Abstract
Introduction
Preclinical data implicate large conductance calcium-activated potassium (BKCa) channels in the pathogenesis of headache and migraine, but the exact role of these channels is still unknown. Here, we investigated whether opening of BKCa channels would cause headache and vascular effects in healthy volunteers.
Methods
In a randomized, double-blind, placebo-controlled, cross-over study, 21 healthy volunteers aged 18–39 years were randomly allocated to receive an intravenous infusion of 0.05 mg/min BKCa channel opener MaxiPost and placebo on two different days. The primary endpoints were the difference in incidence of headache and the difference in area under the curve (AUC) for headache intensity scores (0–12 hours) and for middle cerebral artery blood flow velocity (VMCA) (0–2 hours) between MaxiPost and placebo. The secondary endpoints were the differences in area under the curve for superficial temporal artery and radial artery diameter (0–2 hours) between MaxiPost and placebo.
Results
Twenty participants completed the study. Eighteen participants (90%) developed headache after MaxiPost compared with six (30%) after placebo (p = 0.0005); the difference of incidence is 60% (95% confidence interval 36–84%). The area under the curve for headache intensity (AUC0–12 hours, p = 0.0003), for mean VMCA (AUC0–2 hours, p = 0.0001), for superficial temporal artery diameter (AUC0–2 hours, p = 0.003), and for radial artery diameter (AUC0–2 hours, p = 0.03) were significantly larger after MaxiPost compared to placebo.
Conclusion
MaxiPost caused headache and dilation in extra- and intracerebral arteries. Our findings suggest a possible role of BKCa channels in headache pathophysiology in humans. ClinicalTrials.gov, ID: NCT03887325.
Introduction
Potassium (K+) channels are the most diverse family of ion channels, encoded by more than 80 genes and widely expressed in the human body (1). Preclinical data suggest a role for K+ channels in headache and migraine (2,3). Furthermore, opening of ATP-sensitive potassium (KATP) channels causes headache in healthy volunteers (4) and migraine attacks in migraine patients (5), and dilates cephalic arteries (4,6). Large (big)-conductance calcium-activated potassium (BKCa) channels are expressed in vascular smooth muscle cells (VSMCs) in the extra- and intracerebral arteries, trigeminal ganglion (TG) and trigeminal nucleus caudalis (TNC) (7,8). Cyclic guanosine monophosphate (cGMP) and cyclic adenosine monophosphate (cAMP) signaling pathways stimulate BKCa channels (3,9). Interestingly, migraine triggers such as calcitonin gene-related peptide (CGRP) and pituitary adenylate cyclase-activating polypeptide (PACAP) also stimulate BKCa channels directly or indirectly via cGMP and cAMP (3,10–12). Furthermore, clinical use of BKCa channel openers resulted in headache as a common adverse event (13,14). The headache-inducing and vascular effect of BKCa channel openers has never been systematically studied.
Here, we investigated headache-eliciting and vascular effects of the BKCa channel opener MaxiPost in a balanced, double-blinded, randomized, cross-over study.
Materials and methods
We recruited 21 healthy volunteers through the Danish test subject website (www.forsøgsperson.dk). All participants gave written informed consent before inclusion. The female participants were required to have sufficient contraception (contraceptive pill or intrauterine device/system (IUD/IUS)). Exclusion criteria were previous serious somatic or psychiatric diseases, any type of primary headache or intake of daily medication except oral contraceptives.
The participants underwent a full medical examination including ECG and were informed that MaxiPost might induce headache in some individuals, but the timing or the characteristics of headache were not discussed.
The study was conducted according to the Helsinki II Declaration of 1964, as revised in 2013. The Ethics Committee of The Capital Region (H-18052101) and the Danish Data Protection Agency approved the study (ClinicalTrials.gov (NCT03887325)).
Experimental design
We randomly allocated all participants to receive an intravenous infusion of 0.05 mg/min (40 ml of 25 µg/ml) MaxiPost (BMS 204352) (TOCRIS, CAS number 187523-35-9) or 40 ml placebo (saline) over 20 min on 2 days separated by at least 1 week. The chosen dose was used in previous study investigating the pharmacokinetics of MaxiPost (13). The study drug was prepared and randomized by the Capital Region Central Pharmacy, and the investigators did not have access to the randomization code until the study was completed.
