Abstract
A migraineur can claim to be an infrequent responder (‘non-responder’) to an oral triptan independent of which triptan he or she is presently using. Four trials of an alternative triptan (zolmitriptan/rizatriptan; eletriptan; naratriptan; almotriptan) in patients with a history of infrequent response to oral sumatriptan were compared and contrasted in terms of study design, patient characteristics, and efficacy and tolerability of the triptan under investigation. Unfortunately, none of the reported studies used an appropriate parallel design, which would have had the non-responding triptan (oral sumatriptan) in one arm and without encapsulation. While the four trials differed in terms of study design (open-label vs. placebo-controlled), definition of sumatriptan ‘non-responder’ (retrospective vs. prospective) and pain intensity at baseline (30% severe to 70% severe), all four demonstrated that lack of response to sumatriptan did not predict lack of response to an alternative triptan. Changing triptans resulted in 2-h pain-relief rates of 25–81% in patients with a history of poor response to sumatriptan. It can be concluded that migraine patients who respond infrequently to sumatriptan should be switched to a different triptan, as lack of response to one triptan does not predict likelihood of responsiveness to another. A review of the available evidence suggests that almotriptan may be one of the most appropriate choices for an alternative triptan.
Introduction
Migraine is a common and debilitating recurrent condition that affects 12–20% of women and 6% of men (1–4). Improvements in the understanding of the pathogenesis of migraine, in particular the involvement of pre- and postsynaptic serotonin (5-HT) receptors, have led to the development in the early 1990s of sumatriptan, a selective 5-HT1B/1D receptor agonist (5). A range of such selective 5-HT1B/1D receptor agonists, collectively known as the triptans, are now available for the treatment of migraine, and these are generally the agents of choice to treat a migraine attack (6, 7). Triptans have well-established efficacy in the treatment of acute migraine, acting against nausea, photophobia and phonophobia as well as pain, and are generally safe and well tolerated (8–10). The US Headache Consortium guidelines recommend triptans as initial therapy for patients with moderate to severe headache and for those with headache of any severity with a history of non-response to non-specific therapies (6).
Non-response to acute treatment
Responders and non-responders in randomized controlled trials (RCTs) have traditionally been defined by headache response, i.e. a migrainous headache with a pain intensity of moderate to severe that becomes mild or pain free within 2 h after treatment administration (11). Maximum response rates to oral triptans, in terms of relief from pain at 2 h after treatment, are around 70%; in other words, approximately 30% of patients in single-attack RCTs do not respond to the oral triptan under investigation (9, 12, 13). Consistency of response reflects how reliable the treatment can be expected to be in the clinical setting. Within-patient consistency describes the percentage of individuals who respond to a triptan during a certain proportion of their treated attacks; it is probably of more clinical relevance than across-attack consistency. For example, the intra-individual consistency of response in three out of three attacks for the subcutaneous formulation of sumatriptan is much higher (73%) than that of the conventional tablet (32%) (14–16). Generally, the intra-individual consistency of response to oral triptans is relatively low, with only 40–50% responding in three out of three attacks (14, 16). Accordingly, it is very likely that a migraineur will, from time to time, find that the oral triptan they are using is not reliable (in contrast to the parenteral use of a triptan).
