Abstract
The study set out to investigate the role of corticotrophin-releasing factor (CRF) and orexin-A in chronic migraine (CM) and medication-overuse headache (MOH). Twenty-seven patients affected by CM and 30 with MOH were enrolled. Control CSF specimens were obtained from 20 age-matched subjects who underwent lumbar puncture for diagnostic purposes, and in all of them CSF and blood tests excluded central nervous system or systemic diseases. Orexin-A and CRF were determined by radioimmunoassay methods. Significantly higher levels of orexin-A and CRF were found in the CSF of MOH and to a lesser extent in patients with CM compared with control subjects (orexin-A: P < 0.001 and P < 0.02; CRF: P < 0.002 and P < 0.0003). A significant positive correlation was also found between CSF orexin-A values and those of CRF (R = 0.71; P < 0.0008), monthly drug intake group (R = 0.39; P < 0.03) and scores of a self-completion 10-item instrument to measure dependence upon a variety of substances, the Leeds Dependence Questionnaire (LDQ) in the MOH group (R = 0.68; P < 0.0003). The significantly higher orexin-A levels found in CM and MOH can be interpreted as a compensatory response to chronic head pain or, alternatively, as an expression of hypothalamic response to stress due to chronic pain. A potential role for orexin-A in driving drug seeking in MOH patients through activation of stress pathways in the brain can also be hypothesized.
Keywords
Introduction
Chronic migraine (CM) is one of the major clinical concerns for physicians dedicated to headache in clinical practice. Chronic headache forms evolving from a previous history of episodic migraine involve about 2% of the general population (1). About 60% of patients with chronic daily headache (CDH) (up to 80%) attending specialized Headache Centres complain of CM or transformed migraine according to the Silberstein–Lipton definition (2) and most of them (up to 80%) overuse symptomatic medications (3, 4). Acute migraine medication overuse has been identified as one of the major risk factors for progression of migraine towards chronicity (5).
The neurobiology underlying CM and medication-overuse headache (MOH) is not completely understood. A progressive dysfunction of central pain systems is hypothesized in these two conditions on a predisposed genetic substrate, but few studies have been carried out to clarify this issue (6, 7).
The two neuropeptides orexin-A and -B, also referred to as hypocretin-1 and -2, synthesized by a few thousand neurons in the lateral and dorsal hypothalamus projecting widely throughout the brain, are considered critical components in several functions, including maintaining and regulating arousal, sleep, appetite and drinking, central control of autonomic activity and certain endocrine responses and pain modulation (8–19). They act on two related G-protein coupled receptors, the type-1 and type-2 orexin/hypocretin receptors, which are widely diffused in the brain and spinal cord (20, 21). Among the innervated regions are the periaqueductal grey, locus coeruleus and rostral ventral medulla, which are known to be heavily involved in descending pain inhibitory control and express orexin-1 (OX1) and 2 (OX2) receptors (22–26). Moreover, orexin fibres show robust innervation of the mesencephalic trigeminal nucleus and spinal cord, with high densities in lamina I of the dorsal horn, which is specifically involved in pain transmission (27–30). Several experimental data suggest that orexin-A induces analgesia in acute and inflammatory pain animal models (31–36). The inhibition of pain transmission in case of persistent pain and associated stress is also supported by the finding of a greater degree of hyperalgesia induced by peripheral inflammation and less stress-induced analgesia in prepro-orexin (precursor of orexin-A and -B) knockout mice than wild-type mice (37).
Orexin-A and -B receptor activation has also been shown to be involved in the differential modulation of nociceptive dural input. A recent study has demonstrated that orexin-A, but not orexin-B, is able to inhibit neurogenic dural vasodilation via activation of the OX1 receptor, resulting in inhibition of prejunctional release of calcitonin-gene related peptide from trigeminal neurons (38). Moreover, injection of orexin-A in the posterior hypothalamic area decreased, whereas orexin-B increased the A- and C-fibre responses to dural electrical stimulation as well as spontaneous activity and responses to noxious thermal stimulation of the facial skin (39).