All participants arrived headache free (for 48 h before the start of the study) at the clinic between 8:00 am and 09:00 am After 30 min rest in the supine position, a venous catheter was inserted into the antecubital vein for drug infusion, and baseline measurements of mean arterial blood pressure (MAP) and heart rate (HR) were performed.
Headache characteristics, including localisation, intensity (numerical rating scale (NRS) from 0 to 10 (0, no headache; 1, a feeling of pressure; 10, worst imaginable headache)), and accompanying symptoms (nausea, photophobia and phonophobia), MAP, HR and adverse events were recorded before and then every 10 min until 120 min after the beginning of infusion. After finishing the measurements, the participants were discharged from the hospital and asked to complete a headache diary every hour until 12 h after start of infusion. If the symptoms fulfilled ICHD-3 beta (15) criteria C and D for migraine without aura, they were characterized as migraine-like attacks.
Facial flushing by a speckle contrast imager, middle cerebral artery blood flow velocity (VMCA), left superficial temporal artery (STA) diameter, left radial artery (RA) diameter and end-tidal partial pressure of CO2 (PetCO2) were recorded as described previously (5,6).
Data analysis and statistics
Headache scores are presented as median values, while other values are presented as mean values ± SD. We defined baseline as T0 before the start of infusion of each dose. To calculate the sample size at 5% significance with 80% power, we assumed that 65% of participants would report headache after MaxiPost and 20% after placebo and estimated that inclusion of at least 15 participants would be sufficient in a cross-over study (16). This estimation was reported in previous similar provocation studies with CGRP, PACAP, sildenafil and cilostazol (17–22).
Our primary endpoints were the difference in incidence of headache and the difference in area under the curve (AUC) for headache intensity scores (0–12 hours) and for VMCA (0–2 hours) between MaxiPost and placebo. The difference in AUC for STA, RA, HR, MAP and facial flushing by a speckle contrast imager between MaxiPost and placebo day was our secondary endpoint. Binary categorical data including the incidence of headache, accompanying symptoms and adverse events were analysed with McNemar’s test. We used the trapezium rule to calculate AUC to analyse the differences in response between MaxiPost and placebo (23). We used the Wilcoxon signed rank test to analyse headache intensity scores, paired two-way t-test to analyse other values, and the Mann-Whitney test and independent t-test to assess period and carry-over effects for all baseline variables. All analyses were performed with SPSS Statistics version 23 for Windows and a p-value < 0.05 was considered as the level of significance.
Data availability
The data supporting the findings in the present study are available from the corresponding author, upon reasonable request.
Results
Twenty healthy volunteers (12 women and eight men) completed the study (Figure 1). One participant (ID. 10) was excluded because of headache at baseline. The mean age was 24 years (range 18–39) and mean weight 69 kg (range 53–90). There was no carry-over or period effect for values of headache, HR, MAP, VMCA, STA or RA.

Flow chart of the study.
Headache
Eighteen participants (90%) developed headache after MaxiPost compared with six (30%) after placebo (p = 0.0005); the difference of incidence is 60% (95% confidence interval (CI) 36–84%) (Table 1). The median time to onset for headache was 40 min (range 10 min to 5 h) (Figure 2) after MaxiPost. One participant reported a migraine-like attack. The headache localization was mostly in the frontal and temporal regions (Figure 2).
Clinical characteristics of headache and associated symptoms in healthy participants after MaxiPost and placebo (0–12 hours observation period).
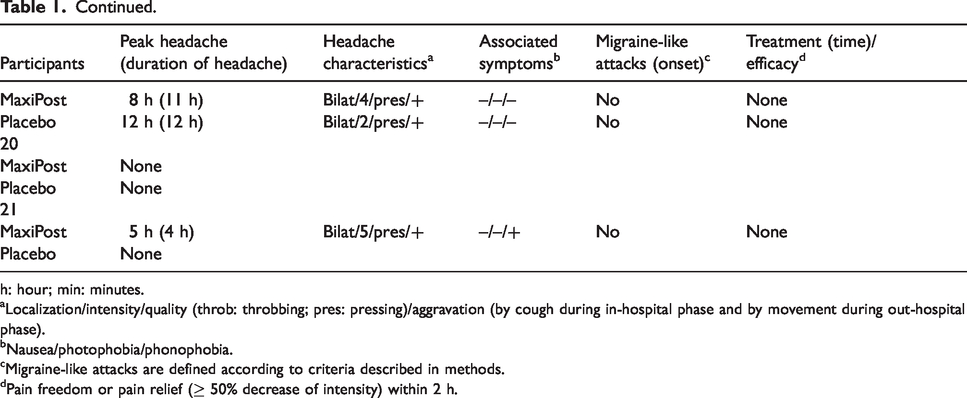
h: hour; min: minutes.
