Abstract
The genuine closed cranial window model, in which the thinned parietal bone constitutes the covering of the preparation, has contributed to a better understanding of the pathophysiological mechanisms in migraine. In its present form, only measurements of the middle meningeal artery (MMA) are performed. The aim of this study was, in addition, to measure pial artery/arteriole (PA) diameter and cortical cerebral blood flux in the same cranial window. The model was evaluated by studying the effects of hypotension and changes in arterial carbon dioxide pressure (PaCO2), because these parameters might influence the interpretation of pharmacological experiments. Out of 23 successful experiments it was possible to measure all three parameters in 19 animals. In four, PA diameter could not be measured, while MMA diameter and local cortical cerebral blood flux (LCBFFlux) always could. Haemorrhage-induced hypotension (-64 + 0.8 mmHg) caused an increase of MMA diameter of 11.8 + 8.4%, PA diameter of 61.2 + 7.7% and a decrease in LCBFFlux of - 36.4 + 2.5%. The decrease in blood pressure did not significantly change the MMA (
Introduction
The use and further development of animal cranial window techniques are important not only for the study of cerebral haemodynamics, but also for studies of pharmacological and basic mechanisms of migraine. Experimental migraine models should be as similar to existing experimental human models as possible, a requirement that seems to be fulfilled by the closed cranial window models. Two different variants have often been used, the ‘open cranial window model’ and the ‘artificial closed window model’(1–3). A genuine closed cranial window model has been developed, in which the parietal bone is grinded to transparency. It preserves the natural protection of the brain, does not affect intracranial pressure or brain tissue and the vessels are kept in their normal environment (4). The latter model has so far only been used to study the response of dural arteries. From human experiments, it is know that dilation of the middle cerebral artery is associated with migraine attacks (5) and marked changes in cortical cerebral blood flow take place during attacks of migraine with aura (6). It would therefore be an advantage if in addition to the middle meningeal artery (MMA) also pial artery/arteriole diameter changes as well as cortical cerebral blood flow could be measured in the same genuine closed cranial window. We describe and validate, in this study, such a modification of Williamson et al.'s genuine closed cranial window technique (4).
The neuropeptides and transmitters believed to play a causative role in migraine are primarily strong vasodilators and, when administered intravenously, could result in a pronounced decrease in blood pressure. Cerebral arteries and arterioles are autoregulated and hence dilate in the face of hypotension (7) but it is unknown if dural arteries autoregulate. The same is true for changes in arterial carbon dioxide pressure (PaCO2). In the interpretation of pharmacological studies, it is important to know how blood pressure and PaCO2 modify these parameters.
The aim of the present study was to assess the feasibility of measuring dural and pial arterial diameter and cortical cerebral blood flux (laser Doppler flowmetry) and to assess the effect of short-term hypotension as well as hypocapnia and hypercapnia in this closed cranial window model in rats.
Methods
Surgical preparation
All experimental procedures were performed in accordance with the domestic guidelines and regulations for animal care and treatment. The study protocol was approved by The Danish Animal Experimentation Inspectorate (file: 2001/561-390).
The experiments were conducted in male Sprague-Dawley rats (300–400 g), anaesthetized by intraperitonal pentobarbital (Mebumal®, 60 mg/kg) and maintained by continuous intravenous infusion of pentobarbital (Mebumal®, 20 mg/kg · h−1). Other combinations of anaesthetics were tried, including inhalation anaesthetics, before deciding on the combination of Mebumal® and N2O. This combination had the least effect on the cerebral blood flow and the cerebral arteries during long-term observation (unpublished data). A tracheotomy was performed and the animals were artificially ventilated (Abovent 7025; UGO Basil, Comerio, Italy) with 30/70% O2/N2O. Body temperature was kept at 37.0–37.5°C using an automatically regulated heating plate (Letica HB101; Panlab, Barcelona, Spain). The femoral arteries and veins were cannulated bilaterally with polythene catheters (Portex® polyethylene catheters ID 0.4; Astratech AS, Tåstrup, Denmark) for measurement of mean arterial blood pressure (MABP) (Transducer TCM4-7; World Precision Instruments, Sarasota, FL, USA), arterial blood samples, infusion of anaesthetics and withdrawal and reinfusion of blood. All measurements were continuously displayed on a computer monitor using data acquisition and analysis software (Perisoft® version 1; Perimed AB, Järfälla, Sweden). Arterial blood gas analysis (ABL520; Radiometer AS, Brønshøj, Denmark) was performed approximately three times during each study. PaCO2, PaO2 and pH were kept within normal limits by adjustments of respiration rate. The inspiratory and expiratory air was monitored by a capnograph (Capnomac AGM103; Datex-Ohmeda Division, Helsinki, Finland). The animal was placed in a stereotactic frame. Skin and connective tissue were removed from the dorsal side of the scull. The right parietal bone was thinned to transparency using a dental drill. During the drilling, cooling was carried out by local application of ice-cold isotonic saline.
