Abstract
Evidence from animals and humans suggests that brainstem nuclei such as the raphe nuclei, the locus coeruleus (LC) and the periaqueductal grey matter (PAG), are involved in the pathophysiology of migraine. In order to understand possible neurotransmitters involved we have, by means of indirect immunocytochemistry, analysed these regions for the occurrence and distribution of calcitonin gene-related peptide (CGRP), substance P (SP), pituitary adenylate-cyclase activating peptide (PACAP) and vasoactive intestinal polypeptide (VIP). CGRP-immunoreactive (-ir) cell bodies, but no fibres, were found to occur in high numbers, constituting 80% of all nerve cell bodies in the LC. A smaller number of these nerve cell bodies (40%) in the LC proved to be PACAP-ir. The LC neurones also stored the vesicular monoamine transporter (VMAT)- and the C-terminal flanking peptide of neuropeptide Y (C-PON)-ir, illustrating their adrenergic nature. Double immunostaining revealed that all VMAT-and C-PON-containing neurones, in addition, stored CGRP. Immunoreactive cell bodies were not seen in the nucleus raphe magnus (NRM) or PAG. Numerous SP-ir nerve fibres were observed in the NRM, the LC and the PAG. Few PACAP-ir nerve fibres were detected in the PAG and few VIP-ir nerve fibres were seen in the NRM and the PAG.
Introduction
The primary cause of migraine attacks is still unknown. The clinical picture of migraine, headache, the accompanying signs (photo- and phonophobia, nausea, vomiting, segmental hyperalgesia and hyperaesthesia) and the headache alteration from one side to the other, suggests a role for brainstem nuclei in the process of migraine (1–3). Experiments have given evidence that certain brainstem nuclei, e.g. the nucleus raphe dorsalis (NRD), the nucleus raphe magnus (NRM), the locus coeruleus (LC), and the periaqueductal grey matter (PAG), are involved in the genesis of migraine headache (4–6). Electrical stimulation of the LC and the NRD in the cat and the monkey results in an altered microcirculation in ipsilateral cortical regions and in the extracranial circulation (4). Stimulation of electrodes implanted into the human PAG may induce headache attacks (7). Furthermore, studies with high-resolution positron emission tomography (PET) have revealed activation of NRD, LC and PAG during spontaneous migraine headache attacks (5–9). Following triptan treatment the pain and the cerebral blood flow alterations disappear but the brainstem activation persists (5). Controlled clinical trials have demonstrated that lipophilic triptans, which penetrate the blood–brain barrier and consequently may influence the brainstem, have less recurrence of migraine attacks (10, 11). These findings support the idea that the pathogenesis of migraine attacks is related to the activity of certain brainstem nuclei, sometimes referred to as a ‘migraine generator or centre’. During migraine attacks the levels of certain neuropeptides are elevated in peripheral blood (12, 13), as signs of the activity in the peripheral parts of the trigeminovascular system.
The aim of the present study was to examine neuronal messengers in the brain stem regions outlined above and to provide an understanding of which messengers may participate in the more central aspects of the migraine attack. We have applied immunocytochemical techniques to study calcitonin gene-related peptide (CGRP)-, substance P (SP)-, pituitary adenylate-cyclase activating peptide (PACAP)- and vasoactive intestinal polypeptide (VIP)-immunoreactivity (-ir) in the NRM, LC and PAG in man, in order to show the neuropeptide distribution in these regions.
Materials and methods
Materials
Brainstem areas were obtained at autopsy from adult subjects in accordance with the University Medical School guidelines for ethics in human tissue experiments. At autopsy, the relevant brainstem regions were removed from five subjects (two female, three male) with an average age of 63 years (range 43–74 years). None of the patients had suffered from any central nervous system (CNS) diseases. The cause of death in all cases was related to a cardiac disease. Tissue was collected within 24 h of death.
Immunocytochemistry
The brainstem nuclei were sectioned and every 10th section was used for counting cells. The total number of counted cells in a nucleus was considered 100%. The proportion of the labelled cells was expressed as a percentage of the total number. For immunocytochemistry, the brainstem samples were immersed overnight in fixative consisting of 2% paraformaldehyde and 0.2% picric acid in 0.1 mol/l phosphate buffer pH 7.2. After fixation, the specimens were rinsed repeatedly in sucrose-enriched (10%) Tyrode solution overnight. The tissues were frozen on dry ice and stored at −70°C. Sections were cut at 10 μm thickness in a cryostat and mounted on chrome–alum-coated slides.
