Abstract
Objective
To develop evidence-based clinical practice guidelines for non-invasive neuromodulation devices in acute and preventive migraine treatment.
Methods
A systematic review was conducted across six databases from 1946 to April 2025. Randomized controlled trials evaluating Food and Drug Administration-cleared or Conformité Européenne (CE)-marked non-invasive neuromodulation devices were included. The quality of evidence was assessed using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) methodology, and recommendations were developed through consensus following GRADE Evidence-to-Decision frameworks. The working group comprised 15 senior members and six junior members.
Results
From 1536 initial records, 15 studies met the inclusion criteria and were finally used to develop evidence-based recommendations. Evidence quality ranged from very low to moderate. Weak recommendations were issued for SAVI Dual, Cefaly, Relivion, and Nerivio in the treatment of acute migraine attacks, and for gammaCore Sapphire, Cefaly, and Nerivio in the preventive migraine treatment. Other cleared devices received no recommendations or have no eligible studies for the GRADE assessment. The primary limitations across studies included imprecision due to small sample sizes and various methodological concerns. Additionally, expert consensus recommendations were developed for devices and clinical scenarios not adequately covered by randomized controlled trials, including potential applications in pediatric populations, vestibular migraine, chronic migraine, menstrual migraine, and medication overuse headache.
Conclusion
Non-invasive neuromodulation devices offer promising alternatives to drug treatment for migraine management. These devices are safe and generally well tolerated and devoid of drug interactions. While current evidence quality varies, ongoing research and technological advancements show encouraging potential. Future studies should adhere to International Headache Society guidelines for neuromodulation device trials, address proper sham controls and blinding assessment, and account for patient adherence challenges in device use. Expanded insurance coverage would enhance cost-effectiveness and device accessibility. These guidelines provide a framework for clinical decision-making while highlighting areas requiring further research.
Keywords
Introduction
Non-invasive neuromodulation represents an emerging treatment option for headache disorders. The International Neuromodulation Society defines therapeutic neuromodulation as “the alteration of nerve activity through targeted delivery of a stimulus, such as electrical stimulation or chemical agents, to specific neurological sites in the body” (1). Unlike invasive devices that require implantation, non-invasive neuromodulation can be applied topically via various modalities, including electrical stimulation, magnetic field, temperature change, and vibration. Compared to invasive neuromodulation, this non-invasive approach offers minimal side effects, eliminates surgical risks, provides greater accessibility to treat migraine early, and allows treatment discontinuation without permanent consequences. Patients may choose non-invasive neuromodulation for acute or preventive migraine treatment based on personal preference or when pharmacological therapies prove ineffective, have adverse effects (AEs), or are contraindicated. These devices can also serve as complementary treatments alongside pharmacological interventions, potentially enhancing efficacy or reducing acute medication use. While neuromodulation devices rarely interact with medications or face contraindications from underlying medical conditions, challenges may arise from technical complexity, cosmetic concerns, or financial constraints (2,3).
Currently, six non-invasive neuromodulation devices have received Food and Drug Administration (FDA) clearance in the United States. At the same time, eight have obtained CE (Conformité Européenne) markings in Europe for treating various headache disorders based on different modes of action (Table 1). In a consensus statement, the American Headache Society (AHS) suggests that “non-invasive neuromodulation devices may be used as monotherapy or adjunctive therapy for acute and preventive migraine treatment.”(17) Recently, the International Headache Society (IHS) issued a guideline for clinical trials on using neuromodulation devices to treat migraine and cluster headache (18,19). However, the historical absence of controlled trial guidelines has led to considerable heterogeneity in trial design and outcomes reporting, complicating evidence interpretation. While there are a few systematic reviews on neuromodulation in migraine treatment (20,21), several new studies have been published in the past few years that were not captured in the prior reviews. In this context, summative evidence that pools data from existing clinical trials is needed to guide clinical decision-making and to appraise the existing evidence.
Summary of FDA-cleared and CE-marked non-invasive neuromodulation devices.

FDA cleared. bCE marked. cEpisodic migraine only. dChronic migraine only. eRegimen not available on website but extracted from Hoffmann et al. (22).
To address these challenges, the IHS established a special working group with two primary objectives: first, to systematically evaluate and grade existing studies on the efficacy of FDA-cleared or CE-marked non-invasive neuromodulation devices; and second, to develop evidence-based recommendations and expert consensus suggestions to guide the use of these devices in the treatment of migraine in adults, adolescents, and, where applicable, children. This comprehensive assessment aims to provide clear, actionable guidance for clinical decision-making among physicians and prescribers. Still, it is not intended to serve as the sole framework for insurance coverage determinations.
Methods
The study was registered on PROSPERO (CRD42023407883) and can be found on the PROSPERO website. The IHS special working group comprised 15 senior members and six junior members. Members were selected in agreement with the IHS Board based on their expertise in the field and their consideration of geographic representativeness.
Formulation of clinical questions
Two clinical questions were formulated for each device using the PICO (Population, Intervention, Comparison, and Outcomes) framework (Table 2) and the format endorsed in the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Handbook (gdt.gradepro.org):
− Is [device] superior to sham for the treatment of acute migraine attacks? − Is [device] superior to sham for preventive migraine treatment?
Inclusion criteria for study eligibility by PICO format.

