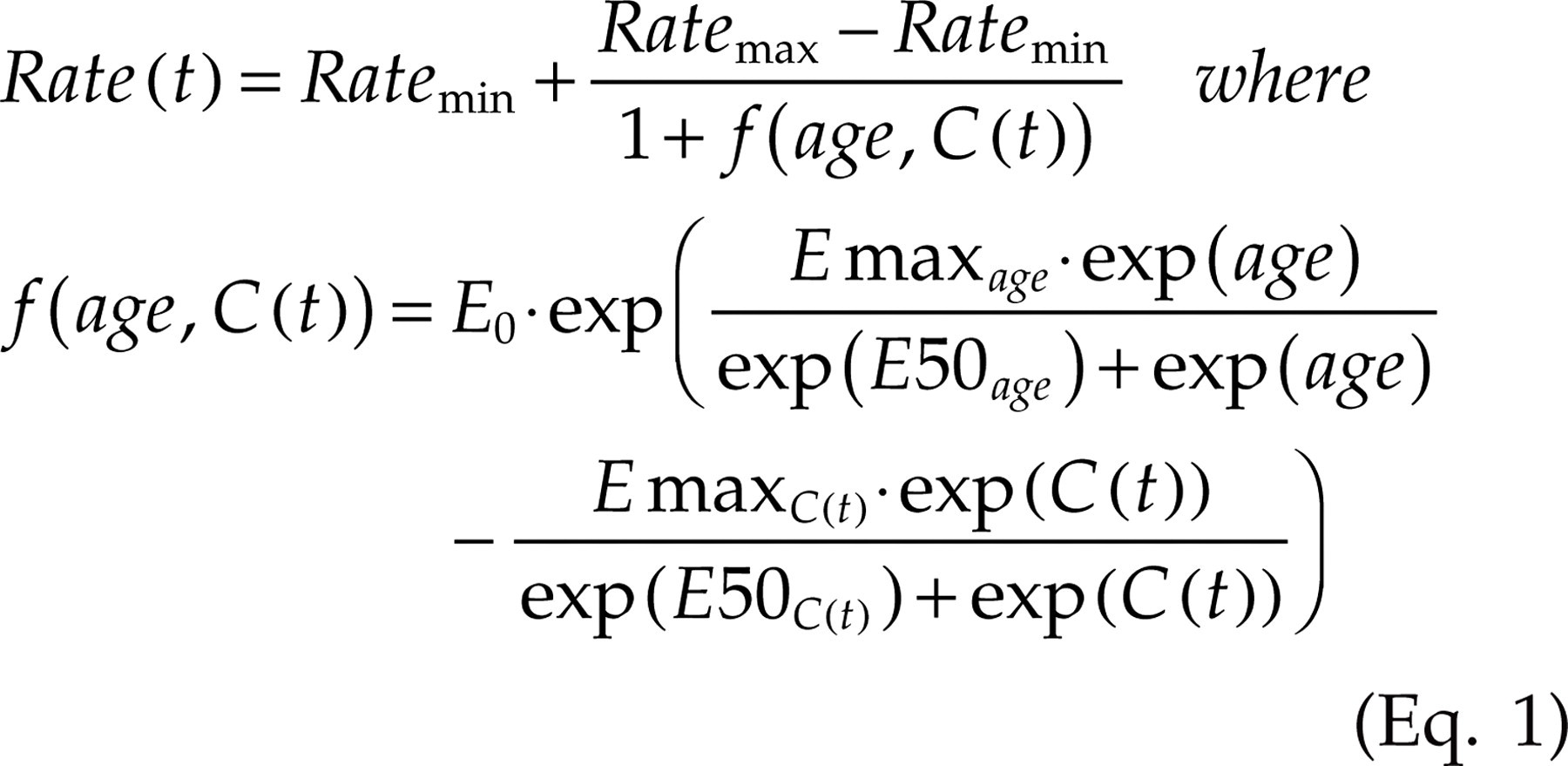Abstract
In migraine, headache severity varies with age. As a consequence, the effectiveness of medication may also depend on a patient's age. The purpose of this study was to assess the combined effect of age and drug treatment on headache characteristics. Using data from clinical trials of sumatriptan in adolescents and adults, we show how the interaction between age and drug exposure can be parameterised as a covariate on a Markov model that describes the decline of headache severity over three clinically defined stages (no relief, relief and pain-free status). The model explains important clinical observations: (i) the rates at which the pain relief and pain-free status were attained were found to be inversely related to age; (ii) in placebo-treated patients, the mean transit time from ‘no relief’ to ‘relief’ is 3 h for young adolescents and increases to 6 h for patients aged ≥ 30 years; and (iii) sumatriptan reduces the transit time to 2 h, irrespective of age. These findings indicate that the therapeutic gain over placebo increases with age. Prospective studies of antimigraine drugs should take this relationship into account when extrapolating efficacy data from adults to adolescents.
