Abstract
The aim of the present study was to examine the effect of KATP channel openers pinacidil and levcromakalim on rat dural and pial arteries as well as their inhibition by glibenclamide. We used an in-vivo genuine closed cranial window model and an in-vitro organ bath. Glibenclamide alone reduced the dural but not the pial artery diameter compared with controls. Intravenous pinacidil and levcro-makalim induced dural and pial artery dilation that was significantly attenuated by glibenclamide. In the organ bath pinacidil and levcromakalim induced dural and middle cerebral artery relaxation that was significantly attenuated by glibenclamide. In conclusion, KATP channel openers induce increasing diameter/relaxation of dural and pial arteries after intravenous infusion in vivo and on isolated arteries in vitro. Furthermore, dural arteries were more sensitive to KATP channel openers than pial arteries.
Introduction
Inherited dysfunction of voltage gated Ca2+ channels can cause familial hemiplegic migraine (1) and it has been suggested that the common types of migraine may also be channelopathies (2). Disturbed channel function may lead to instability in the control of pain and other sensory information emanating from the migraine-producing intracranial structures such as dural and/or pial vessels (3). Ray and Wolff (1940) demonstrated that distension of large cerebral as well as the meningeal (dural) arteries caused a throbbing, unilateral migraine-like pain (4).
Levcromakalim and its racemic form cromakalim cause propagated headache when used for asthma, suggesting a role for ATP-sensitive K+ (KATP) channels in migraine pathogenesis. K+ channels participate in the regulation of cerebral vascular tone (5) and activation or inhibition of K+ channel activity provides an important mechanism to dilate or constrict cerebral arteries (6). KATP channel openers and blockers have been shown to exert pharmacological action in cerebral arterial smooth muscle (6).
Since the development of a genuine closed cranial model it has been successfully implemented in migraine research because it allows the direct measurement of the diameter of dural vessels in anaesthetized animals and investigation of the vascular effects of vasoactive substances (as well as electrical stimulation of the cranial window) (7). The method has been further verified for the possibility of measuring diameter changes of pial arteries and local cerebral blood flow by laser Doppler flowmetry (LCBFFlux) (K. Petersen et al., 2004, unpublished). To date, the role of KATP channels in the middle meningeal artery and the effects in genuine closed cranial window model have not been studied. In the present study we therefore aimed to: (i) explore the vasodilator effects in vivo of pinacidil and levcromakalim, the best described KATP channel openers on dural and pial arteries and cerebral blood flux; (ii) evaluate the effect of glibenclamide and its inhibition of the effect of pinacidil and levcromakalim (8–10); and (iii) explore the effects of pinacidil and levcromakalim and their inhibition by glibenclamide on isolated middle cerebral and middle meningeal arteries in vitro.
Materials and methods
In-vivo studies
Surgical preparation
This study complied with The Danish Animal Experimentation Inspectorate (file: 2001/561–390) on the care and use of animals. All experiments were conducted using the closed cranial window technique as previously described by Williamson (7). Male Sprague-Dawley rats (300–400 g) were anaesthetized throughout the experiments with pentobarbital (Mebumal®, The Danish Hospital Pharmacy, Glostrup, Denmark). Anaesthesia was induced by 60 mg/kg intraperitonal pentobarbital and throughout the course of the experiments maintained by intravenous infusion of 20 mg/kg per h pentobarbital. The depth of anaesthesia was judged by suppression of the hindpaw withdrawal reflex. The animals were mechanically ventilated. The respirator delivered 3–3.5 ml/stroke of O2 30% and N2O 70%, at a ventilation rate of 60–65 strokes/min (Rodent Ventilator; Ugo Basile, Biological Research Apparatus, Comerio, Italy). The femoral arteries and veins were cannulated bilaterally by catheters (0.40 mm i.d.) (Portex ® Astra Tech A/S, Taastrup, Denmark) for administration of test substances, blood sampling and measurement of mean arterial blood pressure (MABP) and arterial blood gases (ABG) (Transducer TCM4-7; World Precision Instruments, Sarasota, FL, USA). The animals were placed in a stereotaxic frame, the skull exposed and the parietal bone thinned by drilling with a saline-cooled dental drill until the dural and pial arteries were clearly visible through the intact skull. Before placing the video camera, warm (37 °C) mineral oil was applied.
Intravital microscopy
The vessels were imaged by a Sony video camera with microscope (Sony DSP digital camera, MS 50 objective; Sony Corporation, Tokyo, Japan) and the real time image was displayed on a television screen. Dural and pial artery diameter were continuously measured using a Video Dimension Analyser (VDA; Living Systems Instruments Inc., Burlington, VT, USA). Data were collected and analysed by data acquisition and analysis software (Perisoft version 1; Perimed AB, Järfälla, Sweden). LCBFFlux (Perimed Periflux®4001; Perimed) was measured by a probe bent 90° (Perimed 410, fibre separation 0.25 mm; Perimed) held by a micromanipulator. The probe was placed free of the bone but in touch with mineral oil, which enhances the accuracy of the LCBFFlux measurement (11). The probe was placed in an area of no or limited visual vessels.
