Abstract
Triptans are 5-HT1 receptor agonists used as anti-migraine drugs. They act primarily on meningeal blood vessels and on trigeminovascular afferents, but they may also exert central effects. We studied the regional effects of acute and chronic treatment with sumatriptan or zolmitriptan on the rate of serotonin (5-HT) synthesis in the rat brain, using the α-14C-methyl-L-tryptophan quantitative autoradiographic method. Sumatriptan at low (300 μg/kg, s.c.) and high (1 mg/kg) doses, as well as zolmitriptan (100 μg/kg), acutely decreased (15-40%, P < 0.05-0.001) 5-HT synthetic rate in many brain regions, including the dorsal raphe nucleus. Chronically, sumatriptan (21 days, approximately 300 μg/kg per day via osmotic minipumps) induced significant increases in the 5-HT synthesis rate in many projection areas but had no effect in the dorsal raphe nucleus. The acute effects on 5-HT synthesis rate would be compatible with activation of 5-HT1 autoreceptors that inhibit serotonin release. In contrast, the increased 5-HT synthesis rate observed after chronic sumatriptan might possibly result from a down-regulation/desensitization of 5-HT1 receptors and/or unmasking of excitatory triptan-sensitive 5-HT receptors. Overall, the present findings indicate that not only zolmitriptan but also sumatriptan affect brain serotonergic neurotransmission.
Introduction
The symptomatic treatment of migraine headache has been greatly improved by the emergence of highly effective anti-migraine drugs acting as agonists at 5-HT1B/1D and, to a lesser extent, 5-HT1A/1F receptors (1, 2). They are commonly known as the ‘triptans’, the first developed molecule of this series being sumatriptan. Two sites of action, 5-HT1B receptors that mediate contraction of intracranial arteries and 5-HT1D/1F receptors that inhibit calcitonin gene-related peptide (CGRP) and/or substance P release from trigeminovascular afferents, are thought mainly to account for their clinical efficacy (for review [3, 4]). Additionally, 5-HT1B/D/F receptors are located in several brain areas (5) including the region of termination of dural trigeminovascular afferents in the spinal trigeminal nucleus (6), where they represent potential sites of action for brain penetrant triptans. Activation of these centrally located receptors, suggested to correspond mostly to 5-HT1D receptors (7), results in inhibition of trigeminal nociceptive neurotransmission following dural stimulation (7–9).
In contrast to zolmitriptan, rizatriptan, eletriptan and naratriptan that act both centrally and peripherally, sumatriptan was originally thought to act only at peripheral sites due to its minimal passage within the central nervous system (CNS) (10, 11). However, more recent findings have provided evidence that sumatriptan can also act at central sites. These include (i) substantial sumatriptan concentrations in spinal fluid of healthy volunteers after its administration (100 mg, p.o.) (12), (ii) lack of correlation between triptan lipophilicity and clinically efficacy (13), (iii) manifestation of so-called ‘triptan symptoms’ following sumatriptan administration
In addition to 5-HT1 receptors in the brain-stem trigeminalis nucleus caudalis, triptans that enter the CNS may interact with regionally distributed 5-HT1 receptor subtypes involved in the regulation of serotonergic neurotransmission, the latter being altered in migraine patients (17). In this respect, triptan-sensitive 5-HT1B/1D receptors are predominantly terminal autoreceptors that inhibit 5-HT synthesis and/or release, and decrease brain 5-HT levels as measured by a variety of experimental methods (18–22). It is thus possible that inhibition of central 5-HT synthesis, particularly by brain penetrant triptans, may contribute to their anti-migraine efficacy and represent an additional pharmacological effect for these compounds. In the present study, we investigated the ability of two triptans with different CNS penetration to affect brain serotonergic activity in vivo. Specifically, we measured 5-HT synthesis rates in rat brains in vivo after acute administration of sumatriptan (300 µg/kg and 1.0 mg/kg, s.c.) or zolmitriptan (100 µg/kg, s.c.), as well as after chronic sumatriptan treatment (approximately 300 µg/kg per day for 21 days). Serotonin synthesis rate was assessed with the α-14C-methyl-L-tryptophan (α-14C-MTrp) autoradiographic method (23), which has been well characterized and shown to provide a good index of regional brain 5-HT synthesis rate (24, 25). The results show that both sumatriptan and zolmitriptan acutely decreased brain 5-HT synthesis rate while chronic administration of sumatriptan increased 5-HT synthesis in several projection areas.