aLocalization/intensity/quality (throb: throbbing; pres: pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
bNausea/photophobia/phonophobia.
cMigraine-like attacks are defined according to criteria described in methods.
dPain freedom or pain relief (≥ 50% decrease of intensity) within 2 h.

Headache. (a) Median (thick, red line) and individual (thin black lines) headache intensity on a numerical rating scale (NRS) on MaxiPost and placebo days in 20 healthy volunteers. The AUC0–12 hour after MaxiPost infusion was significantly larger than after placebo (p = 0.0003). The median time to onset for headache was 40 min (range 10 min to 5 hours). (b) Localization (percentage) of headache after infusion of MaxiPost. The regions of the head are frontal (n = 14), vertex (the upper surface of the head) (n = 5), temporal (n = 14) and occipital (n = 3). Nine participants had headache in both the frontal and the temporal regions and two had diffuse headache.
The AUC0–12 hours for headache intensity was significantly larger after MaxiPost, 14.4 ± 3.1, compared to placebo, 1.8 ± 1.0 (p = 0.0003) (Figure 2).
Hemodynamic variables
There was a significant decrease in VMCA after MaxiPost compared to placebo (AUC0–2 hours was 93.2 ± 4.3 after MaxiPost and 111.0 ± 3.9 after placebo, p = 0.0001), a significant increase in STA diameter (AUC0–2 hours was 162.0 ± 8.9 after MaxiPost and 135.5 ± 6.2 after placebo, p = 0.003) and a significant increase in RA diameter (AUC0–2 hours was 297.7 ± 13.4 after MaxiPost and 271.8 ± 12.9 after placebo p = 0.03) after MaxiPost compared to placebo (Figure 3). Facial flushing measured by the laser speckle was increased (p = 0.005) after MaxiPost compared to placebo (Figure 5). We found a significant increase in HR but no change in MAP after MaxiPost compared to placebo during the in-hospital phase (HR AUC0–2 hours, p < 0.0001 and MAP AUC0–2 hours, p = 0.06) (Figure 4).
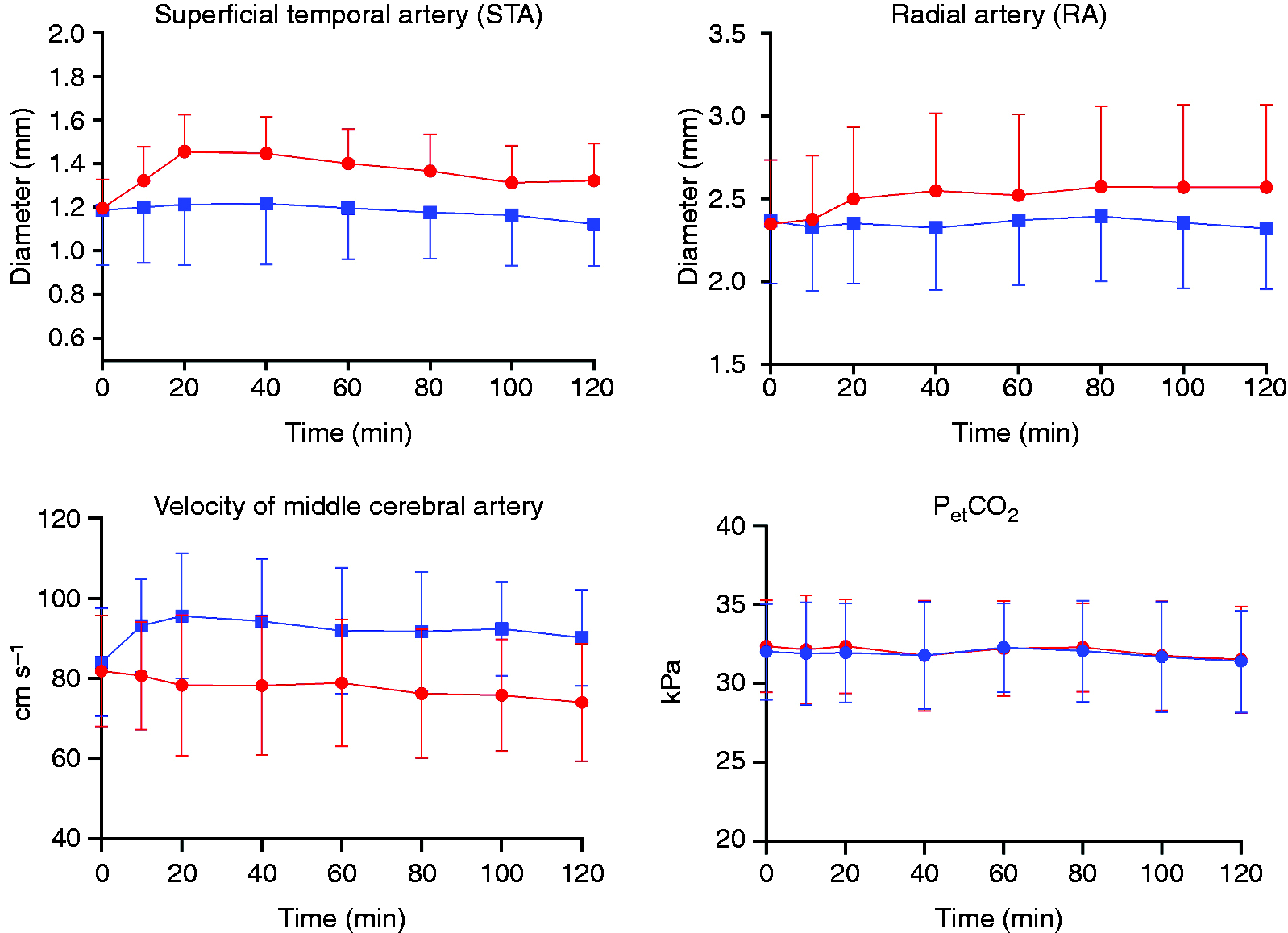
Haemodynamic effects of MaxiPost. Mean changes in middle cerebral artery blood flow velocity (VMCA), superficial temporal artery (STA) and radial artery (RA) diameter, and PetCO2 after MaxiPost and placebo infusion in 20 healthy volunteers. There was a significant change in mean VMCA (p = 0.0001), the STA (p = 0.003) and the RA diameter (p = 0.03) after MaxiPost compared to placebo. No change was observed in PetCO2. Red is MaxiPost and blue is placebo.

