Abstract
Thirty to forty percent of migraineurs do not respond to any given triptan treatment. We identified clinical variables that significantly predict therapeutic non-response and evaluated the efficacy of eletriptan (20, 40 and 80 mg) and sumatriptan (100 mg) vs. placebo in a subgroup of patients with all predictor variables. First-attack data were pooled from 10 randomized, double-blind, placebo-controlled migraine trials (n = 8473). Multivariate regression analyses identified three significant baseline predictors of failure to achieve 2-h pain-free response: severe headache pain, presence of photophobia/phonophobia and presence of nausea. Time of dosing following headache onset did not influence 2-h pain-free response. Among patients with all three risk factors (n = 2010; 24± of total sample), 2-h pain-free response was significantly higher in patients receiving all three doses of eletriptan or sumatriptan vs. placebo (all P< 0.01). Thus, eletriptan and sumatriptan are efficacious in difficult-to-treat patients at high risk for non-response to triptans.
Introduction
In clinical practice, previous treatment response is a useful way of categorizing migraine patients who present for evaluation. Treatment response histories range from patients with new-onset migraine who respond to non-specific analgesics (e.g. non-steroidal anti-inflammatory drugs); to patients who are non-responsive to treatment with more specific acute migraine therapies such as a triptan (5-HT1B/1D agonists) or ergotamine; to patients who are treatment refractory to trials of two or more migraine-specific therapies (1).
An estimated 30–40% of patients do not respond to any given triptan they receive (2). However, this does not necessarily mean these patients are treatment refractory, and a growing body of evidence reports that ‘within-class’ switching is an effective strategy for converting initial non-response to response (3–9). Individuals in this non-responder subgroup are more likely to lapse from medical care than responders (10). Furthermore, because of the reduced productivity associated with untreated migraine and the increased utilization of expensive emergency room services, it would be useful to identify patients who are at high risk of non-response and manage their migraine more effectively. Approaches that can be used include managing patient expectations (even if the first treatment fails, success is likely) and altering the acute treatment strategy by using a triptan that has demonstrated superior antimigraine efficacy (2, 11, 12).
A previous investigation, using multivariate regression methodology on a database of 128 clinical trials (assessing 28 407 migraineurs), has identified severity of baseline headache pain, functional disability and the presence of vomiting as significant predictors of failure to achieve a 2-h pain-free response on sumatriptan [overall model odds ratio (OR) 2.3, 95% confidence interval (CI) 3.3, 6.4] (13). In the current investigation the original findings of this study were replicated for headache severity. Subsequently, the presence of headache severity and other negative response predictors was used to define a high-risk treatment group, based on pooled data, regardless of treatment group. The comparative benefit of eletriptan (20, 40 and 80 mg) and sumatriptan (100 mg) vs. placebo in the subgroup of patients at high risk of failure to achieve a 2-h pain-free response is reported.
Methods
Data were combined from 10 double-blind, placebo-controlled, parallel-group, multicentre studies of eletriptan in the acute treatment of migraine (4, 14–22). (Note that one of the 10 studies has not been published individually.) The current analysis was performed using first-attack data for the following study treatments: eletriptan 20 mg (E20; n = 434), eletriptan 40 mg (E40; n = 3340), eletriptan 80 mg (E80; n = 1854), sumatriptan 100 mg (S100; n = 1126) and placebo (n = 1719).
The combined studies had similar designs, as summarized in previous reports (4, 14–22). Briefly, adult patients were enrolled if they met the International Headache Society criteria for migraine with or without aura (23) and reported a monthly frequency of one to six attacks. Key exclusion criteria were coronary artery disease, heart failure, uncontrolled hypertension or abnormal electrocardiogram; hypersensitivity or known contraindication to treatment with eletriptan or sumatriptan; misuse or abuse of alcohol or other substances, including analgesics or ergotamine; and women who were pregnant or breast-feeding. The study protocols were approved by Institutional Review Boards (Ethics Committees) at each site, and written informed consent was obtained prior to study entry.
