Abstract
The objective of this study was, first, to characterize the usage patterns of specific antimigraine drugs, and second, to compare these in patients with one type of drug with patients with two or more types of drug. Dispensing data on triptans and ergotamine were collected from community pharmacy records over a 1-year period. In a population of approximately 168 000 specific antimigraine medication has been dispensed to 2343 patients (1.4%). Oral dosage forms were prescribed in most prescriptions (77.1%), subcutaneous injections in 9.7%, rectal suppositories in 7.1% and nasal sprays in 6.1%. We identified 292 patients (12.5%) to whom more than one type of drug was dispensed. Multiple drugs patients showed significantly higher drug consumption and deviating patterns of specific antimigraine drug usage, receiving significantly more non-oral dosage forms (32.8% vs. 20.4%, P < 0.001). Our data indicate substantially suboptimal treatment of migraine patients.
Introduction
Estimates of 1-year migraine prevalence in the general population in different European countries and the USA vary from 15% to 18% among women, and 6% among men, overall around 11% (1). Migraine is not only a highly prevalent, but also a very disabling disease (2, 3). Moreover, migraine remains undiagnosed in many patients and treatment is often not optimal (4). The economic burden of migraine to society is also huge (5). New antimigraine drugs became available during the last decade of the past century, opening new therapeutic options for the migraine patient. Sumatriptan was introduced in 1991 and subsequently six other triptans were introduced. Also several new pharmaceutical dosage forms for triptans have reached the market.
The efficacy of triptans as an acute antimigraine treatment has been shown in numerous randomized, double-blind, placebo-controlled trials (6). However, these studies were executed in selected populations under well-controlled conditions. The usage patterns of specific antimigraine drugs in patients and the experiences of patients with these drugs in real life have been studied less extensively. In this study some data were collected to establish the patterns of use of triptans and ergotamine, with special emphasis on patients using more than one specific antimigraine drug in a 1-year period.
In a subsequent study these multiple drugs patients will be questioned to establish the reasons why they kept to their medication, switched to other products or used more than one specific antimigraine drug at the time. The results of this study will be reported later.
Methods
As a result of the reimbursement structure for prescription drugs in the Netherlands, the patient–pharmacy commitment is high and the prescription medication information for each patient in pharmacy records is virtually complete. All 18 community pharmacies in the region around the Maasland Hospital in Sittard participated in the study. The anonymous prescription data of drugs with ATC classification (Anatomical Therapeutic Chemical classification) N02C (‘Anti-migraine drugs’), dispensed in the period 1 October 2001 to 30 September 2002, were provided. Only the data on the use of triptans and ergotamine were selected from these records. The local Ethics Committee approved the study.
The prescription data included: patient identification number, date of birth, sex, dispensing date, drug product, quantity of drug dispensed, and prescribing physician.
Demographic data were obtained from the Dutch Central Bureau of Statistics (Statistics Netherlands CBS, Voorburg/Heerlen, the Netherlands).
Multiple drugs patients were defined as patients to whom more than one type of specific antimigraine drug had been dispensed. Patients with different dosage forms or different strengths of the same drug were also considered as multiple drugs patients.
χ2 statistic with Yates’ correction, standard t-test and a non-parametric test (Mann–Whitney U-test) were used to determine the significance of differences between multiple drugs patients and single drug patients.
Results
The results are summarized in Table 1 and Figs 1–5. The 18 community pharmacies took care of a total population consisting of approximately 168 000 people. In the pharmacy records 2343 (1.4%) patients were identified to whom specific antimigraine medication had been dispensed in the period from 1 October 2001 to 30 September 2002. Of these patients 1843 (78%) were female and 509 (22%) were male. In age distribution a maximum was found in the group of 41–45 years of age both for females and males.
Patients with prescriptions for specific antimigraine drugs in a total population of approximately 168 000 people in a period of 1 year

Multiple drugs patients are those to whom more than one type of specific antimigraine drug has been dispensed in a period of 1 year.
Significantly different from single-drug patients, P < 0.001.
The total number of prescriptions for specific antimigraine drugs was 7542, a mean of 3.2 prescriptions per patient per year. Only one prescription in the 1-year study period was dispensed to 986 patients (42%). We found that 117 patients (5.0%) has received more than 120 dosage units of specific antimigraine drugs (>10 single doses/month), and 45 patients (1.9%) more than 216 (>18 single doses/month).
