Abstract
Central serotonergic neurotransmission was assessed using intensity dependence of cortical auditory evoked potentials (IDAP) in cluster headache (CH) patients during both the active and interictal period. In 15 episodic CH patients and 13 controls previously described methods were used and amplitude-stimulus intensity function (ASF) slopes were computed. In the cluster group mean ASF slope was significantly steeper than in the control group both during the active period (1.53 + 0.90 vs. 0.77 + 0.85, P = 0031) and interictally (1.85 + 1.20 vs. 0.77 + 0.85, P = 0012). In the cluster group IDAPs of active and interictal period did not differ significantly (P = 0378). Duration of the disease or the present bout, distance from the last attack did not correlate with ASF slopes. In conclusion, our results are compatible with decreased level of serotonergic neurotransmission in raphe-cortical pathways. Diminished serotonergic activity in raphe-hypothalamic serotonergic pathways might be hypothesized influencing the activity of hypothalamic neurons and thus play a role in the genesis of cluster headache.
Introduction
Clinical features enable an easy diagnosis for cluster headache in most cases, but despite remarkable new data during the last years its pathogenesis is far from being clear. The circadian rhythmicity of attacks drew the attention to the hypothalamus as a central generator of attacks (1–3). The central hypothesis was further supported by biochemical studies demonstrating hormonal changes (4–6) and by the therapeutical effect of lithium (7). Activation in the ipsilateral hypothalamus during cluster attacks was demonstrated by May et al. (8) in a PET study and a patient with intractable cluster headache was relieved by stereotactic stimulation of the hypothalamic grey (9).
Sumatriptan, the drug considered to be the most effective for the treatment of cluster attacks also has a direct action on hypothalamic targets. This is supported by autoradiographic studies of 3H-sumatriptan binding in both animal models (10) and human autopsy material (11) and also by a case report of patient who continued to have an excellent response to sumatriptan after the complete section of the ipsilateral trigeminal sensory root (12). Moreover, repetitive acute treatment with sumatriptan, naratriptan and eletriptan decreased markedly the attack frequency (13–15) that could be explained by a direct action on the putative cluster generator, i.e. hypothalamus.
Investigating serotonin metabolism in cluster headache, higher plasma levels of 5HT and its metabolite(5-hydroxyindoleacetic acid, 5HIAA), slightly reduced 5HT and significantly reduced 5HIAA platelet levels were found in the active period of the disease (16, 17). A central serotonergic agonist, m-chlorophenylpiperazine (m-CPP) re-duced cortisol and increased prolactin levels in the serum of cluster patients during the bout. Although discussed controversially, these changes might reflect alterations in the central serotonergic system (18).
A surrogate marker of central serotonin neurotransmission, as demonstrated previously in animal models, healthy volunteers and migraine patients with and without the administration of drugs is the intensity dependence of cortical audi-tory evoked potentials (IDAP) (19–24). Therefore the aim of our study was to investigate whether the IDAP changes in cluster headache (CH) patients during both the active and interictal period compared to controls.
Subjects and methods
Subjects
Fifteen episodic cluster patients (11 males, mean age 41 ± 16 years) and 13 control subjects without history of recurrent headache (11 males, mean age 39 ± 9 years) participated in the study. Cluster headache was diagnosed according to the criteria of the International Headache Society (25) (code 3.1.2). Patients were studied during the bout outside attacks without prophylactic treatment and also outside the bout. Before recordings they did not use ergots or triptans to treat their attacks. Patients had a history of cluster headache ranging from 1 week to 33 years, and they were studied 4–60 days after the onset of the bout and 2–30 h after the end of an attack. One patient who participated in the study one week after the onset of his first cluster period presented another bout 6 months later. Oral informed consent of patients was obtained.
Experimental design and recordings
IDAP recordings using 1000 Hz tones (50 ms total duration, 10 ms rise and fall times) were delivered binaurally through earphones at a repetition rate of 0.55 Hz at four intensities (40, 50, 60 and 70 dB) above sensation level in a pseudo-randomised order. The EEG was recorded with a needle electrode at Cz and referenced to linked mastoids. The amplifier system was a Cadwell Sierra apparatus, filters were set at 1 Hz low- and 20 Hz high-cut. For each stimulus intensity 100 artefact-free sweeps were averaged over a 400 ms epoch.
Data analysis
N1 (between 60 and 150 ms poststimulus) and P2 (between 120 and 200 ms poststimulus) components were identified for each averaged recording of 100 responses. Peak-to-peak amplitude of N1-P2 was measured for each stimulus intensity and the linear amplitude/stimulus intensity function (ASF) slope expressed in µV/10 dB was calculated for each recording session. IDAP differences were analysed with
Results
In all recordings N1 and P2 components of auditory evoked potentials (AEP) were clearly identified. There were neither significant latency (Table 1) nor mean amplitude differences in AEP between groups.