Study procedures
All studies were conducted in compliance with Good Clinical Practice guidelines (24). Data collection procedures were similar across all studies. Patients were instructed to take study medication when they experienced a typical migraine attack of moderate or severe intensity that was not spontaneously improving. Patients took study treatment when the aura phase (if present) had ended, and within 6 h of the onset of headache pain. Treatment with study medication was not permitted if the patient had used an analgesic or antiemetic during the previous 6 h, or had taken another triptan or ergotamine-containing or ergot-type medication (e.g. dihydroergotamine) during the previous 24 h. In studies where patients treated more than one attack, only data from the first attack were used. Headache severity was recorded immediately prior to dosing and at a predetermined time postdose. Demographic and clinical characteristics were assessed at the screen visit.
The efficacy outcome used in the current analysis was pain-free response at 2 h postdose.
Statistical analyses
Multivariate logistic regression (Proc Logistic; SAS Inc., Cary, NC, USA), performed on combined data from both the drug and placebo treatment groups, was used to evaluate the extent to which the following variables contributed to 2-h pain-free response rates: gender, age, aura subtype (by history), baseline severity of headache pain, baseline functional disability, baseline nausea or vomiting, baseline photophobia and/or phonophobia, number of previous migraine attacks (number of attacks in the 3 months immediately prior to study initiation) and time of dosing (time between onset of headache and treatment). Factors with P < 0.05 were selected to be included in the shorter model and factors with P > 0.05 were excluded from the model.
Based on the logistic regression, a subgroup of patients at high risk of non-response was identified, as characterized by the presence of three significant predictors of not achieving 2-h pain-free response: severe baseline headache pain, photophobia and/or phonophobia, and nausea. An analysis of variance SAS procedure (GENMOD) was used to evaluate the effect of study treatment on 2-h pain-free response in the high-risk subgroup. This model included pain-free response as the outcome variable, with treatment, age and number of migraine attacks in the past 3 months as covariates.