Oral tablets or capsules were prescribed in most prescriptions (54.0%), orally disintegrating tablets in 23.1%, subcutaneous injections in 9.7%, rectal suppositories in 7.1% and nasal sprays in 6.1% (Fig. 1). Oral tablets or capsules are available in the Netherlands for all specific antimigraine drugs, orally disintegrating tablets for rizatriptan and zolmitriptan, subcutaneous injections and nasal sprays only for sumatriptan, and rectal suppositories for ergotamine and sumatriptan. More than half (52.1%) of the prescriptions were for sumatriptan, 26.1% for rizatriptan, 8.2% for zolmitriptan and 7.6% for naratriptan. Eletriptan and almotriptan, which were not available at the start of the study period and became so only after 2 or 3 months, had a prescription frequency of < 1% (Fig. 2). Ergotamine was prescribed in 4.6%, and the mean number of dosage units per prescription was 20.9. This was significantly higher than for the triptans with mean numbers of dosage units around 10 per prescription, ranging from 6.2 for almotriptan to 13.2 for zolmitriptan (Fig. 3).
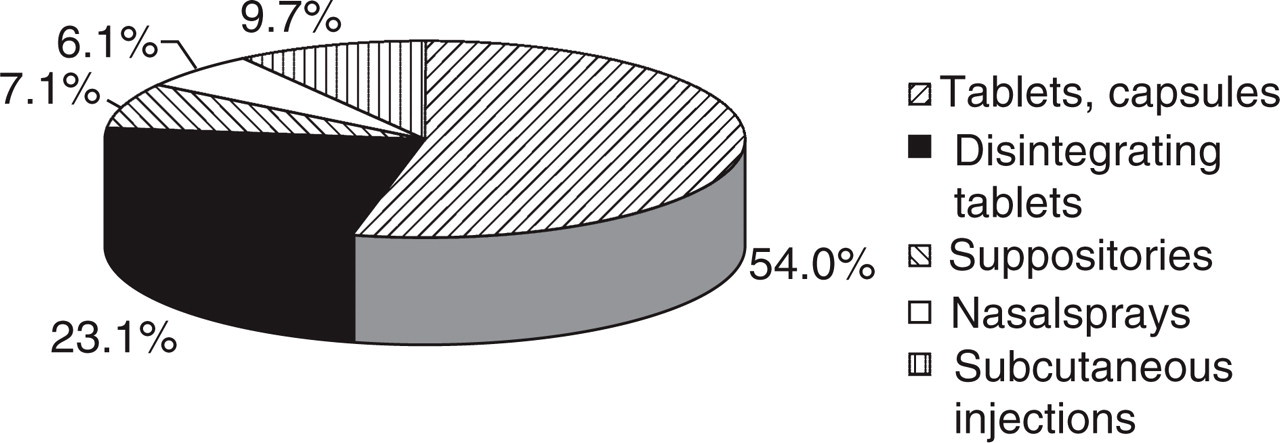
Pharmaceutical forms of 7542 prescriptions for acute specific antimigraine drugs.

Substances prescribed in 7542 prescriptions for acute specific antimigraine drugs.
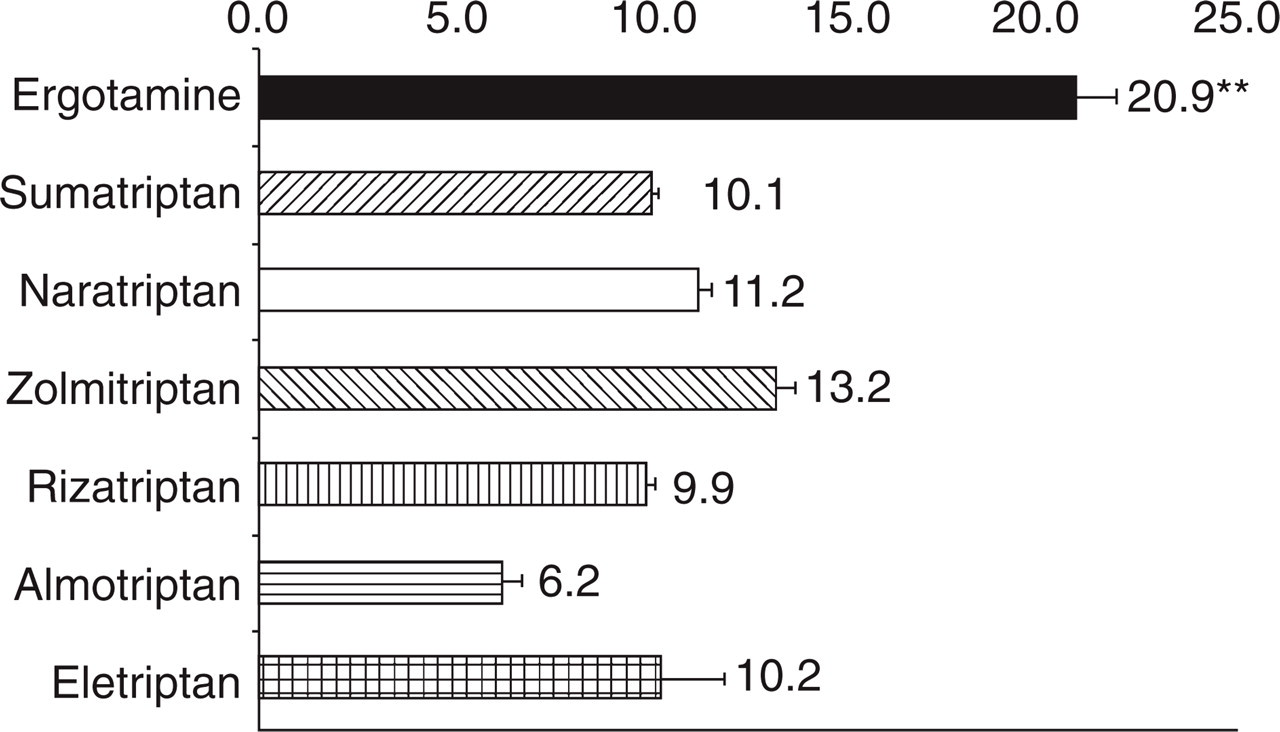
Number of dosage units per prescription (mean ± standard error; ∗P < 0.001 vs. all other drugs).
We identified 292 patients (12.5%) with more than one drug product (‘multiple drugs patients’). These multiple drugs patients received 25.2% of the total number of dosage units prescribed in 25.6% of the prescriptions (Table 1). Mean number of prescriptions per year was 6.6 vs. 2.7 for patients to whom only one type of drug has been prescribed (‘single drug patients’, P < 0.001). Also the mean number of dosage units per year was significantly higher: 70.7 vs. 29.8 (P < 0.001). More than 120 dosage units were dispensed to 38 patients. This was more than in single-drug patients (13.0% vs. 3.9%, P < 0.001). The number of multiple drugs patients receiving more than 216 dosage units was 19, also more than in single-drug patients (6.5% vs. 1.3%, P < 0.001). Relatively fewer patients were in the age group 16–35 years and relatively more in the age group 41–50 years compared with single-drug patients (P < 0.001). These multiple drugs patients used significantly more subcutaneous injections, nasal sprays and suppositories, and fewer orally disintegrating tablets (Fig. 4). Also sumatriptan, eletriptan and almotriptan were used significantly more in these patients, and rizatriptan and naratriptan significantly less (Fig. 5).

