Abstract
Both calcitonin gene-related peptide (CGRP) and nitric oxide (NO) are potent vasodilators that have been shown to induce headache in migraine patients. Their antagonists are effective in the treatment of migraine attacks. In the present study, we hypothesize that vasodilation induced by the NO donor glyceryltrinitrate (GTN) or by CGRP is partially mediated via large conductance calcium-activated potassium (BKCa) channels. The effects of the BKCa channel selective inhibitor iberiotoxin on dural and pial vasodilation induced by CGRP, GTN and endogenously released CGRP by transcranial electrical stimulation (TES) were examined. Iberiotoxin significantly attenuated GTN-induced dural and pial artery dilation
Introduction
Our previous human studies have demonstrated that nitric oxide (NO) and calcitonin gene-related peptide (CGRP) are likely to be involved in migraine pathogenesis (1–4). It has been suggested that the interaction of NO and CGRP is implicated in the development of vascular headaches (5, 6). Both substances are endogenous vasodilators that act via different mechanisms. CGRP is known to cause increased levels of intracellular cAMP, whereas NO stimulates guanylyl cyclase with a subsequent increase in cGMP levels. Inherited dysfunction of voltage gated Ca2+ and Na+ channels and Na+/K+ pump subunits, as well as mutation in neuronal voltage gated Na+ channels, can cause familial hemiplegic migraine (7), and it has been suggested that the common types of migraine may also be caused by channel dysfunction (8).
Whether channels other than Ca2+ and Na+ are involved in mechanisms responsible for migraine is still unknown. Potassium (K+) channels are widely distributed and participate in the regulation of cerebral vascular tone (9). Furthermore, ATP-sensitive K+ channel (KATP) openers are potent vasodilators (10) and may induce headache in humans (11, 12). Large conductance calcium-activated K+ (BKCa) channels are thought to play a critical role in the regulation of cerebral artery diameter (13). These data indicate that K+ channels may be relevant targets in headache research.
It has been suggested that BKCa channels may be activated by protein kinase A (PKA) and protein kinase G (PKG) via the respective cAMP and cGMP pathways in patch-clamp experiments (14, 15). Furthermore, BKCa channels modulate the vasodilator response to both exogenous nitrovasodilators and endogenous receptor-mediated NO release in isolated arteries (16, 17).
We focused on the effect of BKCa channels on NO- and CGRP-induced vasodilation within the dural and pial vessels, because these molecules play a causal role in migraine (1–4).
In order to study the interaction of BKCa channels with CGRP and NO and to compare responses in pial and dural arteries, a rat genuine closed cranial window model was used in combination with studies of isolated middle meningeal (MMA) and middle cerebral (MCA) arteries. The genuine closed cranial model allows a direct measurement of dural and pial artery diameter and local cortical cerebral blood flow by laser Doppler flowmetry (LCBFFlux) in anaesthetized animals. Furthermore, it provides the opportunity to investigate the craniovascular effects of infused vasoactive substances and endogenous CGRP after electrical stimulation (18, 19).
The aim was to: (i) confirm the vasodilator effects
Materials and methods
In vivo studies
Surgical preparation
This study complied with The Danish Animal Experimentation Inspectorate (file: 2001/561-390) on the care and use of animals. All experiments were conducted using the closed cranial window technique as previously described by Williamson (18). Male Sprague—Dawley rats (300–400 g) were anaesthetized throughout the experiments with pentobarbital (Mebumal®; The Danish Hospital Pharmacy, Glostrup, Denmark). Anaesthesia was induced by 60 mg/kg i.p. and throughout the experiments anaesthesia was maintained by i.v. infusion of 20 mg/kg per hour pentobarbital. The depth of anaesthesia was judged by suppression of the hind paw withdrawal reflex. The animals were mechanically ventilated. The respirator delivered 3–3.5 ml/stroke of O2 30% and N2O 70%, at a ventilation rate of 60–65 strokes/min (Rodent Ventilator; UGO BASILE, Comerio, Italy). In the genuine closed cranial window many combinations of anaesthetics were tried by Petersen and colleagues, including inhalation anaesthetics, before deciding on the combination of Mebumal (pentobarbital) and N2O. It has been concluded that this combination had the least effect on the cerebral blood flow and the cerebral arteries during long-term observation (19). The femoral arteries and veins were cannulated bilaterally by catheters (0.40 mm i.d.) (Portex®; Astratech, Taastrup, Denmark) for administration of test substances, blood sampling and measurement of MABP and arterial blood gases (ABG) (Transducer TCM4-7; World Precision Instruments, Sarasota, FL, USA). The animals were placed in a stereotaxic frame, the skull exposed and the parietal bone thinned by drilling with a saline-cooled dental drill until the dural and pial arteries were clearly visible through the intact skull. Before placing the video camera, warm (37 °C) mineral oil was applied.