Results
The combined treatment sample consisted of 8473 patients. The majority of patients (84–86%) were female and aged between 30 and 50 years, reporting two to four migraine attacks per month, typically without aura (Table 1). Severe headache pain was reported in approximately 40% of patients, and approximately 84% reported attack-related impairment as severe, or resulting in bed rest. Demographic and clinical characteristics were similar for each treatment group (Table 1).
Patient baseline demographic and clinical characteristics and characteristics of treated attack
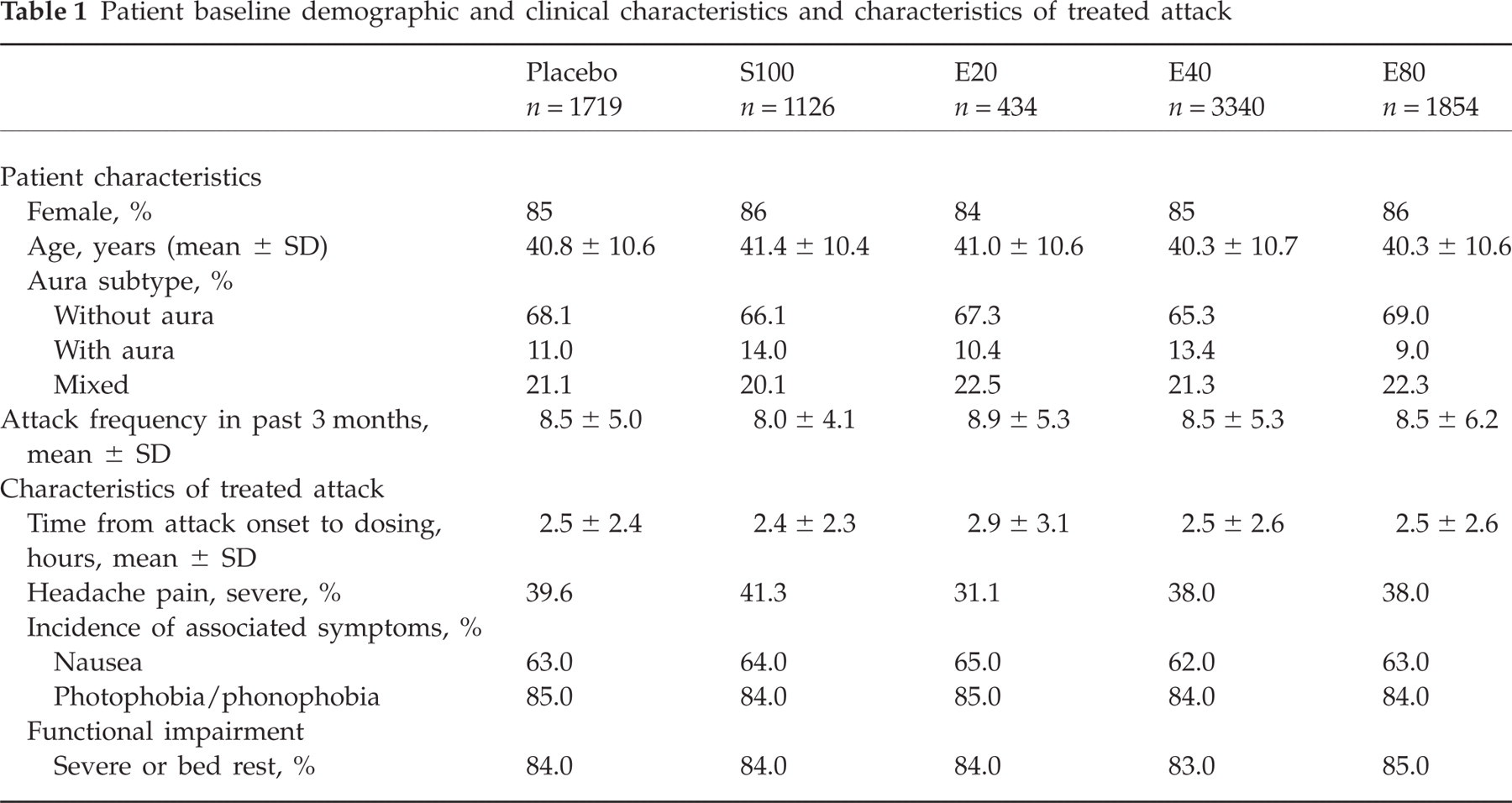
For the total (pooled) sample, treatment with all three doses of eletriptan and S100 was associated with significantly higher 2-h pain-free response compared with placebo (P < 0.0001; Fig. 1).

Pain-free response at 2 h postdose (total sample).
Logistic regression analysis examined the influence of individual demographic and clinical variables on 2-h pain-free response. The strongest predictors of failure to achieve a 2-h pain-free response were: severe baseline headache pain (OR 1.45, 95% CI 1.3, 1.6; P < 0.0001), the presence of photophobia and/or phonophobia (OR 1.37, 95% CI 1.2, 1.6; P < 0.0001) and the presence of nausea (OR 1.24, 95% CI 1.1, 1.4; P < 0.0004; Table 2). The time at which the study drug was taken in relation to headache onset had no influence on 2-h pain-free response (Fig. 2).

Time from headache onset to drug ingestion does not influence pain-free response at 2 h postdose.
Significant pretreatment predictors of failure to achieve pain-free status at 2 h postdose: results of logistic regression analyses (n = 8055)
Effect of study treatment in the high-risk subgroup
A high-risk subgroup was defined based on the presence of the three variables significantly associated with inability to achieve pain-free status at 2 h postdose: the presence at baseline of severe headache pain, photophobia and/or phonophobia, and nausea. The subgroup consisted of 2010 migraineurs (24% of the total treatment sample) in the following treatment groups: E20 (n = 89), E40 (n = 760), E80 (n = 462), S100 (n = 286) and placebo (n = 413).
In this high-risk subgroup, significantly higher pain-free response rates were achieved on E20 (12%; P < 0.005), E40 (22%; P < 0.0001), E80 (28%; P < 0.0001) and S100 (17%; P < 0.0001) than placebo (4%; Fig. 3; Table 3).