Relative use of pharmaceutical dosage forms in single and multiple drugs patients (∗P < 0.05; ∗∗P < 0.001 vs. single-drug patients).

Relative use of substances in single and multiple drugs patients (∗P < 0.05; ∗∗P < 0.001 vs. single-drug patients).
Discussion
Although 1-year migraine prevalence in the general population is reported to be around 11% (1, 2), only 1.4% of the population in the present study had received specific antimigraine drugs. Assuming that the migraine prevalence in the study population does not differ from that in the general population, it can be calculated that only about 13% of migraine patients in the study population are treated with specific antimigraine drugs. Though some patients may be perfectly satisfied using non-specific antimigraine treatment, we consider this a very low percentage probably reflecting a high degree of migraine patients who remain undiagnosed and/or are undertreated.
The female : male ratio (3.5 : 1), and age distribution were virtually the same as in other studies (1, 4). The high proportion of patients (42%) who had only one prescription in the 1-year study period has also been reported previously (7, 8). This could indicate these patients had a low number of migraine attacks per year, were not satisfied with their medication, and/or were not diagnosed correctly (9).
Ten single doses per month has been reported as the lowest critical intake frequency in patients with triptan-induced medication overuse headache (10). We found that 5.0% had exceeded this limit. In multiple drugs patients even 13.0% had received more than 120 dosage units, equal to 10 single doses/month. The mean critical intake frequency in patients with medication overuse headache was 18 single doses of triptans per month (10). Forty-five patients (1.3%) received more than this limit, 19 (6.5%) in the group of multiple drugs patients. These patients are probably in danger of developing medication overuse headache or may already have developed such a chronic headache. However, these figures may be an overestimation because it remains unknown how many of these dosage units were actually taken by the patients. Moreover, some patients may have used these drugs for other indications such as cluster headache, requiring higher dosing. Since the indication is not mentioned on the prescription the number of these patients in the study population is unknown. According to the new classification of headache disorders one of the diagnostic criteria for ergotamine or triptan overuse headache is drug intake on ≥ 10 days/month on a regular basis for ≥ 3 months (11). The proportion of patients in our study meeting this criterion cannot be established because the number of days with drug intake was not recorded.
