Abstract
Tension-type headache is the most common type of primary headaches but no conclusive concept of pathophysiology exists. This may be due to a lack of an appropriate animal model. This study addressed the hypothesis that noxious neck muscle input induces central sensitization of orofacial sensorimotor processing. The effect of hypertonic saline injection into the semispinal neck muscle on the jaw-opening reflex (JOR) was investigated in anaesthetized mice (
Introduction
Although, tension-type headache (TTH) is the most common type of primary headaches the knowledge of its pathophysiology is tiny (1). The best documented abnormality in patients with TTH is increased pericranial myofascial tenderness (2, 3). With manual palpation of head and neck muscles increased pericranial tenderness was found both in patients with episodic and chronic TTH. It was demonstrated that the pericranial tenderness was positively associated with both the intensity and the frequency of TTH (4, 5). Total tenderness scores (TTS) recorded in patients on days with headache were significantly higher than the TTS recorded on days without headache. There was a significant difference between the TTS recorded in TTH patients during the pain free interval and in controls. The muscle hardness was significantly higher in patients on days without headache than in controls (6). Pain-pressure thresholds of the head and neck region in TTH patients and control subjects were recorded with an electronic algometer. The pain-pressure thresholds of the patient group were lower than those of the control group for sternocleidomastoid and trapezius muscles (7). Human experimental models of peripheral muscle pain demonstrated that myofascial tenderness can precede the headache (3). It is generally accepted that myofascial tenderness probably plays a key role in the pathophysiology of TTH. Thus, the international classification of headache disorders stresses the importance of increased neck muscle tenderness in TTH and recommends its evaluation in patients (1). Nevertheless, no conclusive concept of TTH pathophysiology exists. This may be due to a lack of an appropriate animal model.
An approved method to induce muscle pain under experimental conditions is the intramuscular bolus injection of a hypertonic (5%) saline solution (8). This model has been used extensively to characterize the sensory and motor effects involved in muscle pain because the quality of the induced sensation is very similar to acute clinical muscle pain and shows localized and referred pain characteristics. For example, muscle pain induced by a bolus injection of hypertonic saline into the human anterior tibial muscle maintained for a couple of minutes (8, 9). It is conceivable, that such an injection could evoke neck muscle pain and could mimic a pain state in animal experiments with a central facilitation of trigeminal brainstem neurons that corresponds to a TTH attack in patients.
The jaw-opening reflex (JOR) is an established model for the investigation of sensorimotor processing in the orofacial region and the trigeminal brainstem (10–13). This brainstem reflex can be elicited by electric, thermal and mechanical stimulation of the orofacial region (11, 14–16). Primary trigeminal afferents synapse on excitatory sensory neurons of the spinal trigeminal complex. These neurons project ipsi- and contra-laterally to excite digastric motor neurons (17). Thus, the reflex seems to be an adequate model to investigate alterations of excitability in sensory trigeminal neurons with convergent afferent input from different orofacial tissues. So far, it has not been proved whether noxious input from neck muscles has some access to neurons of the JOR reflex arc, but noxious stimulation of cervical muscles and the supplying nerves excited trigeminocervical sensory neurons suggesting convergent input from deep tissues (18, 19).
Consequently, we investigated the effect of a hypertonic saline injection into a neck muscle on the jaw-opening reflex in anaesthetized mice in order to test the hypothesis that noxious input from neck muscles induces a central sensitization of orofacial sensorimotor processing.
Methods
The jaw-opening reflex (JOR) was investigated in 27 adult male C57BL/6 mice (about 12 weeks old; 22–30 g; Charles River Laboratories, Germany). All procedures received institutional approval from the local ethics committees of the university of Aachen (ref. no. 50.203.2-AC 15, 16/03). The principles of laboratory animal care and use of laboratory animals (European Communities Council Directive of November 24, 1986(86/609/EEC)) were followed. All efforts were made to minimize animal suffering and to use only the number of animals necessary to produce reliable scientific data.