For the immunocytochemical demonstration of CGRP, SP, PACAP, VIP, vesicular monoamine transporter (VMAT) and C-terminal flanking peptide of neuropeptide Y (C-PON) commercially available antibodies were used. Details of the primary antibodies used and their sources are given in Table 1 (14–17). Briefly, the sections were incubated with the primary antibody overnight at 4°C in a moist chamber. The site of the antigen–antibody complex was revealed by application of a secondary antibody (1:100, swine anti-rabbit IgG; Dako, Copenhagen, Denmark) conjugated to FITC for 1 h at room temperature. For double immunostaining peptide antisera raised in guinea pigs were used together with antisera raised in rabbits. The second antibodies were labelled with FITC and tetramethylrhodamine isothiocyanate (TRITC) (Dakopatts, Copenhagen, Denmark), respectively. Sections were first incubated with one of the pair of antibodies (VMAT or C-PON) and then with FITC-labelled second antibodies. The sections were then incubated with the CGRP antibody. The latter antibodies were visualized using TRITC-labelled second antibodies. The sections were then examined in a fluorescence microscope using filter settings for viewing FITC and TRITC fluorescence alternately. In the control experiments, the primary antibody was omitted. Control sections were exposed to antiserum that had been preabsorbed with an excess amount of the antigen (10–100 μg of a synthetic peptide per ml diluted antiserum). Additionally, the antiserum was tested for cross-reactions with other peptides (10–100 μg of peptide per ml diluted antiserum). No such cross-reaction was found. Cross-reactions with other peptides or proteins containing amino acid zsequences recognized by the different antisera cannot be excluded. It is therefore appropriate to refer to the product as ‘like-immunoreactive’ (-ir) material. For quantification, 10 complete sections from each brainstem region were evaluated. The proportions of positive cell bodies are detailed below.
Details of antibodies used

CGRP, Calcitonin gene-related peptide; SP, substance P; PACAP, pituitary adenylate-cyclase activating peptide; VIP, vasoactive intestinal polypeptide; VMAT, vesicular monoamine transporter; C-PON, C-terminal flanking peptide of neuropeptide Y.
Results
Nucleus raphe magnus (NRM)
A rich supply of SP-ir nerve fibres (Fig. 1) and few weakly stained VIP-ir nerve fibres were found in the NRM. CGRP-ir or PACAP-ir nerve fibres or SP-ir or VIP-ir cell bodies were not observed in the NRM.

A dense network of fine varicose substance P (SP)-containing fibres is seen in the bed of nucleus raphe magnus
Locus coeruleus (LC)
CGRP-ir neurones occurred in high numbers (80% of all neuronal cells) in each section of the LC region and were distributed homogeneously throughout the LC. A smaller number of neurones (40%) were PACAP-ir (Fig. 2). VIP-ir neurones were not detected in the LC. We found a rich supply of SP-ir nerve fibres in the LC. Autofluorescent lipofuscin granules, emitting orange-yellow fluorescence and dark ferro-pigments, were present within the perikarya as a characteristic of adult human LC tissue. The adrenergic nature of the LC cells was demonstrated by using antibodies towards the VMAT and the C-PON. The majority of the cell bodies in the LC were VMAT- and C-PON-ir. Double immunostaining revealed that all the VMAT-, and the C-PON-containing neurones, in addition, stored CGRP (Fig. 3).