FDA: Food and Drug Administration. CE: Conformité Européenne. MMD: monthly migraine days. MHD: monthly headache days. AMD: monthly acute medication use days. MIDAS: migraine disability assessment. HIT-6: headache impact test. RCT: randomized controlled trial.
Based on their importance for decision-making regarding the optimal management strategy. bIt required baseline and three months of post-treatment assessment. If three months’ data were not available, two months’ data were assessed.
Eligibility criteria
The eligibility criteria are detailed in Table 2. For the evidence assessment, we included only randomized controlled trials (RCTs) and published data from clinicaltrials.gov. While non-RCTs (e.g., open-label, post hoc studies) and RCTs with abstract-only publications were excluded from the primary analysis; these were considered during the expert suggestions process. Selected outcomes (based on the IHS controlled trial guidelines) that are important to patients for decision-making were included. The treatment duration and evaluation endpoint were selected based on the IHS controlled trial guideline (18).
We excluded studies that were: published in languages other than English, focused on populations with other primary or secondary headache disorders, evaluated behavioral interventions, limited to diagnostic procedures, dose titration, review articles, or non-peer-reviewed publications.
Literature review
A librarian performed a literature search in consultation with authors with content expertise (HY, SLO, LS). Published data were retrieved from six sources (OVID MEDLINE, OVID EMBASE, EBSCOHost CINAHL, Wiley Cochrane trials, clinicaltrials.gov, WHO ICTRP) from 1946 to April 1, 2025. The search protocol was verified using the Peer Review of Electronic Search Strategies (PRESS) peer review method (23).
The screening process was conducted in two distinct phases using the PICO Portal (www.picoportal.org). In the first phase, abstracts were screened to identify potentially eligible studies. In the second phase, full texts underwent manual review to confirm eligibility. Both phases of screening were completed independently by two reviewers. Six team members (ASP, DM, HY, J-TY, LS, MAA) worked in pairs to cross-validate the screening process, with any disagreements resolved through consensus discussions with senior team members (SLO, HY).
Data extraction
Data extraction was performed independently by two reviewers (DM, HY), with discrepancies resolved through consensus. Using standardized extraction tables, we collected comprehensive study details, including author information (first author's name and year of publication), complete citation, study characteristics (design, setting, and time period), migraine-specific information (number of participants, diagnostic criteria), methodological details (masking/blinding strategies), study parameters (sample size, treatment/sham type, duration of observations and treatments), population analysis type (intention-to-treat [ITT], modified ITT [mITT], per protocol [PP]), outcomes (primary endpoint or predetermined parameters as shown in Table 2), and safety data (e.g., AEs). All data were extracted as reported, with no imputation of missing values. See supplementary data for more details.
Quality of evidence
The GRADE methodology evaluates evidence quality through structured evidence tables, with initial ratings based on study design, modified by downgrading factors (study limitations, inconsistency, indirectness, imprecision, publication bias) and upgrading factors (large effect size, dose-response gradient, confounding). Using GradePro GDT (https://www.gradepro.org/), we assessed the certainty of the prespecified outcomes and summarized the findings. Effect sizes were calculated as odds ratio (OR) or mean difference (MD). The risk of bias was assessed using the ROB2 (Cochrane's Risk of Bias) assessment tool for RCTs. Six members (ASP, DM, FC, J-TY, LS, MAA) cross-checked each assessment result, and disagreements were resolved by consensus with senior members (HY, SLO). Certainty was downgraded for risk of bias based on ROB2 assessments (Cochrane's Risk of Bias) (24), inconsistency based on heterogeneity, and imprecision using two approaches: confidence intervals relative to minimally important differences and optimal information size requirements (25). Two reviewers independently evaluated GRADE tables, with six team members cross-checking the risk of bias assessments. All disagreements were resolved through consensus with senior members. The final evidence quality was rated as high, moderate, low, or very low for each critical outcome per the GRADE criteria.
GRADE assessment on imprecision
The precision assessment followed the GRADE guideline 34 (25). For dichotomous outcomes, certainty was downgraded to one level if the OR 95% confidence interval (OR 95% CI) crossed the minimally important difference (MID) of 1.2 (determined by group consensus) and two levels if the OR 95% CI upper/lower ratio exceeded 2.5. For continuous outcomes, the certainty assessment used two approaches. Under the confidence interval approach, certainty was downgraded one level if the 95% CI crossed the MID (set at 2 for 0-10 pain rating scale changes and 1.5 for monthly migraine day (MMD) or monthly headache day (MHD) changes, as determined by group consensus). Under the optimal information size (OIS) approach, assuming a group difference of 1, SD of 4, alpha of 0.05, and power of 0.8, the minimum sample size was set at 500. Certainty was downgraded one level for sample sizes between 150–250 (30–50% of OIS) and two levels for samples below 150 (<30% of OIS). We adopted the more conservative assessment when the two approaches yielded different results. Two reviewers (J-TY, HY) independently verified the GRADE tables, with disagreements resolved through consensus with senior members (SLO).
Meta-analysis
Meta-analyses were performed on outcomes reported in multiple studies using the meta and metafor packages in R statistical software (26). Heterogeneity was assessed using Cochran's Q test, with P < 0.1 considered statistically significant. Analyses were conducted using both fixed and random effects models, with results from the random effects model adopted when significant heterogeneity was detected.
Evidence-based recommendations
The GRADE approach provides a systematic framework for developing evidence-based recommendations through a structured and transparent process (27–29). Expert panels follow a two-step process: first, we evaluated four key domains: the balance between desirable and undesirable outcomes based on available evidence across multiple outcomes important for optimal management, overall certainty/quality of that evidence on the desirable vs. undesirable effects, confidence in values and preferences (e.g., cost-effectiveness, usage feasibility, device acceptability), and resource/cost considerations. Second, through consensus discussions, we determined both the direction (for/against an intervention) and the strength (strong/weak) of recommendations. “Strong recommendations” indicate high confidence that benefits clearly outweigh harms for the majority of patients. A strong recommendation implies that most or all individuals will be best served by the recommended course of action. “Weak (conditional) recommendations” imply that the desirable effects probably outweigh the undesirable effects, but not all individuals will be best served by the recommended course of action. “No recommendation” is made when the confidence in effect estimates is so low that the panel feels that a recommendation is too speculative, the trade-offs are so closely balanced that the panel has great difficulty deciding on the direction of a recommendation, or the values and preferences and resource implications are not known or too variable.
Expert consensus suggestions
The expert consensus suggestions were formulated to complement the evidence-based recommendations by addressing clinical scenarios and patient populations inadequately represented in eligible RCTs. These suggestions are intentionally framed as options rather than prescriptive recommendations. No formal level of evidence or strength of suggestion was assigned, reflecting the inherent limitations of the supporting data. These suggestions were informed by additional sources, including ineligible RCTs, open-label studies, post-marketing observational studies, case series, real-world evidence, post-hoc analyses, and conference abstracts. Each suggestion is accompanied by a clear rationale (see Online Supplementary Material) and supported by available data, even when they did not meet the criteria for formal GRADE assessment. Accordingly, these suggestions should be interpreted as considerations for clinical decision-making.
Results
From 1536 initial records, we screened 957 unique records and retrieved 311 full texts for detailed assessment (Figure 1). We excluded 197 records (10 ongoing clinical trials, 92 duplicates or have manuscripts, and 95 studies with ineligible PICO characteristics). Of the remaining 114 full texts reviewed, we found 82 non-RCTs (27 abstracts, 55 publications) and 32 RCTs (six abstracts, 24 publications, two clinicaltrial.gov entries). After excluding six abstracts and studies focused on non-migraine diagnoses (four trigeminal autonomic cephalalgia, one tension-type headache), ineligible study type (three), ineligible intervention (one), and ineligible outcomes (two), 15 studies met the inclusion criteria for evidence assessment. The PICO questions are sorted by device clearance date for migraine treatment. Risk of bias assessment showed that most studies were generally low risk, although a few exhibited some or high concerns (Figure 2).

PRISMA flow diagram of literature search.

Risk of bias summary for each included study.
SAVI Dual delivers a single pulse of transcranial magnetic stimulation (Table 1). In adult patients living with episodic migraine (EM) with aura, SAVI Dual (2 pulses, 30 s apart) is superior to inactive sham for the treatment of acute migraine attacks. A single RCT (including patients living with EM with aura; treated ≥1 attack, 1hr within aura onset) showed the benefits of SAVI Dual over non-active sham, considering the outcomes of pain freedom at 2hr (primary endpoint; OR 2.28, 95%CI 1.15–4.52) and sustained pain freedom at 24hr (OR 2.20, 95%CI 1.03-4.70) (30). The AE rate was comparable to the sham group. The quality of the evidence was low due to very serious imprecision (Table 3). Overall, the desirable effects probably outweigh the undesirable effects based on one RCT with low-quality evidence. The high cost and device accessibility remain problematic.
GRADE evidence table for SAVI dual vs sham in the treatment of acute migraine attacks (mITT).

Inactive sham. bPrimary endpoint. cCI upper/lower ratio > 2.5.
Evidence-based recommendation
The panel conditionally recommends SAVI Dual for the treatment of acute migraine attacks in adult patients with EM with aura.
Strength of the recommendation: weak in favor (↑) Quality of the evidence: low (⊕⊕⊝⊝)
Expert consensus suggestion
SAVI Dual can be an option for the treatment of acute migraine attacks in subjects with EM without aura or chronic migraine (CM) with/without aura (31).
No RCT met the eligibility criteria for this PICO question.
Evidence-based recommendation
There was no RCT to assess the use of SAVI Dual for preventive migraine treatment.
Strength of the recommendation: N/A
Expert consensus suggestion
SAVI Dual can be an option for preventive treatment in subjects with migraine, with or without medication overuse headache (MOH), in adults, adolescents, and pregnant patients (31–36).
Cefaly delivers transcutaneous electrical supraorbital trigeminal nerve stimulation (Table 1). In adult subjects with EM with and without aura, Cefaly is superior to active sham for the treatment of acute migraine attacks. Two RCTs (ACME: 1hr stimulation for headache lasting >3hr; TEAM: 2hr stimulation, within 4hr of migraine onset) showed a moderate effect (OR 1.62, 95%CI 1.09-2.39) for pain freedom at 2hr in the ITT population (primary endpoint for TEAM study) of patients with EM (37,38), although pain freedom at 2hr was not significant in the ACME study (Figure 3). Other endpoints included pain relief at 2hr (OR 1.85, 95%CI 1.30-2.64), sustained pain freedom at 24hr (OR 1.66, 95%CI 1.08-2.54), and pain intensity difference at 2hr (MD 1.02, 95%CI 1.82-0.22). The AEs were infrequent and minor. There were some risk-of-bias concerns in the ACME study, and its primary endpoint was pain intensity change at 1hr, but not the recommended pain freedom at 2hr. Blinding analysis was also not reported in the ACME study. The quality of evidence for the different outcomes ranged from low to high, and overall, it was judged as moderate due to serious imprecision (Table 4). Overall, the desirable effects outweigh the undesirable effects, based on two RCTs of moderate-quality evidence (37,38), although a longer stimulation duration may be needed. Cefaly is currently available without prescription in many countries.