Blood pressure was measured continuously by connecting the catheter inserted in the femoral artery to a transducer (Transducer TCM4-7; World Precision Instruments). Blood gases were measured at the beginning and at the end of each experiment (ABL520; Radiometer Danmark AS, Rødovre, Denmark). PaCO2, PaO2 and pH were kept within normal limits. Blood glucose was measured (Accu-Chek® Compact; Roche, Basle, Switzerland) four times during the experiment. Body temperature was controlled by a heating blanket and kept at 37 ± 0.5 °C. [Panlab s.l. (Letica®), Barcelona, Spain].
Experimental protocols
In all our experiments blood gas analyses were pH 7.45 ± 0.006; PaCO2 38.4 ± 0.05; PaO2 was above 78 mmHg.
The effect of pinacidil and inhibition by glibenclamide. It has been shown that pinacidil at a dose of 0.3 mg/kg causes maximum decrease in BP of 22% in conscious rats (12). We conducted dose–effect studies (n = 4) where pinacidil was infused intravenously at doses of 0.036, 0.12, 0.36, 1.2 mg/kg in a volume of 0.5 ml over 12 min. In cumulative dose–effect studies 0.36 mg/kg gave maximum dilation of dural and pial vessels (Fig. 1). However, when administered as a single dose the effective dose of pinacidil was determined as a dose of 0.38 mg/kg and selected for further study. This dose was repeated four times in three rats to check the reproducibility of the response. Glibenclamide 20 and 30 mg/kg was used because it has been shown that at these doses it is effective in inhibiting cromakalim in anaesthetized rats (8). In order to prevent catheters from being blocked by precipitated glibenclamide they were flushed with 50 µl dimethylsulfoxide (DMSO).

The effect of increasing doses of pinacidil on dural (▪) and pial artery (□) diameter (n = 4). Values given as mean ± SEM, n = 4.
After a stabilization period of about 1–1.5 h, baseline values were recorded and a blood sample taken to determine ABG and blood glucose. First, vehicle (DMSO) was infused intravenously in a volume of 0.1 ml over 20 min. After a 15-min resting interval pinacidil (0.38 mg/kg) was infused, in a volume of 0.5 ml over 12 min. After a washout period of 12 min glibenclamide infusion (20 mg/kg or 30 mg/kg) was given in a volume of 0.1 ml over 20 min followed by a pinacidil (0.38 mg/kg) infusion in a volume of 0.5 ml over 12 min. Additional blood samples were taken during the resting periods.
A special study was done for DMSO. DMSO was infused in a volume of 0.1 ml over 20 min: repeat dose (n = 6) and DMSO with pinacidil (n = 4). There were no significant differences in pinacidil responses before and after DMSO infusion in MABP, dural/pial arteries diameter and LCBFFlux.
The effect of levcromakalim and inhibition by glibenclamide. The maximum effective dose of levcromakalim of 0.1 mg/kg was used. It has been shown that levcromakalim at a dose of 0.1 mg/kg causes a maximum decrease in BP of 23% in conscious rats (8). Reproduction of levcromakalim was studied by giving a four times repeated dose of 0.1 mg/kg with a 12-min resting interval (n = 2). After a stabilization period of about 1 h baseline values were recorded and a blood sample taken to determine ABG and blood glucose. First vehicle (DMSO) was infused intravenously in a volume of 0.1 ml over 20 min. After a 15-min resting interval levcromakalim (0.1 mg/kg) was infused, in a volume of 0.1 ml over 20 min. After a washout period of 15 min, a glibenclamide infusion (20 or 30 mg/kg) was given in a volume of 0.1 ml over 20 min followed by a levcromakalim (0.1 mg/kg) infusion in a volume of 0.1 ml over 20 min. Blood samples for measurements of ABG and blood glucose were taken during the resting periods.
In-vitro studies
Male Sprague-Dawley rats (300–380 g, Denmark) were exanguinated during CO2 anaesthesia. The brain was removed and the first branch of the dural and middle cerebral arteries were dissected out using an operation microscope. Each vessel was cut into 1–2 mm long circular segments and placed in an ice-cold buffer solution gassed with 5% CO2 in O2. The composition of the buffer was (m
Data analysis
For in-vivo studies data are presented as mean ± SEM. The diameter of the dural and pial arteries and LCBFLDF were measured in arbitrary units, MABP in mmHg and presented as percentage change from baseline. Statistical significance was set at P < 0.05. SPSS version 10.0 (SPSS, Chicago, IL, USA) was used for all statistical analysis, GraphPrism® (version 3; San Diego, CA, USA) for graphical evaluation.
For in-vitro studies the amount of relaxation was calculated as percentage of PGF2α precontraction. Values are given as mean ± SEM. Number of experiments = n, one or two segments from each rat. The non-parametric, Mann–Whitney U-test was used to determine statistical significance between two groups of data. Statistical significance was assumed when P < 0.05.