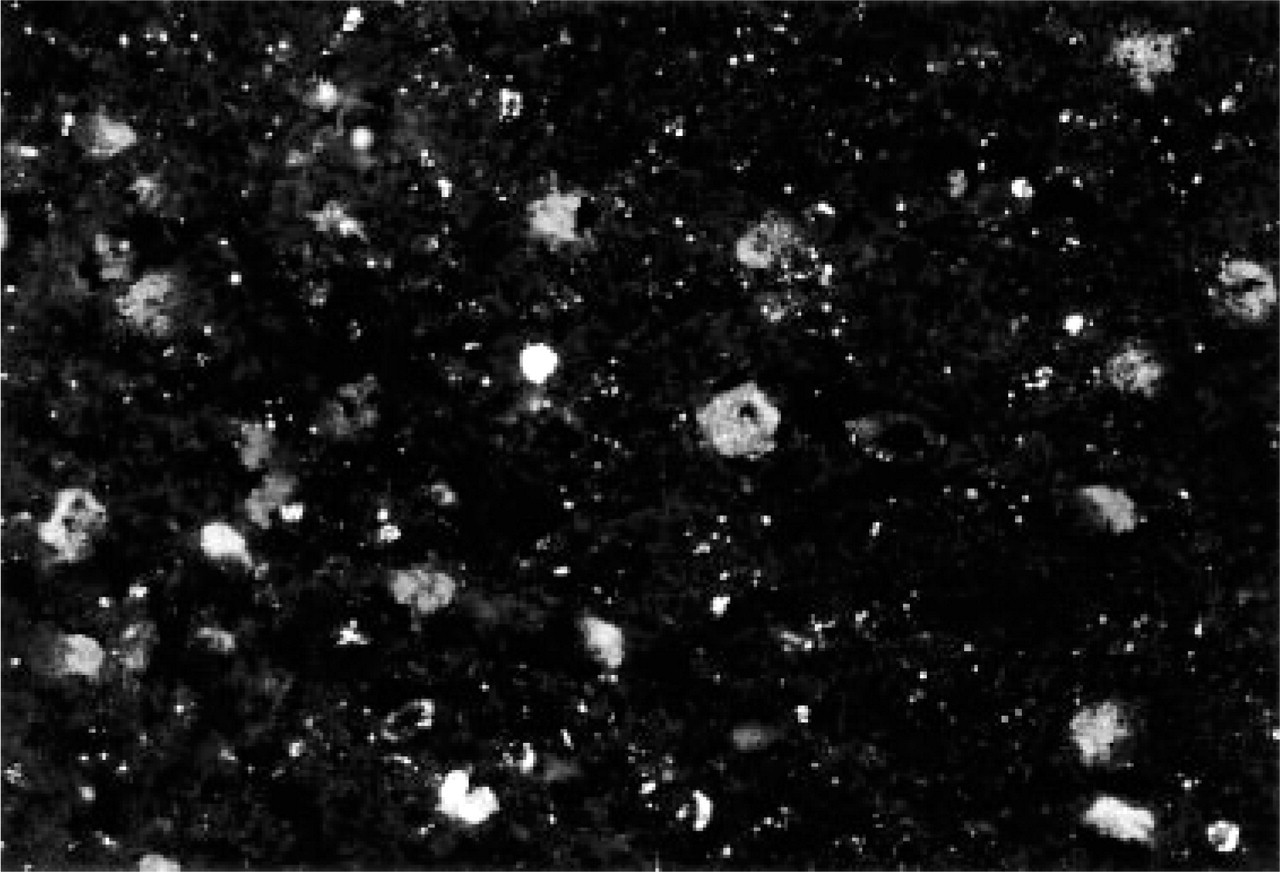
A moderate supply of pituitary adenylate-cyclase activating peptide-immunoreactive (PACAP-ir) nerve cell bodies can be seen in locus coeruleus

Section from locus coeruleus. Double immunostaining for vesicular monoamine transporter (VMAT) (A) and calcitonin gene-related peptide (CGRP) (B) and for C-terminal flanking peptide of neuropeptide Y (C-PON) (C) and CGRP (D) showing the presence of immunoreactive CGRP in a population of cells storing VMAT and C-PON, respectively.
Periaqueductal grey matter (PAG)
A rich network of SP-ir, few PACAP-ir and few VIP-ir nerve fibres were observed in the PAG. CGRP-ir nerve fibres or neuronal cells were not seen in the PAG (Fig. 4).

A rich supply of substance P-immunoreactive (SP-ir) fibres is localized in the periaqueductal grey (A), whereas only occasional fibres seem to contain pituitary adenylate-cyclase activating peptide (PACAP)-ir (arrow) (B).
The immunoreactivity staining with the different antibodies studied in the various regions is schematically represented in Table 2.
Distribution of neuropeptides in brain-stem regions nucleus raphe magnus (NRM), locus coeruleus (LC) and periaqueductal grey matter (PAG)

Arbitrary scale: –, no fibres; +, few; ++, moderate; +++, numerous.
