Abstract
Background
Initiating mechanisms of migraine headache remain poorly understood and a biomarker of migraine does not exist. Inflammation pertaining to the wall of cerebral arteries and brain parenchyma has been suggested to play a role in migraine pathophysiology.
Objective
We conducted the first experimental human study to investigate macrophage-mediated inflammation as a possible biomarker of migraine.
Methods
Using ultrasmall superparamagnetic iron oxide (USPIO)-enhanced 3T magnetic resonance imaging (MRI), we investigated the presence of macrophages in cerebral artery walls and in brain parenchyma of patients with migraine without aura. We used the phosphodiesterase-3-inhibitor cilostazol as an experimental migraine trigger, and investigated both patients who received sumatriptan treatment, and patients who did not. To validate our use of USPIO-enhanced MRI, we included a preclinical mouse model with subcutaneous capsaicin injection in the trigeminal V1 area. The study is registered at ClinicalTrials.gov with the identifier NCT02549898.
Results
A total of 28 female patients with migraine without aura underwent a baseline MRI scan, ingested cilostazol, developed a migraine-like attack, and underwent an USPIO-enhanced MRI scan > 24 hours after intravenous administration of USPIO. Twelve patients treated their attack with 6 mg s.c. sumatriptan, while the remaining 16 patients received no migraine-specific rescue medication. The preclinical model confirmed that USPIO-enhanced MRI detects macrophage-mediated inflammation. In patients, however, migraine attacks were not associated with increased USPIO signal on the pain side of the head compared to the non-pain side.
Conclusion
Our findings suggest that migraine without aura is not associated with macrophage-mediated inflammation specific to the head pain side.
Introduction
Migraine is a global health concern and the second leading cause of years lived with disability (1). Migraine attacks are recurrent and characterized by severe and usually throbbing headache (2). Despite important advances in our understanding of this complex disease, key aspects of migraine pathophysiology remain a conundrum. Notably, it is still unclear where and how the pain of migraine originates.
Ample evidence suggests that migraine headache cannot be attributed to the simple mechanics of vasodilation (3). The pain of migraine may instead stem from localized inflammation, involving leukocyte recruitment and mast cell degranulation, to activate the perivascular trigeminal nociceptors that densely innervate the dural and cerebral arteries. The importance of inflammation is supported by the involvement of prostaglandins in migraine pathogenesis (4) and the treatment efficacy of non-steroidal anti-inflammatory drugs and steroids (5–7). While the selective migraine abortive drug sumatriptan is traditionally considered a vasoconstrictive agent, possible actions also include attenuating plasma levels of CGRP (8) and blocking plasma protein extravasation (9), processes that may well play a role in the inflammatory response. Interestingly, there are remarkable phenomenological similarities between the clinical presentations of migraine and meningitis (8); however, an inflammatory biomarker of migraine has not been detected in humans thus far.
For the past decades, ultrasmall superparamagnetic particles of iron oxide (USPIO) have been used as magnetic resonance imaging (MRI)-detectable macrophage biomarkers. Upon intravenous administration, USPIO nanoparticles are engulfed by macrophages in inflamed tissue and can be detected with MRI (9). USPIO-enhanced MRI has been used to study a variety of inflammatory processes within the central nervous system, including stroke, intracranial tumors, and multiple sclerosis (10–13), with an increase in USPIO signal indicating the presence of macrophages and ongoing inflammation. USPIO-enhanced MRI has not been used to study migraine.
Here, we applied high-resolution USPIO-enhanced MRI at 3T to investigate the walls of cerebral arteries, the dura mater, and pre-specified regions of interest (ROIs) within the brain parenchyma of patients with migraine without aura. We used the highly effective phosphodiesterase-3-inhibitor cilostazol as an experimental migraine trigger (14). We hypothesized that unilateral attacks of migraine without aura would be associated with inflammation on the head pain side, manifesting as increased USPIO signal. We also hypothesized that therapeutic use of sumatriptan, a 5-hydroxytryptamine agonist, would block this inflammatory response. To verify the link between macrophage-mediated inflammation and USPIO enhancement, we investigated the effect of subcutaneous capsaicin injection in the trigeminal area on USPIO-MRI in a mouse model (Figure 1).