Materials and methods
Animals
Sprague-Dawley male rats (Charles River, St-Constant, Canada) weighing between 200 and 250 g were used in the acute treatment study. In the chronic study rat weights ranged from 180 g and 230 g at the beginning and between 300 g and 365 g at the end of the treatment. Rats were housed in the animal facility (room temperature 22 °C, 12-h day–night cycle) for at least 48 h before being used in an experiment. The rats were fasted overnight with water supplied ad libitum before the day of the experiment with α-14C-MTrp to reduce variability in plasma amino acids. To avoid any influence of circadian rhythm on the results, all rats were killed between 14.00 h and 16.00 h. All protocols were approved by the Animal Ethics Committee, according to the Canadian Council for Animal Care guidelines.
Drugs
In the acute studies, sumatriptan succinate (GR43175C; GlaxoSmithKline R&D, Stevenage, UK, and Mississauga, ON, Canada) was dissolved in 0.9% NaCl solution to a final individual dose 1.0 mg/kg or 300 µg/kg. Zolmitriptan succinate (311C90; AstraZeneca Canada Inc., Mississauga, ON, Canada) was dissolved in the same manner in order to reach a final dose of 100 µg/kg for each rat. These doses were selected as they have been proven efficacious in two in vivo animal models used to evaluate anti-migraine drug efficacy, namely neurogenic dural plasma extravasation and c-fos expression in the nucleus trigeminalis caudalis (7, 26, 27). New controls were added for the low-dose sumatriptan experiment to avoid possible seasonal changes in synthesis rates, as the experiment was done at a later time in the year. For the chronic study, osmotic minipumps were loaded with sumatriptan diluted in saline (25 mg/2 ml) as determined by the capacity of the pump (Alzet osmotic pump model 2004; via Durect Corp., Cupertino, CA, USA) for a consistent delivery at a rate of 0.25 µl/h for 21 days. They were implanted subcutaneously under aseptic conditions and all minipumps were checked for effectiveness by examining whether they emptied their content at the end of the study. During the course of the experiment, rats received an average dose of approximately 300 µg/kg per day sumatriptan. For the determination of the 5-HT synthesis rate, α-14C-MTrp was synthesized using the procedure described previously (28) with a specific activity of approximately 55 mCi/mmol.
Autoradiographic procedure
The femoral artery and vein were cannulated with plastic catheters under light halothane (approximately 1.0%) anaesthesia. Two rats were operated upon per day. The posterior limbs of the rats were fixed using a loose-fitting plaster cast, and the rats were allowed to awake. The body temperature of the rats was kept at 37 °C with a heat lamp. In the acute study, rats were then injected with drugs or vehicle, at least 2 h after insertion of the catheters. Drugs were injected subcutaneously, beneath the loose skin of the neck. The order of control and drug-treated rats was randomly altered on a daily basis. Exactly 20 min after drug or vehicle injection, 30 µCi of α-14C-MTrp in 1.0 ml of saline were injected through the catheter into the femoral vein over 2 min by an injection pump. For the chronic sumatriptan study, on day 22 of minipump implantation and at least 2 h after surgical preparation, α-14C-MTrp was injected in the same way as in the acute treatment study.
