Abstract
Studies performed in selected populations show that the use of triptans for migraine is low. Our study was aimed at establishing patterns of triptan utilization in a large community using the drug prescription database of a regional Health Authority in Italy. In a population of 224 065 residents, 0.55% received at least one prescription of triptans in 1 year: 77.9% were female and 22.1% male. Oral dosage forms accounted for 94% of prescriptions. About 60% of patients received a single prescription (containing one or two packages) of one triptan in 1 year. Age distribution showed that 7% of patients were aged >65 years. They received 14% of packages, prevalently sumatriptan and zolmitriptan (the two triptans with the longest commercialization in Italy); 5.7% of patients received 40% of packages. Moreover, 3.2% of triptans users received >120 dosage units in the year in the form of tablets (>10 single doses/month), and were potential triptan abusers. Our data indicate suboptimal treatment of migraine patients and also incorrect treatment of some patients (potential triptans abusers, the elderly).
Introduction
Migraine remains undiagnosed and undertreated in at least 50% of patients, and < 50% of migraine patients consult a physician (1, 2). Less than 50% of migraineurs are not recognized by their general practitioner (GP) as having migraine and <30% of these have management of migraine (3). Possible causes of undiagnosis and undermanagement by GPs are: insufficient time spent with the patient, complexity of diagnostic criteria of the International Headache Society (IHS) (4), variability of the clinical manifestation of headache in migraine patients and high utilization of over-the-counter (OTC) drugs (2, 5, 6). Studies performed in selected populations show that triptans use is low (3–19% of migraine patients) in various countries, particularly in Italy, where triptans had been prescribed in only 3% of migraine patients (2).
