Abstract
Studies performed in selected populations have shown a poor utilization of triptans for migraine. Our study was aimed at establishing patterns of triptans utilization in a large community using the pharmaceutical prescriptions database of two consecutive years in a regional Health Authority in Italy. About 0.5% of the population observed received triptans prescriptions in a year, but > 50% of the cases received only one prescription. On the other hand, 46% of triptan users did not receive a triptan prescription in the following year (past users): in 80% of cases, patients received only 1–2 triptan packages. The evaluation of the discontinued triptan type has shown percentages varying between 30 and 70%. The percentage of triptan users who received a triptan prescription for the first time in the successive year of study (new users) was 52%. These findings together highlight a high turnover in triptans utilization. Less than 15% of subjects received more than one triptan product in the 2 years. In conclusion, we observed a low percentage of triptan users and a low rate of utilization, associated with a high percentage of discontinuation and new utilization (high turnover), without any substantial increase in triptans utilization during the years. All these data probably do not support optimal satisfaction with triptan therapy.
Introduction
Since the commercialization of the new antimigraine drug sumatriptan about 20 years ago, scientific societies, pharmaceutical companies and headache specialists have tried to highlight migraine pathology in the population, arguing that it is probably underestimated. In the following years many other triptans have become available.
The average prevalence of migraine worldwide has been reported to be 10% (1). The efficacy of triptans has been shown in several double-blind placebo-controlled trials (2). A higher efficacy of triptans in comparison with ergotamine and non-steroidal anti-inflammatory drugs (NSAIDs) has also been reported (3,4), supporting triptan use as the gold standard in migraine treatment.
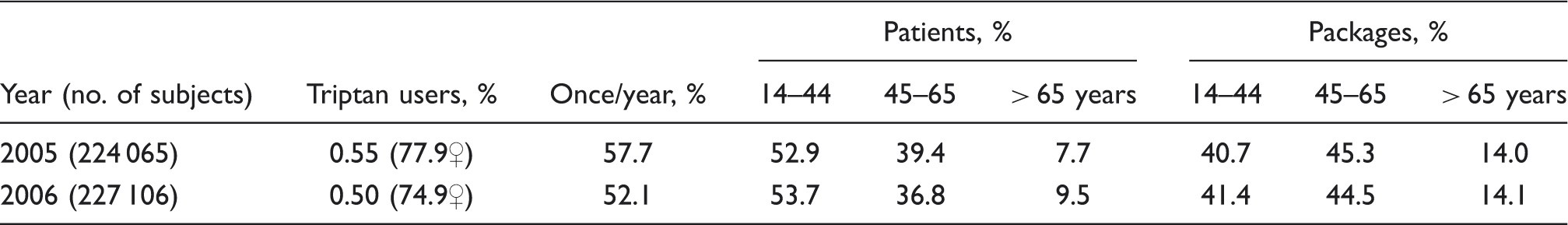
Nevertheless, the percentage of migraine patients utilizing triptans is low (3–19%) in several countries (5). However, the rate of migraine patients using triptans is often estimated from studies of selected migraine populations, taken from specialist consultation. We have poor data about the real utilization of triptans in the general population; the largest published study concerns about five million people of the Italian population (6), and recently we performed another study describing the drug utilization pattern in a population of Tuscany (7). The goal of the present study was to replicate the Tuscany analysis during two consecutive years, aiming to improve the understanding of the pattern of triptan utilization following patients and therapies over time (TRIPTEMP study).
Materials and methods
Due to the reimbursement system of the Italian National Health Service, the practitioner must fill in the prescription with personal data of the patient (name and family name, National Insurance number, address) as well as the medicine, the date of prescription, his own stamp and signature. When the pharmacist delivers the drug to the patient he takes the coupon from the drugs box and attaches it to the prescription, writing the date of delivery and pharmacy's stamp. All this information is set aside on a drug prescription database that we often use to evaluate accurate drug utilization.