Drugs
Pinacidil was a gift from Leo Pharma Nordic (Malmö, Denmark). Levcromakalim and glibenclamide were obtained from Tocris Cookson Ltd (Avonmouth, UK). Pinacidil (was dissolved in 1 N HCl and diluted to 2.5 mg/ml (in-vivo studies) or 10−2
Results
The effect of glibenclamide and DMSO in the genuine closed cranial window model
Glibenclamide (20 mg/kg) alone increased MABP 28 ± 5% from baseline and constricted dural arteries 60 ± 9% compared with DMSO controls (P < 0.005). Pial artery constriction was 3 ± 3% (ns), which is not significantly different from the effect of DMSO alone (Table 1). The constriction of dural arteries, as well as the increase of MABP, was prolonged (30–60 min) and did not return to baseline during the experiments (n = 19). Glibenclamide 30 mg/kg had similar effects (Table 1). In our experiments glibenclamide was dissolved in DMSO. Recent studies have suggested that DMSO, among other antioxidants, can inhibit ATP-sensitive K+ channels (14, 15). We therefore compared the effect of pinacidil on dural/pial vessel diameter before and after DMSO infusion. However, there was no significant difference in vessel diameter before and after DMSO infusion (0.1 ml over 20 min). Furthermore, DMSO given as an infusion (0.1 ml over 20 min) did not cause any significant change in the response to pinacidil (data not shown).
Effect of pinacidil (0.38 mg/kg), levcromakalim (0.1 mg/kg), glibenclamide (20 and 30 mg/kg) and DMSO on dural/pial artery diameter, LCBFFlux and mean arterial blood pressure (MABP)

Values are given as mean ± SEM, n = 19. Statistical analysis comparing responses of glibenclamide 20 and 30 mg/kg vs. DMSO performed by Student's t-test: ns (P > 0.05);
(P < 0.05);
(P < 0.005).
The effect of pinacidil and inhibition by glibenclamide in the genuine closed cranial window model
A 12-min infusion of pinacidil (0.38 mg/kg) increased dural artery diameter by 126 ± 8% and pial artery diameter by 17 ± 3% (n = 10) (Table 1, Fig. 2). The response was significantly lower in pial than in dural arteries (P < 0.05).

Trace showing the effect of pinacidil and inhibition by glibenclamide in the genuine closed cranial window model. (a) Pinacidil 0.38 mg/kg infusion over 12 min. (b) Pause. (c) Glibenclamide 20 mg/kg infusion over 20 min. (d) Pause. (e) Pinacidil 0.38 mg/kg infusion over 12 min. (f) Pause.
Following infusion of the lower dose of glibenclamide (20 mg/kg) dural artery dilation by pinacidil was attenuated from 131 ± 13% to 53 ± 3% (P = 0.004, t = 6.2, n = 5) (Fig. 3a). Pial artery diameter after pinacidil, in the presence of glibenclamide (20 mg/kg), decreased from 18 ± 5% to 3 ± 2% (20 mg/kg, P = 0.077, t = 2.4, n = 5) (Fig. 3c). Pinacidil infusion caused an increased LCBFFlux of 18 ± 4% (n = 10) (Table 1). Infusion of glibenclamide (20 mg/kg) attenuated this increase in LCBFFlux, to pinacidil, from 22 ± 8% to 8 ± 8% (P = 0.142, t = 1.8, n = 5) (Fig. 4a). Pinacidil infusion caused a drop of MABP of 28 ± 2% (35 ± 3 mmHg) (Table 1). Following infusion of glibenclamide the drop of MABP was attenuated from 26 ± 2% to 11 ± 1% (32 ± 2 to 16 ± 2 mmHg) (P = 0.001, t = 9.2, n = 5) (Fig. 5a).
