Abstract
Cortex and periaqueductal grey (PAG) play a major role in the pathophysiology of migraine. Some antiepileptic drugs (AEDs) influence the activity of these structures by modulating high-voltage-activated (HVA) Ca2+ channels and are effective in migraine prevention. The aim of the present study was to investigate the expression of total HVA Ca2+ channels in cortical and PAG neurons and to study the differential action of AEDs on these channels. Isolated neurons were visually identified based on morphological criteria. HVA currents were recorded by whole-cell patch-clamp technique. The distribution ratio of L-, N-, P-, Q- and R-type HVA Ca2+ channels was different between cortical and PAG neurons. In particular, we found that P- and Q-type HVA Ca2+ channels were more expressed in PAG neurons than in cortical cells, whereas L- and R-type HVA Ca2+ channels showed an opposite distribution. Interestingly, N-type HVA Ca2+ channels were equally distributed in these two neuronal populations. A differential sensitivity to AEDs of HVA Ca2+ channels located on cortical and PAG neurons was observed for topiramate (TPM), but not for lamotrigine (LTG) or levetiracetam (LEV). In fact, whereas both LTG and LEV were equally effective and potent in inhibiting HVA Ca2+ currents in the two neuronal populations, TPM showed a much higher potency and efficacy in blocking these currents in PAG neurons than in cortical pyramidal cells. TPM, in fact, inhibited N-, P- and L-type channels in PAG neurons, whereas in cortical neurons this AED modulated only P- and L-type channels. Unlike the other AEDs investigated, valproic acid did not affect HVA Ca2+ currents in cortical and PAG neurons. The negative modulation of specific subtypes of HVA Ca2+ channels by various AEDs can restore normal electrical activity in target brain areas such as cortex and PAG, providing interesting therapeutic approaches in migraine prevention.
Introduction
Migraine and epilepsy share several clinical features (1). Both conditions have a paroxysmal onset and they are often strictly lateralized. They may be associated with transient motor and/or sensory symptoms. Ictal and interictal EEG abnormalities, reflecting increased neuronal excitability, can be found in migraine as well as in epilepsy.
Some antiepileptic drugs (AEDs) are effective in the prevention of migraine (2–5). A rationale for this use is the hypothesis that migraine and epilepsy share several pathogenic mechanisms (4, 5). Unbalanced activity between excitatory glutamatergic transmission and GABAergic inhibition has been postulated in these two pathological conditions. Similarly, abnormal activation of voltage-operated ionic channels such as sodium (Na+) and calcium (Ca2+) channels has been implicated in both migraine and epilepsy (5–8).
Ca2+ channels also play a key role in the physiology of the periaqueductal grey (PAG). PAG is a major nodal point of modulation of craniovascular nociception and, through its role in the descending pain modulation system, contributes to migraine pathophysiology (9–11). Ca2+ channels in the PAG modulate trigeminal nociception, and dysfunctional activity of these channels influences migraine pathophysiology (12). In fact, the pathophysiology of migraine seems to involve also the activation of trigeminal afferents (10). Pain in migraine results from the activation of trigeminovascular afferents from the meninges, and substantial evidence indicates that the trigeminovascular system is activated by cortical spreading depression, which results from neocortical hyperexcitability. The mechanisms that underlie cortical hyperexcitability are unknown, but the phenomenon could be related to excessive excitatory transmitter release resulting from alterations in Ca2+ channel function, as occurs in some forms of migraine (3).
Altered activity of Ca2+ channels in both cortical and PAG neurons can represent a target of AEDs showing efficacy in migraine prevention (5). For this reason, in the present study we have first analysed the differential expression of various subtypes of high-voltage-activated (HVA) Ca2+ channels in neurons isolated from cortex and PAG. In the second phase of the study, we have compared the efficacy and the potency of four different AEDs in inhibiting the various subtypes of Ca2+ channels in cortical and PAG neurons. In particular, we have focused our attention on topiramate (TPM) and valproic acid (VPA), whose efficacy in migraine prevention has been widely confirmed in several clinical studies (5). Moreover, we also have extended our analysis to lamotrigine (LTG), since preliminary clinical findings have raised the possibility that this AED is selectively effective in the prevention of migraine with aura (13–15). Finally, we have included in the study levetiracetam (LEV), a new AED selectively targeting HVA Ca2+ channels (16) and whose efficacy in migraine prevention has been reported in open-label studies (17).