The detailed description of anaesthesia, surgery and electrophysiological recording in mice recently has been published (20). The mice were anaesthetized by an initial intraperitoneal injection of a 0.5% pentobarbital sodium salt solution (Sigma-Aldrich, Germany) with a dose of 70 mg/kg. Depth of anaesthesia was checked by ensuring that noxious pinch stimulation (blunt forceps) of the hindpaw, the forepaw and the ear did not evoke any sensorimotor reflexes. When the mouse was sufficiently deeply anaesthetized the skin of the throat was carefully shaved and Lidocaine gel (AstraZeneca, Germany) was applied to induce local anaesthesia. Dexpanthenol eye ointment (Roche, Germany) was applied to the cornea of both eyes to protect it from drying. The right external jugular vein was catheterized for continuous administration of a 1% methohexital sodium salt solution (Lilly, Germany) with a dose of 60 mg/kg/h corresponding to a flow rate of 0.15 ml/h for a 25 g mouse. A pair of Teflon-coated stainless steel wires (140 µm diameter) was inserted into the right anterior digastric muscle (Dig) to record electromyographic activity (EMG) via a differential amplifier. After tracheotomy, animals were placed in a stereotaxic frame and were artificially respired with a stroke volume of about 0.5 ml and about 180 strokes per minute for the duration of the experiment. The body core temperature was maintained at 37.5°C with a heating blanket and a fine rectal thermal probe (FMI, Germany). One platinum needle electrode each (300 µm diameter) was subcutaneously inserted into the left forepaw and the right hindpaw to record the electrocardiogram (ECG) via a differential amplifier. Two stainless steel needle electrodes (150 µm diameter) were longitudinally inserted into the tongue (parallel, 2 mm distance) in order to apply electric stimuli. The oral cavity was filled up with white Vaseline (Riemser, Germany) to protect the oral mucous membrane from drying. The right semispinal neck muscle or the tensor muscle of fascia lata of the left hindpaw were carefully exposed. A 27G injection cannula needle (0.4 mm diameter) was inserted into the muscle belly. The needle was connected via thin tubing to a glass microsyringe (1 ml) that was fixed into a CMA/102 microdialysis pump (CMA Microdialysis, Sweden). Intramuscular injections of 20 µl hypertonic (5%) or isotonic (0.9%) saline were performed with a flow rate of 20 µl/min. After the surgery and the placement of all electrodes the anaesthetized animal had a rest for at least one hour. In this time period the level of anaesthesia (reflexes) and the heart rate were routinely checked and documented, and depth of anaesthesia was maintained.
All electric signals (EMG, ECG) were recorded by bioamplifiers and led into a data collection system (CED micro 1401) and a personal computer to compile waveform files using the Signal software program (CED, Cambridge, UK).
The jaw-opening reflex was elicited by rectangular electric pulses of 500 µs duration with a stimulation frequency of 0.1 Hz (Fig. 1a). The electric threshold of the JOR was determined by applying increasing and decreasing stimulus intensities from 0 to 2 mA in steps of 100 µA. The lowest stimulus intensity that just evoked a reflex response was defined as the JOR threshold (IJOR). The test stimulus intensity was adjusted to about 125% of the IJOR. The JOR was evoked in blocks of 8 stimuli each (Fig. 1b). These blocks were repeated every 5 min. After the three baseline JOR blocks either hypertonic saline was injected into the right semispinal neck muscle or the left tensor muscle of fascia lata (hypertonic neck

Pattern and stimulus paradigm of the jaw-opening reflex in mice. (a) Electric stimulation (rectangular pulse, 500 µs duration) of the tongue elicited a single sweep of the jaw-opening reflex (JOR) that was electromyographically (EMG) recorded by wire electrodes in the anterior belly of the digastric muscle in an anaesthetized mouse. The EMG is presented in arbitrary units (a.u). (b) The JOR was evoked in blocks of 8 stimuli each with an interstimulus interval of 10 s. The blocks were repeated every 5 min. After the three baseline JOR blocks either hypertonic saline (20 µl, 5%) was injected into the right semispinal neck muscle or the left tensor muscle of fascia lata or isotonic saline (20 µl, 0.9%) was injected into the right semispinal neck muscle. After the intramuscular injection the JOR blocks were repeated every 5 min for one hour.