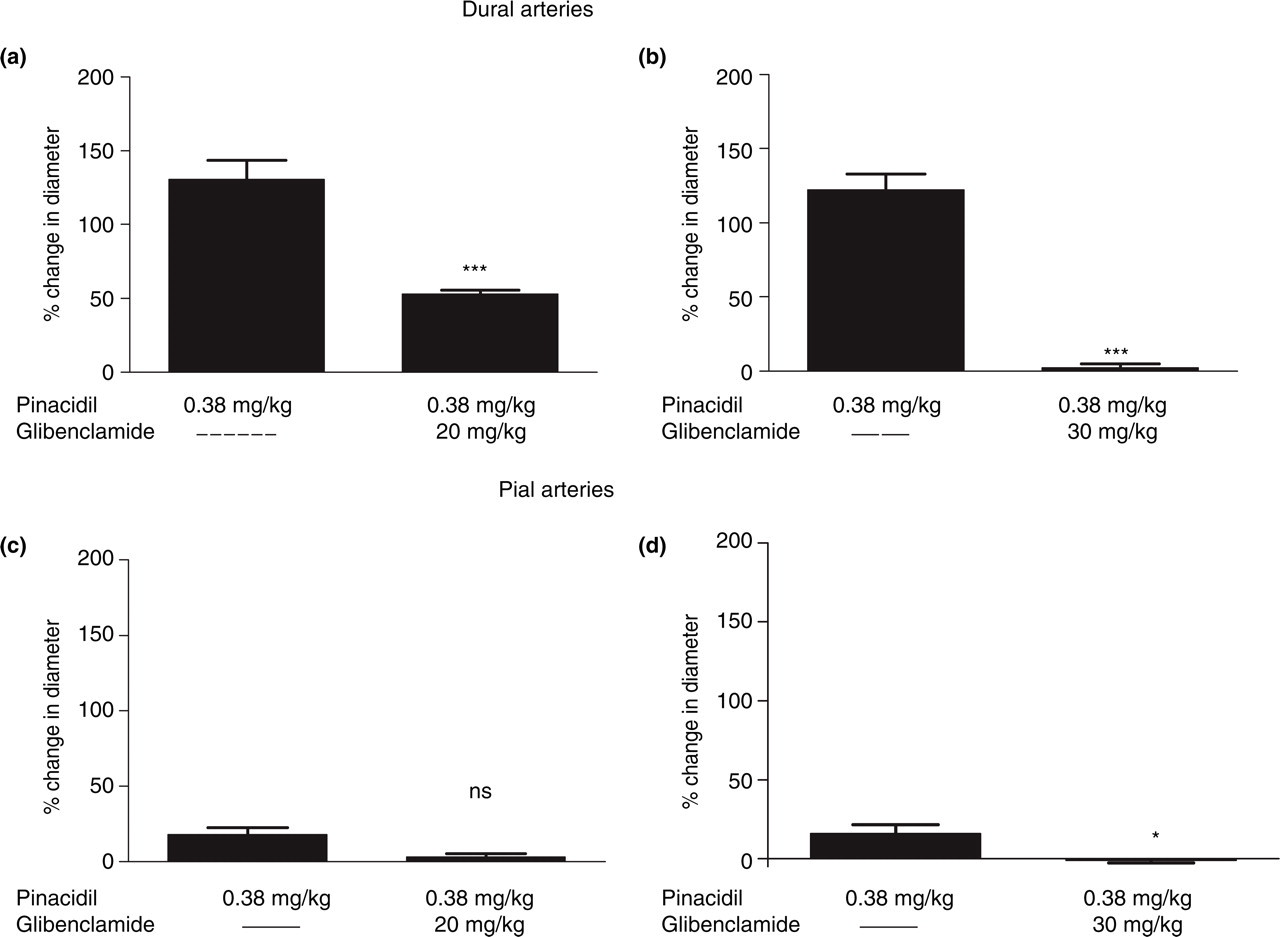
Effect of pinacidil (0.38 mg/kg) on the dural (a,b) and pial (c,d) artery diameter in the presence or absence of glibenclamide 20 mg/kg (a,c) and 30 mg/kg (b,d), n = 10. Values given as mean ± SEM, n = 10. Statistical analysis comparing responses in presence and in absence of glibenclamide performed by Student's t-test: ns (P > 0.05); ∗(P < 0.05); ∗∗(P < 0.01); ∗∗∗(P < 0.005).

Effect of pinacidil (0.38 mg/kg) on LCBFFlux in the presence or absence of glibenclamide 20 mg/kg (a) and 30 mg/kg (b), n = 10. Values given as mean ± SEM, n = 10. Statistical analysis comparing responses in presence and in absence of glibenclamide performed by Student's t-test: ns (P > 0.05); ∗(P < 0.05); ∗∗(P < 0.01); ∗∗∗(P < 0.005).
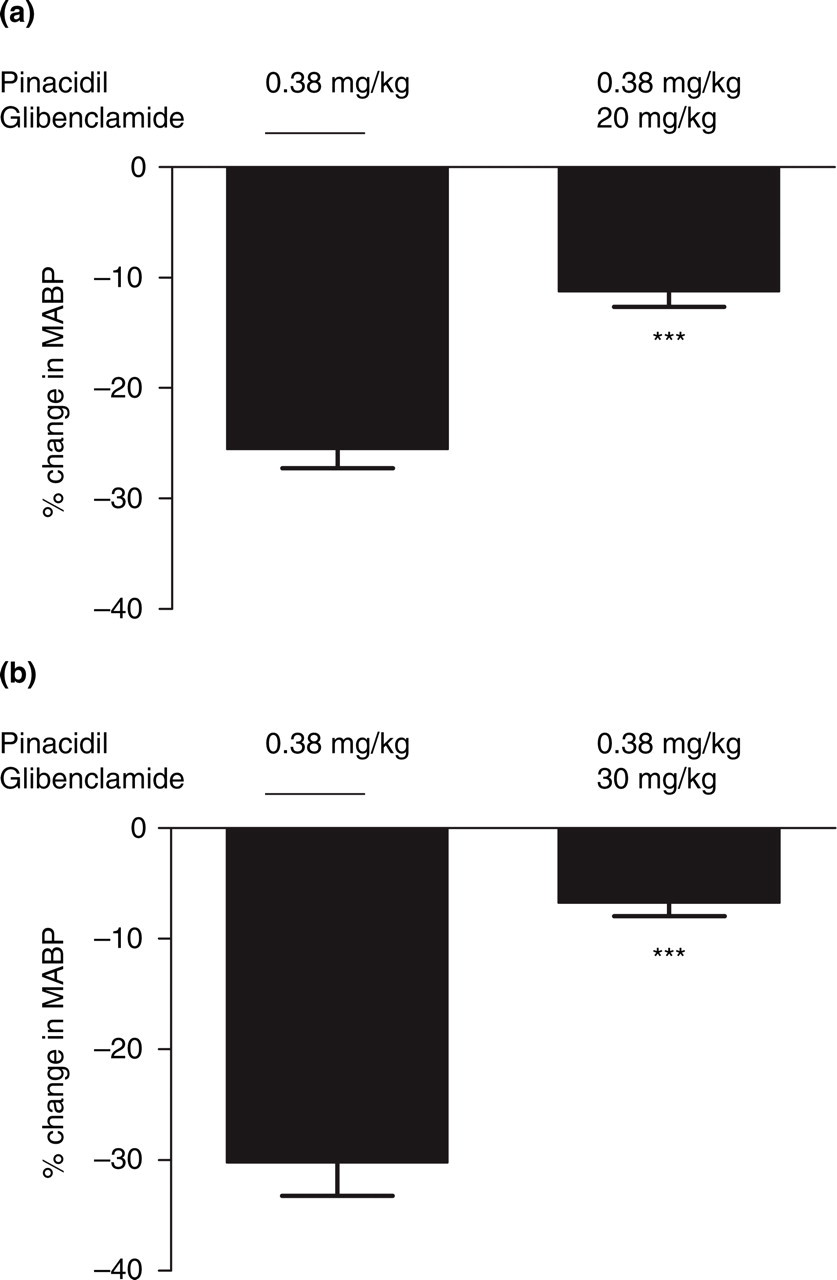
Effect of pinacidil on mean arterial blood pressure (MABP) in the presence or absence of glibenclamide 20 mg/kg (a) and 30 mg/kg (b), n = 10. Values given as mean ± SEM, n = 10. Statistical analysis comparing responses in presence and in absence of glibenclamide performed by Student's t-test: ns (P > 0.05); ∗(P < 0.05); ∗∗(P < 0.01); ∗∗∗(P < 0.005).
Following infusion of the higher dose of glibenclamide (30 mg/kg) dural artery dilation by pinacidil was attenuated from 122 ± 11% to 2 ± 2% (P = 0.001, t = 10.2, n = 5) (Fig. 3b). Pial artery diameter after pinacidil, in the presence of glibenclamide (30 mg/kg), decreased from 16 ± 6% to − 1 ± 2% (30 mg/kg, P = 0.047, t = 2.8, n = 5) (Fig. 3d). Following infusion of glibenclamide (30 mg/kg) the increase in LCBFFlux after pinacidil was attenuated from 15 ± 2% to − 8 ± 5% (P = 0.013, t = 4.2, n = 5) (Fig. 4b). After infusion of 30 mg/kg glibenclamide the drop in MABP after pinacidil was attenuated from 30 ± 3% to 7 ± 1% (39 ± 5 ro 10 ± 2 mmHg) (P = 0.002, t = 7.7, n = 5) (Fig. 5b).