Study procedures for migraine patients who underwent the MRI protocol with or without receiving sumatriptan treatment.
Methods
Participants
The Ethical Committee for the Capital Region of Denmark approved the study (H-15005669), which was conducted in accordance with the Helsinki Declaration of 1964 with later revisions. The study is registered at ClinicalTrials.gov with the identifier NCT02549898. All participants provided written informed consent to participate in the study.
Patients were eligible for inclusion if they were females aged 18–50 years and had a verified diagnosis of migraine without aura as defined by the International Headache Society classification (2). Patients were eligible for inclusion if they reported their spontaneous migraine as unilateral in ≥70% of attacks. All patients were prescreened with cilostazol, and only patients who developed a unilateral migraine-like attack after the first cilostazol induction were included in the study. Exclusion criteria included any other type of headache (apart from episodic tension-type headache ≤5 days per month), any history of somatic or psychiatric conditions requiring medical treatment at the time of study, pregnant or nursing women, drug misuse, daily intake of medication (apart from oral contraceptives), contraindications for MRI (i.e. any type of metal implantation in the body or claustrophobia), history of atopic or drug allergy, evidence of iron overload assessed by biochemical detection of ferritin, iron saturation, and genetic screening of the HFE gene for hereditary haemochromatosis.
We also enrolled healthy controls for which exclusion criteria additionally included any history of migraine with or without aura as well as first-degree relatives with history of migraine.
All participants underwent a full medical examination and pregnancy testing. Enrolment was performed at Rigshospitalet Glostrup between January 2015 and April 2017.
Procedures
All patients reported to the clinic migraine-free for at least 5 days, and headache-free for at least 3 days. Coffee, tea, cocoa, alcohol, other methylxanthine-containing beverages, and tobacco were not allowed for at least 12 hours before study start. Patients underwent a baseline scan and then ingested 200 mg cilostazol (Pletal®, Otsuka Pharmaceutical Europe Ltd) orally. Headache intensity and characteristics were recorded every hour from cilostazol ingestion on a verbal rating scale from 0 to 10 (0; no headache, 1; very mild headache, 10; worst imaginable headache). After onset of migraine, 12 out of 30 patients were allocated to treat their migraine attack with 6 mg subcutaneous injection of sumatriptan (Imigran®, GlaxoSmithKline). Patients who did not treat with sumatriptan were permitted to treat their headache only with promethazine and metoclopramide, in order to exclude any direct drug effect on the cerebral vasculature.
All patients received ferumoxytol administration (Feraheme®, AMAG Pharmaceuticals Inc) intravenously at a dose of 5 mg Fe per kg body weight diluted in isotonic saline in the ratio 1:3. Ferumoxytol infusion took place over 30 minutes through a peripheral intravenous cannula, with continuous monitoring of vital signs including blood pressure, heart rate, and respiratory rate for 2 hours after ferumoxytol administration. All patients underwent an USPIO scan 27 hours after ferumoxytol administration. This timepoint was elected to increase the likelihood that ferumoxytol particles had traversed the blood pool phase to be located within macrophages in the inflamed tissue (9). As part of the MRI protocol, patients were also scanned at migraine onset and after sumatriptan treatment (data published elsewhere).
All healthy controls underwent a baseline scan, USPIO infusion, and an USPIO scan. Two of the healthy controls also ingested cilostazol.
Imaging protocols
Data acquisition and imaging protocols for 3T magnetic resonance imaging of migraine patients.

The MRI protocol included a sagittal 3D black blood volumetric isotropic turbo spin-echo sequence (VISTA), an axial T2*-weighted sequence with 11 echoes with echo time TE1/ΔTE (ms) of 4·6/4·6· an axial T1-weighted sequence with 7 inversion times (seconds) of 0·12, 0·3, 0·6, 1·0, 2·0, 4·0, and 10·0, and a high-resolution MR angiography sequence.