The onset latency, the duration and the integral of the JOR were analysed in each single sweep. Arithmetic mean and standard error were calculated (mean ± sem). One and two way repeated measures analyses of variance were applied in order to test statistically significant differences within and between groups (hypertonic neck, hypertonic hindpaw, isotonic neck). Multiple comparison procedures were performed by the Holm-Sidak post hoc test. The Holm-Sidak Test can be used for both pair wise comparisons and comparisons vs. a control group. It is more powerful than the Tukey and Bonferroni tests and, consequently, it is able to detect differences that these other tests do not. It is recommended as the first-line procedure for pair wise comparison testing. The JOR thresholds before and after intramuscular injection were analysed by the paired
Results
The JOR was elicited in all 32 experiments by electric tongue stimulation with a threshold stimulus intensity of IJOR = 663 ± 47 µA (mean ± sem) (Fig. 1a). Based on the stimulus-response-functions the stimulus intensity (IS) was adjusted to 828 ± 59 µA corresponding to 125 ± 3% of the JOR threshold.
In the control experiments the JOR was elicited before and after an injection of isotonic saline into the right semispinal neck muscle in 11 mice. Before the intramuscular injection the IJOR was 605 ± 68 µA, the JOR onset latency and duration were 5.2 ± 0.2 ms and 4.4 ± 0.3 ms (mean ± sem). According to the one way repeated measures analysis of variance (factor: time) the integral (−7.3 ± 8.5%), the latency (5.5 ± 0.4 ms) and the duration (4.2 ± 0.3 ms) of the JOR were not affected by the injection (Fig. 2). The IJOR of 560 ± 50 µA as measured 60 min after injection was not statistically different from the baseline threshold as analysed by the paired

Injection of isotonic saline into the semispinal neck muscle did not affect the jaw-opening reflex. (a) The grey and black graphs represent averages of eight rectified reflex sweeps 5 min before (grey, pre) and 60 min after (black, post) isotonic saline injection into the right semispinal neck muscle in one mouse. Both averages are nearly congruent. The EMG activity is given in arbitrary units. (b) Integral (○), latency (▵) and duration (□) of the jaw-opening reflex from 15 min before to 60 min after injection of 20 µl isotonic saline into the right semispinal neck muscle in 11 mice. The changes from baseline (time points − 15, − 10, − 5) are expressed as arithmetic mean and standard error. The reflex remained unchanged.
In 11 mice hypertonic saline solution was injected into the right semispinal neck muscle. Under baseline conditions the JOR threshold was 664 ± 105 µA, the JOR latency and duration were 5.1 ± 0.1 ms and 4.6 ± 0.2 ms (mean ± sem). The one way repeated measures analysis of variance (factor: time) documented significant changes of the JOR within one hour after the intramuscular injection: the reflex integral increased by 94.5 ± 31.2% (