Forest plots of meta-analysis of trials assessing the efficacy of Cefaly in the treatment of acute migraine attacks.
GRADE evidence table for Cefaly vs sham in the treatment of acute migraine attacks (ITT population).

Active sham. bPrimary endpoint only for the TEAM study. cCI crosses the MID of 1.2. dN less than 150.
Evidence-based recommendation
The panel conditionally recommends the use of Cefaly for the treatment of acute migraine attacks in adult subjects with EM.
Strength of the recommendation: weak in favor (↑) Quality of the evidence: moderate (⊕⊕⊕⊝)
Expert consensus suggestions
Cefaly can be an option for the acute treatment of pediatric migraine and vestibular migraine (39,40).
Cefaly can be an option for the treatment of acute migraine attacks in adults with CM (41).
In adult subjects with EM with and without aura, Cefaly (20 min stimulation daily) is probably superior to active sham for preventive migraine treatment. The single RCT assessed did not meet one of its primary endpoints (MMD reduction; MD 1.74, 95%CI −0.32-3.8) in the ITT population in subjects with EM (42). The 50% responder rate, the second primary endpoint, favored the Cefaly device over the sham (OR 4.49, 95%CI 1.28-15.72), but this was not true for MHD reduction (MD 2.28, 95%CI −0.01-4.57). The study was not powered for co-primary endpoints. No blinding analysis was done, and the study was not registered. The AEs were usually mild. The quality of the evidence was very low due to very serious imprecision (Table 5). Overall, the desired effects probably outweigh the undesired effects based on one RCT of very low-quality evidence, half of the primary endpoints being positive, and positive secondary endpoints (42). Cefaly is currently available without prescription in many countries.
GRADE evidence table for Cefaly vs sham in preventive migraine treatment (ITT population)

Active sham. bCo-primary endpoint. cN less than 150.
Evidence-based recommendation
The panel conditionally recommends the use of Cefaly for preventive migraine treatment in adult subjects with EM.
Strength of the recommendation: weak in favor (↑) Quality of the evidence: very low (⊕⊝⊝⊝)
Expert consensus suggestion
Cefaly can be an option for preventive treatment of CM and MOH in the pediatric and adult populations (43–48).
GammaCore Sapphire delivers transcutaneous vagus nerve stimulation in the neck (Table 1). In adult subjects with EM with/without aura, gammaCore Sapphire (2 min stimulation on each side) may not be superior to active sham for the treatment of acute migraine attacks. The RCT (subjects with EM, treated ≥1 attack, 20 min within migraine onset, including mild headache) did not meet its primary endpoint (pain freedom for the first-treated attack at 2hr; 30.4% vs. 19.7%, p = 0.067), although significant benefits were seen at 30/60 min (pain freedom) and 2hr (pain relief) (49). For all treated attacks, there were significant differences at 2hr (pain freedom; 22.9% vs. 14.8%, p = 0.026) (50). Most AEs were mild-moderate, with a few subjects withdrawing due to AEs. The active sham might have created some treatment effects, although blinding was successful. The quality of the evidence was low due to very serious imprecision (Table 6). Overall, the desirable effects possibly outweigh the undesirable effects based on one RCT with low-quality evidence (49,50), but remain to be properly studied, as current data showed only positive secondary but not primary endpoints. The high cost and device accessibility remain problematic, although functionally similar non-prescription non-invasive vagus nerve stimulators are sold over-the-counter as wellness products.
GRADE evidence table for gammaCore Sapphire vs sham in the treatment of acute migraine attacks (ITT population).

Active sham. bPrimary endpoint. cCI upper/lower ratio >2.5. dUnit of measure is attacks.
Evidence-based recommendation
There were no solid efficacy data to recommend the use of gammaCore Sapphire for the treatment of acute migraine attacks.
Strength of the recommendation: no recommendation Quality of the evidence: low (⊕⊕⊝⊝)
Expert consensus suggestions
The gammaCore Sapphire can be an option for the treatment of acute migraine attacks, and associated vertigo and nystagmus in subjects with vestibular migraine (49–52).
The gammaCore Sapphire can be an option for the treatment of acute migraine attacks in adolescents (53).
The gammaCore Sapphire can be an option for the treatment of acute migraine attacks in subjects with CM, although perhaps to a lesser degree than in EM and probably ineffective in refractory CM (54,55).
The gammaCore Sapphire can be an option for the treatment of acute migraine attacks in subjects with MOH (56,57).
In adult subjects with migraine, gammaCore Sapphire (2 min stimulations twice, three times daily) is superior to sham for preventive migraine treatment from two RCTs (PREMIUM, PREMIUM II) (58,59). The EVENT study was excluded due to the lack of proper endpoints at three months (60). The treatment effects from two RCTs, when defined as a reduction in mean MMD (primary endpoint for PREMIUM II), were small but significant (MD 0.72, 95%CI 0.31-1.14) in the mITT population (≥67% treatment adherence) in subjects with EM (PREMIUM) and EM/CM (PREMIUM II) (Figure 4) (58,59). Other endpoints include a change in MHD (MD 0.91, 95%CI 0.43-1.40), 50% responder rate (OR 1.73, 95%CI 1.11-2.69), and change in AMD (MD 0.81, 95%CI 0.02-1.61). While PREMIUM I but not II used an active sham, both studies achieved successful blinding. AEs were generally mild, with no serious adverse events (SAEs) in either study. The quality of the evidence was moderate due to serious imprecision (Table 7). Overall, the desirable effects likely outweigh the undesirable effects based on two RCTs with moderate-quality evidence (58,59). However, the beneficial effect of the gammaCore Sapphire may depend on the degree of adherence to the treatment, which is likely to be inferior with real-world use compared to the trial setting. The device cost and accessibility remain problematic.