N/A: not applicable.
TE1/ΔTE.
Quantitative image analysis
3D T1 black blood VISTA images
We used 3D black blood MRI images to investigate vessel wall signal intensity in the MCA and cavernous part of the internal carotid (ICAcavernous) artery. The luminal contours of the chosen vessels were manually delineated in a semi-automated manner (VesselMass, Leiden University, the Netherlands), and the abluminal contour was automated with a predefined wall thickness for each vessel segment (MCA 0.5 mm, ICAcavernous 0.6 mm).
To address any potential spatial and temporal variations in regions of interest on the 3D T1 black blood VISTA sequence that are not normalized to a control region, we first tested for differences between the pain and non-pain sides at baseline, and subsequently quantified USPIO uptake by testing for differences in signal intensity between the pain and non-pain sides at the post-USPIO scan.
T2* and T1-weighted images
We used T2* and T1-weighted MRI images to investigate signal intensity in ROIs within the brain parenchyma, specifically the thalamus, pons, anterior cerebral artery (ACA) territory, middle cerebral artery (MCA) territory, and posterior cerebral artery (PCA) territory. We chose ROIs rather than whole-brain analysis to increase the probability that even discrete differences in USPIO uptake would be identified. ROIs were drawn manually in FSLVIEW (version 3.2.0) and Matlab (version 8.4.0) on R2* (R2* = 1/T2*) and R1 (R1 = 1/T1) maps. Each ROI included both grey and white matter for a given vascular territory. To quantify USPIO uptake, ΔR2* and ΔR1 values were calculated (post-USPIO minus baseline) and compared between the pain side and non-pain sides.
The reader (SK) was blinded to scan sequence, treatment arm, and headache laterality.
As an explorative addition, we performed two post-hoc analyses to investigate bilateral differences in USPIO uptake of all ROIs and vessel segments a) comparing patients who received sumatriptan treatment with those patients who did not, and b) comparing patients who reported ongoing headache at the time of the USPIO scan with patients who were headache free. For the latter comparison, we only included patients who had not received sumatriptan treatment.
For these explorative analyses, we applied the mean of the pain and non-pain side values (a measure of bilateral USPIO uptake for a given ROI) of patients and tested for differences between signal intensity, ΔR2*, and ΔR1 changes for the sumatriptan treated group compared to the non-sumatriptan treated group, and for patients who reported ongoing headache and patients who were headache free.
Qualitative image analysis
An experienced radiologist (DT) visually assessed the MR angiography scans of all patients and healthy controls for presence of dural enhancement. A binary rating system was applied, where 0 signified no enhancement, and 1 signified enhancement. Both the left and right side of the dura mater was visually assessed and rated, both for patients who had received sumatriptan and those who had not. The radiologist was blinded to treatment and headache laterality.
Statistical analysis
We calculated our sample size based on detection of at least 20% change in arterial wall thickness between the pain side and non-pain side during attack, at 5% significance (two-tailed) and with 80% power. The estimated standard deviation of arterial wall thickness changes was assumed to be 16%. Based on these assumptions, we calculated that 10 patients should be included, but increased our sample size to 30 for increased confidence in our findings. We also included four healthy controls for a descriptive investigation of USPIO uptake over time.
All absolute values are presented as mean ± standard deviation. Percent changes are reported as mean and 95% confidence intervals (CI). Headache score is presented as median with interquartile range.
Primary endpoints of the study were a) difference in signal intensity change between the pain side and the non-pain side during unilateral migraine attacks, and b) difference in signal intensity change between the pain side and the non-pain side during unilateral migraine attacks after sumatriptan treatment.
We tested for side-to-side differences in signal intensity, ΔR2*, and ΔR1 values between the pain-side and the non-pain side using the paired t-test. For the exploratory analysis, we tested for differences in bilateral ΔR2* and ΔR1 between the sumatriptan treated and non-sumatriptan treated group and between patients with ongoing headache at the time of the USPIO scan and patients who were headache free using the independent samples t-test. Headache status at time of USPIO scan was reported as a binary variable (yes/no). We made no adjustment for multiple analyses.