Intravital microscopy
The vessels were imaged by a Sony® video camera with microscope (Sony® DSP digital camera, MS 50 objective; Tokyo, Japan) and the real-time image was displayed on a television screen. Dural and pial artery diameters were continuously measured using a Video Dimension Analyser (VDA; Living Systems Instrumentation®, Burlington VT, USA). Data were collected and analysed by data acquisition and analysis software (Perisoft version 1; Perimed Systems®, Järfälla, Sweden). LCBFFlux (Perimed Periflux® 4001; Perimed AB) was measured by a probe 90° bent (Perimed 410, fibre separation 0.25 mm; Perimed AB) held by a micromanipulator. The probe was placed free of the bone but in touch with mineral oil, which enhances the accuracy of the LCBFFlux measurement (20). The probe was placed, using a micromanipulator, under visual control on the surface of the cranial window with no or few observable vessels.
Blood pressure was measured continuously by connecting the catheter inserted in the femoral artery to a transducer (Transducer TCM4-7; World Precision Instruments). Blood gases were measured at the beginning and end of each experiment (ABL520; Radiometer, Br⊘nsh⊘j, Denmark). Because changes in PaCO2, PaO2 and pH can affect the diameter of pial and dural artery as well as LCBFFlux, these values were kept within normal limits (19). Body temperature was controlled by a heating blanket and kept at 37 ± 0.5 °C. (Letica® Scientific Instruments, Barcelona, Spain).
Experimental protocols
In GTN dose–effective studies we have found that a bolus dose of 20 µg/kg caused maximal dilation of dural and pial arteries with a fall in MABP of up to 35 mmHg in anaesthetized rats (not published).
After a stabilization period of about 1–1.5 h, baseline values were recorded and a blood sample taken to determine ABG. First, bolus CGRP (0.3 µg/kg), GTN (20 µg/kg) or TES was given. After a wash-out period of 10–12 min, iberiotoxin infusion 0.1 mg kg−1 was given in a volume of 1 ml over 5 min followed by bolus CGRP (0.3 µg/kg), GTN (20 µg/kg) or TES.
In vitro studies
Male Sprague–Dawley rats (300–380 g; Denmark) were exanguinated during CO2 anaesthesia. The brain was removed and the first branch of the MMAs and MCAs were dissected out using an operation microscope. Each vessel was cut into 1–2 mm long circular segments and placed in an ice-cold buffer solution gassed with 5% CO2 in O2. The composition of the buffer was (m
Data analysis
For
All data are presented as mean ± SEM. Statistical significance was set at
For
Drugs
Iberiotoxin were obtained from Tocris Cookson Inc. (Bristol, UK). Final concentrations were made using normal saline. Rat αCGRP was obtained from Neosystem (Strasbourg, France). GTN was obtained from The Danish Hospital Pharmacy, Glostrup, Denmark (Glycerylnitrat®). Stock solutions of the drugs were kept frozen in small aliquots and diluted in isotonic saline (0.9%) immediately before use.