Methods
Slice preparation
Preparation of brain slices has been described previously (16, 18, 19), in accordance with the guidelines of the European Union Council (86/609/EU) and the Animal Act (1986). Wistar rats (4–6 weeks of age), were sacrificed under ether anaesthesia by cervical dislocation. Coronal slices (5–10 slices per rat, 180–200 µm thick) were prepared from tissue blocks with a vibratome in oxygenated Krebs' solution (composition below) maintained at 33 °C. Slices were allowed to recover for 30–60 min. The composition of the Krebs' solution was (in mM): 126 NaCl, 2.5 KCl, 1.3 MgCl2, 1.2 NaH2PO4, 2.4 CaCl2, 10 glucose and 18 NaHCO3. The pH of the extracellular solution was 7.4.
Preparation of acutely dissociated neurons
Slices from frontal cortex, and dorsolateral PAG were incubated in N-2-hydroxyethylpiperazine-N'-2-ethanesulphonic acid (HEPES)-buffered Hank's balanced salt solution (HBSS), bubbled with pure oxygen. Temperature was kept at 35°C. A single slice was then incubated in HBSS media with 0.5 mg/ml protease XIV added. After 30 min the tissue was repeatedly washed in HBSS and mechanically triturated with a graded series of fire-polished Pasteur pipettes. The obtained supernatant was placed in a Petri dish mounted on the stage of an inverted microscope (Nikon Diaphot, Japan). Healthy cells were allowed to settle for about 10 min. When examining dissociated neurons, only presumed pyramidal neurons, from cortex, and neurons from PAG were chosen for recordings.
Whole-cell patch-clamp recordings
Pyramidal neurons were identified by their peculiar shape and by the typical apical process spared by the enzymatic dissociation (Fig. 1). PAG neurons were identified (Fig. 1) using differential interference contrast optics on an upright microscope (Olympus, Tokyo, Japan), in accordance with previous works (20). In particular, we selected the grey matter near Silvius aqueduct, according to the stereotaxic coordinates (21).
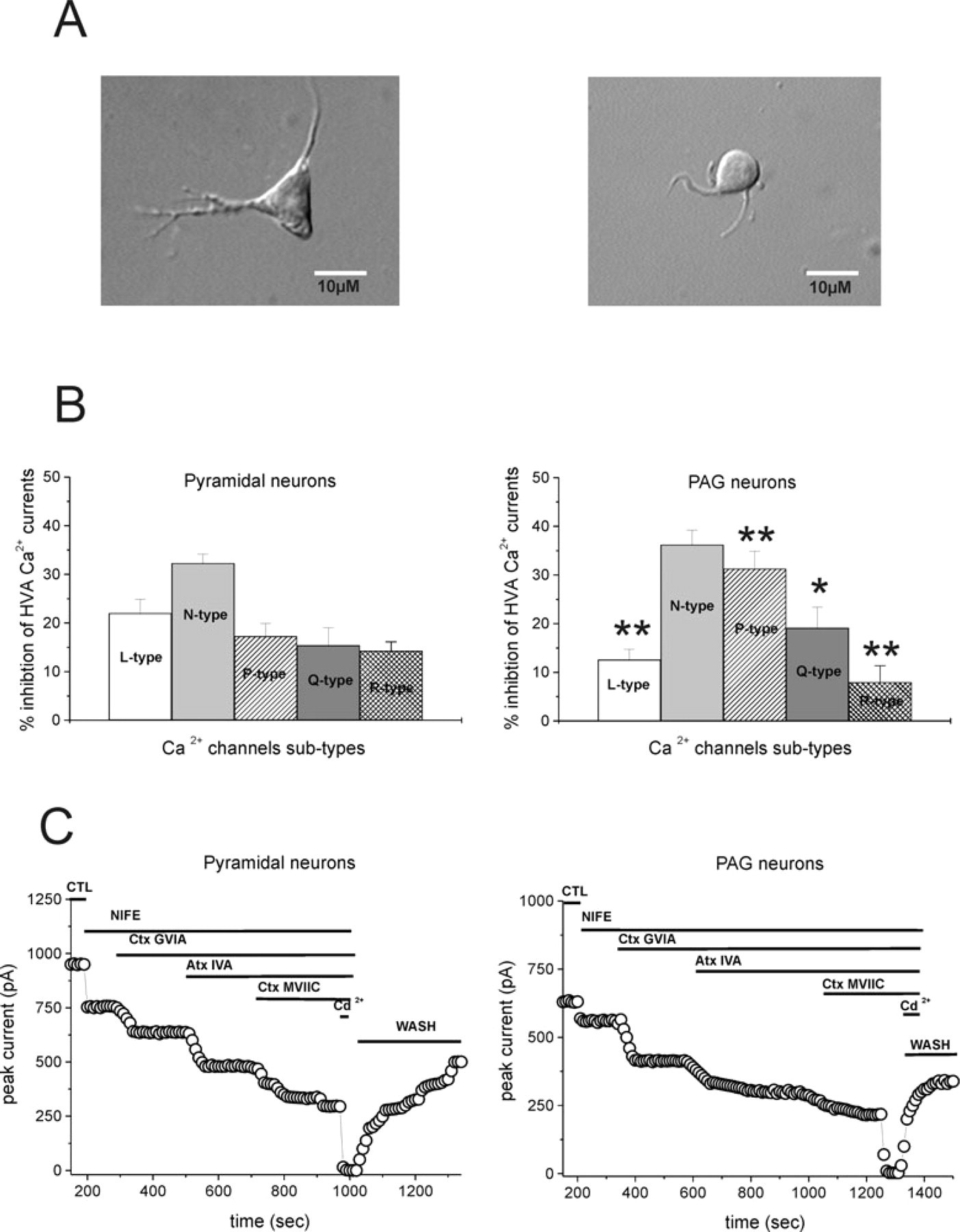
Distinctive morphological and electrophysiological features of two neuronal subtypes acutely dissociated from rats. (A) Representative images of a cortical neuron and a periaqueductal grey (PAG) neuron. Left side shows a picture of a typical cell classified as a pyramidal neuron. Note the triangular shape with two large dendrites emerging from the cell body. Right side shows a picture of a PAG neuron. The