Introduction
Migraine affects all ages. Although its peak prevalence is in the third to fifth decades (1), migraine is the most common cause of primary headache in children and adolescents, with prevalence between 2.7 and 10.6% (2). The characteristics of childhood migraine differ somewhat from migraine in adults. It features a lower incidence of migraine attacks with aura and headache that is bilateral instead of unilateral. As regards the dynamics, the attack frequency is lower and the duration shorter in the young.
The differences between the two age groups extend to the acute treatment of migraine attacks with ‘triptan’ agents (serotonin 1B/D receptor agonists). In particular, it has been difficult to demonstrate superiority of sumatriptan over placebo treatment. The main problem with randomised placebo-controlled trials of sumatriptan in children and adolescents is the high placebo response rate (3). This may partially reflect the intrinsically shorter duration of attacks, yet it is also known that the anticipation of a treatment effect is especially important in paediatric pain studies.
An inverse relationship between placebo response rate and age has been demonstrated in recent papers (1–3). However, it is not clear whether the response rate following active treatment can be attributed to the underlying age-dependent placebo response or if it is independent of age. The matter of age dependency is essential if one intends to design new clinical studies and predict the efficacy of new dosage forms and dosing regimens.
According to current data analysis methodology for migraine trials, the fraction of the antimigraine response in each age category that can be attributed to triptan treatment may be found by simply subtracting the placebo response rate from the response rate after drug treatment. Comparison of the remainders may then explain whether the treatment effect varies with age. However, this approach is based on the assumption that placebo effect and active treatment effect determine the response in an additive manner. In reality, these effects need not be independent, but may share common pathways. In short, the response to treatment is expected to result from a certain degree of interaction between placebo and drug effects.
Clearly, for any evidence of interaction to be extracted from clinical data, a technique is needed that accounts for the presence of non-linearity. Recent publications have reviewed the experimental and statistical issues of interaction and confounders in observational studies (4–6), but no clear examples are available in clinical epidemiology literature that describe a methodology for covariate interaction taking into account pharmacokinetics. Models have been used to describe treatment effect and allow for appropriate adjustment between covariate and drug effect at treatment level (6). Nevertheless, these methods do not explicitly model drug exposure or discriminate between drug-specific and disease-specific parameters.
Recently the usefulness of Markov chains in the analysis of migraine data has been demonstrated (7–9). The aim of this analysis was to develop a Markov model to quantify the relationship between drug exposure, patient age and headache response in migraine attacks, allowing for the interaction between placebo and drug effects. Model parameters are estimated from clinical efficacy studies in adolescents and adults treated with placebo and various oral doses of sumatriptan.
The current investigation illustrates how Markov chain methodology can accommodate the analysis of ordinal categorical endpoints (i.e. headache intensity scores) used in clinical trials and account for demographic and treatment explanatory variables affecting the time course of response to antimigraine drugs. This approach can be applied to other disease conditions in which non-stationarity is observed during treatment (i.e. those characterised by a systematic change in either mean or variance over time).
Materials and methods
Markov model
Migraine response in randomised clinical trials is assessed in terms of the fraction of patients attaining ‘pain relief’ and ‘pain-free’ status (10). Within a migraine attack, the transitions from one response status to the next can be represented by the state-to-state transitions of a Markov chain (9). The dynamics of the transitions in a Markov chain are governed by the Markov property, which states that only the current state determines what the next state will be. The rates of transition may be expressed as constants or as functions of explanatory variables. The current model consists of a three-state Markov chain representing disease dynamics over three clinical states (no relief, pain relief, pain free) (Fig. 1).

Diagram of the Markov model. The circles represent the clinical states ‘no relief’, ‘relief’ and ‘pain-free’. The states are connected to the observed headache severity scores. Bold arrows indicate forward transitions. The associated transition rates are functions of both sumatriptan concentration and patient age.