In this instance the patterns of triptan prescription in the population of Health Authority 11 of Tuscany were investigated. We performed an analysis of prescriptions dispensed during 2005–2006 using: (i) before the Anatomical Therapeutic Chemical (ATC) classification (NO2CC), and (ii) afterwards the personal data of patients who received a prescription of triptans in 2005–2006. In 2005, all triptans (except naratriptan) were available in Italy as oral tablets: rizatriptan and zolmitriptan as soluble oral tablets, sumatriptan as subcutaneous injections, nasal spray and rectal suppositories. In Italy each prescription of triptans contains one or two packages, with dosage units contained in each package ranging from two to six (see 6).
In our previous 2005 study, performed only through the ATC classification, analysis of data was influenced by a double bias, but not so substantial as to influence the findings (7): (i) a small percentage (5%) of triptan packages was excluded for incomplete demographic data, (ii) more importantly, the analysis did not take into account if each patient took one or more kinds of triptans, so it was possible that one patient taking three different kinds of triptans could be considered as three patients. In the present study we have intentionally performed the same analysis (ATC classification) in 2006 as in 2005, for a better comparison of data. However, thereafter, using the patients' personal data, we have carefully extracted the subjects who received at least a triptan prescription in the period 2005–2006, thus eliminating the possibility that a patient taking more than one triptan could be considered more than one patient. The percentage of triptans users who received at least one triptan prescription in 2005 but discontinued triptans in 2006 (past users) was detected; the same was done for patients who started triptan therapy in 2006 (new users). The first triptan prescribed in 2005 and in 2006 (new users) was also analysed. The percentage of patients who utilized more than one kind of triptan was also detected.
Statistical analysis was performed, first to evaluate if the distribution of triptans among new users and past users was significantly different (by using the Z test), then to determine whether the likelihood of switching to a new triptan varied by type of triptan initially used (by using χ2 test). χ2 test was also used to test if the real distribution of triptan use was equal to the expected one under the hypothesis of homogeneity and if the proportion of patients receiving only one triptan prescription/year was homogeneous in all the groups considered (past users, new users, both 2005–2006 users).