Within-patient consistency results obtained with oral triptans in RCTs suggest that migraineurs and their migraine attacks are not totally independent and that responders and non-responders are not distributed at random. Accordingly, the study population of migraine patients can not be considered to be homogeneous but rather a combination of different subgroups of migraineurs who do not react in a uniform way to a given treatment (12, 16, 17). Thus, individual migraineurs or their migraine attacks are more or less likely to respond to a given treatment. In an analysis of data from controlled clinical trials in which three attacks were treated with sumatriptan tablets 50 mg or 100 mg, it was demonstrated that two treatment successes with respect to pain relief and pain free at 2 h were followed by a successful response to the third attack in more than 70% of the patient population; in patients who had experienced two treatment failures in the preceding attacks about 70% also failed to respond during the third attack (14). This phenomenon apparently holds true for other triptans as well (14). In an open-label, five-way cross-over preference study performed in five countries, several triptans were analysed in both triptan-naive and triptan-experienced patients who treated one migraine attack with a triptan as early as possible; the majority of attacks treated (85%) were of moderate to severe intensity (18). Efficacy data were derived from the intention-to-treat population of 372 patients who treated at least one attack. The 2-h pain-free response rates were similar for sumatriptan 50 and 100 mg, rizatriptan 10 mg, and zolmitriptan 2.5 mg at 37%, 40%, 38% and 36%, respectively. The 2-h pain-free response for naratriptan 2.5 mg was 28%. Two hundred and fifty-eight patients (69%) completed all five treatments and expressed a preference (18). Interestingly, subanalyses of the data revealed that a high percentage (58–81%) of the frequent responders (‘good responders’), defined as having responded to any of the triptans during the two preceding attacks with respect to pain relief at 2 h, also tended to respond to treatment of the third attack (14, 18). On the other hand, in the minority of patients who were infrequent responders (‘bad responders’/‘non-responders’), defined as having experienced two treatment failures to any of the triptans in the preceding attacks, a much lower percentage (14–47%) responded to treatment of the third attack. Interestingly, however, the difference in efficacy among the triptans became more marked. Accordingly, the probability of response in an individual migraine is related to whether or not the individual responded during a previous attack. The clinical implication of these findings is that if the migraineur does not respond to treatment of two attacks, they should probably try another treatment alternative or a higher dose for the next attack.
There are at least two plausible explanations for inconsistent treatment effects with oral triptans (17, 19, 20). The most likely explanation is that gastrointestinal motility is inhibited and gastric emptying time delayed during the migraine attack (15, 21–25). There is also the risk that the migraine-induced nausea culminates in vomiting. Triptans appear to prolong gastric emptying time by activating 5-HT1 receptors on gastric myenteric neurons (26–28). For these reasons, the absorption of any triptan taken orally during the migraine attack can be erratic and treatment effects inconsistent (14, 15).
Central sensitization of the trigeminal pain transmission is an additional explanation for inconsistent response to acute treatment with an oral triptan. According to this theory, the migraineur runs a greater risk of sensitization of both second- and third-order neurons of the ophthalmic division of the trigeminal nerve if the migraine attack is allowed to continue without treatment (29–32). This central sensitization leads to cutaneous allodynia corresponding to the innervation area as well as a poorer response to treatment with a triptan (29–32). Whereas the throbbing characteristic of the pain and sensitivity to coughing (peripheral first trigeminal neuron sensitization) disappear with triptan treatment, the cutaneous allodynia and expanded perceptive fields (central second trigeminal neuron sensitization) may last for hours or days. According to Burstein and coworkers, about 50% of migraineurs develop cutaneous allodynia within 1 h and about 75% within 4 h, and in the presence of cutaneous allodynia the triptans do not provide freedom from pain (32). It is thus suggested that this type of central sensitization is driven by both attack duration and pain intensity and that migraineurs should be most responsive to triptan therapy within the first 30–60 min of an attack, before cutaneous allodynia develops (29–32). Whether cutaneous allodynia can also be used clinically as a practical marker that predicts decreased responsiveness to acute treatment with a triptan is, however, still disputed. Both the phenomenon of compromised oral absorption and the theory of trigeminal sensitization provide reasonable explanations for why oral therapy with triptans may provide pain relief in only some migraine attacks.
Although it is considered good clinical practice to try a second or third triptan in a patient in whom a particular triptan has proved non-ideal in all clinical respects, there have been very few studies investigating the efficacy of an alternative triptan in such patients (33–35). The objective of this review is to examine available evidence so as to assess the value of prescribing an alternative triptan to patients who failed to respond to oral treatment with a previous triptan. Such a review should provide guidance to clinicians faced with a patient who does not respond to a particular triptan and help in the selection of an alternative therapy that will offer the greatest probability of achieving an effective response.
Review of non-responder trials
Zolmitriptan and rizatriptan in sumatriptan non-responders
The efficacy and tolerability of zolmitriptan and rizatriptan in patients considered to be sumatriptan non-responders were investigated in a single-centre, open-label, crossover design trial (36). Sumatriptan non-responders were defined as patients who, according to history, had not responded to three or more of five trials of sumatriptan 50 mg, taken early in the headache phase. The first five headaches were treated with either zolmitriptan 5 mg or rizatriptan 10 mg, and the next five with the other agent. Fifty-six patients were enrolled, of whom 48 completed the study; 120 attacks were treated with zolmitriptan and 120 with rizatriptan. The rate of response at 2 h, i.e. reduction from moderate/severe pain to mild/no pain, was 73% in attacks treated with zolmitriptan and 81% in attacks treated with rizatriptan. Patients were pain-free at 2 h in 45% of zolmitriptan-treated attacks and 58% of rizatriptan-treated attacks. Rizatriptan therefore appeared to provide a better response than zolmitriptan in this group of patients, but the number of patients in the study was too small for statistical significance to be assessed. About 19% of patients were unresponsive to both zolmitriptan and rizatriptan.