However, in trials of antimigraine drugs the observed variable is not the clinical state, but rather the headache intensity score, which is defined on a four-point scale. In this scale, scores 0, 1, 2 and 3 indicate ‘no pain’, ‘mild pain’, ‘moderate pain’ and ‘severe pain’, respectively. In order to relate the three clinical states to the headache intensity scores (Fig. 1), the states are coupled to a layer that gives the expected headache scores for each of the states. The expected distributions of the scores over the states can be deduced from clinical guidelines (10), which state that pain relief is defined as a decrease in headache from score 3 or 2 to 1 or 0, whereas pain free is defined as a decrease from a score 3 or 2 to 0. Therefore, the distribution of the first Markov state (representing ‘no relief’) consists of scores 2 and 3, the second state (similar to pain relief) will contain score 1, and the last state (pain free) will be occupied by score 0.
Age and treatment effects
As shown in Fig. 1, the Markov chain allows transitions in two directions: forward (towards ‘pain-free’ status) and backward (towards ‘no relief’). Forward transitions reflect resolution of the attack either through the natural course of the disease or through placebo treatment. Measures of drug exposure such as plasma drug concentration have been shown to be determinants of these rates (7–9). However, as the effect of placebo treatment has been shown to be strongly related to the age of the patient, age is also likely to affect the rate of transition towards pain-free status.
On the other hand, following drug treatment, the response does not seem to depend on patient age, as response rates are comparable between adolescents and adults.
This complicated interaction between age and oral sumatriptan treatment was captured in a new definition of the forward rates of the Markov chain model, which is represented by Equation 1. This relationship bears similarities to models describing the simultaneous actions of two or more drugs (11, 12). In addition to the clinical observations described above, the proposed relationship is also based on the following assumptions: (i) each term strictly describes either a drug-related or a disease-related property; (ii) the existence of upper limits to the transition rates (i.e. they cannot be infinite); (iii) small changes in sumatriptan concentration or age can have a large influence on the transition rate (i.e. ‘non-linearity’); and (iv) backward transition rates were assumed not to be affected by sumatriptan treatment or age.
Equation 1 : Rate(t) is the forward rate of transition from state x to y, Ratemin and Ratemax are the minimum and maximum values of that transition rate, respectively. f is the function that describes the relative contributions of patient age (age) and sumatriptan concentration (C(t)) to the transition rate. E0 is a scaling factor. Both the age effect and the sumatriptan effect are scaled by two parameters: Emax represents the maximum contribution of the effect, E50 is defined as the value of age or sumatriptan concentration (depending on the subscript) at which half of the maximum effect is reached. At time 0, the value of exp(C(t)) is replaced with zero.
Data
Sumatriptan pharmacokinetic (PK) and efficacy data were obtained from the Sumatriptan Naratriptan Aggregate Patient database (13). Details about the data source have been described elsewhere (9). For the aggregated response dataset, studies were selected that met the following criteria: (i) contained migraine patient data (rather than healthy subjects or other headache type data); (ii) contained randomised, double-blind, placebo-controlled oral sumatriptan treatment; (iii) did not include special populations (other than adolescents); and (iv) did not include patients with mild migraine headaches. Headache score measurements in adults performed at 0, 0.5, 1, 1.5, 2, 4 and 8 h post dose were included in the analysis. Whenever available, scores at 0.17, 0.33, 3 and 6 h were included also. It has been shown that data up to 8 h can be described by Markov chain dynamics without losing accuracy in the early time points. However, headache was assessed only up to 4 h post dose in most adolescents. This was not considered to complicate analysis since it reflected the shorter duration of migraine attacks in young patients and sufficient data were available covering the three states of the attack.
For the various studies used in our analysis, escape medication was allowed after either 2 or 4 h post dose. The data used in our analysis were therefore censored at time points of escape medication use. Given that no further headache assessments were recorded after administration of escape medication, model predictions refer to the subset of patients who do not take escape medication (either because they were not allowed to or because they did not request escape medication). For sumatriptan, efficacy data included headache scores from 1231 migraine attacks, 449 of which were recorded in adolescents. The characteristics of the studied patient populations are summarised in Fig. 2 and Table 1.
