Abstract
Infusion of nitric oxide (NO) donors is known to induce delayed attacks of migraine and cluster headache or aggravate tension-type headaches in patients suffering from these primary headaches. Previously we have reported that infusion of NO donors in the rat causes delayed neuronal activity in the spinal trigeminal nucleus, which parallels the above clinical observations. Suggesting that endogenous NO production is involved in the generation of primary headaches, we used this animal model of meningeal nociception to determine whether a prolonged increase in NO levels causes an increase in neuronal activity. In anaesthetized rats spinal trigeminal neurons with afferent input from the exposed dura were recorded. Continuous intravenous infusion of the NO donors sodium nitroprusside (25 μg/kg/h) or glycerol trinitrate (250 μg/ kg/h) for 2h induced a persisting increase in neuronal activity but no change in systemic blood pressure. In this activated trigeminal system the calcitonin gene-related peptide (CGRP) receptor antagonist BIBN4096BS (900 μg/ kg) was infused. Spinal trigeminal activity was significantly reduced within minutes and to a similar extent as previously reported in animals not treated with NO. Slow continuous NO infusion may be a model of the active headache phase, and inhibition of CGRP receptors can reverse the induced neuronal activity.
Introduction
Intravenous infusion or sublingual administration of the nitric oxide (NO) donor glycerol trinitrate (GTN) has long been known to induce mild to moderate headaches in both healthy subjects and patients suffering from primary headaches (1–4). Patients suffering from migraine and tension-type headaches have been found to respond to the administration of NO donors with immediate mild headaches of higher pain scores than healthy persons (4, 5). In migraine patients, with a delay of several hours following the infusion of GTN, this immediate headache is followed by a second headache phase that frequently fulfils the criteria of genuine migraine attacks (3, 6).
Such a biphasic response to NO has also been observed in an experimental headache model in rats. Systemic infusion of the NO donor sodium nitroprusside over 10 min induced an immediate transient and a second delayed increase in activity of neurons in the spinal trigeminal nucleus (STN) with meningeal afferent input (7). Infusion of large doses of GTN provoked c-fos expression, a marker for predominantly nociceptive input in rat brainstem nuclei (8, 9).
Conversely, inhibition of endogenous NO synthesis has been reported to relieve ongoing migraine attacks (10, 11) and reduce the severity of chronic tension-type headaches (12). Accordingly in rats, inhibition of endogenous NO synthesis with NG-nitro-L-arginine methyl ester decreased spinal trigeminal activity (7, 13). These results suggest that the activity of central trigeminal neurons is at least partially controlled by a tonic NO effect. Thus, the first aim of this study was to test if a weak but long-lasting increase in systemic NO levels activates spinal trigeminal neurons with meningeal afferent input.
In culture, the effects of a prolonged increase in NO levels on trigeminal neurons have been studied (14). Exposure to a NO donor for 1 h stimulated the release of calcitonin gene-related peptide (CGRP), a neuropeptide predominantly found in nociceptive neurons. CGRP is known to generally modulate central pain processing (15–17), but it seems to have particular significance for the generation of headaches. Indeed, infusion of CGRP produced migraine-like headaches in migraineurs (10). A key role of CGRP in migraine pain is evidenced by the effectiveness of the CGRP receptor antagonist BIBN4096BS in treating migraine patients (18). In animal models BIBN4096BS has been shown to inhibit ongoing trigeminal activity, as well as responses to electrical and thermal stimulation of peripheral receptive fields (19, 20). However, its effectiveness in a state of increased activity, as possibly caused by endogenous release of NO, has not thus far been demonstrated. Therefore, the second aim of this study was to examine whether increased activity caused by prolonged NO infusion can be reversed by blockade of CGRP receptors.