size of this multipolar neuron is significantly less than that of pyramidal cortical neurons. (B) The histograms show the representative distribution of Ca2+ channel subtypes in pyramidal neurons and PAG neurons. Asterisk represents significance (∗P < 0.05, ∗∗P < 0.01). The pharmacological dissection of high-voltage-activated Ca2+ currents in cortical and PAG neurons was obtained by utilizing specific channel blockers: nifedipine (NIFE) for L-type, ω-conotoxin (Ctx) GVIA for N-type, ω-agatoxin (Atx) IVA for P-type, ω-conotoxin (Ctx) MVIIC for Q-type. (C) Representative time course showing the effect of toxins sequentially added. Drugs were applied for the duration indicated by the respective bars. Note that 200 µ
After enzymatic dissociation two different cell types were obtained, with distinct sizes: ≤ 15 µm and > 20 µm in diameter.
Patch-clamp recordings in the whole-cell configuration were performed utilizing fire-polished pipettes (WPI PG52165-4) pulled on a Sutter Flaming-Brown micropipette puller (Sutter Instruments, Novato, CA, USA). The pipettes had diameters of 1–2 µ
The internal solution to record HVA Ca2+ currents consisted of (in m
Recordings were made with an Axopatch 1D (Axon Instruments, Union City, CA, USA) at room temperature. Series resistance compensation was routinely employed (70–80%). Data were low-pass filtered (corner frequency = 5 kHz). For data acquisition and analysis pClamp 9 running on a PC was used. Barium currents were studied with both voltage steps and ramps (0.3–0.6 mV/ms). In some experiments, leak currents were subtracted online with a p/4 leak subtraction protocol. Control and drug solutions were applied with a linear array of six gravity-fed capillaries positioned within 500–600 µm of the patched neuron. Dose–response curves were achieved by adding sequentially different concentrations of AEDs. Data analysis was performed offline using Microcal Origin and Graphpad Prism software running on a PC. Values given in the text and in the figures are mean ±
Drug source and handling
Nifedipine (NIFE), ω-conotoxin (Ctx) GVIA, ω-conotoxin (Ctx) MVIIC and ω-agatoxin (Atx) IVA were from Alomone Labs (Jerusalem, Israel). LTG was kindly provided by GlaxoSmithKline (Brentford, UK). VPA was purchased from Sigma (Milan, Italy), TPM was provided by Johnson & Johnson (Spring House, PA, USA) and LEV was obtained from UCB-Pharma (Brussels, Belgium).
