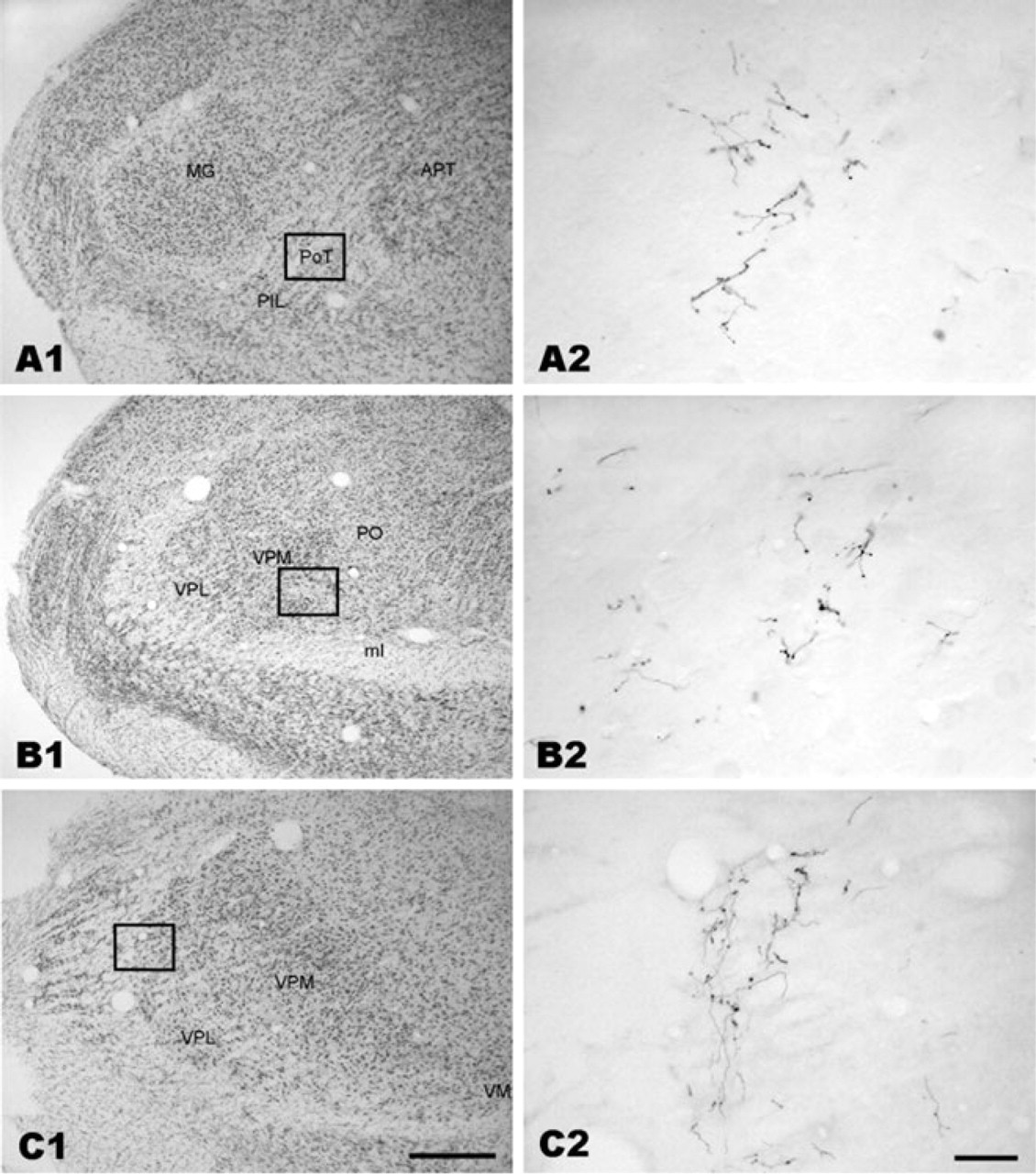
Photomicrographs showing anterograde biotinylated dextran amine (BDA) labelling in the thalamus. (A1, B1) (from rat 26) and (C1) (from rat 85) are from thionin-stained sections. (A2, B2, C2) (from adjacent sections) show BDA-labelled terminals in the posterior nuclear group, the ventral posteromedial nucleus and the ventral posterolateral nucleus in the areas corresponding to the rectangles in (A1), (B1) and (C1), respectively. Scale bar = 500 μm in (C1) (valid for A1, B1 and C1). Scale bar = 50 μm in (C2) (valid for A2, B2 and C2).