Discussion
Electrophysiological and clinical data suggest that specific areas (NRD, LC and PAG) of human brainstem are involved in the genesis of migraine headache (1–4, 7). High-resolution PET studies have visualized an increased blood flow in these areas of the brainstem during the headache phase of a migraine attack (5). At present it is discussed if this represents the site of origin of the attack or if it is merely a reflection of centres activated during the primary headache, involved in the accompanying symptoms such as nausea and vomiting.
The LC is known as a major cardiovascular regulatory region with a dense adrenergic supply of fibres and neurones. This was confirmed by our findings of numerous large adrenergic (VMAT-) and neuropeptide Y (C-PON)-ir cell bodies. These cell bodies contain CGRP-ir (80%) and PACAP-ir (40%). Since the latter two peptides are believed to participate in primary headache (18) a modulatory role also on the cardiovascular role is possible in man. Our findings stand in contrast to a report in which a moderate supply of CGRP-ir nerve fibres but no cell bodies was seen in the LC (19). In addition, by radioimmunoassay, high levels of CGRP-ir have been observed in the human LC (20). This is of particular interest, because LC is central to the control of cerebral blood flow and of peripheral cardiovascular regulation and CGRP has profound haemodynamic effects.
To our knowledge there is no previous demonstration of PACAP-ir in the human LC. In agreement with Pammer et al. (21), tyrosine hydroxylase-, neuropeptide Y- and VIP-ir fibres were not seen in the LC. Anatomical studies have revealed a projection from the PAG to the NRM in the rostroventral medulla, both regions being considered as key areas in pain transmission (22). The NRM sends serotoninergic and the LC sends noradrenergic containing fibres to the trigeminal nucleus caudalis (TNC) (23–25). These anatomical findings indicate that the pain transmission in the trigeminovascular system, and especially in the TNC, which is a key region in primary headaches, can be influenced by projection from the brainstem adrenergic (LC) and serotoninergic (NRM, and PAG) centres (2, 18). In pain transmission, these fibres are believed to be gate controllers, by modulating the incoming pain signals and perhaps limiting the signal to the CNS (26).
PACAP dilates cerebral arteries (27, 28) and can increase the cerebral blood flow (29). Radioligand binding studies have demonstrated high specific PACAP 27 binding sites in the human brainstem (30) and radioimmunoassay has revealed a high concentration of PACAP in the PAG (31). In the present study, we observed that about 40% of the LC neurones expressed PACAP-ir, whereas only few PACAP-ir fibres were seen in the PAG. The existence of PACAP and CGRP in the noradrenergic neurones suggests that PACAP has an important role as a neuromodulator in the sensory and the autonomic nervous systems. Moreover, the LC and PAG may influence the trigeminovascular reflex via a neuromodulator effect.
Previous immunocytochemical studies (21, 32) and binding experiments (33) have demonstrated the occurrence of SP-ir and the localization of SP receptor sites in different human brainstem regions. In the present study, we observed a rich supply of SP-ir nerve fibres in the NRM, LC and PAG. The raphe system appears to be a rich source of SP-ir (32). SP interacts with the catecholaminergic and serotonergic systems, which suggests an influence on the cerebrovascular circulation.
SP is involved in plasma extravasation from the postcapillary venules in the dura mater during migraine pain (34, 35). However, neurokinin receptor blockers do not have any effect on migraine headache attacks (36). Further, there are indications that SP does not take part in vascular nociception in humans (37). However, recent studies have revealed a role for SP/neurokinins influencing mood control, since neurokinin-1 (NK-1) antagonists can be used as antidepressants (38). As such SP blockers may be involved as an adjuvant principle in migraine treatment.
Previous immunocytochemical (39) and in situ hybridization studies (40) have demonstrated VIP-ir in the PAG and NRD of cat and rats. Presently, we found only few VIP-ir nerve fibres in the human NRM and PAG. During some migraine attacks and in cluster headache and chronic paroxysmal hemicrania there is a release of VIP into the external jugular vein (18), but the VIP-ir is probably only from perivascular nerves and parasympathetic ganglia.
Footnotes
Acknowledgements
This study was supported by grant 05958 from the Swedish Medical Research Council.