Based on the above findings and on the well-known involvement of both peptides in hypothalamic regulation of neuroendocrine and autonomic function, it has been hypothesized that orexinergic mechanisms may provide a link for endocrine and autonomical changes, as well as nociceptive phenomena, seen in primary headache disorders (19). Furthermore, recent experimental evidence suggests that corticotrophin-releasing factor (CRF) activates the hypocretin system, relaying orexin-A to brainstem nuclei such as the locus coeruleus, nucleus accumbens, as well as ventral tegmental area (VTA) and extended amygdala, which are critical structures involved in the negative motivational state that drives response to stress, addiction and reward (40). A pivotal role for CRF/orexin-A could therefore be hypothesized in MOH.
Taking into account the implication of orexin-A in nociceptive processing of meningeal input and response to drug abuse, the present study aimed to investigate its levels and those of CRF in the cerebrospinal fluid (CSF) of CM and MOH patients, diagnosed according to International Classification of Headache Disorders (ICHD)-II and ICHD-IIR criteria (41–43), and to verify the relationship between the above-mentioned levels and frequency, duration and intensity of chronic headache, monthly drug intake (MOH) and Leeds Dependence Questionnaire (LDQ) scores, which provide a reliable measure of drug dependency (44).
Methods
Patients
The study protocol was approved by the Ethics Committee of the Azienda of Umbria and all the patients gave their written consent to the study. Twenty-seven consecutive patients (18 women, nine men; mean age ±
Assays
CSF levels of orexin-A and CRF were determined by radioimmunoassay (RIA) methods (Peninsula Laboratories, San Carlos, CA, USA) following an extraction procedure using Sep-Pak C18 cartridges (Waters Associates, Milford, MA, USA). Orexin-A and CRF RIA kits enable the reliable measurement of orexin-A and CRF levels as low as 10 pg/ml. Intra- and interassay coefficients of variation for Orexin-A RIA kit were 4.8% (10 measurements) and 5.8% (10 measurements), respectively. The intra-assay coefficient of variation for the CRF RIA was 7.2%, and the interassay coefficient of variation was 11.9%. No cross-reactivity was observed between orexin-A and human CRF, β-endorphin, vasopressin, met-enkephalin, leu-enkephalin, substance-P, neuropeptide Y or angiotensin II, or between CRF and human somatostatin, thyrotropin-releasing hormone, thyroid-stimulating hormone, follicle-stimulating hormone, luteinizing hormone, adrenocorticotropic hormone, α-melanocyte-stimulating hormone, β-lipotropic hormone, substance P, leu-enkephalin, met-enkephalin or β-endorphin.
Clinical variables which were related to CSF levels of orexin-A and CRF were: number of days with headache per month; intensity of headache measured with visual analogue scale (VAS); duration of chronic headache (years); monthly drug intake; and LDQ scores (44).
Statistical analysis
This included
Results
Details of patient and control groups are reported in Table 1. Drugs abused by patients in the MOH group are shown in Table 2.