We used SPSS (version 23.0) for all statistical analyses. All
Preclinical studies
As a proof-of-concept for preclinical translation of USPIO enhancement specific to the pain side in migraine without aura, we investigated a capsaicin mouse model with MRI after infusion of USPIO. All animal experiments were approved by and conducted under the guidelines of the Danish Animal Experiments Inspectorate. Four BalbC mice from Charles River (Wilmington, Massachusetts, USA) were included at the age of approximately 8 weeks, after one week of acclimatization on site. The model setup included two subsequent subcutaneous injections of 100 µg capsaicin (Sigma Aldrich, CAS NO. 404-86-4, St. Louis, Missouri, USA), diluted in sterile PBS including 10 % DMSO. Injections were placed subcutaneously above the area of bregma with 24 hours between the injections. After the last capsaicin injection, USPIO nanoparticles were injected i.v. through the tail vein in a concentration of 500 µmol Fe/kg, and MRI scan performed at 27 hours post-injection (Figure 2). Two mice received capsaicin injection and USPIO, one mouse received capsaicin and no USPIO, and one mouse received no capsaicin and no USPIO. In order to verify the presence of macrophages in the area of capsaicin deposit, 2D Bioluminescence scans were acquired on IVIS Lumina XR (Perkin Elmer, Waltham, Massachusetts, USA) 25 hours after USPIO injection, using the chemiluminescent marker Lucigenin (bis-N-methylacridinium nitrate) in a concentration of 12.5 mg/kg, diluted in isotonic saline (15). Lucigenin was injected intraperitoneally, and the bioluminescence scan acquired 10–13 min after injection using binning 4 and auto acquisition, with a maximum scan time of 5 min. MRI scans acquired 27 hours post-USPIO injection were performed on a 7T Bruker BioSpec Pharmascanner (Bruker, Ettlingen, Germany), and included a T1-weighted sequence for visualization. Details for T1-weighted scanning sequence: TE/TR 1500/8, image size 256 × 256, FOV 30 × 30 mm, averages 2, slice thickness 0.5 mm, scan time 2 min 18 sec.
Study procedures of the preclinical capsaicin mouse model.
Results
Out of 30 included female patients, a total of 28 patients underwent the full MRI protocol and were included in the final analysis (Figure 3).
Flowchart of study inclusion.
After cilostazol ingestion, all 28 patients developed migraine fulfilling ICHD-3 criteria of migraine without aura (2). Twelve patients treated their attack with sumatriptan, and the remaining 16 patients treated with promethazine and/or metoclopramide. Median headache intensity (interquartile range) was 7 (2.25) before sumatriptan and 3 (3.50) after sumatriptan.
Clinical characteristics of 28 migraine patients prior to post-USPIO scan.

n = 11, data missing for one patient.
n = 14, data missing for two patients.
Four healthy female controls received USPIO infusion and underwent the same MRI protocol as the migraine patients. Two of the controls received cilostazol, and two did not.
Preclinical capsaicin model
Capsaicin injection placed subcutaneously in the first division (V1) of the trigeminal nerve area of the mouse revealed contrast enhancement on the post-USPIO MRI assessed by visual inspection. Bioluminescence imaging using the macrophage marker Lucigenin confirmed a detectable presence of macrophages within the USPIO-enhancing inflammatory site (Figure 4).
T1-weighted images from 7T MRI scan (top row) and images from bioluminescence scan with the macrophage marker Lucigenin (bottom row). Three models are depicted; from the left: A) Capsaicin s.c. injection in V1 area, USPIO infusion, and MRI 27 hours post-USPIO, B) capsaicin s.c. injection in V1 area, no USPIO infusion and MRI 27 hours post-capsaicin, and C) untreated control with no capsaicin injection, USPIO infusion and MRI 27 hours post-USPIO. The bottom row depicts paired mice for each column, the bioluminescence signal is marked by the red circle and the signal intensity shown in flux (p/sec/cm2/sr).
