Abstract
Migraine is the most frequent neurological disorder in the adult population worldwide, affecting up to 12% of the general population and more frequent in women (~25%). It has a high impact on our society due to its disabling nature and, therein, reduced quality of life and increased absenteeism from work. Headache is the primary clinical manifestation and it has been associated with ‘a hereditary or predisposed sensitivity of neurovascular reactions to certain stimuli or to cyclic changes in the central nervous system’ (1). Amongst the many neurotransmitters in the brain, the serotonergic (serotonin, 5-HT) system from the brainstem raphe nucleus has been most convincingly implicated in migraine pathophysiology. The documented changes in 5-HT metabolism and in the processing of central 5-HT-mediated responses during and in between migraine attacks have led to the suggestion that migraine is a consequence of a central neurochemical imbalance that involves a low serotonergic disposition. Although the exact cascade of events that link abnormal serotonergic neurotransmission to the manifestation of head pain and the accompanying symptoms has yet to be fully understood, recent evidence suggests that a low 5-HT state facilitates activation of the trigeminovascular nociceptive pathway, as induced by cortical spreading depression. In this short review, we present and discuss the original and most recent findings that support a role for altered serotonergic neurotransmission in the manifestation of migraine headache.
Keywords
THE BIOLOGY OF THE SEROTONERGIC SYSTEM
5-Hydroxytryptamine or serotonin (5-HT) is synthesized from the essential amino acid tryptophan through its rate limiting enzyme tryptophan hydroxylase (TPH) into 5-hydroxytryptophan that is then decarboxylated by the aromatic
In brain, 5-HT is stored within synaptic vesicles where it is protected from enzymatic degradation. Upon depolarization of 5-HT neurons, 5-HT release occurs through Ca2+-dependent exocytosis and as a function of the firing rate of serotonergic neurons. Accordingly, the release of 5-HT is decreased by drugs such as agonists at presynaptic 5-HT1A or 5-HT1B autoreceptors that reduce the firing rate of serotonergic neurons or 5-HT synthesis. Upon its release, 5-HT is either metabolized into 5-HIAA or recaptured into serotonergic neurons by the serotonin transporter (5-HTT or SERT), a mechanism essential in the maintenance of 5-HT homeostasis.
Genetic basis for altered 5-HT neurotransmission in migraine
5-HT neurotransmission is thus tightly regulated at the level of both its biosynthesis and reuptake. Recent investigations have evaluated the possible link between genetic factors within the serotonergic pathway and migraine susceptibility. Particularly, genetic analyses did not support a role for the TPH gene polymorphisms in the involvement of either migraine with aura (MA) or migraine without aura (MoA) (6, 7), although the AA variant of the A218C polymorphism was less frequent in migraineurs (7). As this variant is unlikely to affect 5-HT biosynthesis, its association with migraine occurrence is still unclear. Similarly, no evidence could be provided for any association between genetic polymorphisms of MAO-A, MAO-B or AADC with migraine susceptibility (6, 8, 9). A similar conclusion has been reached for most 5-HT receptors, because no difference between migraine sufferers and controls could be detected for polymorphisms at 5-HT1A (10), 5-HT2A(11–13) or 5-HT2C (14, 15) receptors. Additionally, the genetic variants of 5-HT1B (16) and 5-HT1F (17) receptors do not influence the clinical response of migraine patients to acute treatment with the 5-HT1 receptor agonist sumatriptan.
The most convincing evidence has been obtained for the association of polymorphisms in the 5-HT transporter with either MA or MoA, although this contribution has not always been confirmed (18). The 5-HTT protein is encoded by the SLC6A4 gene on chromosome 17q12, and two polymorphisms have been reported. The first polymorphism located in the proximal 5′ regulatory region of the 5-HTT gene (5-HTTLPR) was originally associated with migraine susceptibility in a group of women with MA and MoA (13). Subsequently, an increased association was found between this less active short allele of the 5-HTT and predisposition to MA but not MoA (19, 20), whereas another group has reported a link with increased frequency of migraine attacks (21). In contrast, the second polymorphism in the 5-HTT, i.e. the multiallelic variable number of tandem repeats (VNTR), was more frequent in MoA patients (22, 23). Both the 5-HTTLPR and VNTR polymorphisms have been associated with a lower rate of 5-HT reuptake (22, 23).