Nausea and vomiting are common accompanying symptoms in migraine attacks and gastric stasis during a migraine attack may seriously disturb the oral absorption of acute antimigraine drugs (1, 12). Therefore, in many cases oral drug treatment may not be the administration route of first choice. Nevertheless, oral dosage forms were prescribed in the vast majority of prescriptions (77%), compared with the non-oral forms such as subcutaneous injections, rectal suppositories or nasal sprays (Fig. 1), probably reflecting patients’ and/or prescribers’ preference for the oral route of administration.
Since 1996 sumatriptan tablets and the other subsequently introduced triptans have been fully reimbursed in the Netherlands. Until that time ergotamine was the drug of first choice (8). In our study population ergotamine has only a modest place with a prescription frequency of 4.6%. Sumatriptan appeared to be the specific antimigraine drug of first choice, followed by rizatriptan, zolmitriptan and naratriptan. Two new triptans, almotriptan and eletriptan, were marketed during the 1-year study period and only a few prescriptions for these drugs were registered.
The mean number of dosage forms per prescription was about 10 for the triptans (Fig. 3). In a nationwide database of 6 million people similar data were found: 9.1–14.0 defined daily dosages per prescription for the triptans in 2001 (personal communication, F. Dekkers, Genees-en Hulpmiddelen Informatie Project/College voor Zorgverzekeringen, Diemen, the Netherlands). For ergotamine we found a substantially higher mean number of dosage units per prescription (20.9) than for the triptans, where the nationwide database showed 6.6 defined daily dosages per prescription for ergotamine-containing drugs. This discrepancy may partly be explained by the relatively high defined daily dosage for ergotamine of 4 mg, whereas the ergotamine drugs contain 1 or 2 mg per dosage unit. We have expressed these data as dosage units per prescription rather than defined daily dosages per prescription because we think this is more meaningful for acute antimigraine drugs. The similarity of our data to those of the nationwide database suggests that data found in the study population may possibly be generalized for the total population in the Netherlands.
We identified 12.5% as ‘multiple drugs patients’. These patients accounted for about 25% of the total number of prescriptions for specific antimigraine drugs and about 25% of the number of dosage units. Therefore, it can be concluded that these multiple drugs patients received about twice as many prescriptions and dosage units compared with the total study population and even 2.4 times as many compared with the single-drug patients (Table 1). The dosage forms used by these multiple drugs patients, with substantially more subcutaneous injections, more nasal sprays and suppositories compared with the single-drug patients, indicate these patients had migraine attacks which were more difficult to treat (Fig. 4). The increased use of the newest triptans, eletriptan and almotriptan, also support this view. In a subsequent study these patients will be questioned to discover the reasons for these changes in the patterns of use of specific antimigraine drugs and why they used more than one specific antimigraine drug.
Footnotes
Appendix
Co-operating Pharmacists Sittard-Geleen and its environs (Pharmacist, Pharmacy, Location): P. H. M. Paques, Apotheek De Baandert, Sittard; L. I. van der Ven, Apotheek Bloemen, Sittard; F. Sikkes, Apotheek Born, Born; A. J. W. Brunnekreeft-de Brouwer, S. G. J. Buijzen, Apotheek Buijzen, Sittard; A. M. Soeterboek, Apotheek Elsloo, Elsloo; J. A. M. H. Backbier, Apotheek Dr van Kempen, Sittard; M. J. J. Gels, Apotheek Kluis, Geleen; M. J. J. M. Rieter, Apotheek Limbrichterveld, Sittard; Y. H. M. I. Brounts, Apotheek Lindenheuvel, Geleen; L. P. Rutten, Lucas Apotheek, Geleen; B. W. G. Martens, Apotheek Martens, Beek; V. S. J. de Warrimont, Apotheek Neerbeek, Beek; V. la Fontaine, F. H. T. Hoogenberk, Apotheek Oud Geleen, Geleen; P. M. J. Stassen, H. J. L. M. Pieters, Apotheek Pieters, Stein; M. M. van der Kuy-de Ree, P-H. M. van der Kuy, Apotheek Susteren, Susteren; J. W. H. G. Boelens, Apotheek Timmers, Sittard; R. Hillen, Apotheek Voerendaal, Voerendaal; A. F. C. M. Custers, Apotheek Zuidhof, Geleen.