Results
The effect of NO donor GTN and inhibition by iberiotoxin in the genuine closed cranial window model
An infusion of GTN (20 µg/kg) increased dural artery diameter by 100 ± 12% and pial artery diameter by 43 ± 5% (
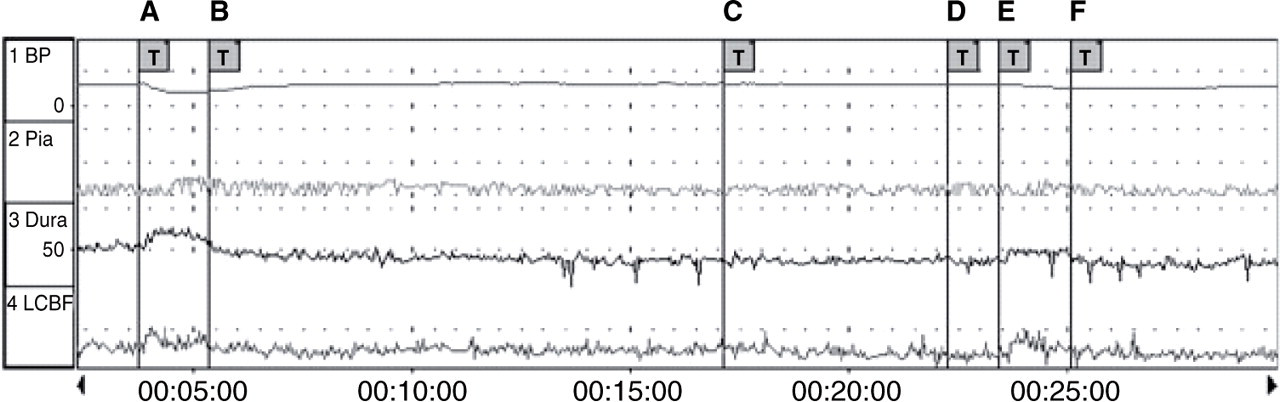
Trace showing the effect of glyceryltrinitrate (GTN) (20 µg/kg) and inhibition by iberiotoxin (0.1 mg/kg) on mean arterial blood pressure, dural and pial artery diameter and local cortical cerebral blood flow using the genuine closed cranial window model. A, GTN (20 µg/kg) bolus; B, pause; C, iberiotoxin (0.1 mg/kg) infusion over 5 min; D, pause; E, GTN (20 µg/kg) bolus; F, pause.

Effect of glyceryltrinitrate (GTN) (20 µg/kg) on (a) dural, (b) pial artery diameter, (c) local cortical cerebral blood flow (LCBFFlux) and (d) mean arterial blood pressure (MABP) in the absence and presence of iberiotoxin (0.1 mg/kg). Values given as mean ± SEM,
Effect of CGRP (0.3 µg/kg), GTN (20 µg/kg) and TES on dural/pial artery diameter, LCBFFlux and MABP


Values given as mean ± SEM. Statistical analysis comparing dura vs. pia responses in the absence and presence of iberiotoxin to calcitonin gene-related peptide (CGRP), glyceryltrinitrate (GTN) and transcranial electrical stimulation (TES) performed by non-parametric Wilcoxon test: NS,
An infusion of iberiotoxin (0.1 mg/kg) over 5 min had no effect on MABP, LCBFFlux or dural/pial artery diameter (
The effect of CGRP and inhibition by iberiotoxin in the genuine closed cranial window model
An infusion of CGRP (0.3 µg/kg) increased dural artery diameter by 81 ± 16% and pial artery diameter by 12 ± 3% (
The effect of TES and inhibition by iberiotoxin in the genuine closed cranial window model
TES increased dural artery diameter by 124 ± 19% and pial artery diameter by 71 ± 36% (
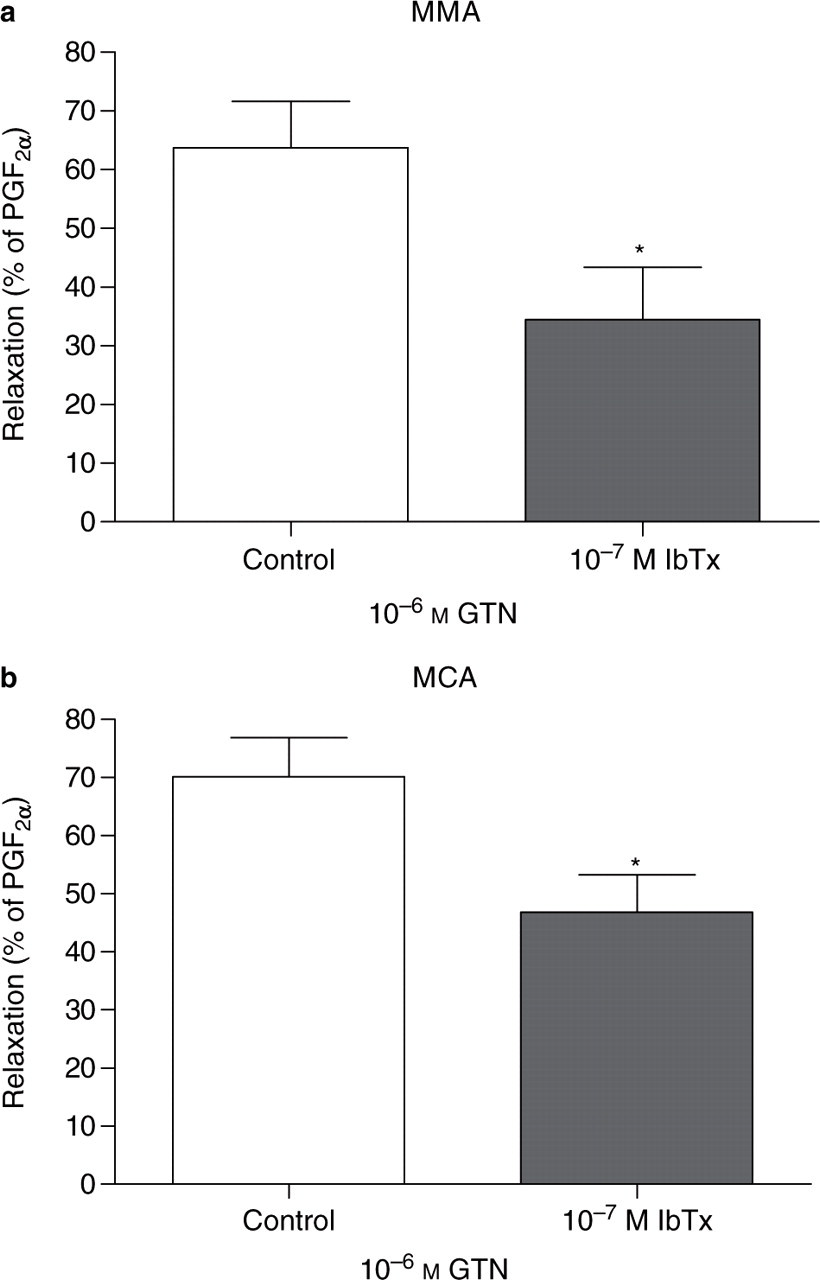
The effect of iberiotoxin on CGRP- and GTN-induced relaxation in isolated dural and cerebral arteries in vitro
When given alone, 10−7
Discussion
The main finding of the present study is that the BKCa channel selective blocker iberiotoxin can attenuate GTN- but not CGRP-induced dural and pial artery dilation
Effect of GTN and inhibition by iberiotoxin
This study has shown that bolus GTN decreases systemic blood pressure and dilates dural and pial arteries
The effect of CGRP and inhibition by iberiotoxin
In line with several other investigations, we have shown that i.v. administration of CGRP decreases systemic blood pressure and dilates dural and pial arteries