Details of patients and control subjects
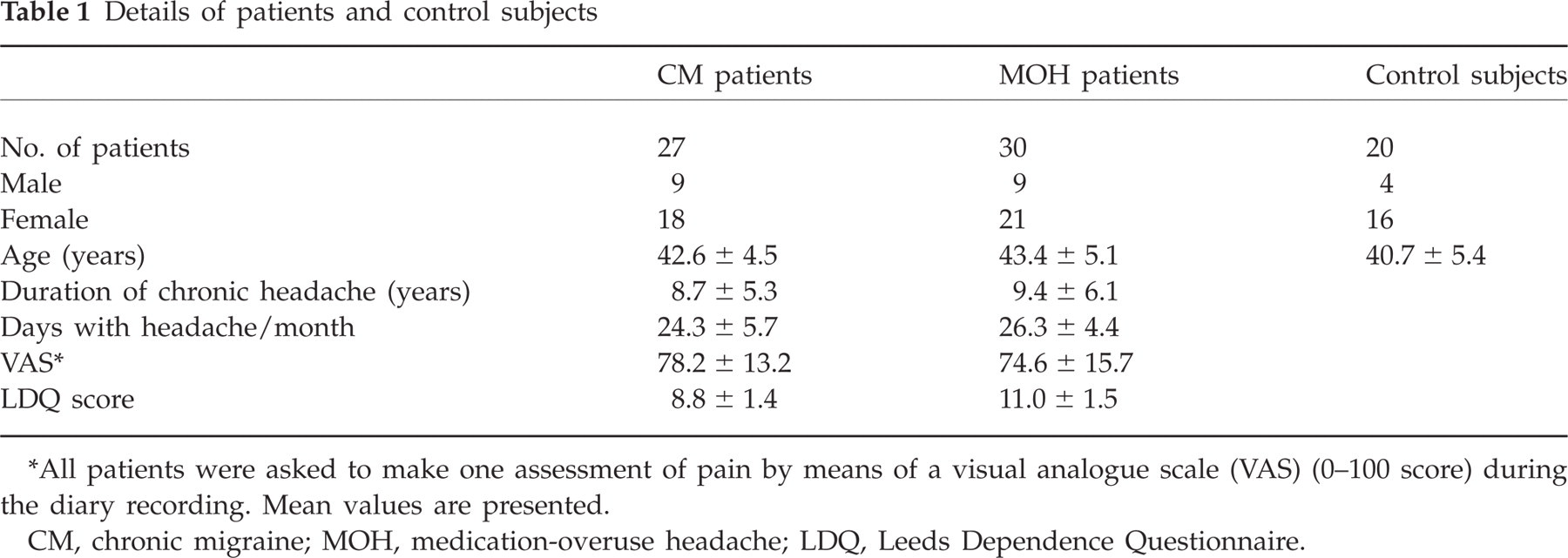
All patients were asked to make one assessment of pain by means of a visual analogue scale (VAS) (0–100 score) during the diary recording. Mean values are presented.
CM, chronic migraine; MOH, medication-overuse headache; LDQ, Leeds Dependence Questionnaire.
Drugs abused by patients of MOH group

The sum of percentages exceeded 100% because more than half the patients abused two or more symptomatic drugs.
CSF examination was within the normal limits and no cells were found in either patients or controls.
CM patients (with and without medication overuse) considered as a whole group had significantly increased CRF and orexin CSF levels over controls (CRF 22.42 ± 2.39 vs. 17.40 ± 2.58, P < 0.0001; orexin 304.17 ± 14.32 vs. 272.34 ± 11.24, P < 0.0001).
Moreover, both CRF and orexin values in the CSF of MOH patients were significantly higher than those of CM (P < 0.02 and P < 0.004, respectively) (Table 3).
Descriptive statistics [mean, standard deviation (

Statistical significance: Bonferroni test.
Orexin-A: CM vs. control subjects P < 0.0002; MOH vs. control subjects P < 0.000000; CM vs. MOH P < 0.004.
CRF: CM vs. control subjects P < 0.0003; MOH vs. control subjects P < 0.000000; CM vs. MOH P < 0.02.
CRF, corticotrophin-releasing factor; CM, chronic migraine; MOH, medication-overuse headache.
No significant differences were found between CRF and orexin values between men and women in either patient or control groups, although a trend toward slightly higher values was observed for women.
In MOH patients a strong positive significant correlation between the CSF levels of orexin-A and those of CRF (R = 0.71, P < 0.0008) was found (Fig. 1).