Injection of hypertonic saline into the semispinal neck muscle induced a facilitation of the jaw-opening reflex. (a) The grey and black graphs represent averages of eight rectified reflex sweeps 5 min before (grey, pre) and 60 min after (black, post) hypertonic saline injection into the right semispinal neck muscle in one mouse. The reflex was clearly facilitated. The EMG activity is given in arbitrary units. (b) Integral (○), latency (▵) and duration (□) of the jaw-opening reflex from 15 min before to 60 min after injection of 20 µl hypertonic saline into the right semispinal neck muscle in 11 mice. The changes from baseline (time points − 15, − 10, − 5) are expressed as arithmetic mean and standard error. The asterisks code the level of significance in comparison to the baseline blocks calculated by the Holm-Sidak post hoc test (∗
Hypertonic saline solution was injected into the left tensor muscle of fascia lata in 10 mice. Before the injection under baseline conditions latency and duration of the JOR were 5.2 ± 0.3 ms and 4.3 ± 0.2 ms. After the intramuscular injection the JOR integral (−8.7 ± 13.3%) and the latency (5.4 ± 0.5 ms) remained statistically unchanged, whereas the duration decreased by − 9.4 ± 6.7% to 4.0 ± 0.4 ms (
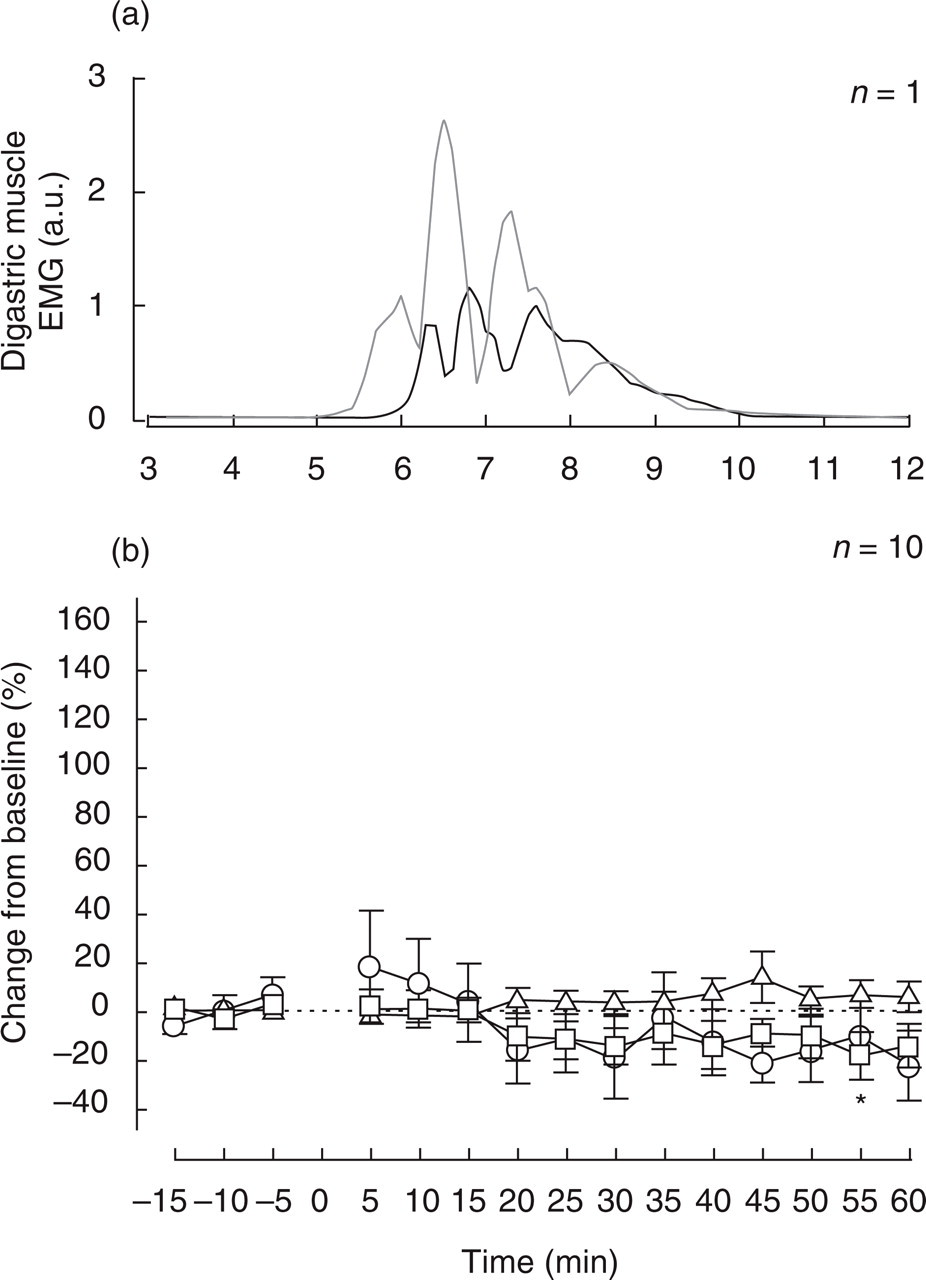
Injection of hypertonic saline into the left tensor muscle of fascia lata induced a slight reduction of the reflex duration. (a) The grey and black graphs represent averages of eight rectified reflex sweeps 5 min before (grey, pre) and 60 min after (black, post) hypertonic saline injection into the left tensor muscle of fascia lata in one mouse. The reflex slightly decreased. The EMG activity is given in arbitrary units. (b) Integral (○), latency (▵) and duration (□) of the jaw-opening reflex from 15 min before to 60 min after injection of 20 µl hypertonic saline into the left tensor muscle of fascia lata in 10 mice. The changes from baseline (time points − 15, − 10, − 5) are expressed as arithmetic mean and standard error. The asterisks code the level of significance in comparison to the baseline blocks calculated by the Holm-Sidak post hoc test (∗
The two way repeated measures analysis of variance (factors: kind of intramuscular injection, time) for all three experimental groups (isotonic neck, hypertonic neck, hypertonic hindpaw) showed significant differences in the mean JOR integrals (
Discussion
Hypertonic saline injection into the neck muscle induced a sustained facilitation of the JOR for at least one hour with increased integral and duration and decreased latency. The reflex threshold decreased to 61% after injection. Isotonic saline injection into the neck muscle or hypertonic saline injection into a hindpaw muscle did neither change the reflex integral nor the threshold.