The BDA injection sites in rats 56 and 58 located in the rostral part of C2 and were smaller than that in rat 26. The injections included a small part of laminae I–II in the lateral part of the dorsal horn and had a rostrocaudal extension of about 600 μm (Fig. 1A,B). Terminal labelling in the pons and in the midbrain of these two cases was generally sparser than in rat 26, but had a similar distribution. In the thalamus of rat 56, anterograde labelling was detected only in the PoT, whereas in rat 58 terminal labelling was evident also in the VPM (Table 2).
BDA injections in laminae I–IV
The group with injections extending through laminae I–IV included four rats (rats 51, 55, 85 and 87; Fig. 1A,C,F). Rats 51, 55 and 87 had closely similar injection sites. They were all located in the ventrolateral part of the dorsal horn and formed a narrow strip across laminae I–IV, with a centre in lamina III (rat 55) or laminae III–IV (rats 51 and 87). The rostrocaudal spread of the injections in these three cases varied between 800 and 1200 μm. The injection site in rat 85 located in the lateralmost part of the dorsal horn but did not spread into the adjacent white matter. It was centred in lamina III and extended rostrocaudally for about 1500 μm. Rats 51 and 55 displayed closely similar anterograde labelling patterns in the brainstem and thalamus, labelling that was less abundant and comprised fewer nuclei than in rats 85 and 87. The nuclei in the brainstem and the thalamus that contained anterograde labelling in both rats 51 and 55 were the PnC, LPB, CnF, DpMe, APT, PoT and the posterior intralaminar thalamic nucleus (PIL) (Table 2). Besides those nuclei, the SubC and the MPB in the pons and the InG in the midbrain were labelled in rat 55 but not in rat 51, whereas the PAG in the midbrain and the suprageniculate nucleus (SG) in the thalamus were labelled in rat 51 but not in rat 55. In rat 51, labelling was detected only on the contralateral side. In rat 55, except for the LPB, the MPB and the SubC, which all showed bilateral labelling, anterograde labelling was confined to the contralateral side. Except for a few nuclei where anterograde labelling was relatively dense, like in the LPB of rat 55, most nuclei contained sparse to moderate labelling.
Rats 85 and 87 showed closely similar anterograde labelling patterns. All nuclei in the brainstem that were labelled in rats 51 and 55 were also labelled in rats 85 and 87, in which brainstem labelling was mostly bilateral. In addition, sparse to moderate labelling was detected in the deep grey layer of the superior colliculus (DpG). In the thalamus of rats 85 and 87 anterograde labelling was confined to the contralateral side, but was more extensive than in rats 51 and 55. In addition to the PoT and the Po, the VPM, VM, SG and PIL, also the ventral posterolateral nucleus (VPL) contained anterograde labelling in rats 85 and 87 (Fig. 4C). The density of terminal-like varicosities was moderate in most thalamic nuclei, although a few, like the VPL, displayed a high density of labelled terminations.
BDA injections in laminae I–IV that extended into the LCN
Two rats had injections forming wedge-shaped strips across lateral laminae I–IV in the caudal C1 (rat 27) or rostral C2 (rat 29), injections that also extended into the adjacent LCN (Fig. 1A,D). The rostrocaudal extent of the injections was 1000 and 1200 μm, respectively. As seen in Table 2, the overall labelling patterns and densities of terminal-like varicosities in these rats were similar to those in rats 85 and 87, except for some additional labelling evident only in rats 27 and 29. The nuclei containing labelled varicosities in the pons and the midbrain of rats 27 and 29 included the SubC, PnC, LPB, MPB, PAG, CnF, DpMe and InG. The DpG was labelled only in rat 29. The labelling was mostly bilateral. The central nucleus (CIC) and the external cortex of the inferior colliculus (ECIC) were found to contain anterograde labelling (confined to the contralateral side) only in rats 27 and 29. In the thalamus, anterograde labelling had distributions similar to those in rats 85 and 87 and was confined to the contralateral side.
Summary
Although the distribution and density of terminal-like varicosities differed between the different injection groups and to some extent also within groups, some common characteristics could still be extracted. For example, in the pons and midbrain, labelling was generally present bilaterally, although some nuclei showed more prominent labelling on the contralateral side. In the thalamus, terminal labelling was confined to the contralateral side in all cases. Overall, the labelling patterns in the pons and the midbrain were similar in cases with an injection that was limited to laminae I–II and in those where the injection extended through laminae I–IV. The PnC, LPB and MPB in the pons, the CnF, PAG, DpMe and APT in the midbrain contained moderate to dense terminal-like labelling irrespective of the injections including laminae I–II or laminae I–IV. In the thalamus, anterograde labelling was more extensive when tracer injections in addition to laminae I–II also included laminae III–IV. Thus, the PIL, VPL and SG contained labelling only when the injections extended into laminae III–IV. The PoT, Po and VPM were labelled after injections into both laminae I–II and laminae I–IV. The CIC and ECIC displayed anterograde labelling only when injections comprising laminae I–IV also involved the LCN (rats 27 and 29). Otherwise, the distributions of anterograde labelling in rats 27 and 29 were similar to those in rats 85 and 87, i.e. the two rats with the most extensive labelling following BDA injections comprising laminae I–IV but not the LCN.