Materials and methods
Anaesthesia and general treatment
The experimental protocol was reviewed by an ethics committee and approved by the local government. Male Wistar rats with body weights of 290–360 g were used. Surgical and recording procedures were performed largely as previously described (21). Rats were anaesthetized with 2.5% isoflurane for surgery periods. Compared with the thiopental anaesthesia as used in previous experiments, a constant depth of narcosis besides a faster induction is a clear advantage. The femoral artery was cannulated to monitor mean arterial blood pressure. Both femoral veins were cannulated to allow an independent application of the CGRP receptor antagonist and the NO donor. The animals were transferred to a stereotactic frame, paralysed with intravenous administration of gallamine triethiodide (40 mg/kg) and artificially ventilated with oxygen-enriched room air (about 30% oxygen). Expiratory CO2 was monitored and maintained at 4.5–5.0%, body temperature was maintained at 37.0–37.5°C with a feedback-controlled homoeothermic system (TKM 0902; FMI GmbH, Seeheim-Ober Beerbach, Germany). Vital parameters (blood pressure, expiratory CO2 level and body temperature) were continuously monitored throughout the experiment. The skin was transected in the midsagittal line. Using a dental drill, a cranial window of 6 × 4 mm (rostral-caudal and medial-lateral, respectively) was carefully cut into the left parietal bone to expose the dura mater. During surgery and throughout the experiment the dura was protected from drying with isotonic saline. The neck muscles were divided and the medullary brainstem was exposed by cutting a window into the atlanto-occipital ligament and the underlying dura mater. After completion of the surgical procedures, the isofluorane level was maintained at 1.5% throughout the experiment, which was sufficient to suppress nocifensive responses evoked by noxious pinch stimuli of the hindpaw and the earlobe. Custom-built carbon fibre glass microelectrodes with an impedance of 1–6 MΩ were inserted into the ipsilateral medulla and advanced or retracted at 2.5-μm steps using a SMS87 microstepper (TC Elektronik, Würzburg, Germany). Single shock stimuli (pulses of 0.5 ms, 5–10 V, 0.2 Hz) were delivered to the exposed dura through bipolar electrodes with their rounded tips touching the dural surface. Neurons in the subnucleus caudalis of the STN with input from meningeal afferents were identified by their discharge in response to electrical stimulation of the dura with a stable latency and by their responses to mechanical probing of the dura. The mechanical threshold was determined with von Frey filaments (0.5–10 mN). The signal was band-passed and amplified with a custom-built amplifier, digitized at a rate of 16 kHz and saved for offline analysis (1401 μ, Spike2 Software; Cambridge Electronic Design, Cambridge, UK). The position of each recording site was determined by measuring the distance caudal and lateral to the obex of the electrode and by reading the depth of the microstepper.
Experimental protocols
The ongoing activity of all units was recorded for a control period of ≥ 30 min. Four experimental groups were designed by infusion of one of two NO donors followed by either saline or CGRP receptor antagonist BIBN4096BS; the control group received identical volumes of saline. The NO donors GTN (250 μg/kg/h; Schwarz Pharma, Monheim, Germany) and sodium nitroprusside (SNP 25 μg/kg/h; Sigma, St Louis, MO, USA) were dissolved in 1 ml saline; either of them was infused at a constant rate of 0.5 ml/h; no change in systemic blood pressure was observed. BIBN4096BS (900 μg/kg in 250 μl, provided by Boehringer-Ingelheim, Biberach, Germany) was infused over a period of 5 min into the second femoral vein catheter to avoid changes in the flow rate of the concurrently injected NO donor. Infusions were controlled by a syringe pump (Harvard Apparatus, Holliston, MA, USA). Experiments were terminated with a lethal dose of intravenous thiopental.
In 11 animals at least 120 min of continuous NO donor infusion was recorded (six with GTN, five with SNP) to investigate the development of neuronal activity. In 12 additional animals, six for each NO donor, BIBN4096BS was infused over a period of 5 min. The infusion was started between 65 and 80 min after the NO donor, when the online display of neuronal activity was stable within the previous 10 min.
Data analysis
Extracellular action potential waveforms were analysed off-line using Spike 2 software. Constant latency within post-stimulus histograms was used to build the template of the action potential shape. Further action potentials were automatically added to the existing templates based on similarity in amplitude during a 2 ms period. Neuronal activity per 30 s was exported for further analysis.