Arterial blood samples were taken at progressively increasing intervals, starting from the beginning of tracer injection to decapitation of rats. The blood samples were centrifuged (4 min) at 9300

Representative qualitative autoradiograms obtained from rats acutely injected (subcutaneously) with saline, sumatriptan (1 mg/kg) or zolmitriptan (100 µg/kg). The autoradiograms were exposed for 3 weeks and illustrate the tissue radioactivity in the brain 150 min after injection of 30 µCi of α-14C-methyl-L-tryptophan (MTrp). CiC, Cingulate cortex; CP, caudate-putamen; DR, dorsal raphe; FrC, frontal cortex; Hi, hippocampus; HT, hypothalamus; PaC, parietal cortex; PG, pineal gland; Th, thalamus. Note that the pineal gland is not present in the saline (control) group in the section selected. The areas delineated by circles correspond to the regions where the readings were made for each structure (at least 12 readings/structure per rat).
Measurement of plasma tryptophan concentration
At three points of blood sampling during the experiments, an additional 50 µl of plasma were deproteinized with 25 µl of 20% trichloroacetic acid and used for the measurement of the plasma Trp concentration. After the sample was mixed with a vortex and centrifuged, 50 µl of supernatant was stored at − 84 °C until it was measured for total Trp concentration. A further 50 µl of plasma were filtered through an Ultrafree-MC filter spinning at 11 200
Calculation of net clearance and 5-HT synthesis rate
The net plasma to brain clearance (K∗; ml/g per min) values of α-14C-MTrp were calculated from the linear relationship between the volume of distribution (DV; ml/g) and the exposure time (Θ; min) (29). An input function for each rat was smoothed using plasma radioactivities obtained from arterial sampling, and the plasma radioactivity at the time the rat was killed was estimated by a least-squares fit by fitting data points after the peaks to a sum of exponentials. Using this mathematically fitted curve of the input function and the measured data points before the peak, the exposure time (Θ) was calculated as a ratio between the integral under the plasma curve and the plasma tracer concentration at the end of the experiment. For the calculation of the 5-HT synthesis rates, we used a lumped constant (LC) of 0.42 measured in vivo in rat brain (30), and shown not to be affected by pharmacological treatments such as probenecid (30) or fluoxetine (unpublished observation). The LC was then used to convert the α-14C-MTrp trapping constant (K∗) into the constant for Trp conversion to 5-HT via the synthetic pathway (24). The synthesis rate (R; pmol/g per min) values were calculated by the equation R = K∗ Cp free-Trp/LC, where Cp free-Trp(pmol/ml) is the concentration of plasma free Trp.
Statistical analysis
The statistical comparison between groups of control and treated rats was achieved by ANOVA followed by post hoc Tuckey comparison test for the physiological parameters (Table 1) or Bonferroni post hoc correction for the analysis of the three groups in study 1 (Table 2). Comparison of ratios of 5-HT synthesis rates between control and treated groups was performed by one-sample two-tailed t-tests. The null hypothesis is that the mean ratio should be 1 ± 0 if there is no effect of treatment. A mean ratio > 1 suggests a significant reduction while a ratio < 1 indicates a significant increase in overall synthesis. After the main effect was found significant (P < 0.05), two-tailed t-test of individual structure between treated and respective control groups was performed. The power calculations of all statistical analyses are shown in Tables. All results are expressed as mean ± SD and statistical significance was defined as P < 0.05.
Physiological parameters and tryptophan concentrations in rats acutely or chronically treated with saline, sumatriptan or zolmitriptan

Values are expressed as mean ± standard deviation (SD). Body weights were taken just prior to cannulation (acute treatments) or just before subcutaneous osmotic minipump injection and just prior to the cannulation procedure (chronic treatment). Parameters are not significantly different from their respective controls, with the exception of weight of the control rats in the high-dose experiment which was significantly lower than that of the sumatriptan- and zolmitriptan-treated rats
(unpaired t-test, t = 2.725, P < 0.05;
unpaired t-test, t = 3.816, P < 0.01). Saline vehicle is a normal 0.9% NaCl solution.
5-HT synthesis rate values (R-values) in rats injected acutely with saline (control, 0.9% NaCl, s.c.), sumatriptan (1.0 mg/kg, s.c.) or zolmitriptan (100 µg/kg, s.c.)