Our results are in agreement with several studies that have examined the effect of CGRP on cerebral arteries
The effect of electrical stimulation and inhibition by iberiotoxin
TES caused dilation of dural and pial vessels without affecting MABP. In our experiments, iberiotoxin could not attenuate these effects. It has been suggested that activation of the trigeminal ganglion by TES primarily releases CGRP (34, 35). This is further supported by the inhibitory effect of CGRP antagonists on TES-mediated dilation of dural and pial arteries (24). Thus, iberiotoxin failed to attenuate dural/pial vasodilation induced by exogenous CGRP and CGRP evoked by TES.
Possible role of BKCa channels in migraine pathogenesis
Within the cranium only a few structures, such as nociceptive afferents at meningeal blood vessels and at the large cerebral arteries, are able to generate nociceptive signals (36). When these vessels are stimulated experimentally during brain surgery, patients experience a throbbing unilateral migraine-like pain (36). Experimental and clinical data suggest that the trigeminovascular system of large dural vessels, rather than pial vessels, is involved in the pathogenesis of headache (36–40). CGRP plays a pivotal role in migraine, as infusion of CGRP can trigger a migraine attack in migraineurs (3) and the CGRP receptor antagonist BIBN 4096 BS is effective in the treatment of acute attacks of migraine (4). In a similar way, NO also seems to be involved in the generation of severe vascular headaches, because administration of the NO donor GTN causes migraine headaches in migraineurs (1) and the nitric oxide synthase inhibitor L-NMMA is effective in the treatment of the acute migraine attack (2). It has been suggested that the activation of the trigeminocerebrovascular system by NO involves interaction with CGRP, thereby causing the dural blood vessel dilation during migraine (6). Our results are in agreement with a previous study, which suggested that activation of BKCa channels may contribute to NO-induced vasodilation in the heart (25).
In summary, our experiments confirm that NO produces marked dilation of meningeal blood vessels, which is believed to produce migraine headache in the clinic. Thus, BKCa channels may be involved in migraine pathogenesis, since inhibition of the BKCa channels within cranial vessels significantly reduces NO (GTN)-induced dural vasodilation.
Acknowledgement
This study was supported by The Else Torps Legat, The Novo Nordisk foundation and The Lundbeck foundation.