Forest plots of meta-analysis of trials assessing the efficacy of gammaCore in migraine prevention.
GRADE evidence table for gammaCore vs sham in preventive migraine treatment (mITT population).
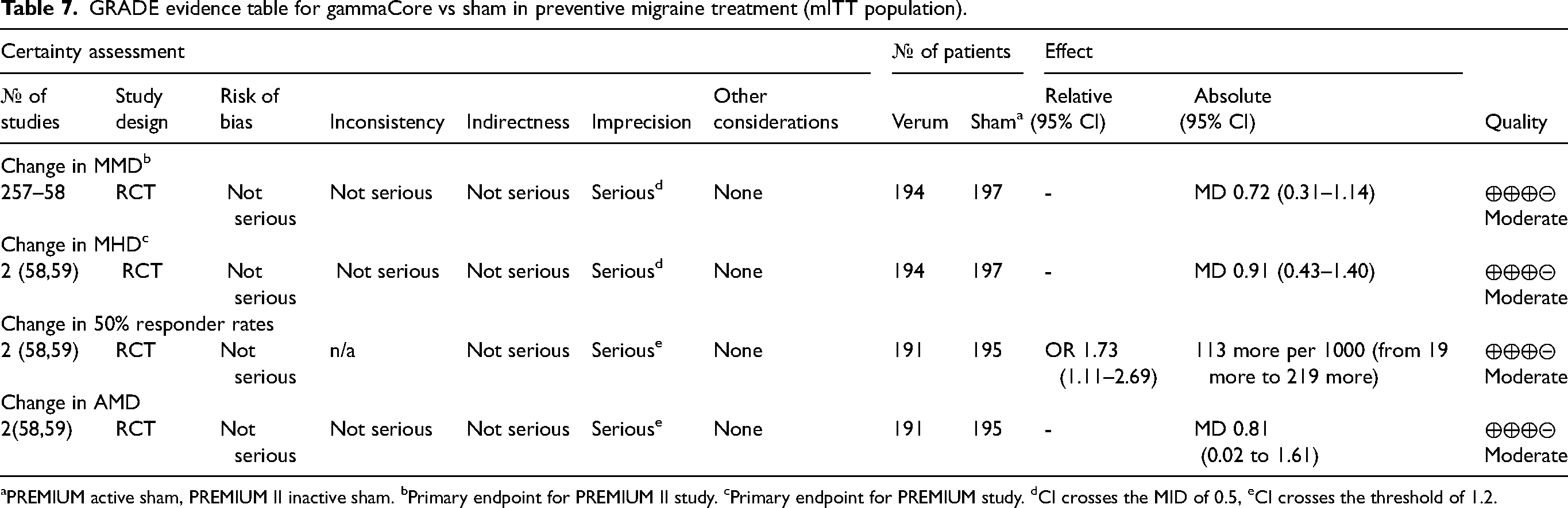
PREMIUM active sham, PREMIUM II inactive sham. bPrimary endpoint for PREMIUM II study. cPrimary endpoint for PREMIUM study. dCI crosses the MID of 0.5, eCI crosses the threshold of 1.2.
Evidence-based recommendation
The panel conditionally recommends the use of gammaCore Sapphire for preventive treatment of EM/CM in adult subjects with migraine, especially if they can maintain treatment adherence.
Strength of the recommendation: weak in favor (↑) Quality of the evidence: moderate (⊕⊕⊕⊝)
Expert consensus suggestions
The gammaCore Sapphire can be an option for preventive treatment of menstrual and menstrually-related migraine when used perimenstrually (61).
The gammaCore Sapphire can be an option for preventive treatment in adult subjects with CM (60).
HeadaTerm is shown to be substantially equivalent to Cefaly PREVENTIVE (Table 1). In the one RCT reviewed (62), there were no eligible outcome data to assess the use of HeadaTerm in the treatment of acute migraine attacks.
Evidence-based recommendation
No recommendation.
Strength of the recommendation: N/A
Expert consensus suggestion
1: HeadaTerm can be an option for the treatment of acute migraine attacks in adult subjects with EM (62).
No RCT met the eligibility criteria for this PICO question.
Evidence-based recommendation
No recommendation.
Strength of the recommendation: N/A
Expert consensus suggestion
Based on a predicate device (Cefaly PREVENTIVE), HeadaTerm can be an option for preventive migraine treatment.
Nerivio delivers transcutaneous electrical stimulation to the upper arm skin to activate endogenous analgesia (Table 1). In adult subjects with EM with or without aura, Nerivio may be superior to an active sham for the treatment of acute migraine attacks based on two RCTs (10,63). A parameter assessment exploratory study by Yarnitsky et al. was excluded because the stimulation parameters differed from the currently cleared device (64). The 2017 RCT (NCT03076515, data not published; stimulation for 25 min at the migraine onset) failed its primary endpoint (pain relief at 2hr) and was terminated early due to lack of adherence (63). The 2019 RCT (30/45-min stimulation, 1hr within symptom onset) showed significant improvement in pain relief at 2hr (primary endpoint) and pain freedom at 2hr in the mITT population (Figure 5) (10). Device-related AEs were primarily topical issues, with no SAEs. Neither study used pain freedom at 2hr as the primary endpoint. Blinding analysis was not reported in the 2017 study, but was successful in the 2019 study. The quality of the evidence was very low due to a serious risk of bias for the 2017 study and very serious imprecision (Table 8). Overall, the desired effects probably outweigh the undesired effects based on two very low-quality studies, although a longer stimulation of 45 min was cleared as the recommended dosing (10,63). Insurance coverage is available for some payers in the US, but device accessibility remains problematic outside the US.

A forest plot of meta-analysis of trials assessing the efficacy of Nerivio in the treatment of acute migraine attacks.
GRADE evidence table for Nerivio vs sham in the treatment of acute migraine attacks (mITT population).

Active sham. bPrimary endpoint for both studies. cCI upper/lower ratio > 2.5.
Evidence-based recommendation
The panel conditionally recommends the use of Nerivio for the treatment of acute migraine attacks in adult subjects with EM.
Strength of the recommendation: weak in favor (↑) Quality of the evidence: very low (⊕⊝⊝⊝)
Expert consensus suggestions
Nerivio can be an option for the treatment of acute migraine attacks in pregnant women or patients with menstrually-related migraine (65,66).
Nerivio can be an option for the treatment of acute migraine attacks in pediatric patients (≥8 years old) (67–70).
Nerivio can be an option for the treatment of acute migraine attacks in adults with CM (71,72).
Nerivio can be combined with behavioral intervention (75).
Tepper et al. conducted a double-blind, sham-controlled RCT of Nerivio 45 min every other day for eight weeks for migraine prevention (73). Of 248 participants (130 CM), 179 (92 CM) qualified for the mITT analysis. Overall, Nerivio reduced MMD by 4.0 ± 4.0 compared to 1.3 ± 4.0 in the sham device group with a therapeutic gain of 2.7 MMD (95%CI 1.5-3.9, p< 0.001). A non-significant 0.8 (95%CI −1.0 to 2.5) and −1.1 (95%CI −5.0 to 2.9) of HIT-6 and MSQ scores were reported, respectively. Treatment adherence was high in this study. Nearly 90% of participants in the Nerivio (89.8%) and sham (89.2%) groups completed more than 75% of the treatments during the eight-week intervention. In a post hoc analysis, Blumenfeld et al. showed that both EM and CM patients benefited, with EM showing significance at two weeks and CM at six weeks (76). No device-related AE was reported in the Nerivio arm except for one in the sham group. The quality of the evidence was very low due to a very serious imprecision and an eight-week follow-up duration (Table 9). Overall, the desired effects probably outweigh the undesired effects based on one very low-quality study. Insurance coverage is available for some payers in the US, but device accessibility remains problematic outside the US.
GRADE evidence table for Nerivio vs sham in the preventive migraine treatment (mITT population).

Active sham. bPrimary endpoint. cN < 500. d95%CI upper/lower ratio >2.5. e95% CI crossed 1.5.
Evidence-based recommendation
The panel conditionally recommends the use of Nerivio for preventive treatment of EM/CM in adult subjects with migraine.
Strength of the recommendation: weak in favor (↑) Quality of the evidence: very low (⊕⊝⊝⊝)
Expert consensus suggestion
Nerivio can be an option for preventive migraine treatment in adolescents (77).
Relivion delivers external concurrent occipital and trigeminal neurostimulation (Table 1). In adult subjects with EM, Relivion (1-hr stimulation within 30–60 min from headache onset) is superior to active sham for the treatment of acute migraine attacks. Two RCTs showed the benefit on pain freedom at 2hr (OR 4.27, 95%CI 1.69-10.75), pain relief at 2hr (OR 2.81, 95%CI 1.06-7.49), and sustained pain freedom at 24hr (OR 6.08, 95%CI 2.06-17.94) in the mITT population, mostly patients with EM (Figure 6) (78,79). Neither study used pain freedom at 2hr as the primary endpoint. AEs were infrequent and usually mild, but with a few early withdrawals due to not tolerating paresthesias arising from stimulation. The quality of the evidence was very low due to very serious imprecision and a serious risk of bias. (Table 10). Overall, the desirable effects possibly outweigh the undesirable effects based on two very low-quality studies (78,79). The device is currently not covered by insurance, and the unit cost is high; the device accessibility remains problematic outside the US.