Uptake of USPIO in intracranial arterial walls
We investigated the middle cerebral artery (MCA) and the cavernous part of the internal carotid artery (ICAcavernous). We tested for side-to-side differences in signal intensity at baseline and found no difference between the pain and non-pain sides (
Both in patients who treated with sumatriptan and patients who did not, we found comparable signal intensities on the pain and non-pain sides for MCA and ICAcavernous (
Ratio of USPIO signal intensity between pain and non-pain side in migraine patients, and right and left side in healthy controls.

Uptake of USPIO in dura mater and brain parenchyma
Visual assessment of the dura mater revealed no difference in enhancement on any side of the brain, neither in migraine patients nor controls.
R2* and R1 values at baseline and post-USPIO scan for pons, thalamus, and anterior cerebral, middle cerebral, and posterior cerebral artery vascular territories.

Comparing the pain side with the non-pain side, we found no difference in ΔR2* or ΔR1 for pons, thalamus, ACA, MCA, and PCA vascular territories ( Mean parenchymal R2* values in patients with unilateral attacks of migraine without aura, pain side (red line) compared to non-pain side (blue line), post-USPIO compared to baseline, with 95% confidence interval error bars. Data displayed for patients treated with sumatriptan (n = 12) and patients not treated with sumatriptan (n = 14). Ratio of ΔR2* between pain and non-pain sides of investigated ROIs in migraine patients who did not receive sumatriptan; pons 1.01, thalamus 1.00, ACA territory 1.04, MCA territory 0.98, PCA territory 0.98. Ratio of ΔR2* between pain and non-pain sides of investigated ROIs in migraine patients who did receive sumatriptan: Pons 0.90, thalamus 0.99, ACA territory 0.96, MCA territory 1.00, PCA territory 1.05.
In controls, we found that the ratio between the right and left sides in ΔR2* and ΔR1 was similar to the ratio between the pain and non-pain sides of the migraine patients (Figure 6 for ΔR2* data, R1 data not shown).
Mean parenchymal R2* values in healthy controls (n = 4), right side (green line) compared to left side (orange line), post-USPIO compared to baseline, with 95% confidence interval error bars. Ratio of ΔR2* between right and left sides of investigated ROIs: Pons 0.97, thalamus 0.90, ACA territory 0.93, MCA territory 0.94, PCA territory 1.01.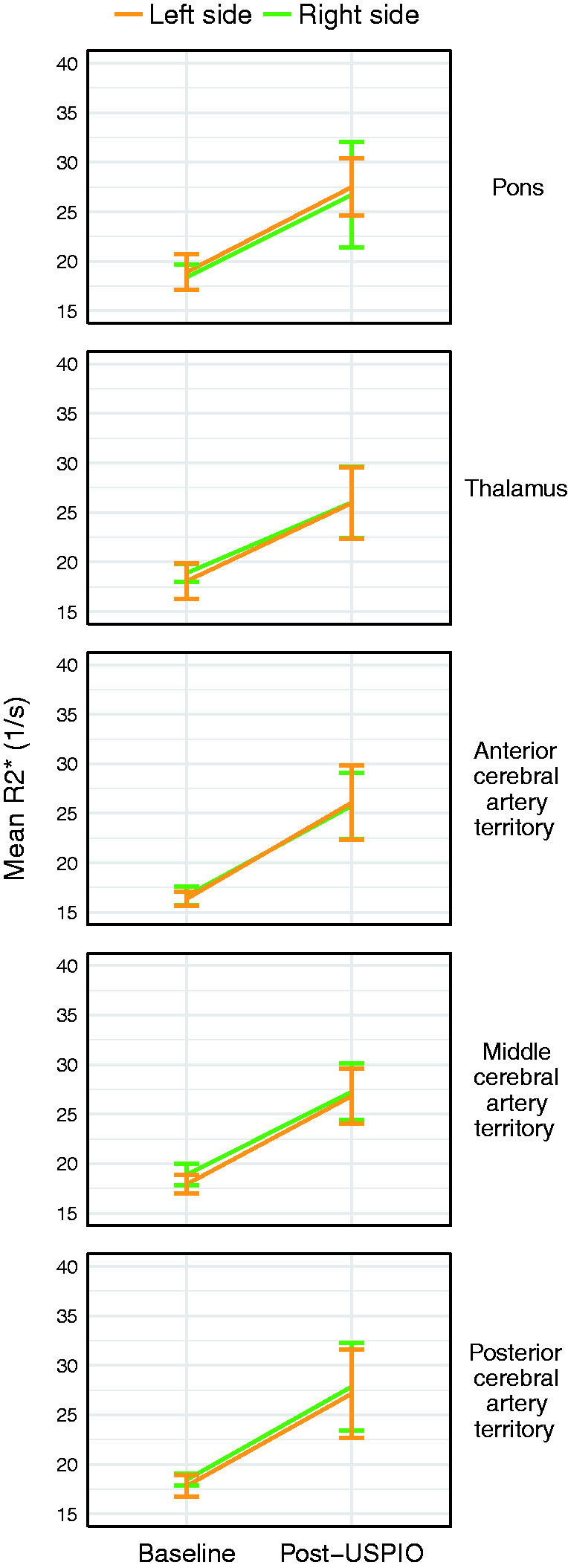
Bilateral uptake of USPIO in migraine without aura
As we found no difference in USPIO uptake comparing the pain and non-pain side in migraine patients, we performed a
Focusing on patients who did not receive sumatriptan, we also performed a