The JOR is an established model for the investigation of somatosensory and nociceptive processing in the orofacial region and the trigeminal brainstem (10–13). This markedly convergent brainstem reflex can be elicited by electric, thermal and mechanical stimulation of the orofacial region like the lips, the tongue, the tooth pulp, the orofacial skin, the cornea and the meninges (11, 14–16). In the present study the JOR threshold was about 660 µA. The thresholds for eliciting the JOR by incisor tooth pulp and face skin stimuli (200 µs duration) were 600 µA and 900 µA (16). With upper lip (100 µs) and vibrissal pad (200 µs) stimulation thresholds were 800 µA and 1.5 mA, respectively (21). The somewhat longer stimulus duration in the present study may account for the lower JOR threshold stimulus intensity. The JOR is under strong descending modulatory control from the nucleus submedius (16), the striatum (22), the periaqueductal grey, the nucleus raphe magnus (10, 20), and the anterior pretectal nucleus (23). The JOR seems to be an adequate model to investigate long-term modulatory effects on orofacial somatosensory and sensorimotor processing in living animals.
A potentiation of the JOR by noxious input from the semispinal neck muscle may be due to excitatory effects on somatosensory neurons of the brainstem participating in the JOR reflex arc. As mentioned above the JOR can be elicited by stimulation of multiple orofacial tissues. So far, it has not been proved whether noxious input from the neck muscle has some access to neurons of the JOR reflex arc, but the convergent nature of medullary dorsal horn neurons with extensive input from deep tissues suggests such an access (18). Therefore, noxious input from the semispinal neck muscle may slightly depolarize neurons of the JOR reflex arc causing a central facilitation (24) of the tongue-evoked JOR. Thus, heterosynaptic convergent input from neck muscle and tongue afferents onto trigeminal brainstem neurons may account for the main result of the present study. Noxious input from a hindpaw muscle did not facilitate the JOR but slightly decreased its duration. This may be due to a lack of convergent access from hindpaw muscle afferents to the neuronal network of the JOR. The slight inhibitory effect on the JOR by remote noxious input from the hindpaw may be mediated by the diffuse noxious inhibitory control system (25, 26).