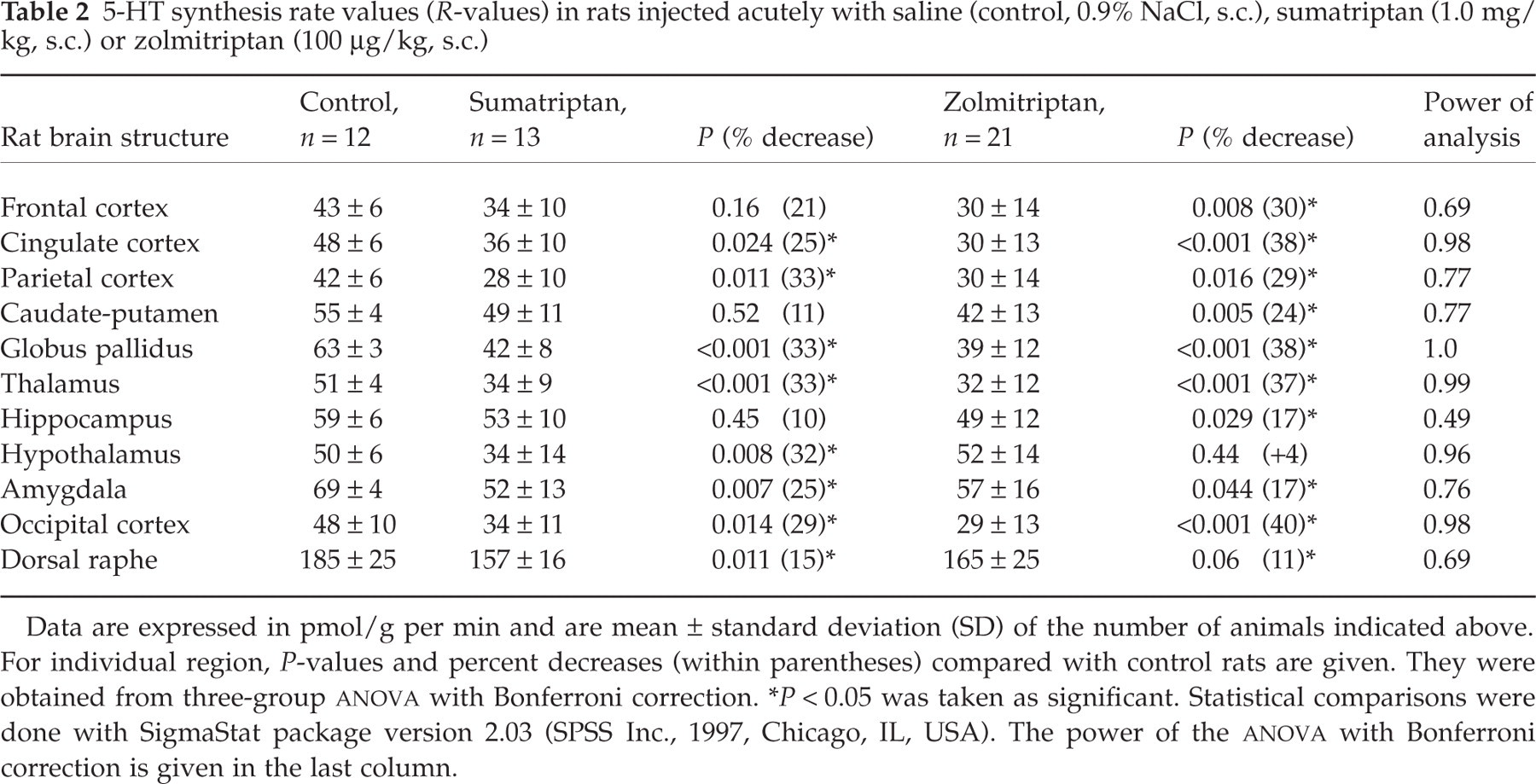
Data are expressed in pmol/g per min and are mean ± standard deviation (SD) of the number of animals indicated above. For individual region, P-values and percent decreases (within parentheses) compared with control rats are given. They were obtained from three-group ANOVA with Bonferroni correction.