For statistical analysis neuronal activity was normalized to a pre-infusion period of 20 min for the NO donors and 10 min for BIBN4096BS. Changes in neuronal activity were analysed by an
Pairwise correlations between location coordinates, latency to electrical stimulation, ongoing activity and relative changes of activity after infusion of NO donors or BIBN4096BS were calculated. Data are given in mean ±
Results
General properties of neurons
In 30 male Wistar rats, one spinal trigeminal neuron with meningeal receptive fields per animal was recorded in the left caudal subnucleus of the STN. The recording sites were located 1.7/2.6/3.9 mm (min/mean/max) caudal to the obex, 1.1/1.8/2.2 mm lateral from the midline and at a depth of 0.3/0.9/1.5 mm from the dorsal surface of the medulla (Fig. 1). The electrical threshold at which discharges could be evoked by electrical stimulation of the dura mater was between 4.5 and 10 V. At threshold, the minimal response latency to dural electrical stimulation varied from 8 to 15 ms, suggesting afferent input from C and A δ fibres. Each electrical pulse was followed by only one response with a constant latency, even at stimulation intensities of two to three times the threshold intensity, indicating that only a single spinal trigeminal neuron was activated. Dural receptive fields were close to branches of the middle meningeal artery. Dural von Frey thresholds varied between different units from 0.5 to 8 mN. Most units had low mechanical threshold facial receptive fields predominantly in the ophthalmic region, as reported in previous studies (21, 22). The rate of mean ongoing activity varied from 0.01 to 1.9 Hz, the mean rate was 0.4 ± 0.1 Hz. Pairwise product–momentum correlations of all acquired data showed that the latency between electrical stimulation and the elicited action potentials in the STN was shorter for neurons located more caudally (R =−0.56, P = 0.004), as previously reported (7).
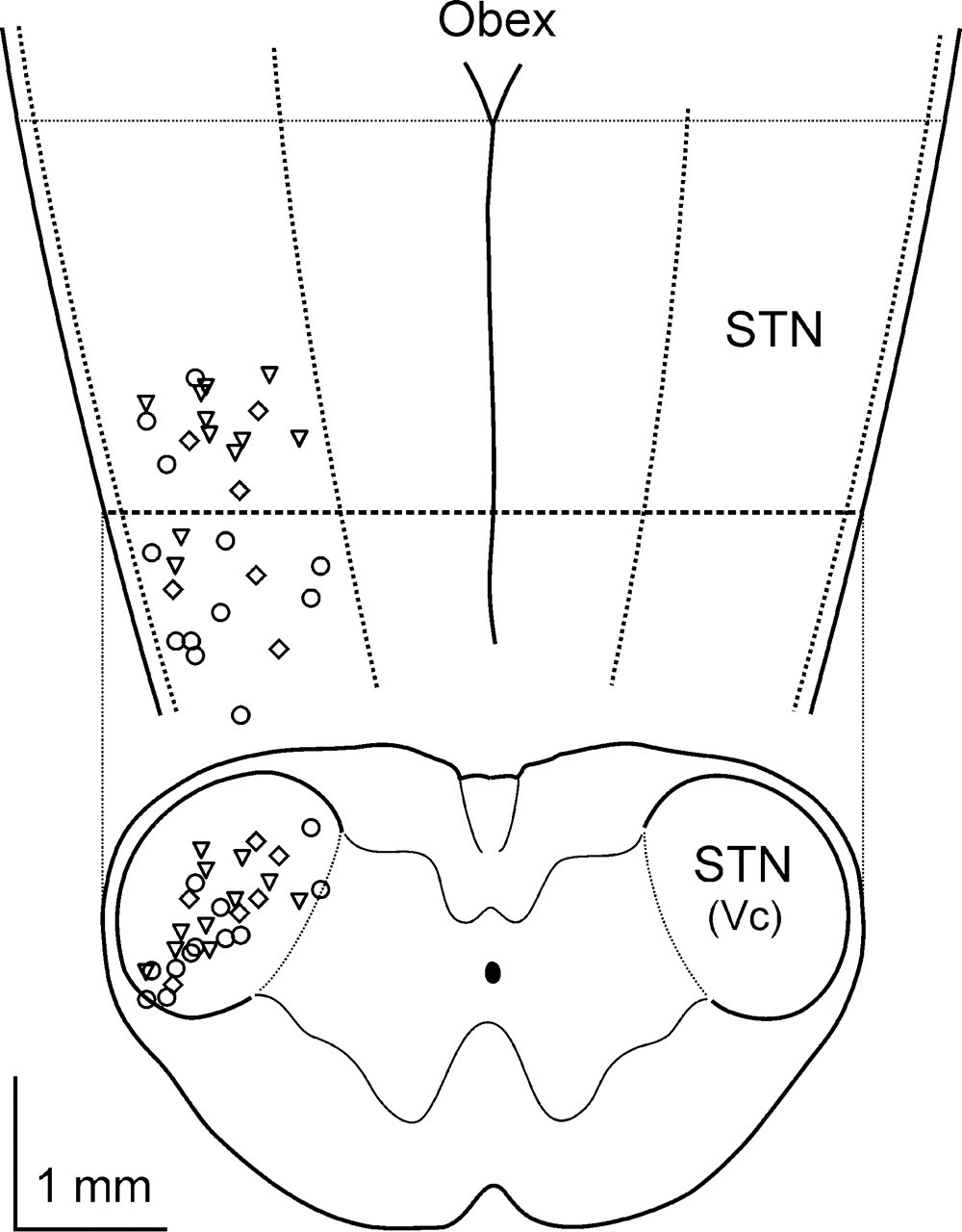
Schematic dorsal and horizontal view of the brainstem. Recording electrode positions are marked with circles (glycerol trinitrate), triangles (sodium nitroprusside) or rhombi (controls).