Discussion
In this study, we applied USPIO-enhanced MRI to investigate inflammation and macrophage infiltration as a biomarker of migraine. The validity of USPIO to detect inflammation and macrophage infiltration in the trigeminal area was confirmed by a preclinical mouse model, which demonstrated that capsaicin-induced inflammation is characterized by macrophage infiltration detectable with USPIO-MRI. In migraine patients, we found no signs of inflammation in the intracranial vessel walls, the dura mater, or the brain parenchyma when comparing the headache side with the non-headache side. We also detected similar USPIO uptake on either side of the brain in patients who treated their attacks with sumatriptan. These findings suggest that unilateral attacks of migraine without aura are
With the latest advent of monoclonal antibodies targeting calcitonin gene-related peptide (CGRP) for migraine preventive treatment, proposed to exert their effect in the peripheral nervous system, it is highly relevant to reconsider where and how trigeminal sensory fibers may be activated to produce the pain of migraine. Apart from the efficacy of anti-inflammatory treatment, inflammation in migraine pathogenesis is corroborated by multiple lines of evidence, including ictal release of the proinflammatory cytokines tumor necrosis factor alpha, interleukin-1 beta, and interleukin-6 (16,17) which may sensitize meningeal nociceptors (18–20). In addition, preclinical studies have shown that cortical spreading depression, the propagating wave of neuronal depolarization implicated in migraine aura, opens neuronal Panx1 channels linking neuronal stress to an inflammatory response mediated by interleukin-1 beta (21). Cortical spreading depression also causes astrocytosis and upregulation of proinflammatory markers indicating neuroinflammation (22). Evident sites to search for inflammatory processes include the dura mater, which is heavily innervated by pain fibers and densely populated with macrophages and mast cells (23,24), pons and thalamus as relay stations in the transmission of the nociceptive trigeminal signal, and broader vascular territories of the brain parenchyma as possible sites for diffuse inflammation. The pial MCA and extracerebral ICAcavernous are also key structures, as both arteries dilate during migraine, and ICAcavernous importantly also constricts after sumatriptan treatment (3). Considering that vasodilation likely plays a supporting rather than leading role in the activation of perivascular nociceptors, inflammation pertaining to the vessel wall could well be the overarching process that leads to migraine pain.