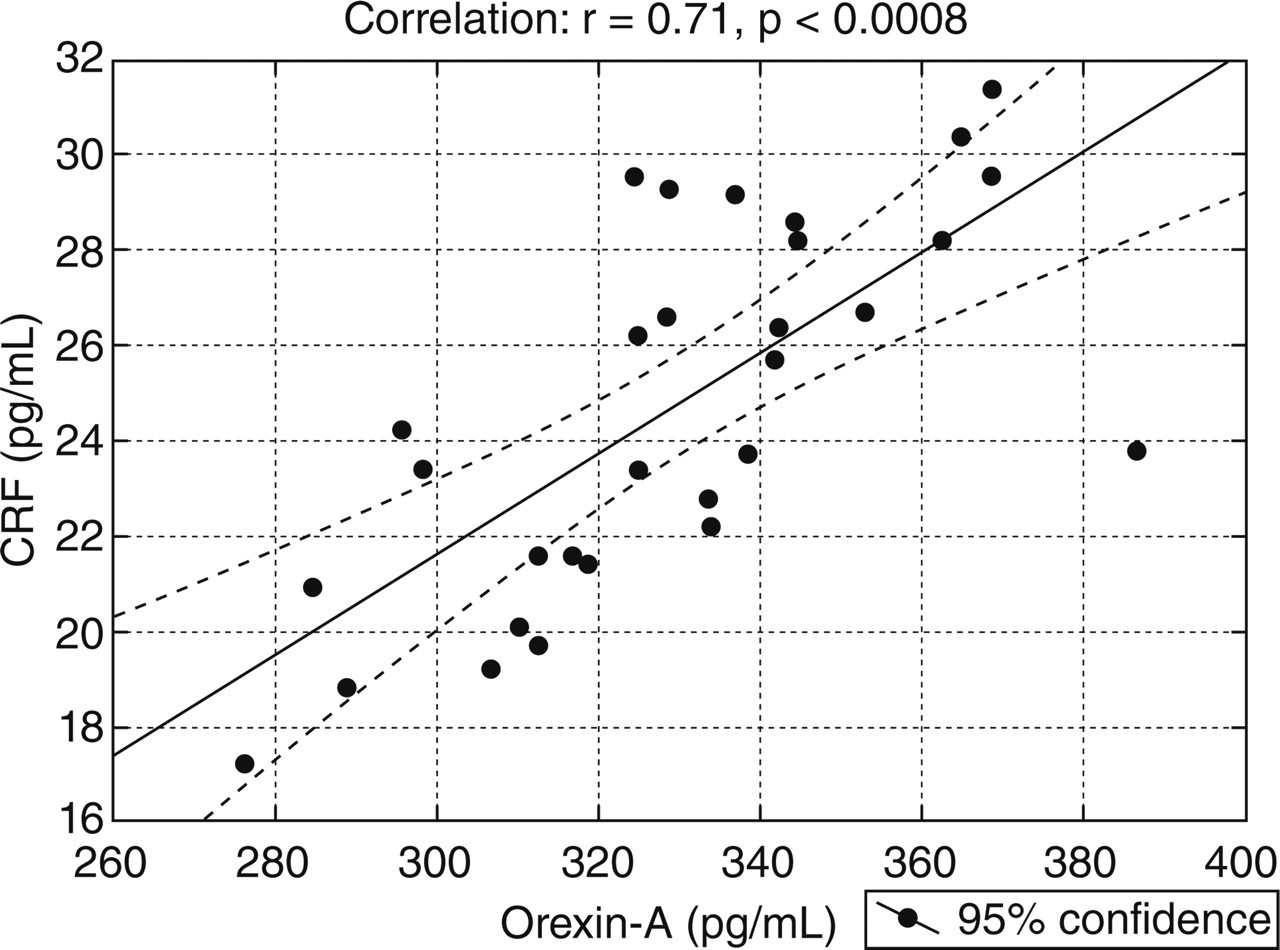
Plot of cerebrospinal fluid (CSF) levels of orexin-A and corticotrophin-releasing factor (CRF) levels, both expressed as pg/ml, in the CSF of medication-overuse headache (MOH) patients.
A significant positive correlation also emerged between CSF orexin-A and monthly drug intake (Fig. 2) and scores of LDQ in the MOH group (Fig. 3). In the same group, CRF also showed a positive correlation with both number of drugs taken per month and LDQ score (R = 0.43; P < 0.01 and R = 0.51; P < 0.002). This may have been the consequence of the already mentioned significant correlation with CSF orexin-A levels.