Discussion
To our knowledge, this is the first study to examine ascending projections from the ventrolateral part of the C1/C2 dorsal horn, an area that has been demonstrated to receive primary afferent terminations from sensory nerves in several cranial blood vessels (2, 3, 7–9). A large number of nuclei in the brainstem and thalamus receive projections from this area, including the lateral and medial parts of parabrachial nucleus (PB), the CnF, PAG, DpMe, InG and APT in the brainstem, the Po, PoT and VPM in the thalamus. Although it appears unlikely that all of these nuclei are directly involved in the expression of primary headaches, at least some of them might play a role in the transmission and/or modulation of nociceptive signals from nerves in the cranial vasculature. The results may in addition provide a guide for further physiological and pathophysiological studies to explore the mechanism of headache.
Methodological considerations
Many different tracers have been used for anterograde labelling of nerve fibres in the CNS, including WGA-HRP, PHA-L, BDA and different fluorophores (22, 24, 26, 27). To obtain largely selective anterograde labelling, BDA and PHA-L are commonly used. There are several common features to these two tracers. First, both can be injected iontophoretically to produce small and well-defined injections sites. Second, anterogradely labelled structures display a ‘Golgi-like’ appearance, making it easier to differentiate between nerve fibres and terminal boutons. In comparison with PHA-L, BDA labelling usually has a lower failure rate and the staining procedure for BDA is simpler (28). We therefore decided to use BDA in the present work. An obvious drawback of the small injections obtained with BDA, as well as PHA-L, is that relatively few neurons will absorb the tracer, resulting in relatively sparse anterograde labelling, especially if the number of projection neurons in the injected area is already small. To compensate for this, several injections may be placed in the area of interest. However, this would also increase the risk of contaminating areas outside of that to be labelled (e.g. laminae I–II), and we therefore chose to make a single injection in each rat. Because of this, the anterograde labelling obtained in the present experiments probably underestimates the density of ascending projections from the ventrolateral dorsal horn at C1/C2. We chose not to use other tracers, such as WGA-HRP or CTb, in this study. The reason for this is that it is very difficult to obtain small and well-demarcated injections, similar to those of BDA, with WGA-HRP and CTb, and that neither WGA-HRP nor CTb will demonstrate fibres and terminals with the same clarity as BDA.
Our original intention was to cover the entire longitudinal ventrolateral part of the C1 and C2 dorsal horns through BDA injections in different cases. Unfortunately, most of the injections targeted to the C1 dorsal horn inadvertently spread outside of the intended region, leaving only two rats with appropriately located injection in the C1 dorsal horn. The sparseness of successful C1 injections may be regarded as a limitation of the present study. However, cranial vessel nerves display extended longitudinal termination areas, but restricted mediolateral ones. Thus, in an evaluation of ascending projections from the vessel nerve target areas, correct mediolateral locations of the injections is more essential than to include injections at all different rostrocaudal levels receiving cranial vessel nerve input.
We injected BDA into the ventrolateral part of the C1 or C2 dorsal horn as this region was found to be a primary target of afferent fibres in several cranial blood vessels (7–9). However, this does not mean that all neurons within this area are involved in headaches. Some neurons are likely to receive and convey inputs primarily or exclusively from different types of low-threshold receptors (for review, see (29)). Neurons in the spinal and trigeminal dorsal horns have different physiological properties, some responding to low-threshold inputs, others to high-threshold inputs and still others to both (wide-dynamic-range neurons) (30). Thus, the terminations detected in this study are likely to be involved in a variety of sensory and motor functions associated with the ventrolateral C1/C2 dorsal horn.