Eight patients receiving zolmitriptan in comparison with none receiving rizatriptan developed nausea (which was mild, necessitating withdrawal in only one patient), whereas eight patients on rizatriptan and one on zolmitriptan experienced dizziness and somnolence. Chest symptoms occurred in seven of 48 patients with zolmitriptan and two of 48 patients with rizatriptan.
Eletriptan in sumatriptan non-responders
The efficacy and tolerability of eletriptan in sumatriptan non-responders was investigated in a double-blind, parallel-group, placebo-controlled study involving migraine patients with a history of unsatisfactory treatment with sumatriptan (37). The trial was conducted in around 50 headache treatment centres in Norway, Sweden, Denmark, Finland and the Netherlands. Inclusion criteria required patients to have discontinued sumatriptan 2 weeks to 2 years previously because of slow onset of action, inconsistent response, poor overall efficacy, recurrence or poor tolerability. The patients were randomly assigned at a 2 : 2 : 1 ratio to eletriptan 40 mg (n = 188), eletriptan 80 mg (n = 171) or placebo (n = 87) to treat up to three migraine attacks. Results in the eletriptan 80-mg group will not be considered in this review as 80 mg is not an approved single dosage in most countries. In the eletriptan 40-mg group, 53% had discontinued sumatriptan as a result of insufficient overall clinical response, and 26% had discontinued owing to adverse events; the equivalent figures were 38% and 36% for the placebo group. During the trial 179 patients (95%) in the eletriptan 40-mg group and 81 (93%) in the placebo group experienced and treated at least one migraine attack with the study medication. At baseline, 30% of the eletriptan 40-mg group and 33% of the placebo group rated their headache pain as severe.
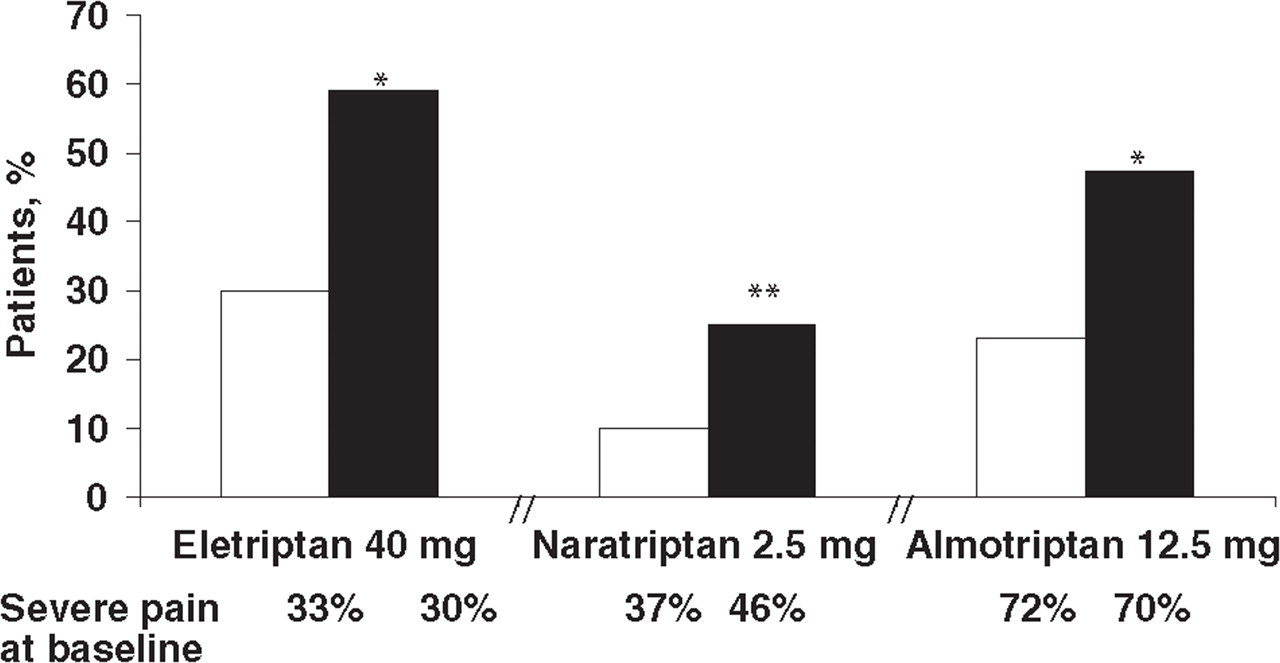
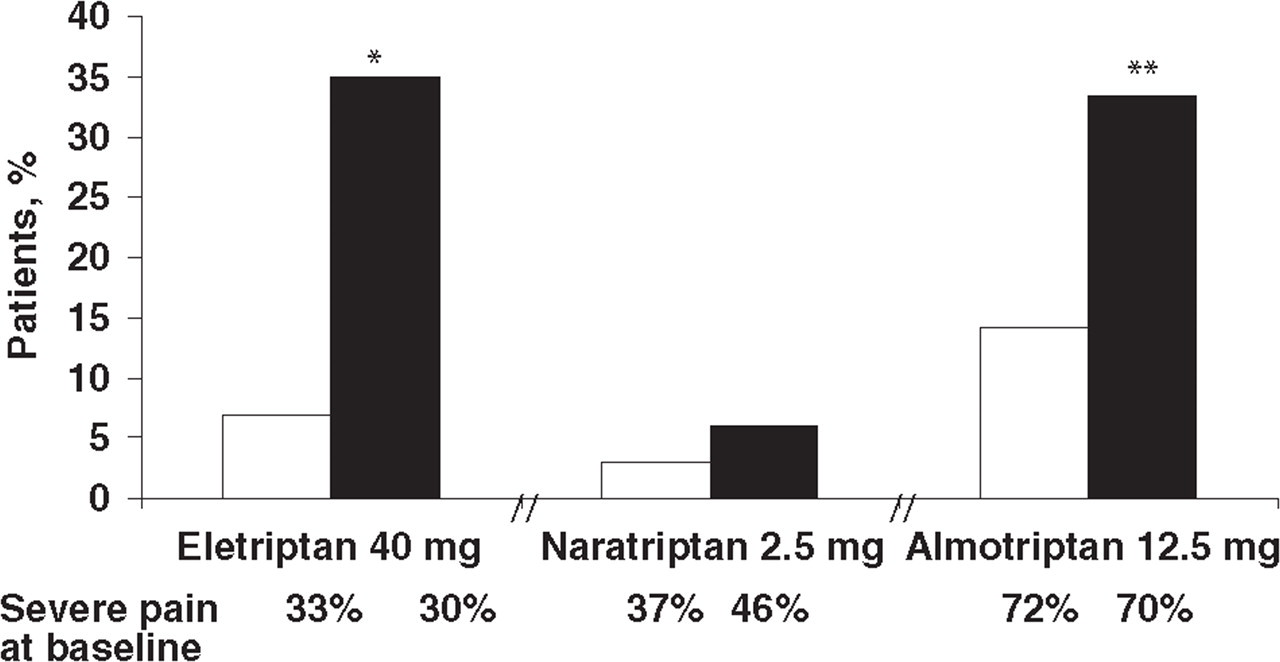
Two-hour pain relief for the first attack was 59% (91/154 patients) with eletriptan 40 mg vs. 30% (22/74 patients) with placebo (P < 0.001) (Fig. 1). The 2-h pain-free rates were 35% for eletriptan 40 mg and 7% for placebo (P < 0.001) (Fig. 2). Headache recurrence (defined as return of headache from a situation of no pain or mild pain between 2 and 24 h after drug administration) occurred in 26% of patients receiving eletriptan 40 mg and 50% of patients receiving placebo, and fewer patients on eletriptan than placebo required rescue medication (24% vs. 63%).