Hu et al. (27) carried out a study in rats to determine if EMG activity of jaw and neck muscles could be influenced by injection of mustard oil into deep paraspinal tissues surrounding the C1-3 vertebrae. Mustard oil injection significantly increased the digastric muscle activity in the majority of animals. The early phase of increase in EMG activity was usually initiated about 20 s following injection and lasted about 1.6 min. The second phase occurred about 11 min later and lasted 11 min. The site of mustard oil injection in the study was quite unspecific and not restricted to one well-defined neck muscle like in our study (right semispinal neck muscle). In contrast to hypertonic saline solution, mustard oil acts as a very strong inflammatory irritant. In our study nociceptive afferents in the semispinal muscle should be activated but no inflammation should be induced. In a recent paper Ashina et al. (28) demonstrated that tender points are not sites of ongoing inflammation in patients with chronic tension-type headache. Consequently, we decided not to apply mustard oil. Furthermore, spontaneous EMG activity usually was not induced after hypertonic saline injection in our study. Even if the effect of mustard oil on jaw muscle activity has some impact on the results of our study, the duration of the effect of about 20–25 min is definitely shorter than the sustained potentiation of the JOR in our study. Hypertonic saline (100 µl, 5%) injected into the mid-region of the masseter muscle in lightly anaesthetized rats effectively elicited hindpaw shaking behaviour for about 2 min (29). This may be an adequate behav-ioural assessment of craniofacial muscle pain. Ro & Capra (30) recorded neurons in the trigeminal subnucleus interpolaris with afferent input from masseter muscle in cats. Following intramuscular injection of 250 µl 5% hypertonic saline solution about half of the cells showed a significant modulation of either the resting discharge and/or the responses to innocuous mechanical stimulation on their cutaneous receptive fields. The most common response to the injection was an induction or facilitation of resting discharge in nociceptive wide-dynamic-range (13/34 cells) and muscle-high-threshold neurons (8/16 cells) which declined as an exponential decay function, returning to preinjection level within 3–4 min after the injection. No low-threshold-mechanoreceptive neuron was facilitated by hypertonic saline injection (30). Electrophysiological recordings of the suprascapular nerve with afferent input from the supra- and infraspinatus muscles in rabbits showed that all 26 nerve fibres in the supraspinatus muscle belonged to group III afferents. Six of 8 units in the infraspinatus muscle were also group III fibres (31). These data suggest that an injection of hypertonic saline into skeletal muscles preferentially activates nociceptive group III fibres. In humans intramuscular injections of hypertonic saline into a jaw-closing muscle increased muscle activity only within 30–60 s after injection (32). Electromyographic recordings of the semispinal neck muscle in three mice showed that intramuscular injection of hypertonic saline induced a slight transient activation of the muscle in one experiment and had no excitatory effect on muscle activity in 2 of 3 experiments (Fig. 5). Therefore an appreciable activation of muscle fibres and muscle spindle afferents by intramuscular hypertonic saline injection may transiently occur for a short time period but does certainly not account for a prolonged muscle pain, tenderness and reflex facilitation.