NO donors
After a control period of 20 min, the NO donor infusion was started and maintained until the end of the experiment. Continuous infusion of SNP increased the activity of spinal trigeminal neurons starting 30 min after the onset of application (n = 11, repeated measurements

Ongoing activity of two spinal trigeminal neurons after infusion of the nitric oxide donor sodium nitroprusside (SNP). In the experiment in the lower panel, the calcitonin gene-related peptide receptor antagonist BIBN4096BS was infused within a period of 5 min.

Continuous infusion of nitric oxide (NO) donors stimulated the ongoing activity of spinal trigeminal neurons. The activity was normalized to the last 20 min before application (open columns). Sodium nitroprusside (SNP, upper panel, n = 11) or glycerol trinitrate (GTN, middle panel, n = 13) increased the activity. Continuous NO donor infusion periods are visualized in grey columns. In control animals (lower panel, n = 6) no change in activity was observed. ∗P < 0.05.
CGRP receptor inhibition
After infusion of BIBN4096BS, neuronal activity was lower compared with animals treated with NO only (two-way

Infusion of the calcitonin gene-related peptide receptor antagonist BIBN4096BS reduced the nitric oxide donor-stimulated activity of spinal trigeminal neurons. The upper panels demonstrate the effect for sodium nitroprusside (SNP, n = 6) and glycerol trinitrate (GTN, n = 6) compared with controls (n = 5 and 6). The lower panel is a summary comparing all BIBN4096BS treated neurons (n = 12) vs. controls (n = 11). The activity was normalized to the last 10 min before BIBN4096BS infusion. ∗P < 0.05.

Changes of spinal trigeminal activity due to the nitric oxide (NO) donors (grey columns) and BIBN4096BS infusion (black columns) is summarized by normalization to a common 20-min control period (open columns). Note that BIBN4096BS reversed 81% of the NO donor-dependent increase of ongoing activity for sodium nitroprusside and 74% of that for glycerol trinitrate. ∗P < 0.05 compared with control period. #P < 0.05 between animals treated or not treated with BIBN4096BS.
Discussion
In the present study, long-lasting infusions of low doses of NO donors caused persisting increases in neuronal activity in the STN. The effects were similar for the NO donors SNP and GTN. Subsequent CGRP receptor inhibition reduced the elevated activity to about 60% within 7.5 min, similar to previous observations in spinal trigeminal neurons without NO donor pretreatment (19).
General properties of neurons in the spinal trigeminal nucleus
All recorded neurons had convergent input from the parietal dura and facial areas. Recording positions, response latencies to electrical stimuli or activation thresholds of dural mechanical stimulation were not correlated with the changes in neuronal activity after infusion. There was also no correlation between the change in activity after NO donors and previous ongoing activity, allowing the conclusion that the increased activity is not conveyed by a small subpopulation of neurons but is rather a common phenomenon of central trigeminal neurons. Also, the relative reduction of activity by CGRP receptor inhibition was independent of previous and NO-stimulated activity. The previously reported negative correlation of the response latency to dural electrical stimulation with the rostro-caudal coordinates of the units could be confirmed (19). Properties of spinal trigeminal neurons included in this study are similar to those of previously published studies.