Patients taking eletriptan experienced more adverse events than patients taking placebo. The majority of adverse events were mild to moderate in severity and transient in nature. The most commonly reported adverse events were asthenia (5% of eletriptan 40 mg patients vs. 2% of the placebo group), nausea (5% vs. 8%), chest symptoms (4% vs. 0%) and vomiting (4% vs. 3%). There were no statistically significant differences between rates of adverse events associated with eletriptan 40 mg and placebo, but the study was not powered to detect such differences.
Naratriptan in sumatriptan non-responders
In a multicentre US trial of the efficacy of naratriptan in sumatriptan non-responders, non-response to sumatriptan 50 mg was not retrospectively based on patient history, as in the trials discussed earlier, but on a single-blind, prospective assessment that was integral to the study design (38). Patients who described themselves as experiencing an unsatisfactory response to oral sumatriptan were recruited into the study and supplied, in a single-blind fashion, with a sumatriptan 50-mg tablet to use for their next migraine attack of moderate or severe pain intensity. Patients entering the study were required to have had at least one migraine attack resulting in moderate or severe pain in each of the 2 months preceding the study. The response to sumatriptan 50 mg was assessed 1–7 days after treatment, and patients who had responded to sumatriptan, by recording a reduction from severe or moderate to mild or no pain at 4 h after treatment, were screened out of the study. Of 347 patients enrolled into the first phase of the study, 124 (35.7%) responded to sumatriptan and did not enter the second phase of the study. A total of 206 of the remaining patients were randomly assigned in a double-blind manner to naratriptan 2.5 mg (n = 99) or placebo (n = 107) for their second migraine attack.
The baseline characteristics of the second migraine attack in the naratriptan and placebo groups were statistically indistinguishable; 46% of the naratriptan patients and 37% of the placebo group had severe pain intensity at baseline. Pain relief (reduction from severe/moderate pain to mild/no pain) at 4 h, the primary efficacy variable of the study, was achieved in significantly more patients in the naratriptan group (41%) than in the placebo group (19%, P < 0.001). The use of this endpoint, however, is not typical, as 4 h is considered to be too long for the patient to wait for pain relief (9). Response at 2 h was also significantly better (P < 0.005) with naratriptan (25%) than placebo (10%) (Fig. 1), but the proportion of patients pain-free at 2 h did not differ significantly between naratriptan (6%) and placebo (3%) (Fig. 2). Use of rescue medication was similar among naratriptan (6%) and placebo (5%) patients.
There were no serious adverse events in the study. In the second phase, more patients on naratriptan than placebo experienced adverse events (12% vs. 5%). Investigators considered that 12 patients had experienced adverse events during the second migraine attack that were related to study medication: of these, nine had received naratriptan and three received placebo.
Almotriptan in sumatriptan non-responders
The response to almotriptan in sumatriptan non-responders was assessed in a trial of similar design to that just described to investigate the efficacy of naratriptan in sumatriptan non-responders (39). The trial was a double-blind, randomized, placebo-controlled study carried out in 60 centres across Germany. Patients describing themselves as experiencing at least two unsuccessful responses to sumatriptan, who had a history of migraine of >1 year and at least one migraine attack that resulted in moderate or severe pain in each of the two preceding months, were recruited into the study. As in the naratriptan trial, the patients’ response to sumatriptan 50 mg was confirmed prospectively under clinical observation. Of 302 patients with a history of non-response to sumatriptan entered into the first phase of the trial, 221 (73%) did not respond to sumatriptan (no reduction from severe/moderate pain to mild/no pain at 2 h after treatment) and were entered into the second, double-blind phase of the trial. The high rate of non-response to sumatriptan in this prerandomization attack confirmed the selection of sumatriptan non-responders as part of the inclusion criteria. Of these non-responders to sumatriptan 50 mg, 99 were randomly assigned, in a double-blind fashion, to almotriptan 12.5 mg and 99 to placebo for their next migraine attack of moderate or severe pain intensity.