The effect of levcromakalim and inhibition by glibenclamide
A 20-min infusion of levcromakalim (0.1 mg/kg) increased dural artery diameter by 130 ± 24% and pial artery diameter by 18 ± 3% (n = 9) (Table 1, Fig. 6). Comparing the relaxant response to levcromakalim in the two types of vessels, we found that the response was significantly (P < 0.05) lower in pial than in dural arteries.

Trace showing the effect of levcromakalim and inhibition by glibenclamide in the genuine closed cranial window model. (a) Levcromakalim 0.1 mg/kg infusion over 20 min. (b) Pause. (c) Glibenclamide 30 mg/kg infusion over 20 min. (d) Pause. (e) Levcromakalim 0.1 mg/kg infusion over 20 min. (f) Pause.
Following infusion of the lower dose of glibenclamide (20 mg/kg) the change in dural vessel diameter evoked by subsequent levcromakalim infusion was attenuated from 147 ± 38% to 49 ± 5% (P = 0.067, t = 2.5, n = 5) (Fig. 7a). The increase in pial vessel diameter after pinacidil, in the presense of glibenclamide (20 mg/kg), was attenuated from 20 ± 3% to 6 ± 3% (P = 0.014, t = 4.1, n = 5) (Fig. 7c). Infusion of levcromakalim caused an increase of LCBFFlux of 17 ± 4% (n = 9) (Table 1). Following infusion of glibenclamide (20 mg/kg) the increase in LCBFFlux after levcromakalim infusion was attenuated from 24 ± 3% to 2 ± 4% (20 mg/kg, P = 0.028, t = 3.4, n = 5) (Fig. 8a). Levcromakalim infusion caused a drop of MABP of 31 ± 2% (40 ± 3 mmHg) (n = 9) (Table 1). Following infusion of glibenclamide (20 mg/kg) the drop of MABP after levcromakalim was attenuated from 30 ± 3% to 12 ± 3% (37 ± 4 to 17 ± 3 mmHg) (P = 0.014, t = 4.1, n = 5) (Fig. 9a).