The efficacy of triptans has been shown in several randomized, double-blind, placebo-controlled trials (7). However, these studies were carried out in selected populations and under controlled conditions. Utilization patterns of specific antimigraine drugs in community patients and the experiences of patients with these drugs in real life settings have been studied less extensively (8). In this study, data were collected to establish the patterns of use of triptans in a large community.
Materials and methods
Under the reimbursement system of the Italian National Health Service, all prescribed drugs included within the essential level of assistance (LEA) are recorded by Regional Health Authorities in association with the demographic characteristics of patients. Therefore, the medication records of individual patients in drugs prescription databases are quite complete. This allows accurate investigation of drug utilization in our setting. The patterns of triptan prescription in the population of Health Authority 11 of Tuscany were investigated. The prescription database was analysed using the Anatomical Therapeutic Chemical classification (NO2CC: triptans) dispensed during 2005.
In Italy each triptan prescription could contain one or two packages.
In 2005, all triptans (except naratriptan) were available as oral tablets in Italy, for rizatriptan and zolmitriptan as soluble oral tablets, for sumatriptan as subcutaneous injections, nasal spray and rectal suppositories.
Results
For a total population of 224 065 residents of Health Authority 11, the total number of triptan packages prescribed was 8982 (Fig. 1).
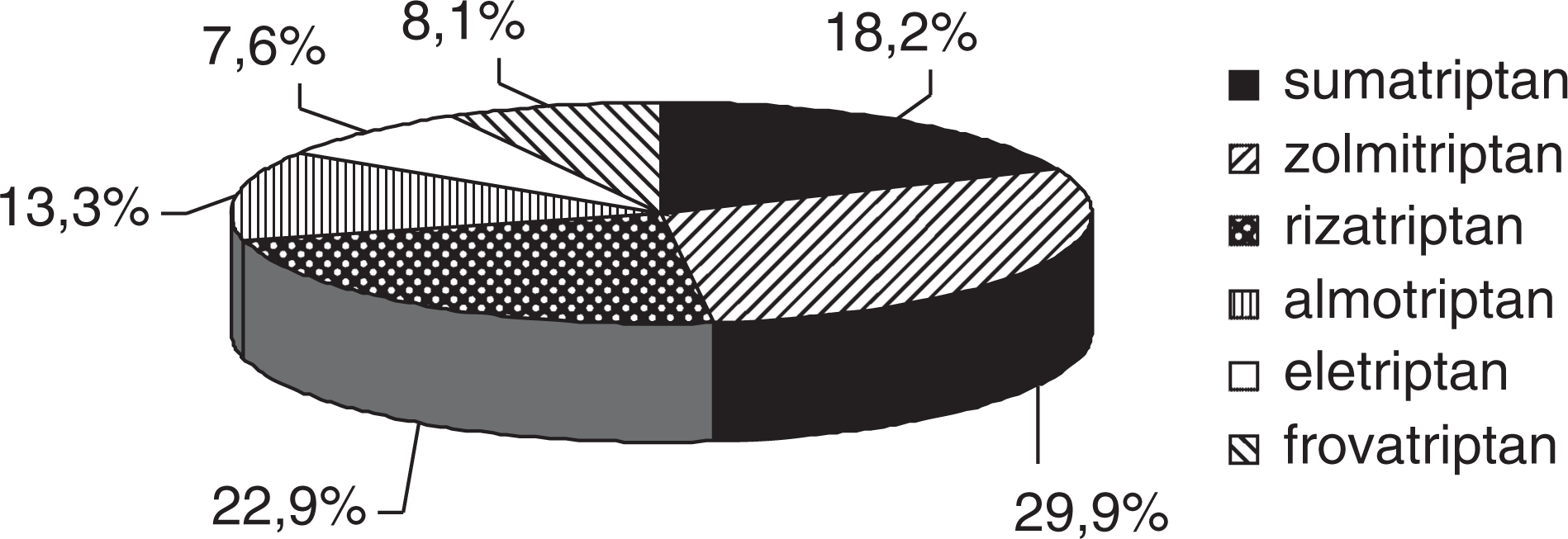
Percentage of 8982 packages of triptans prescribed during 2005.
Oral tablets accounted for 5954 packages (66.3%), soluble oral tablets for 2486 (27.7%), subcutaneous injection for 316 (3.5%), nasal sprays for 147 (1.6%) and rectal suppositories for 79 (0.9%) (Fig. 2).

Pharmaceutical forms of 8982 packages of triptans prescribed during 2005.
In our database, the prescriptions of 455 packages (5% of the total amount) were incomplete in demographic data and were excluded from this study. The percentage of packages excluded was almost uniformly distributed between all triptans, average (2.6–8%). Thus, the following analyses were conducted on 8527 packages prescribed and 28 067 dispensed daily dosages (DDDs) (Table 1).