Characteristics of the migraine attacks

Explanatory variables. Graphical summary of explanatory variables ‘drug concentration’ (left) and ‘patient age’ (right). Three concentration vs. time profiles are shown in the left plot. In the right plot, the distribution of patient ages is shown.
Sumatriptan exposure data
The studies from which the headache scores were derived did not contain PK data. In order to provide all headache observations with sumatriptan concentration values, concentration profiles were generated using data from existing PK studies and a model proposed by Cosson and Fuseau (14). Simulations were then performed to derive systematic exposure to sumatriptan at the tested dose levels. It was assumed that the PK in children is not significantly different from that in adults. Moreover, it was assumed that the PK of patients during attack did not differ from healthy subjects. Unpublished data and publications from Cosson et al. (14) and Christensen et al. (15) provide evidence that these assumptions do not invalidate the analysis.
A dataset was constructed containing the observed headache scores, the patient age data and the simulated sumatriptan concentration data. This dataset was then applied to estimate the parameters in the Markov model.
Disease modelling
In the analysis of the migraine response, the Markov chain model described in the previous section was fit simultaneously to headache scores following placebo and active treatment. Patient age and sumatriptan concentrations were included in the data as explanatory variables. Model fitting involved estimation of the parameters defining the forward and backward transition rates of the Markov chain and the expected distributions of the headache scores in each of the states of the Markov chain.
Software
PK modelling and simulation were performed using the non-linear mixed effects software package
Results
Table 2 summarises the model parameters estimates for the forward transitions within the Markov chain, which reflect the improvement towards pain relief and pain-free state. The expected proportions of headache intensity scores per state that can be derived from the model are given in Table 3.
Parameter estimates for the state-to-state transitions corresponding to a decrease in headache intensity

∗For these parameters, no standard errors could be estimated.
Estimates of the parameters that define the forward transition rates in the Markov model are shown together with their 95% confidence intervals (in parentheses).
Parameters are explained in Equation 1 and in the text.
Expected headache severity scores for each of the three clinical states of the Markov chain
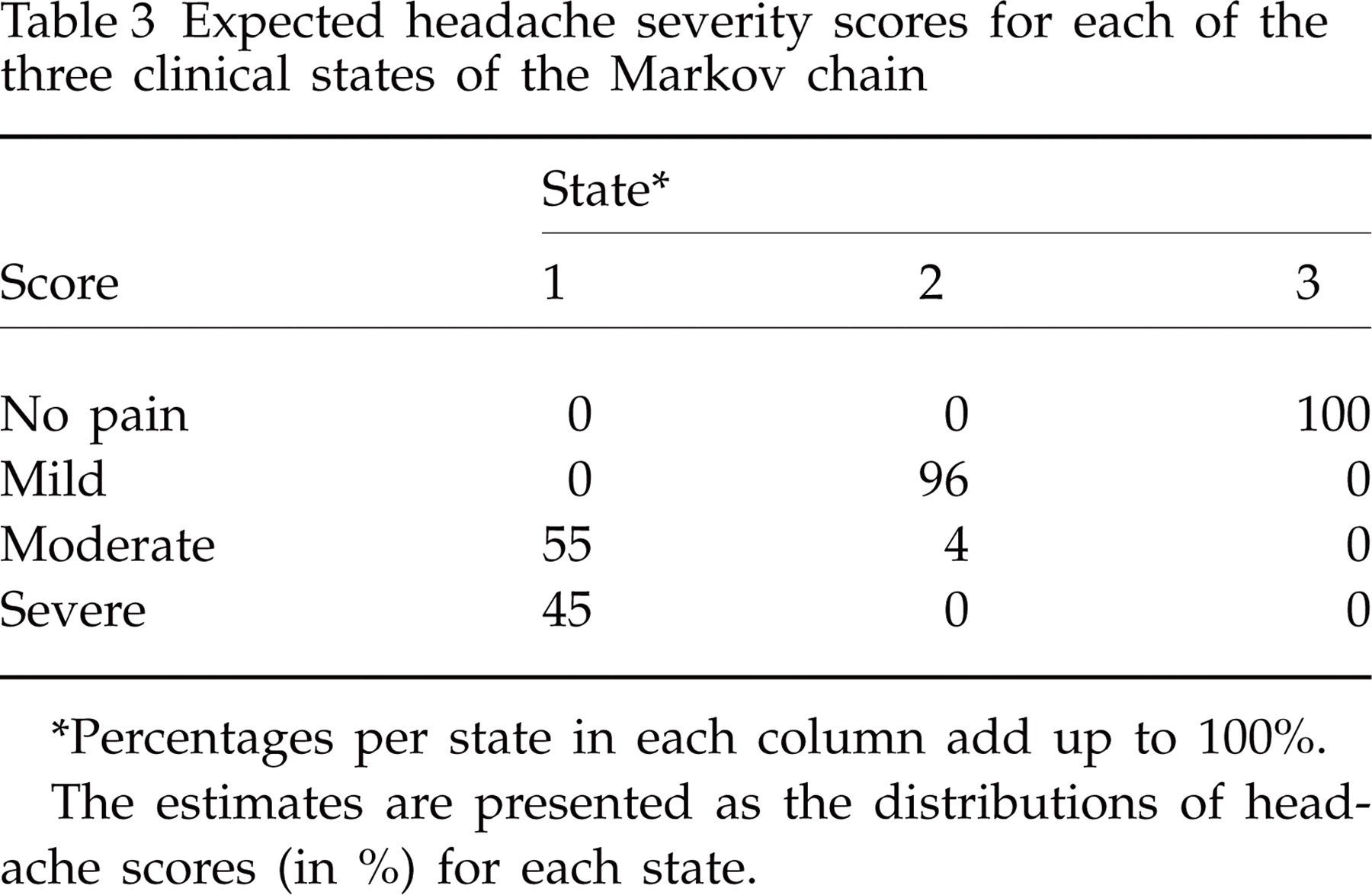