A forest plot of meta-analysis of trials assessing the efficacy of Relivion in the treatment of acute migraine attacks.
GRADE evidence table for Relivion versus sham in the treatment of acute migraine attacks (mITT population).

Active sham. bPrimary endpoint for Tepper 2022 study only, but low recruitment. Daniel et al. (78) study used post-treatment 1hr pain intensity changes as the primary endpoint; data not included as not prespecified data. cCI upper/lower ratio > 2.5.
Evidence-based recommendation
The panel conditionally recommends the use of Relivion for the treatment of acute migraine attacks in adult patients with EM.
Strength of the recommendation: weak in favor (↑) Quality of the evidence: very low (⊕⊝⊝⊝)
Expert consensus suggestion
Relivion can be an option for the treatment of acute migraine attacks in adults with CM (80).
No RCT met the eligibility criteria for this PICO question.
Evidence-based recommendation
No recommendation.
Strength of the recommendation: N/A
Expert consensus suggestion
No RCT met the eligibility criteria for this PICO question.
Evidence-based recommendation
No recommendation.
Strength of the recommendation: N/A
Expert consensus suggestion
No eligible study for suggestion.
tVNS delivers transauricular electrical vagus nerve stimulation (Table 1). In adult subjects with CM, tVNS (4hr stimulation daily) was not superior to active sham for preventive migraine treatment. One RCT failed to meet the primary endpoint (MMD reduction; MD −2.6, 95%CI −5.58-0.38) in the ITT population in patients with CM (83). There was no change in AMD (MD −0.7, 95%CI −1.79-3.19) and no change in MIDAS score (MD −3.1, 95%CI −27.9-21.7), except for a change in HIT-6 score (MD 1.3, 95%CI 2.26-4.86). The quality of the evidence was very low due to some concerns in the randomization process (no information about whether the allocation sequence was random and whether the allocation sequence was concealed until participants were enrolled and assigned to interventions) and very serious imprecision (Table 11). Also, the active sham led to a significant reduction in the MHD compared to the active intervention; no blinding analysis was reported. Overall, the available data do not favor intervention based on the one RCT (83). Although tVNS is available over-the-counter and received CE marking, for migraine treatment, there is currently no evidence to support the use of tVNS for preventive migraine treatment, especially in subjects with CM. The optimal stimulation parameter/setting for migraine prevention remains to be studied.
GRADE evidence table for tVNS vs sham in preventive migraine treatment (ITT population).

Active sham. bPrimary endpoint. cN less than 150.
Evidence-based recommendation
There were no solid efficacy data to recommend the use of tVNS for preventive migraine treatment.
Strength of the recommendation: no recommendation Quality of the evidence: very low (⊕⊝⊝⊝)
Expert consensus suggestion
Transauricular VNS in general, not not limit to the exact tVNS device, may have a role in migraine prevention (13,84,85).
Ozilia delivers intranasal kinetic oscillation stimulation. (Table 1). In subjects with EM, Ozilia (15 min stimulation each nostril) is marginally superior to inactive sham for the treatment of acute migraine attacks. A single RCT demonstrated pain freedom at 2hr (not a primary endpoint; OR 11.00, 95%CI 1.16-103.9) and sustained pain freedom at 24hr (OR 8.80, 95%CI 0.93-83.4) in the PP population of adult subjects with EM (86). The study met the primary endpoint (change in pain intensity 15 min after stimulation completion; 45 min from stimulation onset). The acute use of Ozilia was generally well tolerated, but one patient experienced a vasovagal reaction. Blinding analysis was not reported. The quality of the evidence was very low due to some risk-of-bias concerns and very serious imprecision (Table 12). Overall, the desirable effects possibly outweigh the undesirable effects. Insurance companies currently do not cover the device; the cost and device accessibility can be issues. Since the device requires inserting a catheter into the nostril and attaching a catheter to a pump, the feasibility of use can be problematic, particularly in the acute setting, as it requires a health professional to administer.
GRADE evidence table for Ozilia vs sham in the treatment of acute migraine attacks (PP population).

< 150, CI upper/lower ratio > 2.5.
Evidence-based recommendation
There were no solid data to recommend Ozilia for the treatment of acute migraine attacks.
Strength of the recommendation: no recommendation. Quality of the evidence: very low (⊕⊝⊝⊝)
Expert consensus suggestion
No eligible study for suggestion.
In subjects with migraine, Ozilia (15 min stimulation of each nostril daily) is not superior to inactive sham for preventive migraine treatment based on a single RCT (NCT02243865, not published) (87). A recent study by Hoffman et al. was excluded as it did not have 12-week data (22). The included RCT investigated the preventive efficacy of Ozilia for patients with migraine (EM vs. CM not specified) (87). It did not meet the primary endpoint, as demonstrated by a non-significant MMD reduction (MD 0.2, 95%CI −1.27-1.67) in the PP population. Paresthesia and nasolacrimal secretion were common, but no SAE was reported. No blinding analysis was reported. The quality of the evidence was very low due to the high risk of bias and very serious imprecision (Table 13). Overall, the desirable effect may not outweigh the undesirable effect. Insurance companies currently do not cover the device; the cost and device accessibility can be issues. Since the device requires inserting a catheter into the nostril and attaching a catheter to a pump, the feasibility of use can be problematic.
GRADE evidence table for Ozilia vs sham in preventive migraine treatment (PP population).

aInactive sham. bPrimary endpoint. cN less than 150.
Evidence-based recommendation
There were no solid data to recommend Ozilia for preventive migraine treatment.
Strength of the recommendation: no recommendation Quality of the evidence: very low (⊕⊝⊝⊝)
Expert consensus suggestion
Ozilia can be an option for preventive migraine treatment in adults with CM (22).
Discussion
Based on our systematic evaluation of evidence from eligible RCTs, non-invasive neuromodulation devices demonstrate varying levels of efficacy for migraine treatment. The evidence assessment yielded several key findings (Table 14). For the treatment of acute migraine attacks, the panel conditionally recommends the use of Cefaly, SAVI Dual, Relivion, and Nerivio based on moderate to very low-quality evidence. For preventive migraine treatment, the panel conditionally recommends the use of gammaCore Sapphire, Cefaly, and Nerivio based on moderate to very low quality evidence. Insufficient evidence prevented us from making evidence-based recommendations for several devices, including SAVI Dual (preventive), gammaCoreSapphire (acute), HeadaTerm (acute/preventive), tVNS (acute/preventive), Relivion (preventive), and Ozilia (acute/preventive). To address these evidence gaps, the panel developed expert consensus suggestions (Table 15), since many of these devices may be viable options for specific patient populations, including those with CM, MOH, menstrual migraine, and pediatric migraine, broadening the potential therapeutic applications beyond contexts with formal evidence-based recommendations.
Summary of the evidence-based recommendations.

Recommendations were based on published studies eligible for GRADE evaluation. No eligible outcome/study for SAVI Dual (preventive), tVNS (acute), headaTerm (acute, preventive), and Relivion (preventive).
For episodic migraine with aura. bFor episodic migraine without aura. cFor chronic migraine.
Summary of the expert consensus suggestions.