There were no significant differences between the almotriptan and placebo groups in the analysis of demographic and baseline variables. At baseline for the second attack, 70% of the almotriptan group and 72% of the placebo group had severe pain. The proportion of patients responding to almotriptan (i.e. reduction to mild/no pain at 2 h), 47.5%, was significantly better than with placebo, 23.2% (P < 0.001). Several secondary efficacy endpoints also showed statistically significant differences between almotriptan and placebo. A significantly greater proportion of almotriptan patients were pain free at 2 h, 33.3%, in comparison with 14.1% of those on placebo (P < 0.005), and a significantly greater proportion of patients treated with almotriptan achieved sustained pain-free status (pain free at 2 h with no recurrence of moderate or severe headache and no rescue headache medication 2–24 h postdose), 20.9%, compared with the placebo-treated group, 9.0% (P < 0.05). Use of rescue medication was significantly lower in almotriptan-treated patients than in the placebo group, 26.6% in comparison with 46.9% (P < 0.005). In addition, complete relief at 2 h, defined as no pain and no migraine-associated symptoms (nausea, vomiting, photophobia or phonophobia), was achieved in 17.1% of patients treated with almotriptan compared with 4.4% of patients treated with placebo (P < 0.01) (personal communication).
In the first (sumatriptan) phase of the study, 49 patients (16.2%) experienced 70 treatment-emergent adverse events. In the second phase, seven patients (7.1%) in the almotriptan group (n = 99) reported seven treatment-emergent adverse events and five patients (5.1%) in the placebo group (n = 99) experienced nine treatment-emergent adverse events. The majority of these adverse events were mild or moderate, with only one severe adverse event in the placebo group (nausea).
Comparison of non-responder trials
The four trials described here demonstrate that infrequent or lack of response to oral sumatriptan does not ensure lack of response to all triptans: rates of response (reduction in pain from severe/moderate to mild/none) to alternative triptans at 2 h after treatment of patients who had a historical or prospectively confirmed lack of response to oral sumatriptan ranged from 25% to 81%. In the three placebo-controlled trials, the alternative triptan (eletriptan, naratriptan or almotriptan) provided significantly better 2-h pain relief than placebo (Fig. 1). The almotriptan and eletriptan trials showed that these triptans also provided significantly better 2-h pain-free responses than placebo (Fig. 2). In the naratriptan trial, the proportion of patients pain free at 2 h did not differ significantly between the naratriptan and placebo groups (38), probably as a result of naratriptan's relatively slow onset of action (40). The almotriptan trial also reported sustained pain-free rates, which were significantly better than placebo (20.9% vs. 9%, P < 0.05) (39). These findings provide evidence that patients with acute migraine who do not respond to a particular triptan should be switched to another triptan. There are, however, several important differences between the trials that have implications for therapeutic choices in patients with a history of non-response to sumatriptan (Table 1).
Features of clinical trials of triptans in sumatriptan non-responders