Number of treated patients, number of packages, mean defined daily dosage (DDD)/patient/month (intensity of drug use)

At least one prescription (containing one or two packages) of one triptan was dispensed during 2005 to 1238 patients (0.55% of the population). The percentage of triptan users may be slightly larger: in the case that 5% of packages with incomplete demographic data were prescribed to different patients, this percentage may rise to 0.75%. However, the percentage of triptan users could be even smaller, because in our analysis it was not possible to determine if each patient took one or more type of triptans. Of these patients, 964 were female (77.9%) and 274 male (22.1%), with a female:male ratio of 3.5 : 1 (Table 2). Evidence on age distribution showed that 7.6% of triptan users are >65 years old. We found an average of 6.9 packages per patient per year.
Age and sex distribution of triptan users
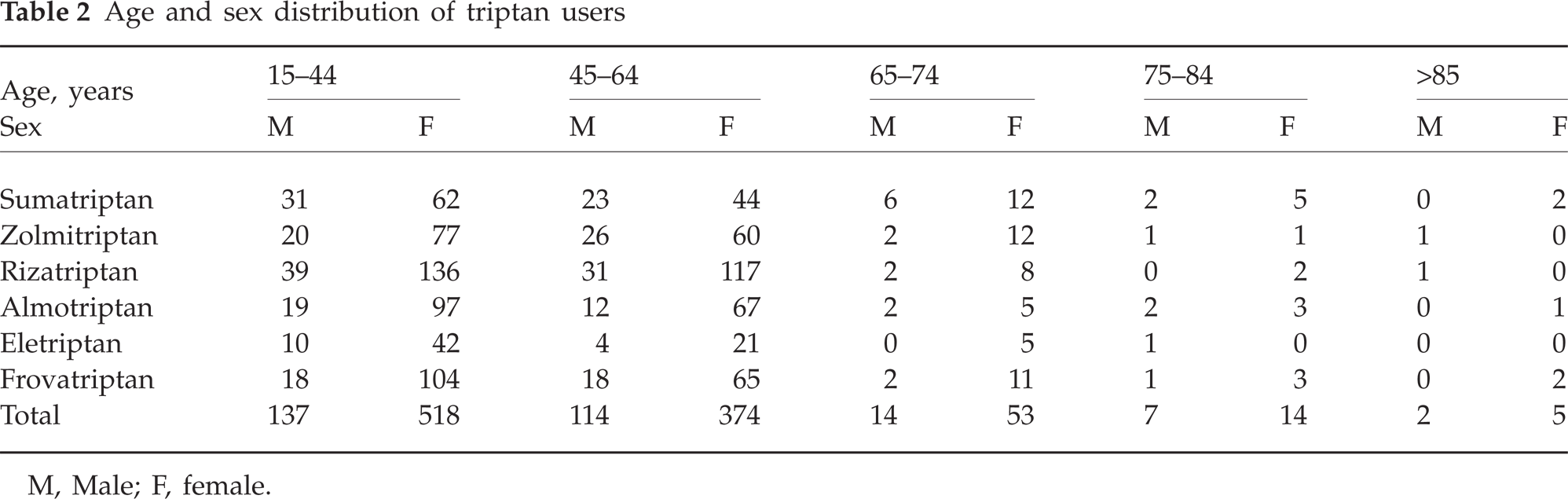
M, Male; F, female.
Of the 8527 packages, 6902 (81%) were prescribed to female and 1625 to male (19%) patients, with a female:male ratio of 4.2 : 1 (Table 3). Patients aged >65 years were dispensed 1199 of 8527 packages (14%), and 1124 of these (94%) were female. Moreover, 924 of 1199 packages prescribed to over 65-year-olds were sumatriptan and zolmitriptan; these are the two triptans with the longest period of commercialization in Italy. Only one to two packages were dispensed to 714 patients (57.7%) and one to six packages to 920 (74%) (Tables 4 and 5). We identified 40 (31 female, nine male) patients (3.2%), mean age 53.8 years (SD = 13.4), who received >120 dosage units in the form of tablets in the year (>10 single doses/month) and 12 patients (0.9%) who received >216 (>18 single doses/month); 5.7% of patients received 40% of packages and 44% of DDDs.
Age and sex distribution of packages prescribed
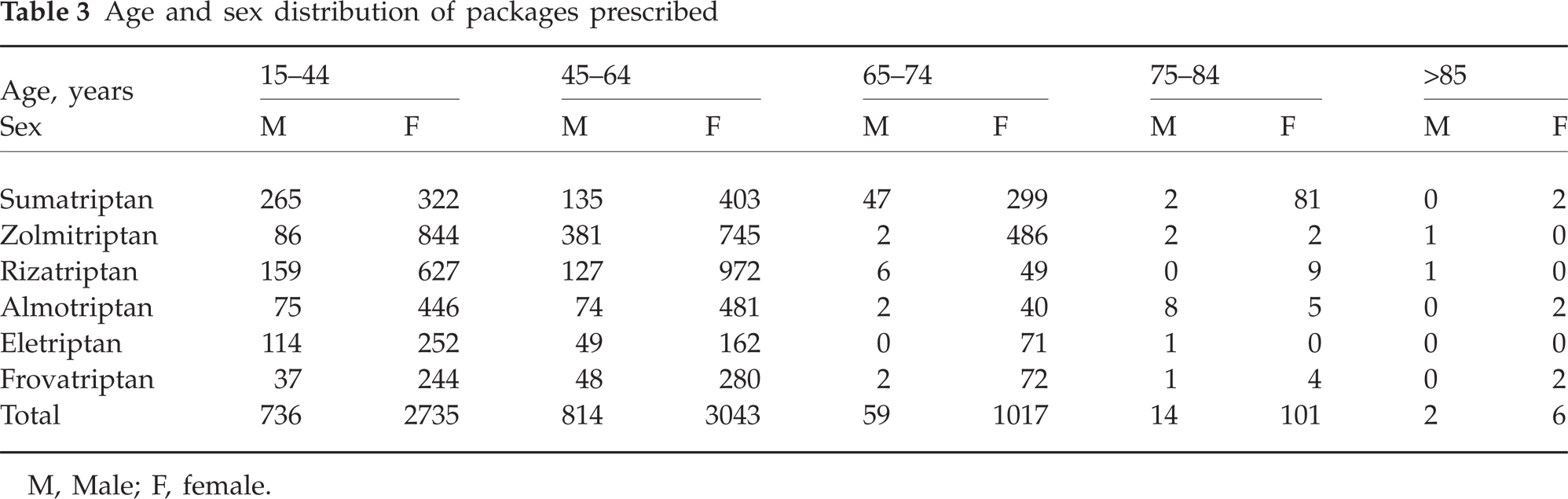
M, Male; F, female.