Results
Comparative data of triptan utilization in 2005/2006 years
Pattern of triptan utilization in 2005–2006 years

In parentheses are the percentages of triptan users in 2005 who discontinued triptan (past users) or continued triptans in 2006, and the percentage of triptan users of 2006 who received first-time prescription in 2006 (new users).
Percentage of total triptan packages prescribed in past users (n = 431) and new users (n = 532)

Patients receiving a triptan prescription only in 2005 (past users) (A), only in 2006 (new users) (B) and in both 2005 and 2006 (C)
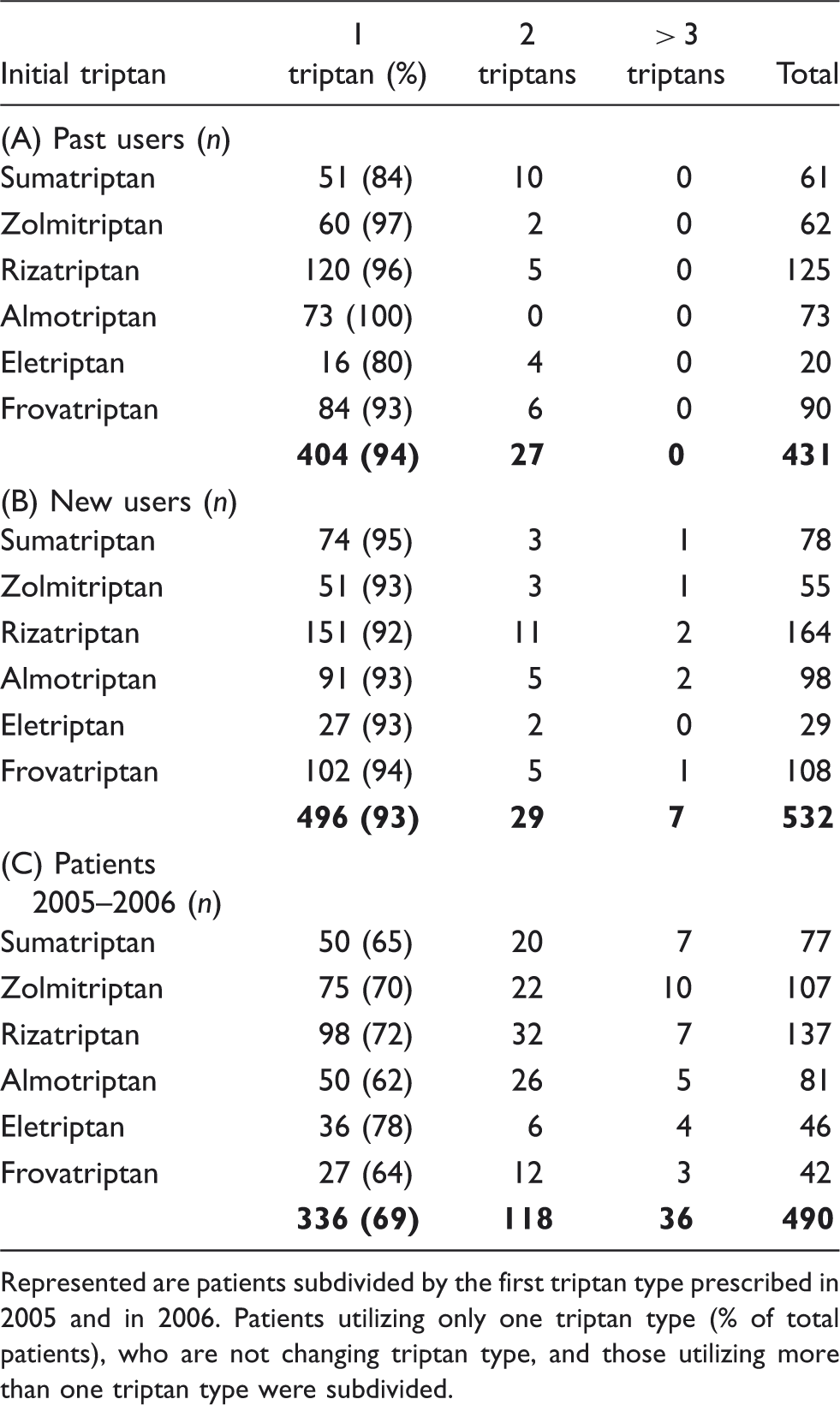
Represented are patients subdivided by the first triptan type prescribed in 2005 and in 2006. Patients utilizing only one triptan type (% of total patients), who are not changing triptan type, and those utilizing more than one triptan type were subdivided.
Percentages of triptan discontinuation based on initial kind of triptan prescribed

Represented are the number of subjects who received a prescription for a triptan type only in 2005 (past users), those who continued triptan treatment in 2006, and the percentage of discontinuation (past users) of total triptan users in 2005.
Analysis of the rate of utilization highlights that 50 subjects in the years 2005–2006 utilized > 120 defined daily doses/year, and they can probably be classified as triptans abusers, responsible for an expenditure that is about one-third (34%) of the total.
Discussion
The efficacy of triptans in migraine attacks has been shown in several double-blind placebo-controlled trials (2). Triptans show superior efficacy and a higher degree of patient satisfaction in comparison with non-triptan therapy (3,4), even if frequently the differences are not dramatic (8). Migraine patients who responded poorly to a triptan may respond better to a different triptan (9,10). Although initial studies reported that sumatriptan non-responders treated their migraine attacks earlier (11), several clinical trials have shown that aggressive treatment, while pain is mild, results in an improved rate of responsiveness (9,12,13). This combined body of evidence suggests that a reasonable degree of convergent validity exists for the superiority of triptans in migraine treatment.