Results
Distribution of HVA Ca2+ channels in cortex and PAG
Patch-clamp recordings were performed from acutely dissociated neurons from layer V of the frontal cortex, and from PAG, identified by means of optical microscopy (Fig. 1A). Barium-sensitive HVA Ca2+ currents were studied in isolation by omitting Na+ and K+ ions from intracellular and extracellular solutions (22). For this study, we operationally defined several HVA calcium current subtypes, based on the proposed nomenclature: L-type current was defined as that blocked by 5 µ
Bath-application of nifedipine (5 µ
Conversely, in the presence of Ctx GVIA (1 µ
Effects of VPA on HVA Ca2+ channels in cortex and PAG
Ca2+ currents were activated by voltage ramps (Figs 2 and 3) or step pulses (not shown). The holding potentials ranged from −70 mV to +40 mV for the ramps and from −10 mV to +10 mV for the tests. Under these conditions, barium currents were dominated by HVA components. VPA (0.01–1 mM) was tested on 37 and 34 PAG and cortical neurons, respectively. Bath-application of VPA, at all the concentrations tested, failed to affect HVA Ca2+ currents (Fig. 2A; P > 0.05).

Effect of valproic acid (VPA) and lamotrigine (LTG) on high-voltage-activated (HVA) Ca2+ currents. (A) Dose–response curve of VPA on HVA currents (0.001–1 m
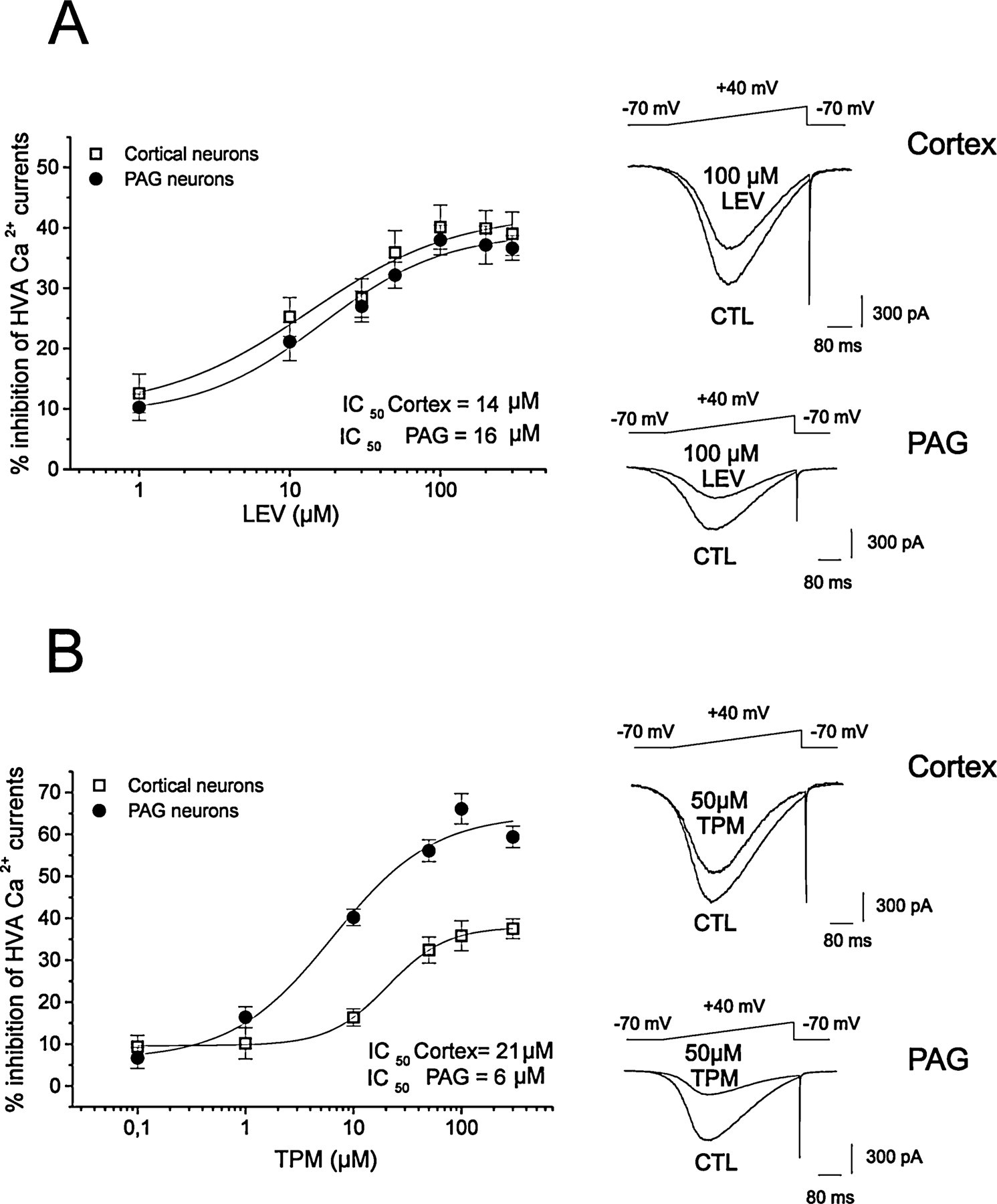