Here, we have conducted the first experimental human study to investigate macrophage-mediated inflammation as a possible biomarker of migraine. Our study is based on the tenet that proinflammatory activation of perivascular trigeminal fibers is mediated by an immune response involving macrophages, which are detectable with USPIO-enhanced MRI. We investigated three structural tiers of the migraine pain pathway (dura mater, intracranial arterial walls, and brain parenchyma), and found no preponderance of USPIO uptake on the pain side compared to the non-pain side, advocating one of three possibilities which will be discussed below; namely, that inflammation is not macrophage-mediated, inflammation is not specific to the pain side in migraine, or inflammation simply does not occur in migraine.
Macrophages are established constituents of the inflammatory response in numerous central nervous system disorders, but it is unclear whether they play a role in migraine pathophysiology. A recent preclinical study demonstrated that cortical spreading depression activates macrophages within both the pia and dura mater (25). In addition, macrophages have been detected in the trigeminal ganglia of a familial hemiplegic migraine type 1 mouse model under inflammatory conditions (26) and are proposed to activate meningeal nociceptors (27). We identified a generalized increase in USPIO uptake but no pain-side specificity of the signal, which may be attributed to USPIO-laden macrophages within the inflamed tissue, or from USPIO within the vascular compartment. While macrophages may be present during migraine, their involvement does not appear to simply relate to activation of perivascular nociceptors on the head pain side. The lack of pain-side specificity of USPIO uptake begs the question whether inflammation in migraine may in fact be a bilateral occurrence rather than a pain-specific phenomenon, particularly after more than 24 hours of headache. However, data from the control subjects also demonstrated a generalized, bilateral increase in USPIO uptake, adding to the notion that the implicated immune response is not specific to migraine pathophysiology.
In our post-hoc investigation of global signal alterations, we found greater global USPIO uptake in the ACA territory in patients who did not receive sumatriptan compared to those who did receive sumatriptan. As studies have demonstrated that ferumoxytol nanoparticles are not taken up by circulating monocytes (28) but rather by local tissue macrophages, it was suggested that USPIO may serve as a viable marker of blood-brain barrier (BBB) integrity, where enhancement represents an inflammatory reaction involving both BBB leakiness and presence of phagocytic cells (29). While our results can neither confirm nor refute an altered permeability of the BBB during migraine due to the detected global increase in USPIO, we can testify that any such disruption displayed no pain-side specificity. Importantly, regardless of whether USPIO nanoparticles are located intracellularly, interstitially or intravascularly, it is thought provoking that only patients who did
Besides central nervous system disorders involving inflammation, USPIO-enhanced MRI has been successfully applied to study conditions with vascular inflammation, including atherosclerotic plaques (31,32), abdominal aortic aneurysms (33,34) and cerebral aneurysms (35,36). This broad range of applicability indicates that USPIO-enhanced MRI may also serve as a highly valid approach to study perivascular inflammation in migraine. To corroborate our use of USPIO-enhanced MRI to detect macrophage-mediated inflammation in the innervation area of the trigeminal nerve, we conducted a preclinical capsaicin model, as subcutaneous injection of capsaicin activates transient receptor vanilloid 1 receptors on nociceptor terminals and leads to localized aseptic inflammation mediated by CGRP. This model is a validated preclinical assay used to test anti-migraine treatment, including anti-CGRP monoclonal antibodies (37). Our results from the preclinical model clearly demonstrated the presence of macrophages in the inflammatory focus detectable with USPIO-enhanced MRI, an interesting finding as capsaicin-induced inflammation is traditionally considered aseptic, without macrophage involvement.
The preclinical capsaicin model followed the MRI timepoints from the human study, with USPIO-enhanced MRI scans conducted 27 hours after ferumoxytol infusion. We elected this timepoint in accordance with previous studies that have applied USPIO to study CNS inflammation, as it is well established that USPIO acts as a blood pool agent immediately upon administration, and only after ≥ 24 hours is detectable after uptake by inflammatory cells (10,12,13). We extended the post-USPIO scan time to 27 hours after ferumoxytol infusion in order to increase our confidence in the detectable USPIO signal.