∗P < 0.05 was taken as significant. Statistical comparisons were done with SigmaStat package version 2.03 (SPSS Inc., 1997, Chicago, IL, USA). The power of the ANOVA with Bonferroni correction is given in the last column.
Results
All physiological parameters and plasma Trp concentrations were strictly comparable between all treated rats and their respective controls, except for the slightly lower body weight of control rats in the high-dose sumatriptan and zolmitriptan study (7 and 11%, P < 0.05 and P < 0.01, respectively) (Table 1). In the chronic sumatriptan study, the average gain in body weight was comparable between saline and sumatritan-treated rats (Table 1). Representative qualitative autoradiograms illustrating the distribution of tissue radioactivity in rat brain after tracer injections are presented in Fig. 1. As seen in all groups, areas with a known high synthetic rate of 5-HT such as the raphe nuclei and the pineal gland (5-HT synthesis in this structure being an intermediary in melatonin synthesis) appear densely labelled. Most projection areas are more weakly labelled, though heterogeneity can be detected as seen in cortex, thalamus, hippocampus and caudate nucleus.
Rates of 5-HT synthesis (R-values) calculated using the mean values of plasma free Trp concentration in each group indicated a significant difference between controls and both acute sumatriptan groups. These treatments resulted in significant decreases in 5-HT synthesis rates in several brain regions for both the high (1.0 mg/kg, s.c.) and low (300 µg/kg, s.c.) sumatriptan doses (Tables 2 and 3), as indicated by the significant increase of the ratios from 1 ± 0. The mean ratios were 1.34 ± 0.15 (t = 7.04; P < 0.0001; n = 11) for the high and 1.28 ± 0.15 (t = 6.33; P < 0.0001; n = 11) for the low dose of sumatriptan. Similarly, acute zolmitriptan administration (100 µg/kg, s.c.) induced significant decreases in 5-HT synthesis rate across almost all brain regions studied (Table 2). The mean ratio was 1.37 ± 0.23 (t = 5.35; P < 0.0002; n = 11) for the zolmitriptan acute treatment. Chronic 21 days sumatriptan treatment (approximately 300 µg/kg per day, s.c.), in contrast, resulted in significant increases in 5-HT synthesis rate in several brain regions (Table 4). The mean ratio was 0.84 ± 0.09, a value significantly lower than one (t = 5.75; P < 0.0002). The regional changes in 5-HT synthesis rates in different rat brain regions measured in all drug conditions compared with saline controls are shown in Tables 2–4. Regional differences in 5-HT synthesis rate varied depending on compound, dosage or regime. Specifically, at the higher dose of sumatriptan most structures measured exhibited significantly lower 5-HT synthesis rate compared with controls. The larger decreases (25–34%) were found in cortical and other projection areas (thalamus, hypothalamus, amygdala) with the exception of the hippocampus and caudate-putamen where there was no significant change in 5-HT synthesis. After zolmitriptan, the hypothalamus was the only region investigated that did not show a significant decrease in 5-HT synthetic rate (Fig. 1 and Table 2). For both compounds, decreases in 5-HT synthesis rate in the dorsal raphe nucleus were proportionally smaller in magnitude than those noted in projection areas (15 and 11% for sumatriptan and zolmitriptan, respectively, Table 2) (Fig. 1), and the power of the analysis was also lower (0.69, Table 2). The low dose of sumatriptan had a restricted effect, with significant decreases in 5-HT synthesis rate being observed only in the hippocampus, hypothalamus and amygdala (Table 3). In contrast, chronic administration of a comparatively low dose of sumatriptan induced significant increases (17–41%) in 5-HT synthesis rate in the majority but not all cortical projection areas, while no change was observed in the dorsal raphe, caudate putamen, hippocampus and hypothalamus (Table 4).
5-HT synthesis rate values (R-values) in rats injected acutely with saline (control, 0.9% NaCl, s.c.) or sumatriptan (300 µg/kg, s.c.)