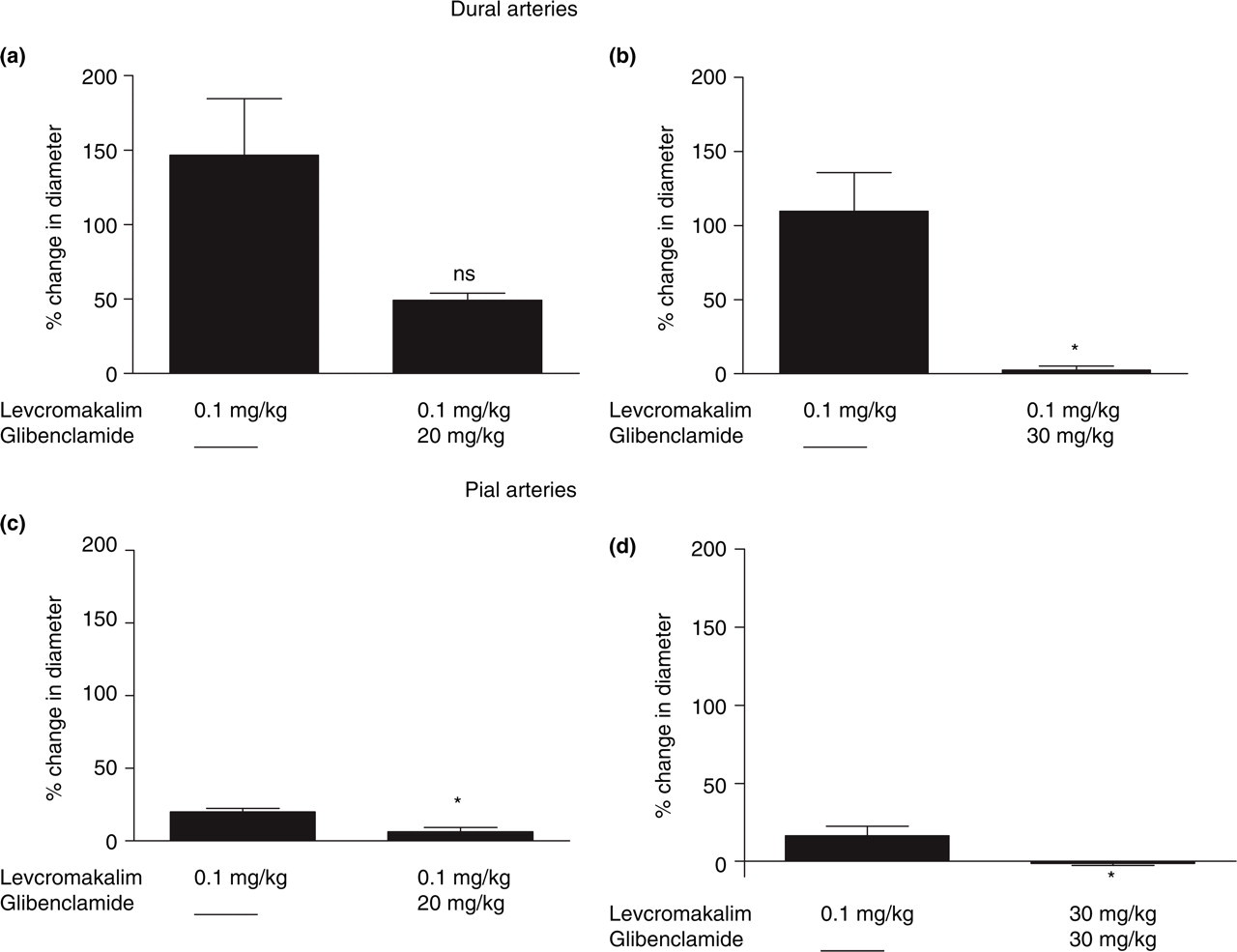
Effect of levcromakalim (0.1 mg/kg) on the dural (a,b) and pial (c,d) artery diameter in the presence or absence of glibenclamide 20 mg/kg (a,c) and 30 mg/kg (b,d). Values given as mean ± SEM, n = 9. Statistical analysis comparing responses in presence and in absence of glibenclamide performed by Student's t-test: ns (P > 0.05); ∗(P < 0.05); ∗∗(P < 0.01); ∗∗∗(P < 0.005).

Effect of levcromakalim (0.1 mg/kg) on LCBFFlux in the presence or absence of glibenclamide 20 mg/kg (a) and 30 mg/kg (b). Values given as mean ± SEM, n = 9. Statistical analysis comparing responses in presence and in absence of glibenclamide performed by Student's t-test: ns (P > 0.05); ∗(P < 0.05); ∗∗(P < 0.01); ∗∗∗(P < 0.005).