In two rats with BDA injections in laminae I–IV, the injections also extended into the LCN. The only nuclei found to contain anterograde labelling in these rats, but not in rats with injections confined to laminae I–IV, were the CIC and the ECIC in the midbrain. It therefore appears likely the anterogradely labelled fibres in these nuclei originate in the LCN. Ascending projections from the LCN has mostly been investigated in cats and other carnivores (e.g. (27, 31, 32)). However, the organization of ascending LCN projections in rats appears to be basically similar to those in carnivores, including abundant termination in the intercollicular area and the deep layers of the superior colliculus in the midbrain, and in the VPL of the thalamus (33). Anterograde labelling in these regions was also detected in this study, and it appears likely that at least a proportion of such labelling in rats 27 and 29 originates in the LCN.
The glass capillaries used to inject BDA were inserted into the dorsal horn through the thin rim of white matter dorsal to the ventrolateral dorsal horn. It is therefore possible that primary afferent fibres in the trigeminal tract descending to this level may have taken up the tracer. Thus, labelling in the ipsilateral nuclei of the spinal trigeminal tract may have included labelling of spinal trigeminal tract fibres and were therefore not analysed in detail.
Another possible source of contamination is ascending fibres in the dorsolateral funiculus. However, this possibility appears unlikely, as no spread of tracer to the dorsolateral funiculus was detected in any of our cases. Further, if ascending spinal fibre tracts had absorbed the tracer, we would have expected a certain amount of terminal labelling in the ipsilateral thalamus (34). We did not detect any such labelling in any of the cases.
Termination sites in the brainstem and thalamus
In agreement with previous studies on spinal and trigeminal projections in rat, cat and monkey (20, 23, 35–37), we found extensive anterograde labelling in the medial and lateral parts of the PB, CnF and PAG following BDA injections into the ventrolateral C1/C2 dorsal horn. The dominating thalamic targets following such injections were the Po, PoT and VPM (21, 22, 24). In comparison with cases where the injection was confined to laminae I–II, laminae I–IV injections (e.g. rats 85 and 87), as expected, resulted in both higher labelling density and labelling in additional areas of the brainstem and especially the thalamus. This additional labelling probably originates from deep dorsal horn projection neurons. In some nuclei, e.g. the VPL, anterograde labelling was evident only when the tracer injection extended into laminae III–IV (cf. (24)).
Several brainstem and thalamic nuclei likely to be important for the processing and modulation of nociceptive signals were devoid of anterograde labelling in our study. These include the nucleus of the solitary tract, the locus coeruleus and the raphe nuclei in the brainstem, and the intralaminar nuclei and the nucleus submedius (Sm) in the thalamus. Nor was any terminal labelling detected in any part of the hypothalamus. Except for the Sm, all the above-mentioned nuclei or regions have been shown to receive projections from the cervical or medullary dorsal horns in different species (18, 20, 21, 24, 30). The absence of anterograde labelling in these nuclei in our experiments may be due to either an absence of such projections from this particular part of the dorsal horn, or that our small injections failed to include neurons with such projections. With respect to the Sm, this nucleus appears to receive only sparse projections from the spinal dorsal horn in rats (18, 24), and trigeminal projections to the Sm originate primarily from neurons in the rostral ventral part of the caudal spinal trigeminal nucleus (38). The absence of anterograde labelling in Sm in the present experiments is therefore not unexpected.
Functional significance: possible relation with headache
As stated above, the anterograde labelling detected following BDA injections extending through laminae I–IV probably includes labelled projections from laminae III–IV neurons. The functional role of such projections is unclear, although the extensive input from low-threshold receptors to these laminae suggests that such projections mainly serve non-nociceptive roles. It has recently been demonstrated that dural nerves contain a substantial number of myelinated axons with response characteristics different from those of presumed nociceptors (39, 40). Such axons were presumably labelled following CTb application on the MMA in our recent study (9), and were found to terminate in the lateral parts of laminae III–IV in the rostral cervical spinal cord, thus in the area included by our present BDA injections into laminae I–IV. The functional role of these fibres has not been determined with certainty, but physiological data suggest that they could signal light mechanical stimulation of the dura during, for example, sudden head movement (40). Some of the projections presently detected following the laminae I–IV injections could be involved in the transmission of such signals to higher centre.
Which of the areas presently detected to contain anterograde labelling may serve a role in primary headache-related pain, and what is the particular role of each area? Considering that thin primary afferents from several cranial vessels terminate in laminae I–II (7–9) and that lamina I projection neurons serve a critical role in conducting nociceptive signals to higher centres (41), it appears likely that at least some of the brainstem and thalamic areas critical for primary headaches are to be found among those anterogradely labelled following our BDA injections into laminae I–II. As ascending projections from the superficial dorsal horn originate almost exclusively from neurons in lamina I (e.g. (42)), it appears most likely that the terminations we detected following our laminae I–II injections are from lamina I projection neurons. A number of the areas found to receive significant lamina I input from the lateral dorsal horn in segments C1 and C2 are discussed in further detail below. However, it should be stressed that also the areas found to receive less abundant lamina I input, and therefore not included in the sections below, may also serve important roles in primary headaches.