Our study has a number of limitations, a pivotal point being the lack of a sensitive method to quantify USPIO uptake in the dura mater. As a proxy, we used blinded visual evaluation by an experienced radiologist with preliminary negative findings, as he noted no abnormal signal alterations in the dura mater after USPIO, and also no side-to-side differences. While other pathological conditions that involve the dura mater may lead to macroscopic enhancement that can be directly visualized (38), we cannot exclude a microscopic enhancement that may have evaded our analysis. We are still in need of an optimized method to interrogate the dura mater and to assess cellular processes relating to this structure. We encountered technical challenges related to delineating vessel walls in the analysis software, which we addressed by applying a standardized thickness of the wall for comparison between patients and between scans. To our knowledge, this is the first study to measure USPIO uptake in intracranial vessel walls using this approach.
As we found no side-to-side difference in USPIO uptake, we must consider that our sample size may not have been large enough to glean any pain-side specificity between the treatment groups. Future investigations could also look into the accumulated headache burden over time to decipher whether a larger headache burden correlates with greater USPIO uptake, reflecting accumulated inflammatory effects. Importantly, we also cannot exclude that inflammatory processes related to migraine may be too subtle to detect with USPIO-enhanced MRI or may not involve macrophages to begin with. For future study, a larger cohort of healthy controls may allow for quantitative comparison of USPIO uptake with that of migraine patients to better grasp the pathological feat of a difference in signal.
In conclusion, unilateral attacks of migraine without aura are not associated with macrophage-mediated inflammation limited to the head pain side, as detected with USPIO-MRI. Future studies should broaden their focus to include non-macrophage mediated inflammatory mechanisms in order to delineate the pathophysiological footprint of migraine.
Clinical implications
We applied ultrasmall superparamagnetic nanoparticles of iron oxide (USPIO) as MRI-detectable macrophage biomarkers to investigate inflammation in patients with migraine without aura. Despite a proof-of-concept preclinical study validating the use of USPIO-MRI to detect macrophage-mediated inflammation, we found no signs of inflammation in the brain when comparing the headache side with the headache-free side in patients. Our findings offer new avenues in our understanding of migraine pathophysiology, suggesting that macrophage-mediated inflammation cannot serve as a biomarker for migraine headache.
Footnotes
Acknowledgements
The authors would like to thank AMAG Pharmaceuticals Inc for their kind donation of ferumoxytol, Assistant Professor Tobias Saam and Dr Rob Van Der Geest for technical support with MR sequences and analysis software, Dr Frauke Wolfram for review of MRI scans, the hematology laboratorium at Herlev Hospital for genetic HFE analyses, and Professor Michael A Moskowitz for his valuable discussion of this work.
Author contributions
SK, FMA and MA designed and initiated the study, and drafted the manuscript. SK, FMA, CC, SY, and ACRO acquired data and performed the clinical experiments. SK, FMA, CC, UL, DT, HSB, HDL, HL, and MA contributed to data processing, analysis, and interpretation. FPF and AK designed, conducted and interpreted the preclinical model. All authors provided input to the manuscript revision and approved the final manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SK has acted as an invited speaker for Novartis. FMA reports a travel grant from Allergan. FMA has acted as an invited speaker for Teva, Novartis, and Eli Lilly. FMA acts on the advisory boards of Novartis and Eli Lilly. MA reports personal fees from Allergan, Amgen, Alder, Eli Lilly, Novartis and Teva, and participated in clinical trials as the principal investigator for Alder ALD403-CLIN-011 (Phase 3b), Amgen 20120178 (Phase 2), 20120295 (Phase 2), 20130255 (OLE), 20120297 (Phase 3), GM-11 gamma-Core-R trials, Novartis CAMG334a2301 (Phase 3b), Amgen PAC1 20150308 (Phase 2a), Teva TV48125-CNS-30068 (Phase 3). MA has no ownership interest and does not own stocks of any pharmaceutical company. MA serves as associated editor of
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from Lundbeck Foundation (R155-2014-171) and Novo Nordisk Foundation (NNF11OC101433 and NNF15OC0017132).
*These authors contributed equally to this work.