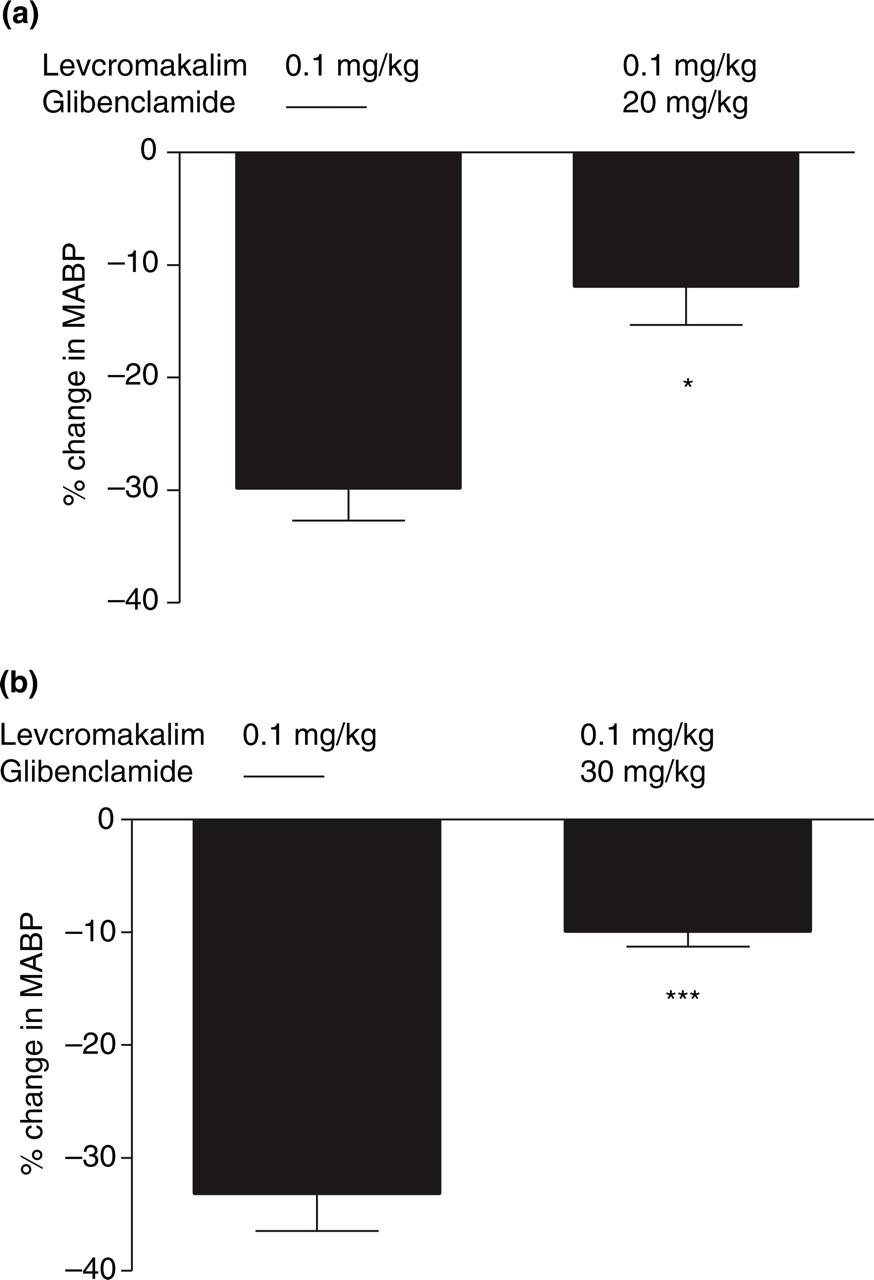
Effect of levcromakalim (0.1 mg/kg) on mean arterial blood pressure (MABP) in the presence or absence of glibenclamide 20 mg/kg (a) and 30 mg/kg (b). Values given as mean ± SEM, n = 9. Statistical analysis comparing responses in presence and in absence of glibenclamide performed by Student's t-test: ns (P > 0.05); ∗(P < 0.05); ∗∗(P < 0.01); ∗∗∗(P < 0.005).
Following infusion of the higher dose of glibenclamide (30 mg/kg) dural artery dilation by levcromakalim was attenuated from 110 ± 26% to 3 ± 3% (P = 0.033, t = 3.8, n = 4) (Fig. 7b). The increase in pial artery diameter after levcromakalim, in the presence of glibenclamide (30 mg/kg), decreased from 16 ± 6% to − 1 ± 1% (P = 0.036, t = 3.6, n = 4) (Fig. 7d).
Following infusion of glibenclamide (30 mg/kg) the increase in LCBFFlux after levcromakalim infusion was attenuated from 9 ± 6% to − 5 ± 5% (P = 0.005, t = 7.2, n = 4) (Fig. 8b). Following infusion of glibenclamide (30 mg/kg) the drop of MABP after levcromakalim was attenuated from 33 ± 3 to 10 ± 1% (44 ± 5 to 15 ± 2 mmHg) (P = 0.003, t = 8.7, n = 4) (Fig. 9b).