Effect of NO donors
In our previous experiments injection of 50 μg/kg SNP over a period of 10 min was followed by a biphasic response, an immediate and a delayed increase in firing rate in the STN neurons (7). This is reminiscent of the biphasic pattern in the time course of headaches provoked by NO donors in migraine patients (4) and in chronic tension-type headache patients (23). Therefore, briefly elevated NO levels induce activity directly, but seem also to induce a process that stimulates the development of delayed activity and headache, respectively. The short half-life of NO suggests the involvement of a transcriptional process. Subcutaneous injection of the NO donor nitroglycerin has been found to increase the number of neuronal nitric oxide synthase (NOS) immunoreactive neurons in the caudal trigeminal nucleus in rats (24), and inducible NOS expression has been observed in macrophages of the rat dura mater following intravenous GTN infusion (25). Thus, injection of NO donors can increase the number of NO-producing cells in peripheral and central trigeminal tissues and induce endogenous NO release in a feed-forward manner. The delay in activation after a single NO donor infusion may be explained by the time needed for the recruitment of such a mechanism.
We suggest that the slow continuous NO donor infusion mimics elevated endogenous NO production. Although the dose of the NO donor SNP was 0.42 μg/kg/min compared with 5 μg/kg/min in the previous study, persistently increased neuronal activity appeared faster in the present study. This may support an important role for continuously elevated NO production in inducing and maintaining increased trigeminal activity. Furthermore, the low injection rate did not cause a decrease in systemic blood pressure, as observed with the previously used higher infusion rate. The slow infusion of low doses of NO donors may be an advantageous option also for clinical migraine models (see below).
In the present study two different NO donors were applied to confirm that the observed changes in trigeminal activity are mediated by NO. SNP breaks down spontaneously, releasing NO and cyanide ions, whereas GTN is enzymatically degraded to NO and 1,2-glyceryl dinitrate by mitochondrial aldehyde dehydrogenase, glutathione S-transferases, cytochrome P450 or xanthine oxidoreductase (26). Since the effects of the two NO donors were similar, we conclude that the observed changes in trigeminal activity are indeed mediated by NO.
CGRP receptor inhibition
In migraine patients infusion of CGRP has provoked characteristic delayed headaches (27). Correspondingly, the novel non-peptide CGRP receptor antagonist BIBN4096BS has been shown to be effective for the acute treatment of migraine (18). In humans, plasma CGRP concentrations have been reported to be increased during both spontaneous and NO-provoked migraine attacks, returning to baseline after cessation of the attack (28, 29), although there is also conflicting evidence about this measurement (30). In an animal model elevated CGRP levels upon electrical stimulation of the cranial dura mater in cats have been reported (31).
Recordings of trigeminal nucleus caudalis neurons in animal experiments reflect the effectiveness of CGRP receptor inhibition. Iontophoretic administration of BIBN4096BS has been shown to inhibit nociceptive transmission in the cat STN (20). We have previously demonstrated that intravenous injection of BIBN4096BS suppresses the ongoing activity of spinal trigeminal neurons as well as the activity evoked by heat stimulation of the exposed cranial dura (19). However, it was unclear if blockade of CGRP receptors could also reduce the activity driven by NO. Indeed, the present study shows that BIBN4096BS can reduce the increased activity. The experimental protocols included 300 μg/kg and subsequently 900 μg/kg BIBN4096BS in the previous study, which does not allow direct statistical comparison with the results of the present study. However, the time course of inhibition is similar, and the relative inhibition by 900 μg/kg BIBN4096BS after the NO donors in the current study is between the inhibition by 300 μg/kg and the cumulative 1200 μg/kg in animals not treated with NO. We conclude that CGRP receptor inhibition can effectively reduce ongoing spinal trigeminal activity, regardless of whether it is spontaneous or induced by NO donors.