Data are expressed in pmol/g per min and are mean ± standard deviation (SD) of the number of animals indicated above. For individual region, P-values and percent decreases (within parentheses) compared with control rats are given. P < 0.05 was taken as significant. The power of the analysis given in the last row was calculated from two-group comparisons, control and treated groups.
∗P < 0.01.
5-HT synthesis rate values (R-values) in various rat brain structures after chronic treatment (21 days) with saline (control, 0.9% NaCl, s.c.) or sumatriptan (∼300 µg/kg per day)
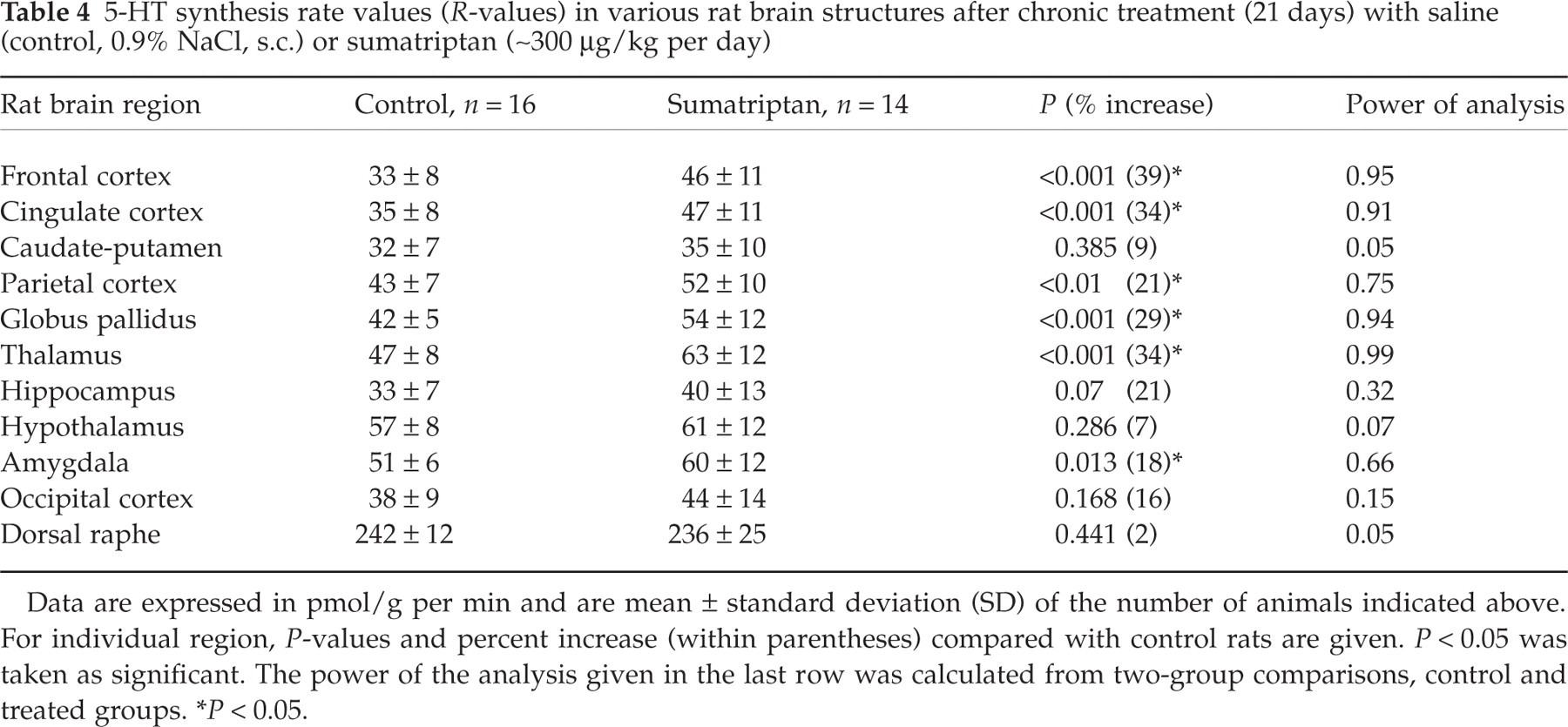
Data are expressed in pmol/g per min and are mean ± standard deviation (SD) of the number of animals indicated above. For individual region, P-values and percent increase (within parentheses) compared with control rats are given. P < 0.05 was taken as significant. The power of the analysis given in the last row was calculated from two-group comparisons, control and treated groups.