Suggestions were based on expert consensus from published studies not eligible for GRADE evidence.
EM: episodic migraine. CM: chronic migraine. MOH: medication overuse headache.
We recognize that evaluating medical devices presents unique challenges beyond traditional evidence assessment frameworks. Device trials face inherent methodological constraints: implementing reliable sham controls proves difficult, recruiting adequate sample sizes poses significant challenges, and manufacturers operate under different financial and resource constraints than pharmaceutical companies. Furthermore, while regulatory pathways such as 510(k) clearance or CE-marking ensure basic safety standards, they may not fully validate clinical efficacy.
This complex landscape necessitated a balanced approach to formulate practical guidelines. While grounding our primary recommendations in systematically evaluating eligible RCTs using GRADE methodology, we complemented this with expert consensus suggestions of additional clinical data from ineligible RCTs, open-label studies, and real-world studies. This allowed us to provide more complete guidance while maintaining transparency about the strength of supporting evidence. By distinguishing between evidence-based recommendations and expert consensus suggestions, we offer clinicians a fuller context for decision-making without compromising methodological rigour. Our approach acknowledges that overly restrictive evidence requirements for guideline generation could have unintended consequences, potentially limiting access to beneficial treatments, particularly for patients with intractable conditions seeking non-pharmacological options. Just as some pharmacological treatments remain valuable in clinical practice despite limited supporting evidence, medical devices may offer important therapeutic alternatives even when evidence thresholds have not been met. Our framework upholds scientific standards while recognizing the practical realities of device development and clinical implementation, ultimately serving both the imperatives of evidence-based medicine and clinical practice needs.
Understanding the role of peer-reviewed publication vs. clinical guidelines
An important consideration for clinicians is understanding the fundamental difference between journal publication standards and clinical recommendation development. While peer-reviewed journals serve as critical gatekeepers for scientific rigor, their manuscript acceptance does not constitute an endorsement of clinical recommendations. Journal peer review primarily evaluates methodological soundness, statistical appropriateness, and scientific contribution within the context of the specific study design. A well-conducted RCT may merit publication even if it has limitations affecting its translation to clinical practice, such as small sample sizes, short follow-up periods, or populations that differ from typical clinical patients.
In contrast, clinical guidelines require a more comprehensive evaluation beyond individual study quality. The GRADE methodology systematically considers the internal validity of studies and their directness to the clinical question, consistency across multiple studies, precision of estimates, patient values and preferences, resource utilization, and implementation feasibility during the evidence-to-recommendation generation. This distinction explains why high-quality studies published in reputable journals may still receive weak recommendations in clinical guidelines. For example, a methodologically sound RCT demonstrating efficacy of a neuromodulation device may be appropriately published while simultaneously receiving a weak recommendation due to concerns about cost-effectiveness or device accessibility. Clinicians should therefore view published studies as essential building blocks of evidence rather than direct practice directives, with guideline synthesis providing the comprehensive evaluation necessary for clinical decision-making.
Regulatory perspective
In the US, medical devices are stratified into three regulatory categories (Class I, II, or III) based on risk, as defined in Section 513 of the Federal Food, Drug, and Cosmetic Act (88). The classification reflects the level of regulatory control necessary to ensure safety and effectiveness. Most non-invasive neuromodulation devices are designated as Class II, indicating a moderate risk. Prior to initiating clinical investigations in the US, manufacturers must secure an Investigational Device Exemption (IDE) from the FDA. IDE requirements depend on the study's risk profile: significant risk studies necessitate comprehensive FDA IDE approval and IRB oversight, whereas nonsignificant risk studies require only IRB approval. This framework ensures appropriate human subject protections while allowing manufacturers to generate the necessary clinical evidence for marketing. The FDA encourages early engagement through its pre-submission program to clarify optimal regulatory pathways and evidence expectations.
For novel devices lacking predicates, manufacturers must pursue the De Novo classification process, which establishes a new device type and regulatory framework. The De Novo pathway requires more robust clinical evidence than traditional 510(k) submissions, including data demonstrating reasonable assurance of safety and efficacy, but generally less than that required for Class III premarket approval (PMA). Once granted, a De Novo device may serve as a predicate for subsequent 510(k) submissions of similar products. Modifications to existing devices may qualify for the “special 510(k)” pathway if their intended use and fundamental technology remain unchanged. Manufacturers must demonstrate substantial equivalence to a legally marketed predicate in terms of intended use, technological characteristics, and safety/effectiveness. Most 510(k) submissions demonstrate substantial equivalence through bench testing, engineering data, and comparison to predicate devices, typically without the need for large clinical trials. When clinical data are required for 510(k), manufacturers can often rely on smaller studies, observational studies, or real-world studies rather than the large RCTs mandatory for drug approvals. However, higher-risk devices pursuing De Novo or PMA pathways may require more robust clinical evidence, though still typically less extensive than pharmaceutical trials.
When expanding indications for cleared devices, the regulatory pathway is contingent upon similarity to the original intended use. Indications analogous to the original cleared use may proceed via traditional 510(k) with moderate clinical evidence, whereas significant indication changes demand more extensive data. This is exemplified by several newly cleared indications that were established based on open-label studies rather than RCTs. Completely novel indications that raise new questions of safety or efficacy may necessitate pursuing a new De Novo classification or PMA submission.
In Europe, medical devices require CE marking for marketing within the European Economic Area (89). The CE marking process involves assessment by Notified Bodies: independent organizations designated by EU member states to conduct conformity assessments. Devices are categorized into four risk classes (I, IIa, IIb, and III), with most non-invasive neuromodulation devices falling into Class IIa or IIb. Under the new Medical Device Regulation (MDR), which superseded the previous Medical Device Directive, manufacturers must demonstrate compliance with Enhanced Essential Requirements, including more rigorous clinical evaluation, risk management, and quality system requirements. Historically, European regulations allowed greater flexibility in clinical evidence requirements compared to the FDA, but the MDR has substantially increased scrutiny of clinical evidence and post-market surveillance. The MDR also introduced Unique Device Identification (UDI) requirements and enhanced transparency through the EUDAMED database.
Future study design perspective
Evaluating non-invasive neuromodulation devices for migraine presents significant methodological challenges that affect evidence quality and guideline development. Most reviewed RCTs in this domain demonstrate low or very low-quality evidence, primarily due to imprecision from small sample sizes yielding wide confidence intervals and various risk-of-bias concerns. Notably, all eligible RCTs were initiated before the publication of IHS's 2021 neuromodulation device controlled trial guidelines (18), resulting in methodological approaches not aligned with the trial guidelines. In acute treatment studies, Nerivio, Relivion, and Ozilia trials selected primary endpoints other than the currently recommended 2hr pain freedom. Many device protocols permitted treatment within 1hr of symptom onset (including during mild headache phase), differing from the typical drug trial designs, which require treatment initiation when the headache intensity is at least moderate (80). In preventive treatment evaluations, several studies employed treatment monitoring durations below the current 12-week recommendations. While acknowledging resource constraints manufacturers face in conducting adequately powered studies, the panel emphasizes that future research should align with current IHS methodological standards to strengthen the evidence base and advance the field with higher-quality data.