Number of male triptan users according to the number of packages prescribed

Number of females triptan users according to the number of packages prescribed

Discussion
In this study it was found that 0.55% of the residents in our district received at least one prescription of a triptan in 1 year. Based on the reported migraine prevalence in Italy of 12% (2, 9), only 6% of our patients received specific triptan antimigraine drugs; these data are similar to those found in other epidemiological studies (2). Another study has shown that 1.4% of the population utilizes specific antimigraine drugs (triptans and ergotamine), with a female:male ratio exactly the same as in our study (3.5 : 1) (8). This percentage of triptan utilization is very low and is perhaps due to a high number of migraine patients who remain undiagnosed and/or undertreated.
The number of patients (about 60%) who received only one prescription (containing one or two packages) in 1 year is larger than the 40% reported in previous studies (8, 10, 11). However, prescriptions in Italy could contain one or two packages, and it was not possible to separate out the single patient receiving only one package, as in the cited studies. The high percentage of single prescriptions of triptans could indicate that many migraine patients had a low frequency of attacks. In fact, it has been reported that only 20% patients had >14 migraine days per year (12). Another explanation of the low percentage and frequency of triptan use is that many migraine patients cannot be completely satisfied by triptan therapy with regard to efficacy and/or side-effects; in these situations, patients prefer other usual medications such as OTC or non-steroidal anti-inflammatory drugs (NSAIDs). Although triptans are undoubtedly very efficacious for migraine attack, a clear superiority over NSAIDs in comparative studies did not appear as a primary end-point (13). A recent study has indeed shown superiority of an OTC drug over sumatriptan (14).
Oral formulations account for the majority (94%) of packages dispensed, probably because of patients' and/or doctors' preference for the oral route of administration. This percentage is higher than the 77% found in a previous study (8).
Some data of our epidemiological study on triptan utilization merit further examination. First, 7% of triptan users and 14% of packages were prescribed for subjects aged >65 years: this is a population at major risk of hypertension and cardiovascular diseases. Moreover, 77% of the packages prescribed for the over 65-year-olds were sumatriptan and zolmitriptan, the two triptans with the longest commercialization in Italy. Sumatriptan users aged >65 years constituted 14.4% and utilized 27.7% of all sumatriptan packages. A previous study has shown that only 3% were sumaptriptan users aged >65 years (15). It should be remembered that the use of sumatriptan in patients aged >65 years is contraindicated. Furthermore, a low percentage of triptan users (5%) utilized a high percentage (40%) of the total amount of the prescribed triptans.
Ten single doses per month has been reported as the lowest critical frequency of intake in patients with triptan-induced medication overuse headache (4). In our study 40 patients (3.5%) exceeded this limit because they received >120 dosage units in 1 year. These patients cannot be classified as suffering from triptan-overuse headache (8.2.2 of the IHS classification) because their clinical characteristics (headache present on >15 days/month, with frequency increased during triptan overuse and impaired within 2 months after triptan discontinuation) and the number of days with triptan intake are not known. However, they are probably at risk of developing triptan-overuse headache or, rather, they have already developed this chronic headache. If the critical frequency of intake for patients with medication overuse headache is reported to be 18 single doses of triptans per month (16), 12 out of our 40 patients consumed >216 single doses per year.
In conclusion, our study has shown very low triptans utilization in the population, which was sometime skewed because of the frequent use in the elderly and a large dosage utilization by a few subjects (triptan abusers). The main problem is that most pharmacists and GPs are neither familiar with nor use migraine therapies endorsed by evidence-based guidelines (17, 18). However, an intensive pharmaceutical care campaign targeting overuse with a history of chronic substance use and inappropriate use of triptans failed to reduce their consumption (19).