Plot of cerebrospinal fluid (CSF) levels of orexin-A (pg/ml) and monthly intake of symptomatic drugs in medication-overuse headache (MOH) patients.

Plot of cerebrospinal fluid (CSF) levels of orexin-A (pg/ml) and total Leeds Dependence Questionnaire (LDQ) scores in medication-overuse headache (MOH) patients.
A trend toward a correlation between orexin-A and CRF levels in the CSF was observed in CM patients without symptomatic drug abuse, but this did not reach the level of statistical significance (P < 0.31; P < 0.06).
In neither the CM nor MOH groups was there any correlation between CSF orexin-A and CRF levels and age, number of days with headache per month, headache intensity, headache index or duration of headache.
Discussion
The higher CRF and orexin-A levels found in the CSF of our CM and MOH patients support the involvement of the hypothalamus in both chronic headache disorders. Increased CRF levels in CSF, in particular, concur with previous findings of an abnormal pattern of hypothalamic hormonal secretion in CM, including increased cortisol concentrations, a decreased nocturnal prolactin peak, and a delayed nocturnal melatonin peak in these patients (45). Neuroendocrine hypothalamic derangement has also been emphasized by Patacchioli et al. (46), who demonstrated increased morning and evening levels of cortisol and also cortisol/dehydroepiandrosterone-sulphate ratio, an inverse marker of psycho-physical well-being, in CM patients, with respect to controls. Conversely, in the same patients, testosterone/cortisol ratios, an anabolic/catabolic index of physical performance, were significantly lower.
Based on the above previous findings, we therefore hypothesized that the significantly higher CRF concentrations detected in the CSF of both CM without medication overuse and, to a greater extent, in MOH, compared with those of the control group, may be interpreted best as an expression of hypothalamic response to stress, due to chronic pain.
Neurophysiological research aimed at mapping the route of their ascending axonal projection from trigeminal territories to the thalamus has demonstrated that highly prioritized nociceptive trigeminal signals reach the hypothalamus through both the direct trigeminohypothalamic tract and indirect reticulohypothalamic tract routes, ensuring that this critical information could be processed by the hypothalamus, which is responsible for several neuroendocrine and autonomic responses, including CRF and orexin secretion (47). Descending projections from the hypothalamus to brainstem nuclei, and trigeminal and trigeminal dorsal horns have been implicated in the descending modulation of nociceptive processing (19).
Recent experimental data in this regard indicate that the CRF peptidergic system directly innervates hypocretin-expressing neurons and depolarizes hypocretin neurons, an effect which is blocked by a CRF-R1 antagonist. Furthermore, activation of hypocretinergic neurons induced by high arousal states, including stress, is impaired in CRF-R1 knockout mice (40, 48, 49). These data therefore suggest that the CRF-R1 receptor mediates stress-induced activation of the hypocretinergic system.
There are also experimental findings suggesting that orexins per se in the CNS may be involved in the activation of central CRF neurons induced by stress (50).
The increased levels of orexin-A detected in the CSF of both CM and, particularly, MOH patients may therefore also be involved in stress responses to chronic pain even if a compensatory mechanism of this neuropeptide to counteract chronic head pain cannot be excluded, based on the observations that it inhibits neurogenic inflammation and exerts an analgesic effect through OX1R receptor activation both at the trigeminal and spinal level (51).
However, the lack of a relation between CRF/orexin-A levels and headache burden in our CM and MOH patients suggests that this stress response to chronic pain, since it is activated, is then independent of headache frequency, intensity and chronic headache duration.
On the basis of experimental findings of a relationship between CRF production and hypothalamic orexin secretion, we also verified the dependency of orexin levels on CRF release. We found a significant positive correlation only for the MOH patient group, whereas only a trend toward a correlation in CM without medication overuse was observed, which did not reach the level of statistical significance.