PERIPHERAL AND BRAIN 5-HT LEVELS IN MIGRAINE
Shortly after its discovery, 5-HT was implicated in the pathophysiology of migraine based on the original finding by Sicuteri and colleagues (24) of an increased urinary excretion of 5-HIAA during migraine attacks. Although this finding has not been confirmed in all populations of migraine sufferers (25), consistent and reproducible increases in plasma 5-HT content concomitant with decreases 5-HIAA levels have been found during migraine attacks. Conversely, low plasma 5-HT and high 5-HIAA levels have been measured in migraine patients between attacks, irrespective of MA and MoA (24–27). The altered levels of 5-HT and its metabolite seen in plasma and urine are thought to reflect dysfunctions occurring not only in blood platelets, but primarily in the brain (27), consistent with the reported increase in cerebrospinal fluid 5-HIAA levels in migraine patients (25). Altogether, these observations lead to the hypothesis that a chronically low serotonin disposition may form the biochemical basis of migraine aetiology, and that a sudden increase in 5-HT release is part of the triggering events that culminate in migraine attacks.
MIGRAINE AS A CHRONIC LOW SEROTONIN SYNDROME
It has recently been found that short-term reduction of brain 5-HT levels through rapid tryptophan depletion induces nausea, dizziness and motion sickness in controls, but not in migraineurs, suggesting no aggravating effect on low 5-HT-related symptoms in these patients (28). Moreover, after subjects completed the tests for pain sensitivity and motion sickness, tryptophan depletion augmented headache in migraine sufferers (29). These findings have been taken as evidence that reduced brain 5-HT synthesis and, therein, 5-HT neurotransmission intensifies symptoms related to migraine and thus might contribute to migraine pathophysiology (29). Interestingly, a recent neuroimaging study performed interictally in migraineurs, using a highly selective 5-HTT ligand and single photon emission computed tomography, has reported increased availability in brainstem 5-HTT in these patients, pointing to dysregulation of the brainstem serotonergic system (30). Such alteration would be compatible with decreased levels of 5-HT at the synaptic cleft, due to decreased synthesis and/or release, although increased catabolism cannot be excluded.
The most convincing arguments for migraine being a syndrome of low serotonergic disposition have been provided by physiological studies that found the increased intensity-dependence in the amplitude of auditory and visual evoked potentials in migraineurs interictally, a characteristic which is indicative of low 5-HT transmission (31) and abnormal processing of cortical sensory information. This most reliable phenotypic marker of migraine patients has been attributed to a deficit in cortical habituation that would result, at least in part, from a reduced preactivation level of sensory cortices (32) as set by several neurotransmitters, including brainstem aminergic pathways. The implication of serotonergic pathways in this response has been further confirmed by the observation that brain penetrant 5-HT1B/1D receptor agonists—that can activate inhibitory prejunctional 5-HT1B/1D autoreceptors on cortical 5-HT afferents and, therein, decrease 5-HT synthesis—increase the amplitude of auditory evoked potentials in both normal subjects and migraine sufferers (33).
Just before and during a migraine attack, the cortical evoked potential habituation deficit seen interictally normalizes (34, 35), suggesting that cortical excitability increases to normal levels. Activation of the brainstem area that contains state-setting aminergic nuclei projecting to the thalamus and cortex has been suggested to contribute to this normalization. Arguments in favour of increased 5-HT availability include the observations that drugs that release 5-HT from neurons and blood platelets (fenfluramine and reserpine) and some 5-HT reuptake inhibitors (zimeldine and femoxetine) are able to provoke migraine attacks more frequently in migraine subjects than in controls (25, 27, 36), as also reported recently in a patient treated with sertraline who developed frequent migraine attacks (37). However, fenfluramine and reserpine used for an extended period can confer resistance to migraine headache, and 5-HT given during an attack will induce headache relief. It seems therefore that the mobilization of 5-HT from intracellular stores at an early stage is required to trigger migraine attacks (38–40).
Interestingly, a positron emission tomography (PET) study performed on a group of patients with MoA has shown that prophylactic treatment with the β-blocker propranolol or nadolol increased the rate of brain 5-HT synthesis, further supporting that migraine is related to low serotonin neurotransmission (41). Such a statement is also in line with the ability of fluoxetine (42), an inhibitor of 5-HT reuptake that increases brain serotonergic functions, and propranolol (43) to correct the habituation deficit or intensity dependence of visual evoked potentials when used in migraine prophylaxis. Additionally, a PET study in migraine patients during spontaneous attacks has shown an increase in cerebral blood flow in the brainstem, including in an area corresponding closely in location to the serotonergic dorsal raphe nucleus, that persisted after sumatriptan, whereas headache, phono- and photophobia had completely vanished (44). This observation suggested activation, at the onset of a migraine attack, of brainstem nuclei—possibly including serotonergic neurons—that may be part of a ‘migraine generator centre’. Our recent findings of an increased rate of 5-HT synthesis in several brain regions in migraine patients within 6 h of a spontaneous attack—compared with interictal levels—would strongly support increased 5-HT availability early in the attack (45).