PAG
Accumulating evidence indicates that the PAG is able to inhibit and facilitate nociceptive responses. It receives input from nociceptive neurons in the spinal cord and sends projections to thalamic nuclei that process nociception (24, 36, 43, 44). The PAG is also a major component of a descending pain inhibitory system. Activation of this system inhibits nociceptive neurons in the dorsal horn of the spinal cord (for reviews, see (45, 46)). It responds not only to somatic pain stimulation (e.g. (16)) but also to cranial vasculature noxious stimulation (10–13). Animal experiments in rat, cat and monkey have shown that following electrical stimulation of the superior sagittal sinus or capsaicin injection into the cisterna magna, a dramatic increase in c-Fos expression is evident in the ventrolateral PAG (10, 12, 47). High-resolution fMRI has demonstrated that patients with episodic migraine and chronic daily headache have impaired iron homeostasis in the PAG during attacks, indicating a role of the PAG as a possible ‘generator’ of migraine attacks (48). Our present finding of bilateral projections to the lateral/ventrolateral PAG from the C1/C2 superficial spinal dorsal horn strongly supports the assertion that the PAG is an important locus for the generation and/or modulation of pain in primary headaches.
PB
The PB, especially its lateral part, has long been recognized for its involvement in the processing of somatic and visceral noxious information in the CNS ((49–53); for review, see (54)). It receives abundant input from spinal and trigeminal lamina I neurons (35–37, 53) that are driven by Aδ- and/or C-fibres (53, 54). After application of capsaicin into the cisterna magna, c-Fos immunoreactivity is significantly increased in the PB (12). Together with our present data, this finding indicates important roles for the LPB in primary headaches, possibly including a function as a ‘relay’ for nociceptive signals directed to the thalamus as well as to centres involved in homeostatic mechanisms (55).
CnF
Possible involvement of the CnF in the initiation of locomotion was recognized many years ago. Electrical stimulation of the CnF, and the adjacent pedunculopontine nucleus, induced controlled locomotion (56). Subsequently, a role more related to sympathetic vasomotor excitation was recognized for the CnF (57). Although a putative role in nociception was noticed two decades ago (e.g. (58, 59)), it was not until more recently that such a function of the CnF was further substantiated (16, 17, 60, 61). In man, fMRI has demonstrated significant activation in the CnF, PAG and PB, when visceral or somatic pain is induced by repeated electrical stimulation (16, 17, 61). Previous studies as well as our present findings support that the CnF receives afferent inputs from presumed nociceptive spinal and medullary lamina I dorsal horn neurons (20, 58, 62). The CnF also receives massive input from the PAG (63). The available evidence thus supports that the CnF is involved in mechanisms associated with pain, including those related to headaches.
Po and PoT
The Po has in different species been shown to receive massive spinal and trigeminal projections from both superficial and deep dorsal horn neurons (18, 20, 22, 24, 64). Po neurons usually have a large receptive field and are excited by more than one receptor type (65). The PoT is a caudal extension of Po, located between the medial portion of the medial geniculate nucleus and the APT. Gauriau and Bernard (66) have shown that 45% of the PoT units are nociceptive specific, 19% are nociceptive non-specific and 36% are tactile. They have also demonstrated that PoT neurons convey nociceptive signals from lamina I and deep dorsal horn neurons to the second somatosensory and insular cortices. As we observed in this study that the input to PoT includes afferents from the craniovascular-receptive zone in the dorsal horn, sensory signals from cranial vessels, including nociceptive signals, might be relayed through the PoT towards cortical areas possibly involved in some aspects of pain perception.
VPM
The VPM primarily relays sensory signals from the trigeminal system to the cerebral cortex. In rats, trigeminal projections to the VPM include those from the superficial laminae in the caudal spinal trigeminal nucleus (24). Considering the close relationship between the caudal spinal trigeminal nucleus and the rostral-most level of the spinal dorsal horn, it appears reasonable that lamina I input from the latter region may target the VPM rather than the VPL, as indicated by our present findings. Thus, the VPM also may serve as a thalamocortical relay for nociceptive signals originating in cranial blood vessels.
Footnotes
Acknowledgements
This project was supported by the Swedish Research Council (Project nos. 5958 and 14276), and by AstraZeneca's Headache, Pfizer AB and GlaxoSmithKline's Migraine stipends.