Implementing proper sham controls and blinding procedures represents a fundamental methodological challenge in device trials. While an optimal sham device should replicate the active intervention's sensory characteristics without delivering therapeutic effects, emerging evidence suggests this ideal is difficult to achieve in practice. Recent research has revealed that presumed non-therapeutic sham gammaCore can unexpectedly modulate the trigeminal-autonomic reflex (90), challenging its validity as a true sham. This phenomenon extends to a tVNS study, where sham stimulation (1 Hz) unexpectedly outperformed verum stimulation (25 Hz) (83), with both frequencies affecting functional connectivity in the vagal nerve pathway and periaqueductal gray (14,85). Thus, it is important to recognize that the stimulation dosing and parameters for both verum and sham devices may not have been fully optimized. Furthermore, our practical recommendations were based only on the evaluated dosings, ranging from a single zap to 4hr per use for acute treatment, and from daily use to once a week for preventive treatment. More prolonged stimulation may render treatment effective, such as in Cefaly's ACME (1hr) vs. TEAM (2hr) or Nerivio 2017 (25 min) vs. 2019 (30–45 min) studies. Still, the therapeutic effect of this treatment duration/frequency remains to be adequately evaluated. Rigorous sham validation, systematic dose-response assessment, and comprehensive blinding verification are essential to strengthen the methodological foundation prior to clinical implementation.
Device adherence presents distinct analytical challenges that require careful methodological consideration, particularly for preventive interventions. Unlike pharmaceutical trials, where mITT typically includes all subjects receiving at least one treatment dose, device trials often impose longer adherence thresholds in the mITT population. This approach may introduce selection bias, limit generalizability to real-world settings, and affect statistical analysis. Research from behavioral intervention studies suggests that nonadherence patterns may correlate with treatment assignment or early intervention effects (91), potentially introducing systematic bias. Also, the PREMIUM II trial illustrates these challenges, where stringent adherence criteria (≥66% weekly adherence and ≥70 days completion requirements) substantially reduced the analyzable population (59), affecting both study power and outcome assessment. It is, therefore, important to prospectively define adherence criteria and study populations rather than making these determinations during analysis. Careful consideration must be given to defining and analyzing adherence in ways that balance methodological rigour with real-world applicability.
Society's perspective
The IHS positions neuromodulation devices as valuable components in the migraine treatment landscape, particularly for patients who prefer non-drug therapies, respond inadequately to medications, experience AEs, or have contraindications to pharmacological treatments. Through the 2021 guidelines for clinical trials with neuromodulation devices (18), the IHS emphasizes the importance of strict and rigorous evidence evaluation and generation, while acknowledging that these technologies may still serve both acute and preventive treatment purposes for many patients. The IHS supports using various FDA-cleared or CE-marked neuromodulation technologies, recognizing their therapeutic efficacies and advantages in terms of minimal side effects, lack of systemic interactions, and no risk of MOH. From a clinical usage perspective, the IHS encourages clinicians to prioritize on-label indications and FDA-cleared parameters when possible, recognizing that thoughtful off-label use may be appropriate when supported by sound medical judgment. In alignment with the broader position on “Setting higher standards for migraine prevention,” (92) the IHS advocates for continued research to refine neuromodulation approaches while positioning these devices as important options that can help address the significant burden of migraine when properly evaluated and applied based on sound scientific evidence. IHS also recognizes that expanding insurance coverage for these devices is critical, as data shows increased device utilization in systems with better coverage, such as the Veterans Administration Health Care System and certain commercial insurers (93,94). Removing cost barriers would likely yield reduced healthcare utilization and improved workplace productivity (95).
It 's important to note that some IHS recommendations in these guidelines may not fully align with current FDA-cleared indications for specific devices. Regulatory clearance and clinical practice guidelines serve different purposes and may result in different scopes of recommendations. FDA clearance establishes safety and efficacy for specific indications through a regulatory pathway. At the same time, clinical practice guidelines synthesize available evidence to provide broader clinical guidance that may encompass both on-label and off-label applications. This divergence underscores the importance of clinical judgment in applying regulatory guidance and professional society recommendations, recognizing that FDA clearance provides a regulatory framework while clinical guidelines offer evidence-based recommendations and expert consensus suggestions that may inform but do not supersede individual clinical decision-making and the physician-patient relationship.
Conclusion
Non-invasive neuromodulation devices offer an innovative alternative for migraine management, providing valuable options for patients seeking non-pharmacological treatments. Most existing devices received weak recommendations due to limitations in study quality and precision, as well as device accessibility and insurance coverage. Despite varying levels of evidence-based recommendations, these devices may be integrated into clinical practice based on expert consensus suggestions. Future research should prioritize adherence to IHS guidelines to ensure study validity and optimize patient outcomes.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251388377 - Supplemental material for International Headache society evidence-based guidelines on the use of non-invasive neuromodulation devices for the acute and preventive treatment of migraine
Supplemental material, sj-docx-1-cep-10.1177_03331024251388377 for International Headache society evidence-based guidelines on the use of non-invasive neuromodulation devices for the acute and preventive treatment of migraine by Hsiangkuo Yuan, Serena L. Orr, Mohammad A.M. Al-Karagholi, Messoud Ashina, Fred Cohen, Hans-Christoph Diener, David W. Dodick, Rigmor Højland Jensen, Michael J. Marmura, Daniele Martinelli, Manjit S. Matharu, Anja S. Petersen, Patricia Pozo-Rosich, Simona Sacco, Lucy Simmonds, Cristina Tassorelli, Stewart J. Tepper, Gisela M. Terwindt, Shuu-Jiun Wang, Jiunn-Tyng Yeh and Stephen D. Silberstein in Cephalalgia
Footnotes
Acknowledgments
The authors thank Rachel Zhao for the literature search and Marcus Vaska for the PRESS peer review. This manuscript used Grammarly (Grammarly, Inc., San Francisco, California, USA) and Claude.ai 3.7 Sonnet (Anthropic PBC, San Francisco, California, USA) to proofread and improve readability. After using this tool/service, the authors reviewed and edited the content as needed and took full responsibility for the content of the publication.
Data availability statement
The data used for this article may be obtained from the authors with a reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HY, in the past 24 months, received funding from the AHS Early-Stage Investigator Research Award; institutional support for serving as an investigator from Teva, AbbVie, Ipsen, Pfizer, Parema, Shiratronics, Johnson & Johnson; consultant/advisory fees from Salvia, AbbVie, Pfizer, Cerenovus; and royalties from Cambridge University Press and MedLink Neurology.
SLO receives royalties from Cambridge University Press. She serves on the editorial boards of Headache, Neurology, and the American Migraine Foundation. She also has research funding from the Canadian Institutes of Health Research, the Alberta Children's Hospital Research Institute, and the American Headache Society.
MMA-K declares no competing interests.
MA is a consultant, speaker, or scientific advisor for AbbVie, Astra Zeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Pfizer, and Teva; a primary investigator for ongoing Lundbeck and Pfizer trials; and is the past president of the International Headache Society. MA is supported through the Lundbeck Foundation Professor Grant (R310-2018-3711), Novo Nordisk Foundation Grant (Grant ID: 0101709), the Danish National Research Foundation Grant (DNRF188) and serves as associate editor of the Journal of Headache and Pain and associate editor of Brain.