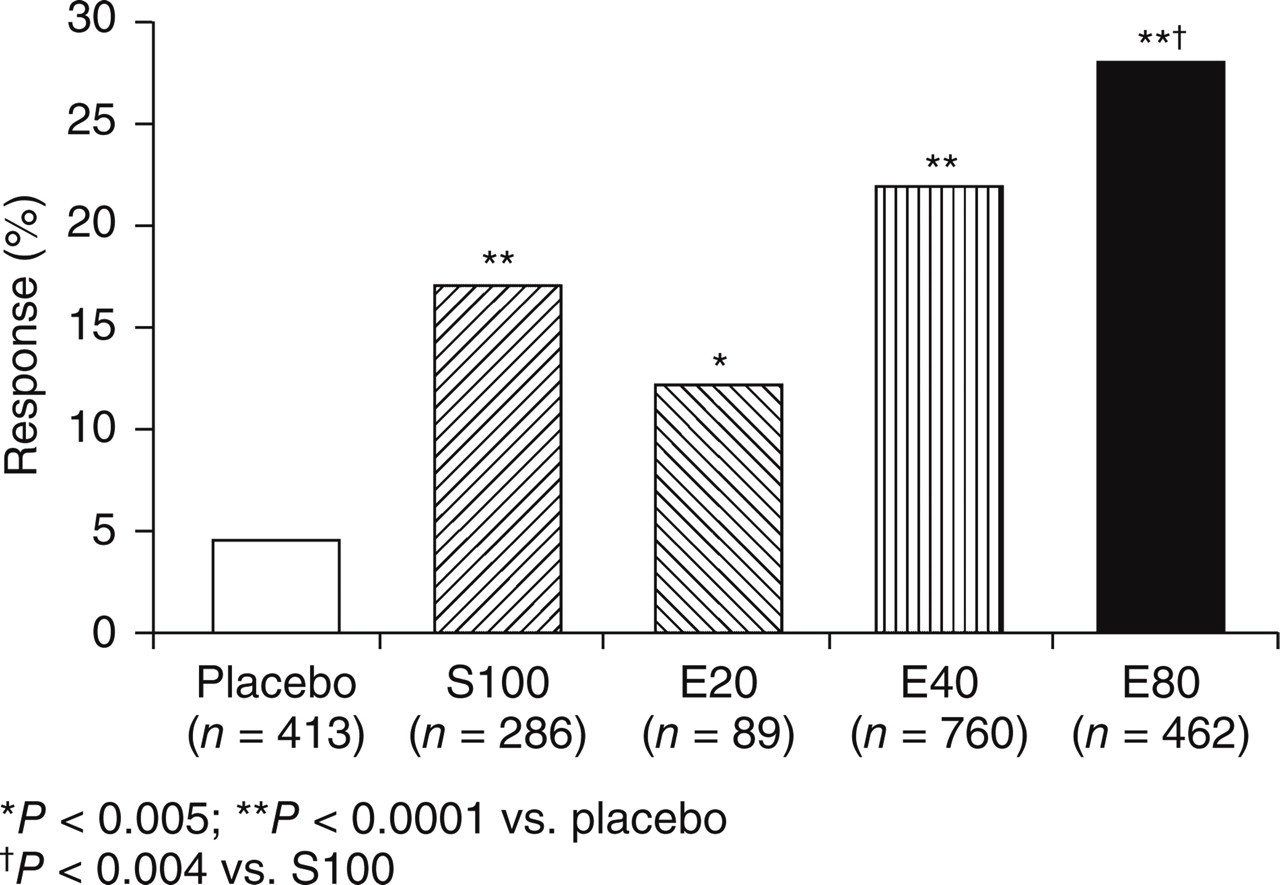
Pain-free response at 2 h postdose: results for the high-risk subgroup.