∗P < 0.05.
Discussion
The present results show that 5-HT synthesis rate is affected in several brain regions in rats treated with two anti-migraine drugs of the triptan family. In the acute studies, a similar pattern and extent of decrease in 5-HT synthesis rate was found between the high-dose sumatriptan (1 mg/kg) and zolmitriptan (100 µg/kg), while the lower sumatriptan dose (300 µg/kg) induced decreases in restricted brain areas. In contrast, chronic administration over a 21-day period of a comparable low dose of sumatriptan (approximately 300 µg/kg per day) resulted in an increase in central 5-HT synthesis rate in some but not all projection areas, with no change in the cell body region of the dorsal raphe nucleus.
Sumatriptan and zolmitriptan exhibit high affinity (approximately 10–100 nM) at 5-HT1B/1D receptors and a slightly lower affinity (50–230 nM) at 5-HT1A/1F receptors as determined in binding and functional assays (1, 16, 31). At functional receptors inhibiting forskolin-stimulated cAMP production, sumatriptan is less potent (about five-fold) than zolmitriptan, but both are full agonists and exhibit highest affinity at 5-HT1D compared with the other 5-HT1 receptors (approximately three- and 22-fold, respectively) (3, 16, 31). The decreased 5-HT synthetic rates observed after acute zolmitriptan and sumatriptan administration result most likely from an agonist effect at terminal 5-HT1B/1D autoreceptors. Indeed, these receptors inhibit 5-HT release from serotonergic nerve terminals (19–21), and their acute activation by non-selective 5-HT1 receptor agonists has recently been shown to decrease 5-HT synthesis rate in most projection areas from the raphe nuclei (32). Although we found rather similar effects with the high-dose sumatriptan (1 mg/kg) and zolmitriptan (100 µg/kg), some differences were apparent such as the lack of effect of sumatriptan in the caudate putamen and hippocampus, and of zolmitriptan in the hypothalamus. These are not easily explained, as the caudate putamen and hippocampus are two regions rich in 5-HT1B receptors (33, 34), and the hypothalamus is a region where blood-borne molecules are known to enter the brain quite readily (35). Despite these inconsistencies that may reflect differences in agonist potency of the compounds and/or animal status, our results indicate that both triptans, perhaps to a slightly different extent, can reach centrally located 5-HT1 receptors and modulate 5-HT synthesis. This conclusion would support the decrease in 5-HT levels reported in the hypothalamus after s.c. administration of 0.6 mg sumatriptan (22), and in the cerebral cortex, hippocampus or striatum following locally injected sumatriptan (16, 36, 37). The contribution of 5-HT1F receptors in these effects is probably minimal as they are mainly located on non-serotonergic neurones (38).
High-dose sumatriptan and zolmitriptan also decreased, but to a smaller extent, 5-HT synthesis rate in the dorsal raphe nucleus. Both compounds exhibit significant affinity at the 5-HT1A receptor, the main somatodendritic autoreceptor in the raphe nuclei that negatively controls 5-HT neuronal firing, 5-HT release (39–41) and, as recently demonstrated, 5-HT synthesis rate (42, 43). The observed decreased 5-HT synthesis rate in the raphe nucleus elicited by the two triptans could thus be compatible with their interaction with 5-HT1A autoreceptors or, possibly, with more sparsely distributed dendritic 5-HT1D receptors ([18] for review) that control 5-HT release independently from cell firing (19).