Vital parameters. Mean heart rate (HR) and mean blood pressure (MAP) after infusion of MaxiPost (red) and placebo (blue). MaxiPost increased HR (p < 0.0001) significantly, whereas MAP decreased without reaching significant level (p = 0.06) compared with placebo.

Facial skin. Blood flow changes were increased after MaxiPost (a) compared to placebo (b), (p = 0.005). Only data from one patient are shown.
Adverse events
Participants reported adverse events included flushing (n = 15 after MaxiPost and n = 4 after placebo) (p = 0.001), palpitations (n = 4 after MaxiPost and none after placebo) (p = 0.12), unusual tiredness (n = 3 after MaxiPost and one after placebo) (p = 0.5) and warm sensation (n = 4 after MaxiPost and one after placebo) (p = 0.25).
Discussion
The novel finding of the present study was that MaxiPost caused vasodilation and induced headache in healthy volunteers. These findings implicate BKCa channels in the pathogenesis of headache and set the stage for further investigations in participants with migraine.
Effect of MaxiPost on cranial arteries
BKCa channels are widely expressed in the body including smooth muscles (24), neurons (25), secretory endocrine cells (26) and sensory cells (27). The gating of BKCa channels is activated by phosphorylation, membrane depolarization, or increase of intracellular (IC) Ca2+ (28). Localized IC Ca2+ transients, termed Ca2+ sparks, activate BKCa channels in VSMCs (29). BKCa channel activation (opening) generates outward K+ currents, leading to hyperpolarization and thus vasodilation (8,24). Preclinical experiments demonstrate that the BKCa channel has a critical role in the regulation of the vascular tone of extra- and intracerebral arteries (11,30). In the present study, MaxiPost caused marked and prolonged dilation of the STA and RA. Also, the difference in mean VMCA with stable PetCO2 after MaxiPost compared to placebo indicated dilation of MCA. Collectively, our data shows that BKCa channel activation caused extra- and intracerebral artery dilation.