Site of action of NO and CGRP
NO is able to affect cutaneous and subcutaneous nociceptive fibres, but did not yield consistent effects (32, 33), supposedly depending on the subset of primary sensory neurons (34). Peripheral effects of NO on trigeminal afferents are also controversial. In the hemisected rat skull preparation some NO donors have been shown to increase the release of CGRP from the cranial dura in vitro (35). In a study on primary meningeal afferents in the anaesthetized rat, the NO donor SNP applied onto the exposed cranial dura sensitized seven neurons out of 19 to mechanical stimuli, seven were desensitized, and the remaining five were unaffected (36). In summary, no consistent net effect on mechanical sensitivity and no change in ongoing activity were elicited by dural NO donor application.
There is no evidence for a peripheral effect of CGRP. In vivo recordings of primary afferent neurons have not shown any sensitizing or activating effect (37). There was no immunohistochemical evidence for CGRP receptors located on peripheral axons of trigeminal neurons innervating the rat cranial dura, although the same study found these receptors on central axons projecting to the STN (38). Local inhibition of CGRP receptors in the cranial dura did not reduce the activity of spinal trigeminal second-order neurons with afferent input from the treated dura (19).
On the other hand, NO is known to facilitate the release of neurotransmitters including CGRP in the dorsal horn (39–41). NO is involved in the induction and maintenance of central sensitization after peripheral tissue injury and inflammation (42). Intrathecal application of SNP, locally applied to the exposed medulla, increased the spinal trigeminal activity comparable to intravenous infusion (7). Assuming that NO release in the STN is an important mechanism to facilitate CGRP release, and given that CGRP is involved in nociceptive transmission, blockade of central CGRP receptors should be effective in inhibiting spinal trigeminal activity. Indeed, local administration of CGRP at the central synapse by means of iontophoresis has been shown to modify the transmission of peripherally evoked activity to the second-order neurons in the STN (20). Compared with the high potency of CGRP receptor antagonist BIBN4096BS, the effective intravenous dose was rather high (19). It may be that the antagonist is required at a high concentration, because it is barely blood–brain barrier permeable or because its affinity to the CGRP receptor is lower in rodents compared with other species (43, 44). CGRP is sparsely found in ascending pathways beyond the level of the STN, making an effect at a thalamic or cortical site rather unlikely.
Role of afferent activity
Slowly infused GTN has been reported not to elicit a significant increase in c-Fos+ cells in the STN without noxious stimulation (45) but, after noxious stimulation, NOS inhibition has been shown to reduce the number of c-Fos+ spinal dorsal horn neurons (46). Therefore, it may be suggested that NO production in the trigeminal nucleus is effective in facilitating nociceptive transmission only if there is at least a minimum of ongoing nociceptive input, as may be the case in our experiments with an open cranial window. Indeed, recent experiments from our group provide evidence for the assumption that the peripheral input is important for maintaining the central activity in the STN (47). Deprivation of peripheral inputs by microinjection of lidocaine into the trigeminal ganglion was followed by a substantial decrease in both the receptive field size and the activity of the recorded units. Thus, in the present study both the NO donor infusion and the preparation may be independent stimuli for the activation of central trigeminal neurons that can be reduced by CGRP receptor inhibition. This assumption is supported by the fact that the relative change of activity due to CGRP receptor inhibition in control (but trepanized) animals vs. animals treated with NO was similar (7). As a working hypothesis, the unavoidable trepanation necessary to reach meningeal receptive fields may change the intact state of the trigeminal system into a pathological condition, which may be more comparable to the latent affected state of the trigeminal system of patients suffering from primary headaches. Some of these conditions may be associated with up-regulation of NO synthesis. Elevated concentrations of NO metabolites in blood samples from the internal jugular vein measured during migraine attacks may indicate an increased NO turnover (48, 49). In addition, increased plasma nitrite concentrations have been found also between attacks of migraine and cluster headache (50). Similarly, the cerebrospinal fluid of patients suffering from chronic daily headache contains elevated concentrations of nitrites (51).
For a clinical interpretation of our data, we may speculate, first, that NO provocation is effective in activating the central trigeminal system in a delayed manner predominantly if it is already sensitized, which may also be the case in patients suffering from primary headaches; second, that CGRP receptor inhibition is effective in reducing this central activity, particularly under these sensitized conditions.
Footnotes
Acknowledgements
The authors thank Jana Schramm for her excellent technical assistance. BIBN4096BS was kindly provided by Boehringer-Ingelheim. The study was supported by the BMBF (German Headache Consortium).