Video microscopy
The obtained cranial window was covered with mineral oil (37°C). A branch of the MMA and a cortical cerebral artery/arteriole (pial) were visualized using a video-microscope (Sony DSP digital camera, MS50 objective; Sony Corporation, Tokyo, Japan) and the real-time image was displayed on a TV screen (4). Measurement of the vessel diameter was achieved by a video dimension analyser (V94; Living Systems Instrumentation Inc., Burlington, VT, USA). Local cerebral blood flux was measured continuously using laser Doppler flowmetry (LDF) (Perimed PF4001; Perimed AB). The probe (Perimed 410; fibre separation 0.25 mm, Perimed AB) was placed using micromanipulators and a microscope. The probe was placed above and without direct contact with the bone, but in touch with the mineral oil, which enhances the accuracy of the local cortical cerebral blood flux (LCBFFlux) measurement (3). The probe was placed in an area with no or few observable vessels. Control of the identification of the pial artery/arteriole (PA) was ensured by removal of the remaining bone and dura in some of the animals.
Experimental protocols
Effect of haemorrhage induced hypotension on cerebral artery diameter and LCBF
Flux
After preparation the rat was left to stabilize before beginning the protocol (1–1.5 h). Syringes flushed with heparinized saline, 10 IE/ml (1000 IE unpreserved heparin dissolved in 100 ml 0.9% NaCl) were used for the withdrawal of blood. Arterial blood was withdrawn steadily without any resistance. Approximately 0.5 ml blood induced a 10–20 mmHg decrease in MABP.
The blood withdrawals were performed successively with increasing intervals of 10 mmHg; when the effect on MABP stabilized the withdrawn blood was reinfused and the catheter flushed by 100 µl of saline. After all parameters had returned to baseline a time span of 5 min were inserted before the consecutive withdrawal. Effort was made to perform two protocols of six withdrawals in each animal with a 15-min pause between the two protocols.
Effect of hypocapnia and hypercapnia on cerebral pial artery diameter and LCBF
Flux
The hypocapnia was induced by hyperventilation, i.e. an increase in respiration rate from 70 to 130 strokes/min and a stroke volume increase from 4.0 to 6.0 ml, induced a decrease in PaCO2 of approximately 15 mmHg. Hypercapnia was induced by addition of 6% CO2. Upon stabilized parameters, the arterial gases were analysed, and when changes in MABP were stable another blood analysis was done. The protocol was repeated when all parameters had returned to baseline and repeated twice in each animal.
Statistical analysis
The diameter of the arteries and the LCBFFlux were measured in arbitrary units (AU) and MABP in mmHg. The maximum effect (peak) of the blood withdrawal and hypocapnia and hypercapnia on dural, pial diameter, and LCBFFlux was calculated as the percentage change from the prestimulation baseline, which was defined as the average of the 60 s preceding the stimulation. Dural and pial arteries were measured in arbitrary units, since the experimental set-up was dependent on the individual course of the arteries chosen for each experiment and a different magnification was used to optimize the image and analytic possibilities of the video dimension analyser. A standardized artery measurement would therefore be impractical. Data are presented as mean ± SEM. The significance level was chosen at 0.05. SPSS version 10.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis, GraphPrism® (GraphPad Software Inc., San Diego, CA, USA) for construction of the graphs. The statistical analysis of the relation between the applied protocol (blood withdrawal, hypocapnia or hypercapnia) and the measured parameters (MABP, vessel diameter and LCBFFlux) was performed using linear regression and a general linear model with the applied protocol as the covariate factor, the measured parameter as depending factor and the difference within the group of animals as a random factor.
The control of consistent baseline and arterial gases (only for the haemorrhage protocol) was done using a univariate analysis of variance with repeated measurements. Animal and number of stimulation or gas analyses was chosen as main factors. If significance was found a
Results
Twenty-three rats were included in the study. The following exclusion criteria were applied: bleeding beneath the skull due to the drilling, resistance in withdrawal of blood that could not be overcome by flushing and adjustment, unstable baseline measurements and significant changes in blood gases. Of the measured parameters only invalid MABP measurements (e.g. dysfunctional artery catheter) could exclude an animal. All four parameters could be measured in 19 animals. In four animals the diameter of the cortical pial vessels could not be accessed; MMA diameter and LCBFFlux could be measured in all animals.