BRAINSTEM SEROTONERGIC NEUROTRANSMISSION AND VASCULAR HEAD PAIN
The end-point of the migraine process, namely the generation of head pain, requires activation of pain-sensitive trigeminovascular fibres that will initiate the overall nociceptive process through the release of vasodilator substances and increased plasma protein extravasation. In animal models, such activation can be induced by cortical spreading depression (CSD) (46), a condition of neuronal depolarization and ionic shifts that has been visualized in migraine sufferers by functional brain imaging techniques (47). Whether the activation of trigeminovascular fibres could be achieved by a change in the firing of dorsal raphe neurons remains speculative, and the mechanisms involved surely are not well described. However, although there is no direct anatomical connection between raphe neurons and meningeal blood vessels (48, 49), these vessels can respond to changes in central 5-HT neurotransmission. Of particular interest is the observation that a lesion of the dorsal raphe nucleus induces supersensitivity to serotonin in isolated cerebral arteries (50). This situation would be compatible with the suggested supersensitivity of neuronal 5-HT1 receptors in migraine sufferers, possibly due their chronic low 5-HT disposition. This was evidenced by the greater efficacy of zolmitriptan—a selective 5-HT1 receptor agonist—on the intensity dependence of cortical auditory evoked potentials in migraineurs interictally compared with controls (33). Thus, the possibility that an abrupt increase in brainstem 5-HT neuron activity and/or platelet discharge of 5-HT following a stressful stimulus activates sensitized neuronal and vascular 5-HT receptors, and triggers the pain-generating process cannot be excluded. Furthermore, rats in low 5-HT state following treatment with the 5-HT depleting drug para-chlorophenylalanine (an inhibitor of TPH) display enhanced CSD waves and an increased number of activated neurons within the trigeminal nucleus caudalis (51). These results strongly support a link between low 5-HT neurotransmission and migraine headache in showing that a low brain 5-HT disposition facilitates CSD-induced trigeminal nociception, probably though increased cortical excitability and sensitivity of the trigeminovascular pathway.
CLINICAL IMPLICATIONS
Based on the growing body of evidence for a direct role for dysfunctions of central 5-HT availability in migraine headache, a better understanding of the mechanisms of some of the drugs currently used in the preventive or symptomatic treatment of migraine headache should be possible. In this regard, it is interesting to note that, similar to migraine, depression is also considered to be a disorder of low brain serotonergic activity, and epidemiological studies have reported comorbidity of migraine with depression (52). Most antidepressant drugs are aimed at enhancing and stabilizing 5-HT neurotransmission (53) and, of interest, the tricyclic antidepressants (TCAs) have been shown to be effective in migraine prophylaxis at a fraction of the dose used in depression. It has also been reported that pharmacologically controlled depressed patients submitted to a rapid tryptophan depleting diet experienced a depressive relapse together with symptoms reminiscent of migraine, such as increased nausea or vomiting, drowsiness and, in some cases, headache (54). In rats, chronic administration of a low dose of amitriptyline, a TCA effective in migraine prophylaxis, selectively decreased the 5-HT synthetic rate in the brainstem dorsal raphe nucleus, while having no effect on that in projection areas (55) (Fig. 1A). Such effects would lead to preserved 5-HT neurotransmission in cortical and lower brain areas involved in the regulation of the pain pathways and, possibly, prevent the burst increase in 5-HT signalling that accompanies the onset of the attacks, two mechanisms of action that could possibly contribute to its antimigraine efficacy.