Latencies of N1 and P2 components (mean ± standard deviation)

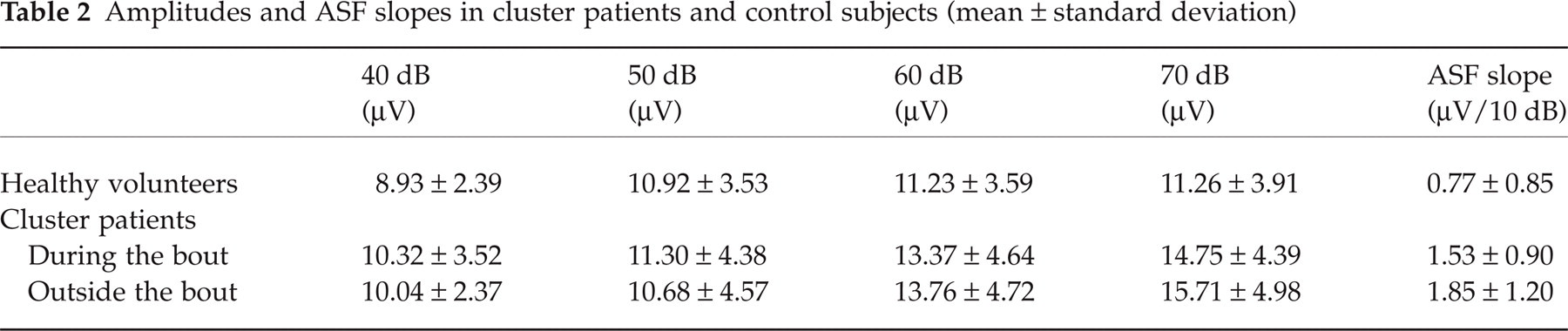
ASF slopes of auditory evoked potentials (AEP) were steep in cluster patients both when pain-free during the bout and outside the bout compared to control subjects (Table 2, Fig. 1). Data of patients both during and outside the bout differed significantly from control subjects (P = 0.031 and 0.012, respectively) whereas no statistical difference was found between cluster patients examined during different periods of cluster headache (P = 0.378). No correlation could be demonstrated in cluster patients between IDAP slopes and disease duration, time from onset of the bout or from the end of the last attack (P = −0.16, –0.12, 0,21, respectively). The number of common migrainous features (nausea, vomiting, photo/phonophobia) and nicotine consumption were also checked but no correlation was found with AEP ASF slopes, either (P = 0.11 and −0.26, respectively).

ASF slopes in healthy volunteers and cluster patients showing significant difference between healthy volunteers and both groups of patients but not between cluster patients during and outside the bout. ∗P < 0.05 compared to healthy volunteers.
Amplitudes and ASF slopes in cluster patients and control subjects (mean ± standard deviation)
Discussion
We demonstrated abnormalities of the cortical processing of sensory information as measured by intensity dependence of AEPs in cluster headache patients during and outside the bout.
This is a similar pattern to IDAP changes in migraine with and without aura patients in the interictal period (21, 23, 24) that can be explained by a dysfunction of the raphe-cortical pathways. There is little evidence of a cortical dysfunction in cluster headache when compared to migraine. To our knowledge no studies on visual, auditory or cognitive habituation to repetitive stimulation have been performed. Evers et al. (26, 27) suggested involvement of cognitive processing in cluster headache assessed by visually evoked event-related potentials. They found significantly increased latencies both during and outside the bout but no loss of cognitive habituation when comparing ERP components of the first and second half of the measurement. Cluster patients, however, show increased sensitivity to light and sound during but not outside the bout compared to healthy controls. Vingen et al. (28) in a study of cluster, and in a previous one of migraine patients (29), assessed light and sound sensitivity using visual and auditory stimulation of increasing intensity to elicit discomfort and pain. Intensity thresholds of discomfort were measured bilaterally and unilaterally as well. Interestingly, unilateral pain in both migraine and cluster headache was associated with lower thresholds on both suggesting involvement of central components, e.g. raphe–cortical pathways.
Serotonergic projections form the raphe nuclei project not only diffusely to the cortex but also to the hypothalamus, especially to the suprachiasmatic nucleus (SCN), the primary pacemaker for circadian rhythms. Evidence of serotonergic influence to SCN comes from animal experiments and is further supported by clinical observations. In vitro serotonin, for instance, influences directly the electrical activity of SCN (30). Behavioural/arousal state information of the brain reaches the SCN from the dorsal raphe nucleus (DRN) via serotonergic projections. This was confirmed by animal experiments: electrical stimulation of DRN resulted in phase-advance of the circadian rhythm (31) on one hand and the presence of functional serotonin 1B/1D autoreceptors was suggested in guinea pig DRN (32) on the other. The activity of the human circadian pacemaker is probably also influenced by serotonin as suggested by clinical observations. Selective serotonin reputake inhibitors frequently disrupt sleep rhythms (33), depressed patients have abnormal ACTH-cortisol and growth hormone rhythms and abnormally short REM sleep latencies (34). Moreover, SCN is a sexually dimorphic nucleus that might explain male preponderance in cluster headache (35).
One cannot entirely exclude the possibility that changes in serotonergic neurotransmission shown in our study are due to the pain itself. Ter Horst et al. (36) suggested pain-related changes in hypothalamic as well as in dorsal raphe and in locus ceruleus activation in both migraine in cluster headache, based on data of their study in rats examining trigeminal nociception-induced cerebral Fos expression. The fact, that in our study IDAP changes did not correlate to time to onset of the bout or to the end of the last attack and disease duration does not support the possibility of a pain-related modification. Furthermore, IDAP changes outside the bout were even more pronounced – although not significantly so – than during the bout that would favour a general and not an attack- or pain-related change in serotonergic transmission.
Taken together, one might hypothesize that in cluster headache dysfunction in raphe-hypothalamic serotonergic neurotransmission might change the excitability of some hypothalamic neurons, e.g. in the SCN. This could lead to a change in its pace-maker activity and thus, play a role in the genesis of cluster-attacks. Hypothalamic neurons might be, however, more excitable in patients with chronic cluster headache, as in PET studies during nitroglycerin induced attacks hypothalamic activation was present only in chronic (8) and not in episodic cluster patients (37). Reasons for this difference are not yet clear. Besides the ’hypothalamic generator’ peripheral factors, such as an inflammatory process in the cavernous sinus and/or narrower sinus cavernosus loggia might have an influence as well (38, 39).