With the lower dose of sumatriptan (300 µg/kg), decreased 5-HT synthesis rate was restricted to the hippocampus, hypothalamus and amygdala. This limited effect is probably explained by the too small amounts of sumatriptan that reached the brain to interact equally well with all 5-HT1 receptor subtypes. Indeed, sumatriptan exhibits lower affinity than zolmitriptan for all 5-HT1 but 5-HT1A receptors (3, 31), and it is a weaker functional agonist than zolmitriptan at these sites (16). Notably, the highest decrease in 5-HT synthesis observed in the hypothalamus agrees very well with the previously reported diminished 5-HT concentration and/or increased 5-HT turnover in this structure following 0.6 or 0.9 mg/kg (s.c.) sumatriptan administration (22).
The increase in 5-HT synthesis rate observed in several projection areas after chronic treatment with sumatriptan is rather surprising. Evidence in the literature for direct regulation of 5-HT1B/1D receptors by chronic agonist administration has not been reported. However, chronic administration of selective serotonin re-uptake inhibitors or tricyclic antidepressant drugs, which ultimately increases the amount of 5-HT in synaptic clefts, has been shown to down-regulate and desensitize somatodendritic and terminal inhibitory 5-HT1A and 5-HT1B autoreceptors (40, 44–46). Given these data, it is thus possible that chronic exposure to sumatriptan results in down-regulation/desensitization of 5-HT1A/B/D receptors, which could lead to loss of feedback autoregulation and increased 5-HT synthesis rate. Alternatively, the relatively high affinity (approximately 500 nM) of sumatriptan for 5-HT7 receptors (47) might possibly explain our findings, at least in part. 5-HT7 receptors are primarily enriched in regions that receive 5-HT innervation such as the cerebral cortex, amygdala, hypothalamus and hippocampus (47–49), and they have been suggested to act as autoreceptors regulating serotonergic neurotransmission (47). Yet, acute treatment with a 5-HT7 receptor antagonist did not support such a role in the raphe nucleus or the cerebral cortex (50). It cannot be excluded, however, that chronic administration of sumatriptan allows the putative excitatory effects of 5-HT7 receptors to become detectable following down-regulation/desensitization of co-distributed inhibitory 5-HT1 receptors (33, 34, 48). Chronic antidepressant exposure has indeed been shown to enhance 5-HT7-mediated cAMP formation in cortical astrocytes (51), and unmasking of 5-HT4 receptor-mediated excitatory responses together with decreased responsiveness of dominant inhibitory 5-HT1A receptors has recently been reported following chronic corticosterone treatment (52). Further studies with selective 5-HT7 receptor agents will be needed to prove this possibility in our model.
Interestingly, overuse or misuse of sumatriptan does occur in migraine patients (53–55). Approximately 4% of patients take the drug daily or more than 10 times each week while approximately 20% of patients use sumatriptan more than 10 times each month (54). While 5-HT is clearly involved in migraine pathogenesis, its exact role is still unclear. Should activation of the migraine generator in the brain-stem be a triggering factor (56, 57) in some migraine patients, one might predict that an increased 5-HT synthesis rate as a result of chronic sumatriptan might have a prophylactic effect. That is, putatively low baseline levels of serotonin production might be normalized and stressors might no longer be as readily able to elicit attacks. In at least one study, naratriptan, used daily, has been found to reduce the frequency and severity of daily headache (58). However, chronic sumatriptan use can also worsen headaches (53), and it is not excluded that this effect might be due to increased 5-HT neurotransmission.
In conclusion, the present findings indicate that sumatriptan and zolmitriptan, depending on their concentrations, can acutely and/or chronically affect central 5-HT synthesis rate and thereafter 5-HT neurotransmission, effects that might potentially be related to their anti-migraine activity.
Footnotes
Acknowledgements
The authors are grateful to GlaxoSmithKline (UK and Canada) and AstraZeneca (Canada) for their generous supply of sumatriptan and zolmitriptan, respectively. This study was supported by grants from the Heart & Stroke Foundation of Québec (E.H.) and NIH RO1-NS29629 (M.D.). The authors thank Ms L. Michel for preparation of the final manuscript.