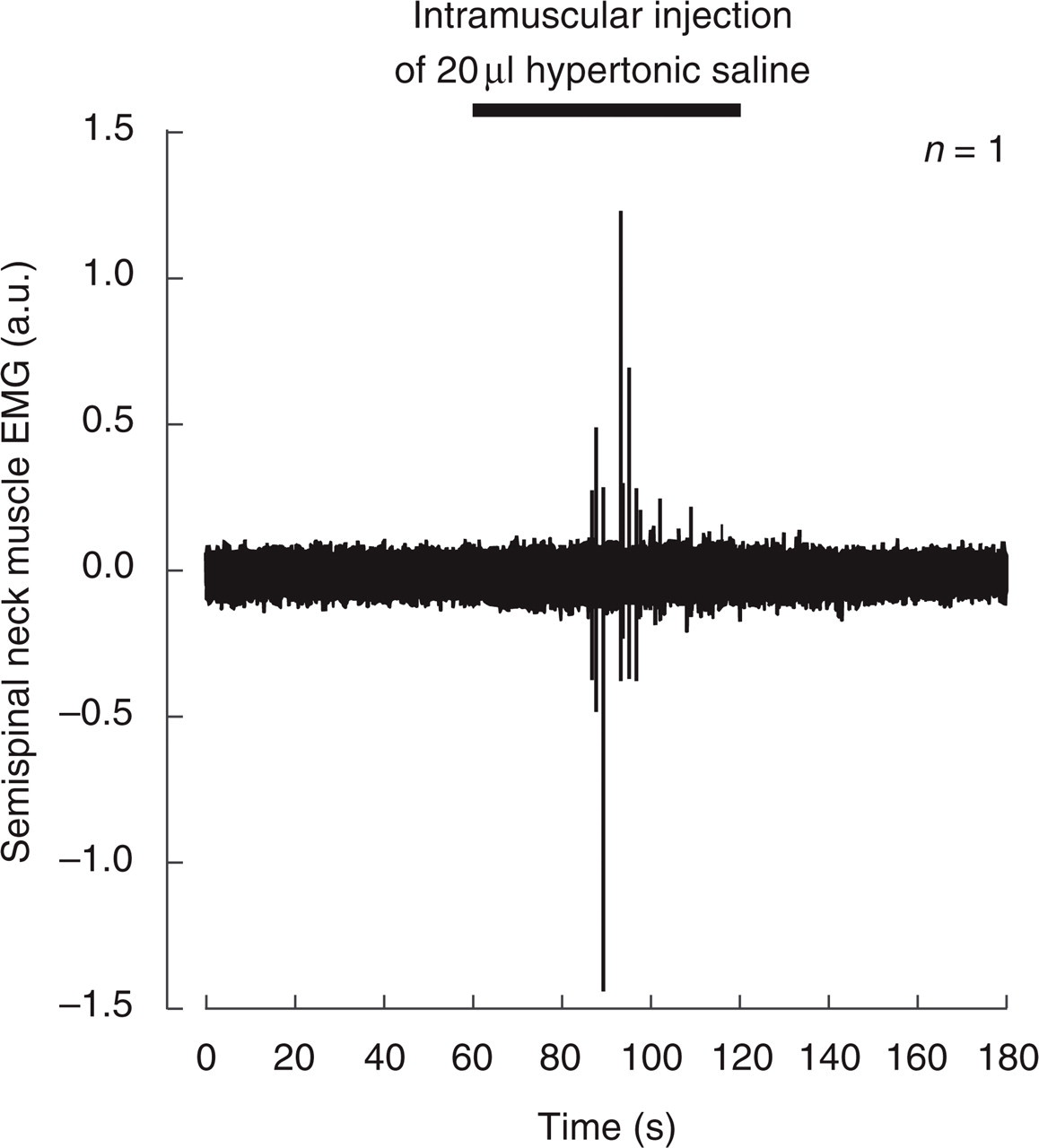
Injection of hypertonic saline into the semispinal neck muscle induced transient muscle activation. Electromyographic (EMG) recording of right semispinal neck muscle activity in one mouse before, during and after an intramuscular injection of 20 µl hypertonic(5%) saline. The EMG recording was performed by intramuscular wire electrodes. The EMG activity is given in arbitrary units. During the injection of hypertonic saline some phasic activation (spikes) of the semispinal muscle was detected.
A bolus injection of 1 ml 5% hypertonic saline into the anterior tibial muscle or the soleus muscle in healthy volunteers instantaneously evoked muscle pain for a duration of about 7 min (9). In the present study the JOR started to be potentiated within 15 min after the injection of hypertonic saline into the neck muscle and this potentiation sustained for at least one hour without any tendency to decrease. Thus, the long-lasting potentiation of the JOR was called long-term potentiation (33). In contrast to former animal and human studies the present effect of the JOR in mice represents a long-term potentiation but not a transient facilitation of the JOR. A psychophysical study in healthy volunteers tested the effect of an injection of bradykinin, serotonin, histamine and prostaglandin E2 into the trapezius muscle on pain and tenderness in the trapezius muscle and the shoulder and neck region (34). This mixture produced a prolonged moderate pain (25 min) and mild tenderness (60 min) without inducing unacceptable side-effects. The administration of this infusion mixture into the trapezius muscle induced a local, prolonged and mild to moderate tenderness in the temporal region and on the finger of TTH patients, too (35). The response to this mixture tended to be increased in patients as compared to the control group. The difference in the effect duration of intramuscular injections comparing spinal and orofacial muscles may be due to some special properties of the neck muscles. Minaki et al. (31) found in rabbits that the shoulder contained more nociceptive units than the knee and facet joints and suggested a higher sensitivity of the shoulder area to noxious stimulations. Muscular electric pain thresholds and pressure-pain thresholds were significantly lower in the trapezius than in the anterior tibial muscle in healthy human volunteers (36). The authors concluded higher pain sensitivity in pericranial muscles.
Finally, the long-term potentiation of the sensorimotor jaw-opening reflex by noxious input from a neck muscle in anaesthetized mice may be an appropriate model to investigate neck muscle nociception. This study may be the origin for a future animal model of tension-type headache.
Footnotes
Acknowledgements
This research project was supported by grants of the German Headache Consortium (Federal Ministry of Education and Research, 01 EM 0117, project A2) and the Interdisciplinary Centre for Clinical Research BIOMAT of the Medical Faculty of the University of Aachen.