FC received honoraria for consulting/speaking/writing from Teva, Pfizer, AbbVie, Eli Lilly, Springer Nature, and Medlink Neurology.
HCD received honoraria for oral presentations from: Betapharm, Lundbeck, Orion, and WebMD. The German Research Council (DFG) supports headache research by HCD. HCD serves on the editorial boards of Cephalalgia, Lancet Neurology, and Drugs.
DWD received consulting fees from Amgen, Atria, CapiThera Ltd, Cerecin, Ceruvia Lifesciences LLC, CoolTech, Ctrl M, Allergan, AbbVie, Biohaven, GlaxoSmithKline, Lundbeck, Eli Lilly, Novartis, Impel, Satsuma, Theranica, WL Gore, Genentech, Nocira, Perfood, Praxis, AYYA Biosciences, Revance, and Pfizer; received honoraria from American Academy of Neurology, Headache Cooperative of the Pacific, Canadian Headache Society, MF Med Ed Research, BioPharm Communications, CEA Group Holding Company (Clinical Education Alliance LLC), Teva (speaking), Amgen (speaking), Eli Lilly (speaking), Lundbeck (speaking), Pfizer (speaking), Vector Psychometric Group, Clinical Care Solutions, CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Academy for Continued Healthcare Learning, Majallin LLC, MedLogix Communications, Medica Communications LLC, MJH Life Sciences, Miller Medical Communications, WebMD Health/ Medscape, Wolters Kluwer, Oxford University Press, and Cambridge University Press. Non-profit board membership: American Brain Foundation, American Migraine Foundation, ONE Neurology, Precon Health Foundation, International Headache Society Global Patient Advocacy Coalition, Atria Health Collaborative, Arizona Brain Injury Alliance, and Domestic Violence HOPE Foundation/Panfila; received research support from Department of Defense, National Institutes of Health, Sperling Foundation, American Migraine Foundation, Henry Jackson Foundation, and Patient-Centered Outcomes Research Institute (PCORI). Stock options/shareholder/ patents/board of directors: Ctrl M (options), Aural Analytics (options), Axon Therapeutics, ExSano (options), Palion (options), Man and Science, Healint (options), Theranica (options), Second Opinion/ Mobile Health (options), Epien (options/board), Nocira (options), Matterhorn (shares/board), Ontologics (shares/board), King-Devick Technologies (options/board), Precon Health (options/board), AYYA Biosciences (options), Axon Therapeutics (options/board), Cephalgia Group (options/board), and Atria Health (options/employee). Patent 17189376.1-1466:v, Title: Onabotulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis (Non-royalty bearing). Patent application submitted: SynaQuell (PreCon Health).
RHJ is the principal investigator in clinical trials, and has given lectures to TEVA, Lundbeck Pharma, AbbVie, Pfizer, and Eli Lilly. Director of Danish Headache center, Director in Lifting The Global Burden (unpaid), Chair of Master of Headache Disorders at University of Copenhagen (personal payment). Her institution received a restricted research grant from Lundbeck Pharma and Novo Nordisk Foundation
MJM, within the past 24 months, has received compensation for consultation from Lundbeck. He has received institutional support for serving as principal investigator from Teva, Pfizer and AbbVie.
DM declares honoraria from Lundbeck; honoraria for participation in an advisory board from AbbVie.
MSM is chair of the medical advisory board of the CSF Leak Association; has served on advisory boards for AbbVie, Eli Lilly, Lundbeck, Pfizer, Salvia, and TEVA; has received payment for educational presentations from AbbVie, Eli Lilly, Lundbeck, Organon, Pfizer, and TEVA; has received grants from Abbott, Medtronic, and Ehlers Danlos Society; and has a patent on system and method for diagnosing and treating headaches (WO2018051103A1, issued).
ASP received personal payment for lectures on headache for general practitioners from Pfizer, she has participated in a two-day neurological symposium sponsored by Lundbeck Pharma, and her institution got a restricted research grant from Lundbeck pharma.
PPR has received, in the last three years, honoraria as a consultant and speaker for: AbbVie, Almirall, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis, Organon, Pfizer and Teva. Her research group has received research grants from AbbVie, Novartis and Teva; as well as, Instituto Salud Carlos III, EraNet Neuron, European Regional Development Fund (001-P-001682) under the framework of the FEDER Operative Programme for Catalunya 2014-2020 - RIS3CAT; has received funding for clinical trials from AbbVie, Amgen, Biohaven, Eli Lilly, Novartis, Pfizer, Teva. She is the Honorary Secretary of the International Headache Society. She is an associate editor for Cephalalgia and Neurologia. She is the founder of ![]() . PPR does not own stocks from any pharmaceutical company.
. PPR does not own stocks from any pharmaceutical company.
SS received personal fees as speaker or advisor: Abbvie, Bayer, Boheringer, Eli Lilly, Lundbeck, Pfizer, Teva. Intellectual: president European Stroke Organisation, editor-in-chief Cephalalgia and Cephalalgia Reports, assistant editor for Stroke.
LS received funding from The Wellington Hospital, London
CT received personal fees for participating in advisory boards or for lecturing at sponsored symposia for AbbVie, Dompé, Eli Lilly, Ipsen, Lundbeck, Medscape, Pfizer and Teva. She is principal investigator or collaborator in clinical trials sponsored by AbbVie, Eli Lilly, Ipsen, Lundbeck, Pfizer and Teva. She has received research grants from the European Commission, the Italian Ministry of Health, the Italian Ministry of University, the Migraine Research Foundation and the Italian Multiple Sclerosis Foundation.
SJT received research funding: Abbvie, Aeon, Amgen, Annovis, Axsome, Biohaven, Cassava, Cognition, Eli Lilly, Inhibikase, Ipsen, Lundbeck, Merz, Neurolief, Pfizer, PrecisionMed, Revance, Scilex, Suven, UCB; consultant and/or Advisory Boards (honoraria): Abbvie, Aeon, Alphasights, Amgen, Aruene/eNeura, Atheneum, Axsome Therapeutics, Bausch Health, Becker Pharmaceutical Consulting, Catch Therapeutics, ClearView Healthcare Partners, ClickTherapeutics, CoolTech, CRG, Decision Resources, Defined Health, DRG, DocDelta, Dr Reddy's, Eli Lilly, ExpertConnect, FCB Health, Fenix, Gilmartin Capital, GLG, Guidepoint Global, Health Advances, Health Science Communications, HMP Communications, Impel, Initiator Pharma, InteractiveForums, IQVIA, Keyquest, Ki Health Partners, Krog and Partners, Lundbeck, M3 Global Research, Magellan Health, Magnolia Innovation, Miravo Healthcare, MJH Holdings, Neurofront Therapeutics, Neurolief, Nocira, Novartis, P Value Communications, Pain Insights, Inc, Palion Medical, Perfood, Pfizer, Pulmatrix, Putnam Associates, Rehaler, SAI MedPartners, Satsuma, Scilex, Slingshot Insights, Spherix Global Insights, Strategy Inc, Synapse Medical Communication, System Analytic, Taylor and Francis, Tegus, Teva, Theranica, Third Bridge, Tonix, Trinity Partners, Unity HA, Vial, XOC; speakers Bureau: AbbVie, Dr Reddy's, Eli Lilly, Lundbeck, Pfizer, Scilex, Teva, Tonix; CME honoraria: American Academy of Neurology, American Headache Society, Annenberg Center for Health Sciences, Catamount Medical Education, Consortium for Research Education Social Awareness and Training In Neurosciences, Diamond Education Foundation, Forefront Collaborative, Haymarket Medical Education, HMP Global, Medical Education Speakers Network, Medical Learning Institute Peerview, Migraine Association of Ireland, Miller Medical Education, National Association for Continuing Education, North American Center for CME, The Ohio State University, Physicians’ Education Resource, PlatformQ Education, Primed, Vindico Medical Education, WebMD/Medscape
GMT reports consultancy or industry support from Novartis, Lilly and Teva, Allergan/Abbvie, Lundbeck, Pfizer and Interactive Studios, as well as independent support from the European Community, Dutch Heart Foundation, Dutch Research Council, Dutch Brain Foundation and Dioraphte.
SJW has served on the advisory boards of Taiwan Pfizer, AbbVie and Hava-Biopharma; has received honoraria as a moderator from AbbVie, Pfizer and Biogen; and has been the principal investigator in clinical trials sponsored by AbbVie, Novartis, Lundbeck and Pfizer. He has received research grants from the National Council of Technology and Science of Taiwan, Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, Taipei Veterans General Hospital, and Taiwan branches of Eli Lilly and Novartis.
JTY declared no conflict of interest.
SDS provides consultation to Alder, Allergan, Amgen, Autonomic Technologies, Avanir, Curelator Inc., Depomed, Dr Reddy's Laboratories, Ensured Inc., electroCore Medical LLC, eNeura Therapeutics, Insys Therapeutics, Lilly USA LLC, Supernus Pharmaceuticals, Teva Pharmaceuticals, Theranica, and Trigemina Inc.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