Inhibitory action of levetiracetam (LEV) and topiramate (TPM) on high-voltage-activated (HVA) Ca2+ currents. (A) LEV dose-dependently inhibited HVA currents both on cortical and periaqueductal grey (PAG) neurons. The IC50 was 14 µ
Effects of LTG on HVA Ca2+ channels in cortex and PAG
The modulatory effect of LTG was tested on 41 cortical pyramidal cells and 37 PAG neurons. LTG (0.1–200 µ

Characterization of the pharmacological modulation of high-voltage-activated (HVA) Ca2+ currents in cortical and periaqueductal grey (PAG) neurons by lamotrigine (LTG) and levetiracetam (LEV). (A) Ca2+currents were activated by step pulses ranging from −10 mV to +10 mV. The histograms shown on right and left side represent the mean ±
A significant interaction was found between LTG and the N-type channel blocker. In the presence of 1 µ
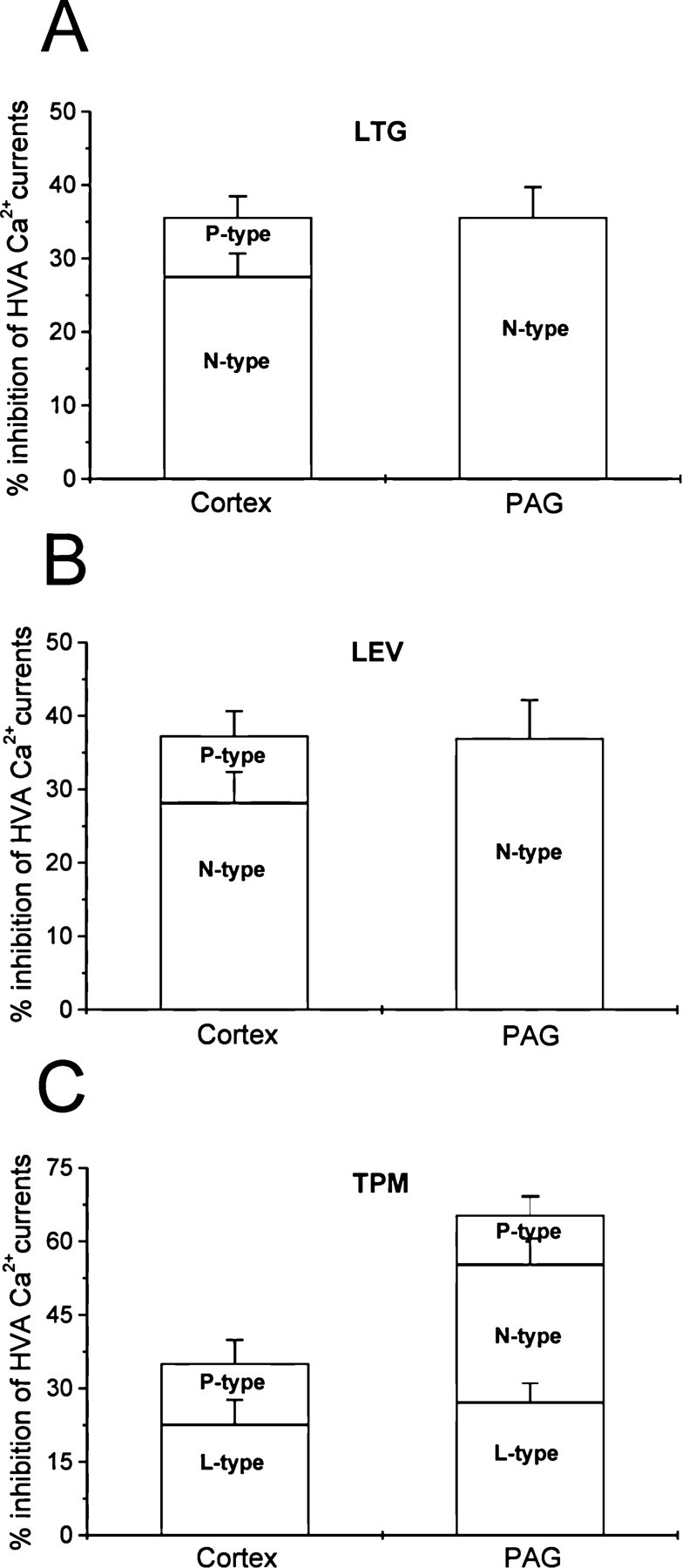
Summary of pharmacological modulation of high-voltage-activated (HVA) Ca2+ currents by antiepileptic drugs in cortical and periaqueductal grey neurons. The histograms summarize the distinct effects of lamotrigine (LTG) (A), levetiracetam (LEV) (B) and topiramate (TPM) (C) on the Ca2+ channel subtypes pharmacologically isolated using selective channel blockers.
In contrast, Ctx GVIA fully occluded the inhibitory action of LTG (100 µ
Effects of LEV on HVA Ca2+ channels in cortex and PAG
Levetiracetam (0.1–300 µ
In cortical neurons the effects of LEV were occluded by N-type and P-type selective blockers (Fig. 4C,D), whereas in PAG neurons LEV inhibition was saturated by Ctx GVIA 1 µ