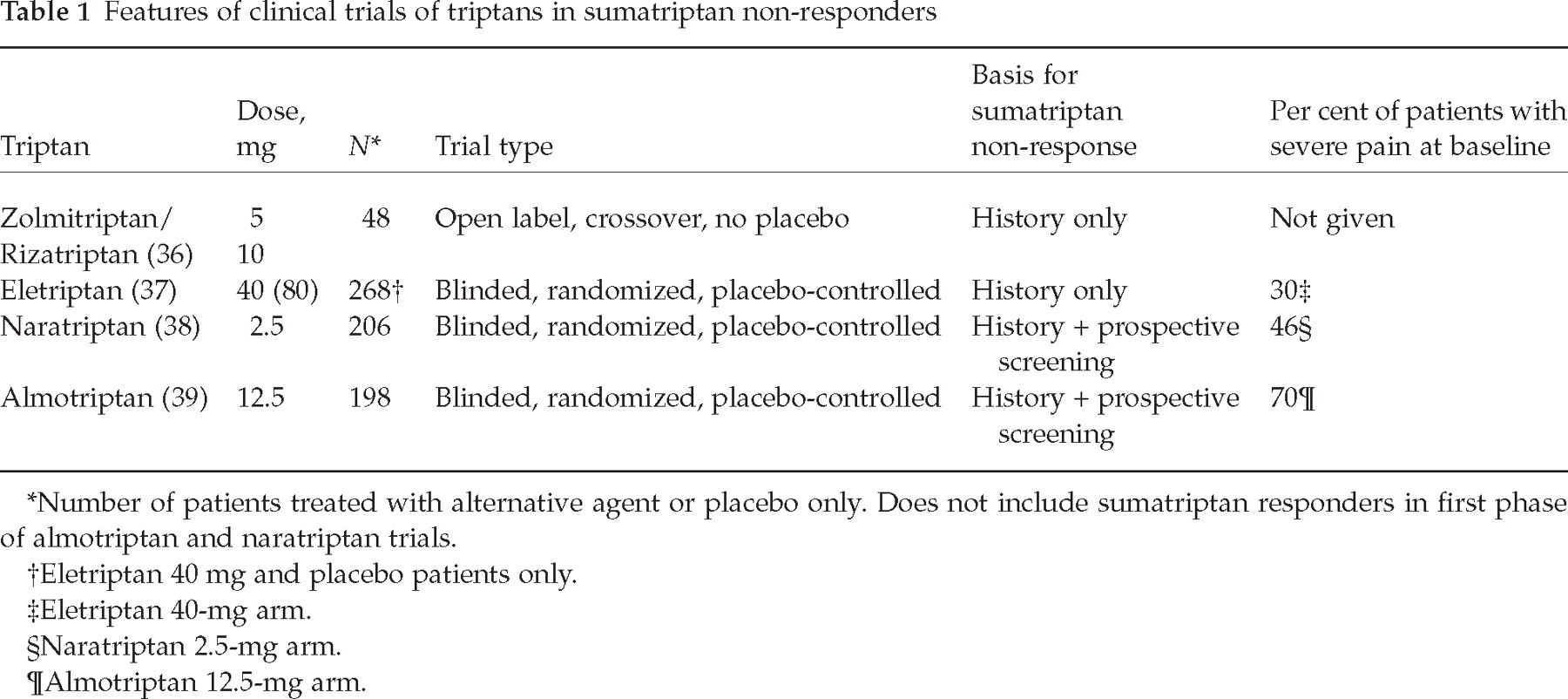
Number of patients treated with alternative agent or placebo only. Does not include sumatriptan responders in first phase of almotriptan and naratriptan trials.
Eletriptan 40 mg and placebo patients only.
Eletriptan 40-mg arm.
Naratriptan 2.5-mg arm.
Almotriptan 12.5-mg arm.
The basic design of the four trials differed. The rizatriptan/zolmitriptan study had an open-label design compared with the double-blinded, randomized, placebo-controlled trials of eletriptan, naratriptan and almotriptan. The rizatriptan/zolmitriptan study also involved a small number of patients: while demonstrating that migraine sufferers with a history of non-response to sumatriptan might nevertheless respond to rizatriptan or zolmitriptan, it was insufficiently powered to assess which alternative triptan provided the better response (36) A limitation of the placebo-controlled trials was that they did not include a sumatriptan arm (not encapsulated) in the active drug vs. placebo attack.
The four trials also differed in their definition of non-response to sumatriptan. Sumatriptan non-response was based on retrospective history in the rizatriptan/zolmitriptan and eletriptan studies (36, 37), whereas non-responders were prospectively selected for in the naratriptan and almotriptan trials (38, 39). Prospective screening results in a non-responder population of greater certainty than does a retrospective history of non-response, as was demonstrated in the naratriptan and almotriptan trials where, under clinical observation, approximately 30% of patients with a history of non-response to sumatriptan did in fact respond to the drug in the first phase of the trials (although it must be borne in mind that there was no placebo control arm in the first phase of these trials against which to compare the response) (38, 39). It is reasonable to expect a higher response rate in the rizatriptan/zolmitriptan and eletriptan trials as a result of the less rigorous methods of patient selection.
Perhaps as a result of the differences in study design, the non-responder trials differed in the severity of the patients’ pain at baseline. Thirty per cent of patients treated with eletriptan 40 mg had severe pain at baseline, in comparison with 46% of those treated with naratriptan 2.5 mg and 70% of those treated with almotriptan 12.5 mg. This may be the result of the greater rigour of the definition of sumatriptan non-response in the almotriptan and naratriptan trials (prospective) compared with the eletriptan trial (retrospective). The patient cohort in the almotriptan study was the most severely affected by their migraine attack and probably the most difficult to treat successfully.
Implications for treatment
Patients with migraine are a heterogeneous population, and it may be that the differences among patients and their responses to triptan medication are at least as important as the differences among the triptans (41, 42). Clinical trials of oral triptans have shown a consistency of response within patients (43), which suggests that lack of response to a particular triptan is also consistent (38). The results of the trials in sumatriptan non-responders reviewed here clearly demonstrate that consistency of response is limited to individual triptan medications and not across the triptan class, as failure to respond to one triptan does not predict failure to a different triptan.