Effect of haemorrhage-induced hypotension on cerebral artery diameter and on LCBF
Flux
The protocol was applied successfully in 14 animals. The MMA and PA diameters varied between 36.3 and 52.5 αU and 63.0 and 75.2 αU, respectively. The mean baseline LCBFFlux and MABP were 102 ± 3.8 AU and 117.1 ± 1.4 mmHg. The prestimulation baseline did not change significantly between blood withdrawals for MABP (
Moderate hypotension did not significantly alter MMA diameter (
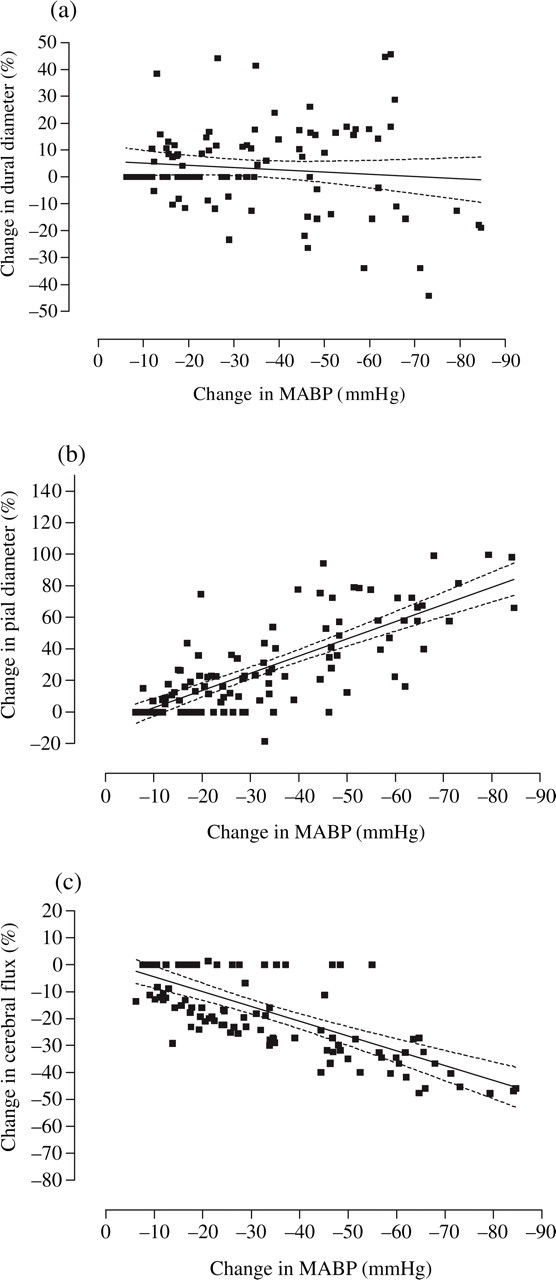
(a–c) Per cent changes in middle meningeal artery (MMA) (a) and pial artery/arteriole (PA) (b) diameter and local cortical cerebral blood flux (LCBFFlux) (c) are plotted against degree of induced hypotension (mmHg). In each of the graphs the fitted regression line with 95% confidence interval is displayed.
Effect of arterial hypotension on the measured parameters

Changes in the measured parameters are given as percentage change from baseline and are listed in hypotensive groups of 10 mmHg (Mean + SEM).
Effect of hypocapnia and hypercapnia on cerebral artery diameter and on LCBF
Flux
Hyperventilation and induced hypercapnia were performed successfully in nine animals. The MMA diameter varied between 34.2 and 43.3 AU and the PA diameter between 62.1 and 67.7 AU. Baseline and peak values are shown in Table 2. Scatter plots of the results are presented in Fig. 2a–c. Comparison between resting values of arterial blood gases did not reveal any significant difference. Both hyperventilation and increased CO2 caused a short-lasting decrease of MABP followed by a return to baseline. Measurements of the change in blood vessel diameter and LCBFFlux were performed after MABP had returned to baseline.

(a–c) The effect of hypocapnia and hypercapnia (change in mmHg) is displayed for each measured parameter except mean arterial blood pressure (MABP). Middle meningeal artery (MMA) (a) and pial artery/arteriole (PA) (b) diameter and local cortical cerebral blood flux (LCBFFlux) (c) are in percentage change.
Effect of hypocapnia and hypercapnia on the measured parameters

Normocapnic (baseline) values and peak hypocapnic and hypercapnic values and the CO2-reactivity index for the measured variables are shown (Mean + SEM). PaCO2, Arterial carbon dioxide pressure; MABP, mean arterial blood pressure; MMA, middle meningeal artery; PA, pial artery/arteriole; LCBFFlux, local cortical cerebral blood flux.