Effects of TPM on HVA Ca2+ channels in cortex and PAG
The effect of TPM (0.5–200 µ
In another set of experiments we tested the channel subtype involved in the inhibitory effect caused by TPM. In cortical neurons application of both NIFE and Atx IVA totally occluded the effects of TPM (33.1 ± 3.3% and 33.4 ± 4.1%, respectively), suggesting that in cortical neurons the modulation of TPM was mediated by L- and P-type Ca2+ channels (n = 7 for each group) (Fig. 5A).
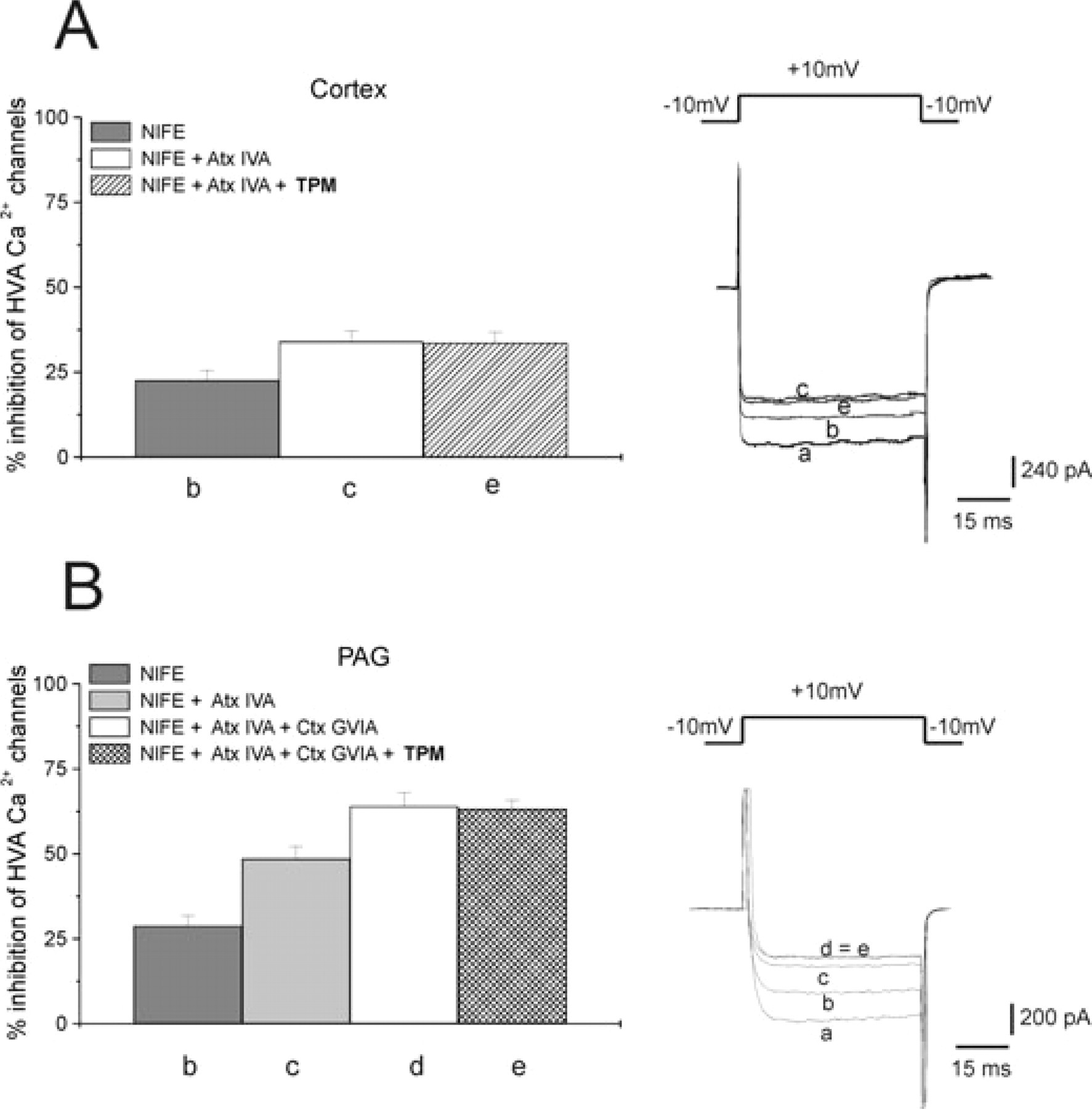
Characterization of the pharmacological modulation of high-voltage-activated (HVA) Ca2+ currents in cortical and periaqueductal grey (PAG) neurons by topiramate (TPM). (A) The selective L-type Ca2+ channel blocker nifedipine (NIFE, 5 µ
Conversely, in PAG neurons, a cocktail with NIFE, Ctx and Atx was necessary to obtain full occlusion of the TPM effect (n = 9, 65.8 ± 4.8% of inhibition) (Fig. 5B). These data suggest the involvement of N-, L- and P-type channels in the modulation of TPM on PAG neurons (Fig. 6).
Discussion
In the present study, the expression of total HVA Ca2+ channels and the specific action of AEDs on these channels have been evaluated in cortical pyramidal cells and compared with the effect on PAG neurons. The expression and pharmacological modulation of HVA Ca2+ channels in cortical and PAG neurons may differentially affect the pathophysiology of pain in migraine. Direct evidence of a prominent role for PAG in migraine has been provided by functional imaging studies of patients (11, 23–25). Stimulation or lesions in the PAG can produce migraine-like headache in non-migraineurs (26–28).
The PAG has been shown to modulate nociception in various experimental animal models of pain (29, 30). Moreover, it has been reported that P- and Q-type Ca2+ channels in the PAG facilitate trigeminal nociception (31). The existence of L-, N-, P-, Q- and R-type HVA Ca2+ channels in acutely dissociated PAG neurons has been previously reported (32).
We found that P- and Q-type HVA Ca2+ channels are more significantly expressed in PAG neurons than in cortical cells, whereas L- and R-type HVA Ca2+ channels show an opposite distribution. Interestingly, our findings suggest that N-type HVA Ca2+ channels are equally distributed in these two neuronal populations.
Although in the present study we made a major effort to dissect distinct Ca2+ channels properly using the best available blockers, it is possible that partial overlapping in the effects of some blockers could influence the obtained results (in particular those dealing with the pharmacological distinction between P and Q channels). We hope that in the future more selective drugs will allow us to achieve an even more accurate pharmacological dissection of Ca2+ channels.