Triptans vary in their pharmacokinetic properties, such as bioavailability, onset of action, half-life, and drug–drug interactions, and in their tolerability (33, 40, 44). For example, naratriptan has the slowest onset of action of the available triptans, frovatriptan has the longest half-life and almotriptan has the highest (oral) bioavailability (33). Almotriptan and naratriptan have the most favourable adverse event profiles (33). A meta-analysis of the results of 53 double-blind, placebo-controlled studies with over 24 000 patients compared six oral triptans with sumatriptan 100 mg with regard to efficacy, consistency of effect, and tolerability (13, 45). This meta-analysis found that although all oral triptans are effective and well tolerated, they differ in ways that are clinically significant for individual patients. The authors concluded that finding the best therapy might involve trial and error, such that if the first triptan fails the patient can be successfully switched to another. The authors also reported that rizatriptan 10 mg, eletriptan 80 mg (a dose not approved in most countries), and almotriptan 12.5 mg provide the highest likelihood of consistent success.
Triptans are safe medications provided they are not used in patients with cardiovascular disease or major risk factors, and no triptan is demonstrably safer than the others (45). Yet, a recent survey of migraine sufferers reported that 71% delayed or avoided taking their medication due to concerns about adverse events (46). A review of triptan formulations by Rapoport et al. concluded that naratriptan, almotriptan and frovatriptan may be good choices for patients with a history of susceptibility to adverse events (33). The meta-analysis of oral triptans reported a mean placebo-subtracted rate of any adverse events of 13% for sumatriptan 100 mg; only almotriptan 12.5 mg and naratriptan 2.5 mg had significantly lower rates (1.8% and 2.4%, respectively), which were also not different from placebo (13). When looking specifically at placebo-subtracted CNS adverse events, most triptans had rates similar to that of sumatriptan 100 mg (6.3%), except for eletriptan 80 mg, which had significantly higher values (14.6%) and almotriptan 12.5 mg, which had significantly lower values (−1.5%, also not different from zero) (13). It has been suggested that lipophilicity is correlated with CNS adverse events; drugs with lower lipophilicity (a measure of their ability to cross the blood–brain barrier) are associated with lower rates of CNS adverse events (47, 48) (Table 2). In regard to chest adverse events, only almotriptan 12.5 mg had significantly lower placebo-subtracted rates than sumatriptan 100 mg (0.4% vs. 1.7%) and also not different from zero. It has been suggested that the low incidence of chest adverse events associated with almotriptan is the result of its lower intrinsic activity on pulmonary arteries and veins compared with other triptans (49).
Properties of oral triptans (47)

t max, Time to maximum concentration; t 1/2, biological half-life; C max, peak plasma concentration.
Log DpH = 7.4 quantifies triptan lipophilicity (increasing numbers indicate greater lipid solubility).
Female/male values.
Patients who failed to respond to one oral triptan formulation may be more likely to be discouraged from continuing therapy if they fail again on a second triptan. For this reason, it is important to select an alternative triptan with a high likelihood of success. The excellent efficacy and tolerability demonstrated by almotriptan, combined with its performance in sumatriptan non-responders described here in the most rigorously performed clinical trial, make it an excellent choice for migraine patients who failed to respond to their previous triptan.
Conclusions
It is concluded that migraine attacks (and migraineurs) can be more or less likely to respond to oral treatment with a given triptan. Different approaches, i.e. early treatment at mild intensity, increasing the dose, changing the formulation, and combination with antiemetics, can be taken to try to improve the intra-individual consistency of response to oral triptans in migraineurs. For the subgroup of patients who despite these attempts do not respond to a particular triptan, the poor responsiveness is likely to be consistent in subsequent attacks (38, 43).
Migraine patients who do not respond to, or are dissatisfied with, oral treatment with sumatriptan should be switched to a different triptan, as lack of response to one triptan does not predict likelihood of responsiveness to another. Faced with such a patient, a physician should draw on clinical experience and the best available evidence to select the triptan most likely to achieve an effective and well-tolerated response, so as to minimize the chances of a second triptan failure that might cause the patient to discontinue treatment. The studies reviewed here suggest that almotriptan 12.5 mg could be an appropriate choice.