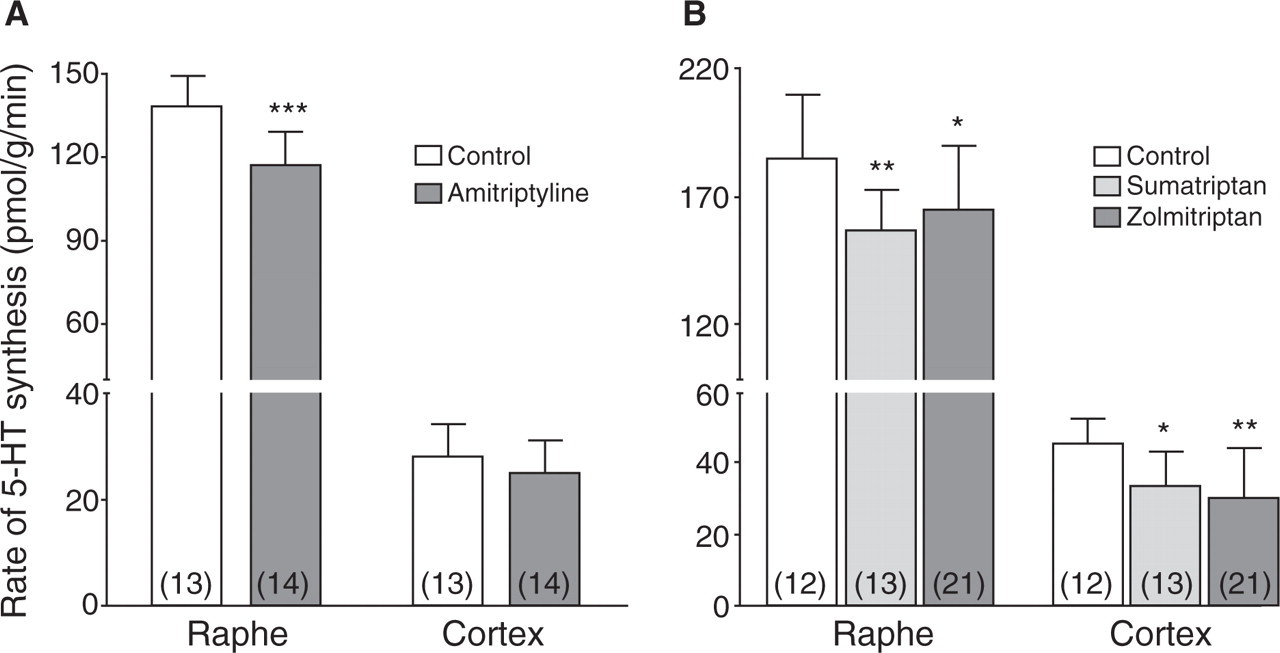
Effects of chronic treatment with amitriptyline (2 mg kg−1 day−1 for 21 days) (A) or acute treatment with sumatriptan (1 mg/kg, s.c.) or zolmitriptan (300 µg/kg, s.c.) (B) on the rate of serotonin synthesis in either the dorsal raphe nucleus or the cerebral cortex (including the cingulate, frontal, parietal and occipital subdivisions). Data were taken (55, 61), respectively. Numbers of animals are shown within parentheses for each group. ∗P < 0.05; ∗∗P < 0.01; ∗∗∗P < 0.001.
In the acute treatment of migraine headache, the efficacy of triptans—selective 5-HT1 receptor agonists—has been ascribed to their ability to constrict cerebral blood vessels and modulate trigeminovascular nociception (56). Yet, triptans, including sumatriptan, the gold standard of this category of drugs, access brain parenchyma where they can exert various actions (33, 57–59). This is in agreement with the widespread distribution of 5-HT1 receptors in human brain, including in brainstem centres involved in the transmission of pain centrally, as well as in several subdivisions of the cerebral cortex (60). Hence, acute administration of sumatriptan or zolmitriptan in rats decreased 5-HT synthesis rate in dorsal raphe nucleus and in several projection areas including cerebral cortex, hippocampus and thalamus (61), probably by interaction with somatodendritic 5-HT1A and terminal 5-HT1B/1D autoreceptors (Fig. 1B). We have recently observed a similar reducing effect of sumatriptan on the 5-HT synthetic rate when administered to migraine sufferers within 6 h of their spontaneous migraine attacks (45). Together with other recent studies showing central sites of action for triptans, these findings indicate that triptans, acting through different 5-HT1 receptor subtypes located on distinct neuronal pathways or parts (cell body vs. terminal areas) of 5-HT neurons, can concurrently but independently modulate 5-HT synthesis and pain pathways (59, 62–65). Such a decrease in the 5-HT synthetic rate could be an additional effect of triptans in the acute treatment of migraine headache. Further work will be needed for a better evaluation of the clinical outcome or therapeutic benefits of such interactions of the triptans with the brain serotonergic system.
ACKNOWLEDGEMENTS
E.H. thanks Drs T. Pringsheim and Y. Sakai, as well as Mr C. Dobson, MSc, for performing the work discussed in this paper, and the Canadian Institutes of Heath Research (CIHR), the Heart and Stroke Foundation of Québec and GlaxoSmithKline Canada for research grant support.