The differential sensitivity to AEDs of HVA Ca2+ channels located on cortical and PAG neurons is one of the major findings of the present study. Whereas LTG and LEV were equally effective and potent in inhibiting HVA Ca2+ currents, TPM showed a much higher potency and efficacy in blocking these currents in PAG neuron than in cortical pyramidal cells. As suggested by the pharmacological analysis obtained using selective toxins for each HVA Ca2+ channel subtype, this difference was achieved because in PAG neurons TPM inhibited N-, P- and L-type channels, whereas in cortical neurons this AED modulated only P- and L-type channels.
It has been reported that the therapeutic action of TPM in migraine possibly involves various cellular mechanisms such as the reduction of glutamatergic transmission, the increase of GABAergic responses and inhibition of Na+ currents (5). However, our findings support the hypothesis that the inhibition of Ca2+ channels in the PAG represents a possible mechanism to explain the therapeutic action of this drug in migraine.
Abnormal activity of Ca2+ channels in the cortex can trigger the induction of spreading depression (SD) (33–35). SD represents a pathophysiological signal that has been associated with the induction of migraine. In particular, it can represent an electrical correlate of the neurological symptoms in migraine with aura (33–35). AEDs could selectively increase the threshold for the induction and reduce the progression of SD to counteract the frequency and intensity of migraine attacks. Accordingly, it has been recently reported that both TPM and VPA inhibit cortical SD (36, 37).
Our study has shown that VPA, unlike TPM, did not affect Ca2+ channels in cortical and PAG neurons. Thus, we must assume that alternative mechanisms are involved in VPA-induced inhibition of SD. VPA may suppress cortical excitability via actions on multiple sites, including ion channels and neurotransmitter receptors. Importantly, long-term treatment with VPA alters whole-brain expression of a variety of genes, including ion channels, presynaptic Ca2+-binding proteins and neurotransmitter receptors (37).
The similar inhibitory action of LTG and LEV on HVA currents in both PAG and cortical neurons seems to provide a cellular mechanism for their possible use in migraine prevention. However, clinically available data for this use are still scattered. In fact, although double-blind, randomized trials have failed to show a significant therapeutic effect of LTG in migraine prophylaxis (13), two small open clinical studies have found that LTG is effective in preventing migraine aura symptoms (14, 38). Accordingly, a controlled 3-year prospective open study has confirmed that LTG significantly reduced both frequency of migraine aura and aura duration (15). Similarly, an open study has suggested that LEV is a safe and effective treatment for migraine with aura (17). However, controlled trials are needed to confirm the observed results. If applied in a pathophysiological model of migraine, our electrophysiological data suggest the possibility that both LTG and LEV might selectively inhibit cortical SD via inhibition of Ca2+ channels and potentially normalize pathological neuronal activity in the PAG. Further pathophysiological studies are required to confirm these possible mechanisms.
Although the cortex and PAG are major sites in migraine pathophysiology, another major area of interest in migraine is the trigeminocervical complex. Interestingly, there is evidence that Ca2+ channel blockers, including AEDs such as topiramate, are able to inhibit trigeminovascular responses (39–42). Moreover, it should be stressed that PAG also has a critical role in inhibiting afferent trigeminal nociceptive traffic (43). Thus, we can assume that brainstem dysfunction might lead to disinhibition of trigeminal afferents and be important in the pain process of migraine. This consideration supports the possibility that AEDs prevent migraine by acting also on the trigeminocervical complex via either direct or indirect action.
Abnormal neuronal activity of Ca2+ channels can represent one of the major targets of AEDs in migraine (5). Accordingly, recent genetic studies on familial hemiplegic migraine (FHM) suggest that abnormalities in Ca2+ channels also play a critical role in migraine pathogenesis. In fact, a missense mutation of a gene, which encodes the α1A subunit of the P/Q-type Ca2+ channel, has been discovered in patients suffering from FHM (44). This finding raises the possibility that sporadic forms of migraine could also be considered channelopathies. Genetic migraine models will be useful in examining the critical mechanisms of migraine attacks and in identifying novel migraine prophylactic targets and therapies (45).
Thus, as suggested in the present study, the negative modulation of specific subtypes of HVA Ca2+ channels by various AEDs can restore normal electrical activity in target brain areas such as the cortex and PAG, providing interesting therapeutic approaches in migraine prevention.
Acknowledgements
We thank Massimo Tolu, Franco Lavaroni and Cristiano Spaccatini for their excellent technical support.