Discussion
In the current study, multivariate logistic regression, performed on the combined treatment and placebo groups, identified three baseline clinical variables (severe baseline headache pain, photophobia and/or phonophobia, and nausea) as defining a subgroup of patients at high risk of non-response (i.e. inability to achieve 2-h pain-free response). Three other variables (female gender, older age, and lower attack frequency) were also associated with failure to achieve a 2-h pain-free response. Despite recent research suggesting that early dosing significantly improves treatment efficacy (25), the current analysis found that time of triptan administration did not influence either headache response or 2-h pain-free response. The results from these placebo-controlled trials show that baseline headache severity, and not time from onset of the attack until drug intake, more accurately predicts acute treatment outcomes. Thus, early dosing may be effective for a slowly developing migraine if treatment is initiated when headache pain is of mild-to-moderate intensity, but may not have any additional benefits over later dosing in managing attacks where the initial pain intensity is severe. This result is consistent with a previously reported study that found that time of drug administration (relative to headache onset) has less influence on treatment response than baseline pain intensity (26).
The results of the current analysis found eletriptan to be efficacious in the difficult-to-treat subgroup of patients at high risk of non-response. Notable was the steep dose–response curve exhibited by this high-risk subgroup: 12%, 22% and 28% of migraineurs on E20, E40 and E80, respectively, achieved 2-h pain-free response. In contrast, among triptan-naive patients (those who had never used a triptan) the dose–response curve has a much more shallow slope: 20%, 28% and 31% of migraineurs on E20, E40 and E80, respectively, achieved 2-h pain-free response (27).
A previous multivariate analysis of the sumatriptan-controlled trial database has also identified severe baseline headache pain as the most significant negative predictor of 2-h pain-free response (13). The subgroup of patients with severe baseline headache pain in the sumatriptan database analysis had a 2-h pain-free response rate of 19% on S100. This is very similar to the rate of 17% on sumatriptan observed for the high-risk subgroup in the current study, even though the latter subgroup of high-risk patients had additional negative response predictors (13).
These results support the findings of previous studies, confirming severe baseline pain intensity as an important negative predictor of a pain-free outcome at 2 h after the administration of a triptan. The important and pragmatic clinical implication is that patients, where possible, should be strongly encouraged to treat a migraine headache before pain intensity becomes severe to achieve optimal acute treatment outcome.
Competing interests
This study was supported by Pfizer Inc. M.A. and B.P. are employees of Pfizer Inc. H-C.D. has received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, CoLucid, Böhringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Grünenthal, Janssen-Cilag, La Roche, 3M Medica, MSD, Novartis, Johnson & Johnson, Pierre Fabre, Pfizer, Schaper and Brümmer, Sanofi-Aventis and Weber & Weber. He has received financial support for research projects from Allergan, Almirall, AstraZeneca, Bayer, GlaxoSmithKline, Janssen-Cilag and Pfizer. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union. D.W.D. is a consultant/advisor for Allergan, Eli Lilly, Endo, GlaxoSmithKline, Medtronic, Merck, Neuralieve, OrthoMcNeil and Pfizer. He has received research support from Advanced Bionics, AstraZeneca, and Medtronic, and is principal investigator of Phase IIII clinical trials with Allergan and St Jude. P.J.G. has collaborated with, advised and received support for research from Advanced Bionics, Almirall, Amgen, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Medtronic, MSD, Pfizer, and Pozen. R.B.L. has consulted for, received lecture honoraria from and conducted studies funded by Advanced Bionics, Allergan Inc, Bristol-Myers Squibb, GlaxoSmithKline, Johnson & Johnson, Merck, NeuraLieve, Pfizer and ProEthics.
Acknowledgements
This study was supported by Pfizer Inc. The research question was conceived by H-C.D., D.W.D., P.J.G. and R.B.L. Pfizer collated the database, and ran the statistical analysis. The primary data output was analysed collectively during an expert working group that included all authors. The authors would like to acknowledge Anthony Donoghue for statistical programming support. Editorial support was provided by Edward Schweizer, MD and GCL, London, and was funded by Pfizer Inc.