We hypothesized that in MOH patients greater activation of the CRF-mediated orexin-A system could be involved not only in the stress response to pain but also in the negative motivational state that drives drug dependence, as already shown in experimental models suggesting the pivotal role for the orexin/hypocretin neuronal system in reward-based learning and memory processing and addiction (52, 53). This is supported by the relation between the number of drugs abused per month and the scores of a self-completed 10-item instrument to measure dependence upon a variety of substances, the LDQ. A recent study using this questionnaire has suggested that CDH patients have a stronger need for analgesics, with total scores similar to those calculated for drug addicts. The pattern of answers to items were, nevertheless, different. Addicts needed a constant effect of the drug much more than CDH patients in order to feel well and to avoid abstinence symptoms, and had higher scores in items assessing compulsive use. Patients with CDH had higher scores for the item exploring the primary importance of the effect over the type of analgesic used, suggesting that the dependence on acute medication in these patients is rather a consequence of headache, originating from the necessity for analgesics to cope with everyday life (44). The association between CSF levels of CRF and orexin-A in MOH patients and LDQ scores could therefore be indirect evidence of the implication of these neuropeptides in drug-seeking behaviour characterizing the above patients.
Current evidence indicates a dichotomy in orexin function, such that orexin neurons in the lateral hypothalamus regulate reward processing for both food and abused drugs, whereas those in the perifornical and dorsomedial hypothalamus regulate arousal and are part of the circuitries that mediate the response to stress (50, 54).
The former orexin system could be more extensively activated in drug addiction, and perhaps in MOH, as suggested by our findings. Molecular adaptations, due to drug overuse, can be induced not only in the orexin-expressing lateral hypothalamus region, integrating environmental cues with consummatory rewards and whose activation drives relapse of drug-seeking behaviour, but also in several orexin projection regions such as the locus coeruleus, nucleus accumbens, amygdala and VTA (8, 55–58). In particular, experimental findings support the pivotal role of the orexin system in inducing neuroplasticity within dopamine neurons in VTA in response to abused drugs and also to any strong motivational stimuli independently of whether they are perceived as positive or negative (52, 59–61).
Although a demonstration of the specificity of CRF/orexin system activation in mediating drug abuse is at this time lacking, its gatekeeper role in inducing neuroplastic changes in experimental models of addiction suggests orexin receptors and the CRF system as potential novel therapeutic targets in the treatment of drug abuse. This can be relevant not only in the case of addiction but also in MOH.
Despite the present study having furnished the first evidence, to our knowledge, of the involvement of the CRF/orexin system in CM with and without medication overuse, it has some limitations. A small number of patients were included in each group. The lack of gender differences in the CSF measurement values of CRF and orexin need to be verified in independent patients and controls.
We could not establish if the findings of increased CRF and orexin-A values were specific for CM and MOH or were common to other chronic painful disorders. Patients with painful neuropathies and myopathies or fibromyalgia could be helpful in clarifying this issue, because they often receive lumbar puncture for diagnostic purposes in the neurological clinical setting.
Moreover, since all patients were in a pain phase at the time of the lumbar puncture, we cannot discriminate if increased CRF and orexin-A levels in CSF in CM and MOH patients are the result of a transformed pain memory or an expression of permanent activation of pain fighting systems.
In addition, CRF/orexin-A levels need to be verified in the CSF of patients with other chronic headache forms, in particular tension-type headache with and without medication overuse, as well as with episodic primary headaches (migraine and tension-type headache), for which, however, the extreme difficulty in obtaining CSF samples, especially in the headache-free periods, should be borne in mind. It would also be interesting to determine the above levels in cluster headache patients, especially in active phase, considering the well-known role played by functional and structural alterations in the hypothalamus in this latter pathological condition.
Moreover, the implication of orexin-B in both episodic and chronic headaches, including MOH, should be clarified in future research.
Finally, to determine if the findings in MOH are due to drug-seeking behaviour, CSF values should also be obtained in the same patients after suspension of abused drugs, comparing data from patients who return to an episodic headache pattern with data from those who do not. It would also be interesting to evaluate the CRF/orexin system in patients with other drug dependencies, but without pain states.
Footnotes
Acknowledgements
We thank John Toomey for the editing and Marisa Morson for technical assistance.