Hypocapnia contracted both MMA (−45.0 ± 4.8%,
Discussion
The genuine closed cranial window model was introduced to the field of migraine research by Williamson et al. (4) and has subsequently contributed significantly to our understanding of migraine mechanisms (8–10). We have modified and expanded the model to a closer resemblance to existing experimental human migraine models in which both cerebral blood flow and middle cerebral artery blood flow velocity are measured (5, 11). In the original version of the animal model only dural vasodilation was examined. Our data showed that it is possible in the same closed cranial window to measure dural as well as pial arterial diameter and laser Doppler determined cortical cerebral blood flux. Data on PA and LCBFFlux with pharmacologically and haemorrhage-induced hypotension are available (7, 12–14). However, data on the reactivity to these stimuli of dural vessels are limited (4). We showed that acute haemorrhage-induced hypotension did not significantly affect the diameter of the MMA, while the PA increased in diameter and LCBFFlux decreased. Hypercapnia and hypocapnia increased and decreased the diameter of both arteries and LCBFFlux.
The dura mater encephali of the rat is translucent (2–5 µm thick) and consists mostly of collagen fibres. The amount of blood vessels is limited, but some are relatively large such as the MMA (15). Dura mater is a pain-sensitive structure richly innervated by sensory C-fibres containing vasoactive peptides, e.g. calcitonin gene-related peptide (CGRP) and substance P (16–18). Increase in meningeal blood flow and MMA diameter can be induced by either electrical stimulation or application of vasoactive peptides (4, 19). The effect of haemorrhage-induced hypotension has to our knowledge not been studied before. We found that MMA was not significantly affected and hence, vasodilation upon intravenous administration of drugs that cause hypotension, e.g. CGRP, is not secondary to induced MABP decrease, but to a direct effect of CGRP on MMA.
Previous studies have shown that rat pial arteries and arterioles increase in diameter upon induction of hypotension, an increase correlated with the degree of hypotension (13, 14). Our results on the reactivity of these vessels to hypotension are in accordance with existing data (7, 13). The effect of blood pressure changes is thus important to separate from a direct effect on pial arteries in pharmacological experiments.
Both pial arteries and arterioles contribute to the maintenance of a constant cerebral blood flow within the lower and upper limit of the autoregulation, 60–160 mmHg (20). Each type of vessel contributes with approximately 40% of the total vascular resistance (21). In cats, a hierarchy of responsiveness to changes in MABP can be described for the different sizes of arterioles (7), but a similar organization could not be found in rats (21). The inner diameter of the cortical arterioles in the latter study were not larger than 60–70 µm, an approximate size also found by others (22). The pial vessels investigated in the present study had a similar diameter [60–75 µm (inner diameter)], indicating that the superficial cortical cerebral pial arterioles that can be measured in a cranial window model do not usually exceed 75 µm and can be classified as first and second order arterioles, 30–100 µm (21).
We measured cerebral blood flow using LDF. The method has been validated in both artificial and genuine closed window models for the measurement of CBF and correlates with data obtained from 14C-iodoantipyrine and hydrogen clearance methods (23, 24). Comparing an artificial and genuine closed window model in rats revealed no significant difference in the LCBFFlux increase induced by whisker stimulation (25). In comparison with the few blood vessels in the dura and this finding, the contribution of the overlying structures are believed to be of minor importance (26). In the present study LCBFFlux correlated with the degree of induced hypotension. To mimic pharmacological experiments the blood pressure was lowered at a fast rate of − 1.57 ± 0.13 mmHg/s. The maximal withdrawal rate for the maintenance of a preserved autoregulation is approximately − 0.4 mmHg/s (20). Furthermore, the duration of the withdrawal in the present study (approximately 1 min) was not sufficient to elucidate the autoregulatory capacity. Therefore, although tempting due to a different primary aim and design, this parameter could not be assessed in the present study. It is however possible to measure LCBFFlux in the genuine closed cranial window, although bone and dural circulation may disturb the overall estimation.
The sensitivity of cerebral arteries and CBF to hypocapnia and hypercapnia is well established. The vasoconstrictor effect of hypocapnia (27, 28) on PA and LCBFFlux was confirmed in our experiments. Hypercapnia dilated the cerebral arteries and increased LCBFFlux, as in previous studies (27, 29). The CO2-reactivity index (Table 2) and effect of hypercapnia on CBF was higher than reported previously (24) and might have been affected by the choice of anaesthetic.
The effect of PaCO2 changes on the MMA has not been assessed in detail in previous experiments using the genuine closed cranial window (4, 19, 25). In the present study, the MMA reacted by a significant increase in diameter to hypercapnia and by decrease to hypocapnia. Since PaCO2 changes affect the diameter of PA and MMA as well as LCBFFlux, it is essential to keep arterial blood gasses within normal limits in experiments with the genuine closed cranial window model.
In conclusion, our study has shown that it is possible to measure the diameter of a pial arteriole as well as cortical cerebral blood flux, in addition to the diameter of the middle meningeal artery in a genuine closed cranial window. With this modification, the model closely resembles established experimental human migraine models. Our results show that PaCO2 must be tightly controlled and that the effect of blood pressure changes must be known to interpret pharmacological experiments in this model.