∗Percentages per state in each column add up to 100%.
The estimates are presented as the distributions of headache scores (in %) for each state.
The values of parameter estimates in Table 2 indicate that the maximum effects of sumatriptan concentration on the transition rates are clearly larger than the maximum effects of age (EmaxC(t) is 39 and 19, whereas Emaxage is 6.9 and 1.9 for first and second forward transitions, respectively). This implies that according to the model, in the presence of drug, age becomes less important in determining the response. The backward transition rates (i.e. towards states of more severe headache) were defined by a single parameter (Ratemin ). The estimated values for Ratemin (including 95% confidence intervals) were 0.051/h (0.033, 0.078) and 0.041/h (0.033, 0.053), for the transitions 2 to 1 and 3 to 2, respectively. From the model, it can be derived that the mean transit time from ‘no relief’ to ‘relief’ is 3 h for young adolescents and increases to 6 h for patients aged ≥ 30 years; and sumatriptan reduces the transit time to 2 h, irrespective of age.
Furthermore, in addition to the estimation of pharmacological potency for sumatriptan, we were able to derive the so-called ‘apparent potency’ for age. These estimates provide information about the age and sumatriptan concentration that correspond to half of their maximum contributions to the transition rates. For age, E50age was 23 and 22 years, for the first and second forward transitions, respectively. For concentration, E50C(t) , was 11 and 34 ng/ml. It is at these midpoints that treatment response is most sensitive to changes in age and sumatriptan concentration.
The level of agreement between the proposed model and the observed headache data is evaluated in Fig. 3, where observed and predicted response vs. time are compared for adolescents and adults, pain free and pain relief responses, and at placebo, 25, 50 and 100 mg. In general, there is good agreement between observations and model predictions. The size of the markers in the plots represents the relative amounts of data available for the different dose levels. Most data were available for the adult study population at the 100 mg dose level. Due to the small amount of data at the other levels, goodness-of-fit is less clear for dose levels other than 100 mg. Note that at the 25 mg level in adults, the observed headache responses are higher than those at the 50 mg level. Obviously, the model cannot take into account these fluctuations in the mean response. At the same time, this high variability illustrates why it can be difficult to identify and describe explanatory variables accurately.