Studies of K ATP openers on isolated cerebral arteries in vitro
A single concentration of pinacidil (3 × 10−6

Relaxant effects of pinacidil (3 × 10−6
Levcromakalim (3 × 10−6
Discussion
In our experiments glibenclamide alone caused constriction of dural arteries but had no significant effect on pial arteries. A possible explanation for the findings could be that KATP channels contribute with a dilatory influence on rat meningeal arteries in the resting state. However, it might also be caused by release of vasoactive substances from the periphery. The pial arteries differ markedly in this respect from the dural arteries. The finding is further supported by a lack of effect of glibenclamide on LCBFFlux which closely reflects cortical blood flow and hence the response of cortical and pial arterioles. Thus, most cerebral arteries and cerebral arterioles are insensitive to glibenclamide. These studies suggest a plausible effect of KATP channel blocker glibenclamide on dural vs. pial arteries and arterioles.
Effect of KATP channel openers on dural vs. pial arteries
In the present study, we found that the two KATP openers pinacidil and levcromakalim induced dilation of dural and pial arteries in vivo and in vitro. The effect on pial arteries is in agreement with several previous studies that have examined the effect of KATP-channel openers on peripheral and cerebral blood arteries from various species, in vitro (16, 17) and in situ (5, 18–20). However, to our knowledge no studies have examined cranial arterial diameter in vivo after i.v. infusion of KATP channel openers. Furthermore, we are the first to describe the effect of KATP channel openers on dural vessels. We found, in vitro as well as in vivo, that dural arteries were significantly more sensitive to the tested KATP channel openers than pial arteries. It has previously been described that the response to KATP channel openers in rat basilar artery is greater than in the middle cerebral artery (21). Moreover, a recent study done in a genuine closed cranial window rat model showed that pial arteries were less responsive to CGRP infusion than dural arteries (K. Petersen et al., 2004, unpublished). In our experiments this difference cannot be explained by a tighter blood–brain barrier in the pial arteries since our in-vitro studies showed similar results. In agreement with previous studies, our results on anaesthetized rats showed that levcromakalim was a three-fold more potent vasodilator than pinacidil (12).
To confirm the presence and functionality of KATP channels in dural and pial arteries in vivo and in vitro we studied the effect of specific KATP channel openers pinacidil and levcromakalim. Dilation of blood vessels in response to activation of ATP-sensitive K+ channels in vitro and in vivo appeared to be specific since glibenclamide inhibited responses to activation of ATP-sensitive K+ channels (9, 10, 18, 22).
Effect of K ATP channel openers on MABP and LCBF Flux
In our study pinacidil and levcromakalim markedly increased LCBFFlux and produced a marked drop of MABP in anaesthetized rats. These effects were inhibited by glibenclamide in a dose-dependent manner. Previous studies have shown that LCBFFlux can be used as a relative measure of CBF (23, 24). We found that the magnitude of the response of pial arterial diameter and of LCBFFlux was of the same magnitude and considerably smaller than the increase of dural artery diameter. Williamson et al. found that selective blood pressure decrease induced by senktide (n = 3) did not alter middle meningeal artery diameter (25). In agreement with this, a recent study showed that induction of acute haemorrhagic hypotension, even to a large degree, did not significantly affect the middle meningeal artery diameter (K. Petersen et al., 2004, unpublished). Since pinacidil and levcromakalim produced a marked drop of MABP in our study, the pial artery response could be secondary to hypotension, i.e. autoregulation (26, 27). On the other hand, the increase of pial artery diameter during hypotension may, at least in part, be due to KATP and Kca channel activation (28).
Possible role of KATP channel openers in migraine pathogenesis
Within the cranium only a few structures are able to generate nociceptive signals, such as vessels within the meninges and the large cerebral arteries (4). When large cerebral vessels and vessels in the dura and pia mater are stimulated experimentally during brain surgery, patients experience a throbbing unilateral migraine-like pain (4), indicating that these structures are involved in migraine pain. Graham and Wolff suggested that ergotamine produced relief of migraine through constriction of cranial blood vessels (29). This theory is still current, with the development of the clinically effective 5-HT1B/1D agonist sumatriptan initially being based on this vascular hypothesis (30). Migraine pain has also been suggested to be caused by the development of a sterile neurogenic inflammation within the meninges (31). However, this theory also involves dilation of dural arteries.
Although KATP channel openers have not been examined in an experimental headache model (32–35), many clinical trials investigating the effect of levcromakalim, pinacidil and nicorandil on respiration and cardiovascular function mention headache as one of the primary side-effects (36–42). The present results demonstrate that KATP channel openers preferentially dilate dural arteries, a mechanism thought to be involved in migraine pathogenesis as part of neurotransmitter release from nerve terminals. Thus, we have demonstrated a possible mechanism of the headache-generating effect of KATP channel openers.
In conclusion, our studies, in vitro as well as in vivo, show that dural arteries are significantly more sensitive to KATP channel openers and blockers than pial arteries. The reason for this difference is not clear but it suggests a heterogeneous distribution of KATP channels in the cranial circulation.
Footnotes
Acknowledgements
This study was supported by the Else Torps Legat, The Novo Nordisk foundation and the Lundbeck foundation. We thank Dr Messoud Ashina for his constructive advice and critical comments during the preparation of this manuscript.