MaxiPost-induced headache
Preclinical studies on the role of BKCa channels in pain sensation have yielded conflicting results. Some studies reported that BKCa channel (dorsal root ganglion) activation modulated inflammatory pain (31), and reduced nerve injury-induced allodynia (32). However, other studies showed that BKCa channel (glial cells) activation resulted in enhancement of pain sensation (33–36).
Clinical trials on the pharmacokinetic effects of BKCa channel openers reported headache but no extracephalic pain as a frequent adverse event (13,14). Further details about the median time to onset for headache, type, localization, intensity, duration, and accompanying symptoms were not reported. In the present study, 90% of participants reported immediate headache that peaked four hours after infusion start. The induced headache was mild and lasted for several hours. Interestingly, cilostazol and levcromakalim – molecules targeting intracellular signaling pathways – led to similar headache intensity and duration as MaxiPost (4,37).
Several scenarios may explain MaxiPost-induced headache. Vasoactive intestinal peptide (VIP) increased the STA diameter by 45% (38) but only provoked very mild headache and in only a few participants during the immediate phase (0–30 min after infusion start) (39). In contrast, the STA diameter increased by 18% after cilostazol (40), 27% after levcromakalim (4), and 22% after MaxiPost administration but provoked more headache. Interestingly, VIP caused short-lasting dilation (30 min) (38,39), while administration of cilostazol, levcromakalim, or MaxiPost caused long-lasting (>2 h) dilation (4,40). A recent study showed that intravenous CGRP infusion caused long-lasting vasodilation (>2h) (41). Also, infusion of glyceryl trinitrate (GTN) led to prolonged decrease in blood flow velocity (>3 h) (42). It is unknown to what extent a long-lasting vasodilation may sensitize and activate trigeminal afferents and thereby cause headache. In animals, BKCa channels are expressed in TNC, and they modulate sensory processing in the trigeminovascular system (43). Several studies reported that BKCa channel openers hyperpolarized the resting membrane potential in neurons and reduced the frequency of spontaneous action potentials (32,43,44). Interestingly, hyperpolarization-activated cyclic nucleotide-gated cation (HCN) channels are involved in pain sensation (45). Furthermore, modulation of HCN channels in sensory neurons contributes in sensory processing and causes a spontaneous and persistent pain (46). Whether BKCa channel activation leads to change in the threshold potential of HCN channels in the trigeminal region is yet to be elucidated.
Conclusion
BKCa channel activation induced headache and caused long-lasting vasodilation in healthy volunteers. The present study implicates BKCa channels in cephalic pain and suggest a possible role of BKCa channels in migraine pathogenesis.
Key findings
Intravenous infusion of BKCa channel opener MaxiPost induced headache and caused long-lasting vasodilation in healthy volunteers.
Footnotes
Acknowledgements
The authors thank all participating subjects and lab technicians Lene Elkjær, Winnie Grønning for expert assistance. Further thanks to Novartis Pharma AG for their collaboration.
Contributions
MMK and HG initiated the study and contributed to study design; protocol development; participant enrolment; acquisition, data processing, analysis, statistics, and interpretation, as well as drafting and revision of the paper. CAWN contributed to participant enrolment, data acquisition and critical review of the paper. CS contributed to data acquisition and critical review of the paper. JMH, JS and CLL contributed to statistical analyses and critical review of the paper. MA initiated the study and contributed to study design, protocol development, statistics, data interpretation, and drafting and revision of the paper.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MMK has received travel grant from ElectroCore, LLC and acted as an invited speaker for Novartis. MA is a consultant, speaker or scientific advisor for Allergan, Amgen, Alder, ATI, Eli Lilly, Novartis, and Teva, primary investigator for Alder, Amgen, Allergan, Eli Lilly, Novartis and Teva trials. MA has no ownership interest and does not own stocks of any pharmaceutical company. MA serves as associate editor of Cephalalgia, associate editor of Headache, and co-editor of the Journal of Headache and Pain. MA is President of the International Headache Society. HG, CAWN, CS, JS, CLL and JMH declare no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is performed as part of a research collaboration with Novartis Pharma AG.