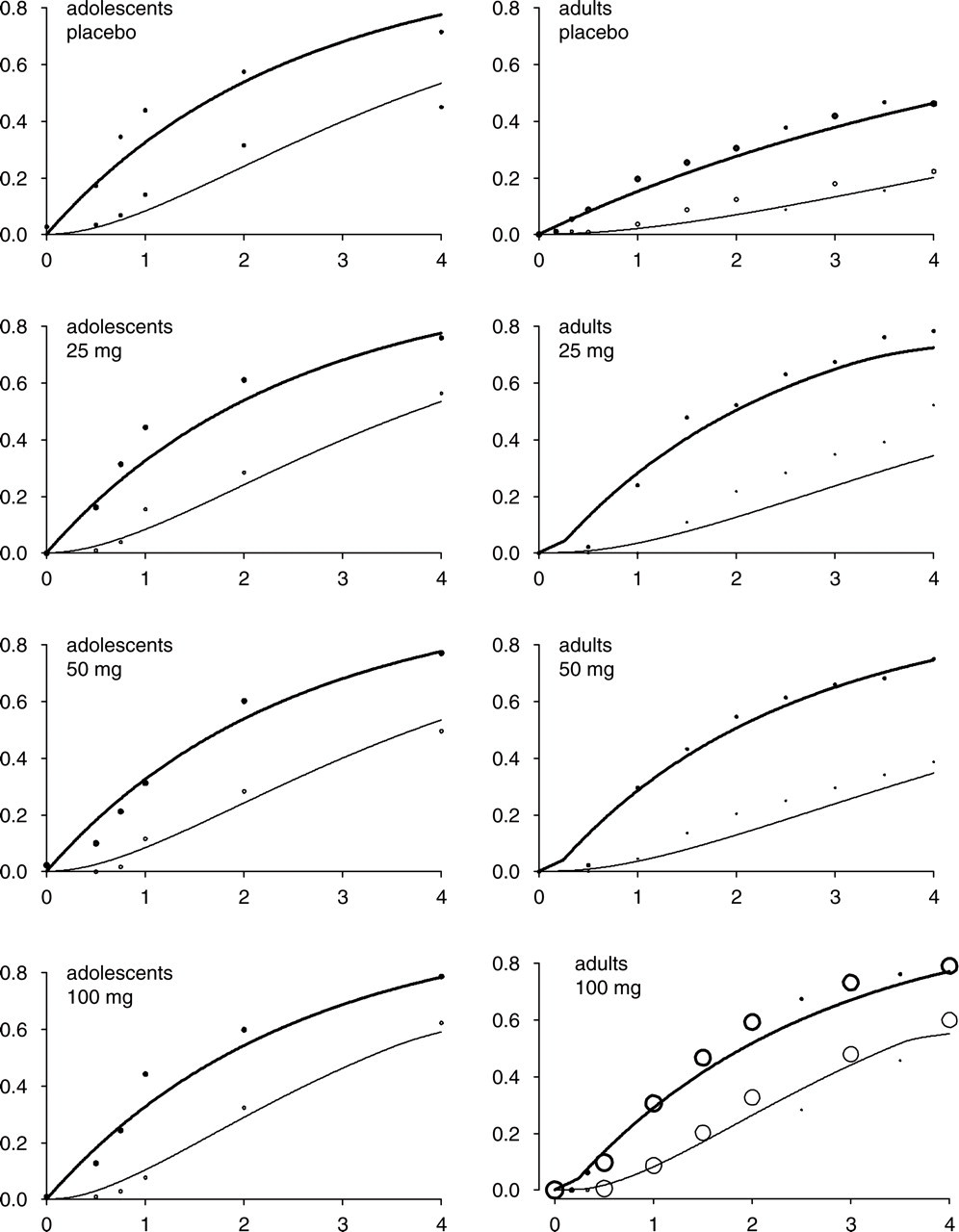
Fraction of patients with headache response vs. postdose time in hours. Observed responses are shown as markers and predicted mean responses as lines. Bold face lines and markers represent pain relief response, normal face lines and markers represent pain-free response. Predictions for both age categories (adolescents and adults) were obtained using the same model. The relative sizes of the markers indicate the proportions of the data that were used in the creation of each panel.
Figure 4 shows predicted response surfaces at 1 h post dose. Particularly in the pain relief surface, the relationship between age and concentration can be clearly recognised.

Response surfaces. Predicted response surfaces for pain-free and pain relief responses at 1 h post dose.
Discussion
A Markov model was developed to describe and predict the headache response to placebo and antimigraine drugs across patients of different ages. Model parameterisation was defined in such a way that discrimination between disease and drug-specific properties is evident and the interaction between age and drug exposure on treatment efficacy can be quantified. Also, it is evident from the estimation of confidence intervals that the precision with which model parameters could be estimated was variable. This is due to the complex model structure as defined in Equation 1, and to the imbalance in the datasets, which contained variable numbers of patients per dose and age groups. The effects of limited data in the low-dose groups were particularly relevant for the estimation of parameters relative to the transition from state 2 to state 3, which were not precisely estimated.
The effect of age on pain response can be best assessed at the midpoints of the relationships between age and the state-to-state transition rates (E50age ). E50age was 23 years for the transitions from ‘no relief’ to ‘pain relief’ and 22 years from ‘relief’ to ‘pain free’, respectively. This means that the placebo response differs most strongly between the age groups at either side of these points. It should be noted that these points are not equal to the age (i.e. 18 years) that represents the formal divide between studies in adolescents and studies in adults. On the basis of these results, it can be argued that the decrease of placebo effect with age represents a continuum and does not merely reflect differences between the designs of both types of study. In fact, this finding raises the question of the rationale for paediatric research protocols that often apply predefined age ranges based on regulatory guidelines (17), irrespective of evidence that discrepancies exist in age ranges for incidence and prevalence of disease.
Age-related discrepancies in migraine characteristics are not limited to the paediatric population (18). Elderly patients have also been found to differ from the middle-aged population in the expression of their migraine symptoms. This difference was not taken into consideration in the present analysis, first because the current lack of efficacious antimigraine medication for children and adolescents warrants a focus on this particular population, second, because it would have complicated parameter estimation even further.
Unfortunately, age-related variables other than patient age were not available in the datasets that were used for the analysis. These variables are potentially more sensitive explanatory variables of migraine response than the patient's age. On the other hand, Bigal et al. (18) have suggested that some changes in migraine symptomatology may well represent a direct result of ageing. For example, age-related changes in blood vessels could render vascular pain-generating mechanisms less pronounced in older patients, which may explain the reduced occurrence of the one-sided pulsating character of migraine in this population.
In addition to the effect of age, drug exposure has a critical role on pain response. Our findings reveal that the midpoints (E50C(t)) of the relationship between sumatriptan concentration and transition rates are within the concentration range of the 100 mg oral dose of sumatriptan, which means that the maximum effect of the oral formulation can be reached with this dose. Moreover, the values of the maximum effect of drug concentration on transition rate (EmaxC(t)) are relatively large when compared with the maximum effects of age (Emaxage). The consequence of this difference is that when sumatriptan concentrations become similar to or higher than E50C(t), the transition rates towards improved headache response are exclusively determined by drug concentration, and age has no further influence on response. This can be seen clearly in the predicted responses in Fig. 3, where during the first 20 min before any drug is absorbed, the slope of the pain relief response is shallower in adults than in adolescents. After 20 min, when a significant amount of drug has been absorbed, any difference in response between the age groups disappears. As individual concentration data were not available for most studies in our analysis, we used population-based mean concentration values. In fact, Visser et al. (19) have found that lack of headache relief after sumatriptan does not appear to be explained by PK differences between patients. We argue, therefore, that mean PK can be used to relate exposure to treatment response, provided that the results of the analysis are not used to predict the responses of individual patients, and the patient population used in the analysis is not biased towards inclusion of non-responders.
Although age and drug concentration are interacting variables according to the relationship in Equation 1, they almost independently determine the response. Thus, in adolescents the response after placebo naturally does not depend on concentration, but conversely, the responses at ≥ 25 mg do not depend on age. A stronger opinion on whether or not these explanatory variables are interactively determining the response may be gained by performing a (meta-)analysis of data from studies investigating administration routes other than oral. Indeed, such studies show that the drug response in adolescents is higher than their placebo response, but equal to the response in adults. Interestingly, intranasally administered sumatriptan is more effective in children than the oral formulation. Furthermore, a significant treatment effect was found when oral sumatriptan medication was given early in the migraine attack rather than at the time of maximum pain intensity.
The utility of this Markov model with explanatory variables is not limited to the study of migraine attacks or efficacy end-points. Due to its structure, the model is thought to be more generally applicable to scenarios where a demographic variable bearing relation to disease progression interacts with treatment effect (response, adverse events). Questions that could be addressed in a quantitative manner include: (i) can effective doses be found for all age categories?; (ii) can treatment response be improved by changing the treatment regimen?; and (iii) what is the influence of PK variability on the age–efficacy relationship? Potentially, this approach opens new avenues to exploring more extensively the role of pharmacokinetics in clinical and observational studies.