The previous findings place triptans in the first position for migraine treatment, and therefore triptans should be utilized by the majority of migraine patients. However, other data have shown the non-optimal efficacy of these drugs. A consistent finding is that more than 30% of patients (and up to 40% of attacks) fail to respond to a particular triptan, either because of suboptimal efficacy or tolerability issues (9,14). Furthermore, even when patients are considered responders to a triptan, less than two-thirds of patients respond to this triptan in three out of three attacks (9,10). Baseline headache severity, rather than time from onset of the attack until drug intake, was reported as the most significant predictor of response failure (15).
The small differences in preference/efficacy between triptans are minor details considering their low use compared with NSAIDs or over-the-counter (OTC) drugs. In fact, utilization of triptans by migraine patients in different countries is low, ranging from 3 to 19% (5). Prescription data show a very low percentage (0.4–1.4% of the general population) of utilization worldwide (6), which, if related to migraine prevalence, is concordant with that in migraine patients. From the analysis of these data, a substantial increase in triptan utilization over the years does not result (6). A prescription register survey in Denmark revealed that nearly 1% of the population used sumatriptan (the only triptan available at the time of the study) in a year (16). Five years later, the percentage of triptan users in the same country was 0.52% of the population (17). Even the incidence of sumatriptan first-time users (new users) was reported not to increase during the years (18). The majority of patients (80%) utilizing a triptan, most of whom (80%) had already tried a different triptan, when asked in a clinical trial performed at headache centres, declared their willingness to try another acute medication (19).
In our study we have analysed the pattern of triptan utilization in a large community during two consecutive years, confirming previous data on the same population (7). We have not found substantial differences concerning the percentage of triptans users, the percentage of patients receiving only one triptan prescription (containing one to two packages) in a year, the age distribution of users or of the amount prescribed, during the two consecutive years of the study. In particular, 52–57% of patients have received only one prescription in a year. The percentage of triptans users is 0.5% of the population studied, as stated in the previous work (7). Fifty-two per cent of patients received a prescription of one triptan in 2006, but none in the previous years (new users). Other studies show an even higher percentage (63%) of new users during a 6–12-month period (20,21). Of new users, 55.7% have received a prescription of one package, and 76% of one to two packages in a year. This percentage is very similar to previous studies showing only one prescription in a year in 56.1% (22) and 51.1% (20) of new users.
Out of all triptans users in 2005, 46.7% did not receive a prescription of triptan in the following year, and may have discontinued triptan use (past users). Most of these patients have received only one (60.4%) or two (19.2%) packages; therefore, 80% of patients have received one to two packages in a year. Moreover, only 6% of past users received a prescription for no more than one other triptan, different from that first prescribed in the year. Instead, those patients who continued the treatment in the following year received at least one other triptan type in 30% of cases. These findings support the following statements: (i) in most cases, patients have discontinued triptan treatment without testing another kind of triptan, (ii) 30% of patients who continued the triptan treatment during the 2 years of the study, but received prescriptions for other triptans, were probably unsatisfied with the previous triptan. The percentage of subjects receiving a prescription of more than one triptan kind in the 2 years of study was 14.8%. A similar percentage (12.5%) was found in a study performed by community pharmacies (23).
The percentage of past users is similar (55%) to that reported in a cohort of patients successfully established on sumatriptan therapy for 12 months, who subsequently had no further triptan therapy in the 15 months' follow-up period (24). Studies in migraine patients in primary care practice show that 30% of migraine patients discontinued triptan therapy, principally for lack of efficacy (25). Rahimtoola et al. (26) found inefficacy and/or the occurrence of side-effects as the main reasons for discontinuing treatment after only one prescription. Perhaps some patients tried a triptan but thereafter did not require it again. This assumption was supported by the fact that although new triptan users are a large proportion of total users, the percentage of triptans users during the years has remained low.
Analysis of triptan discontinuation focused on triptan kind shows some significant differences. Excluding eletriptan, the highest percentage of discontinuation concerns the triptans commercialized more recently, which are also more frequently prescribed in new users. It has been reported that a high percentage (54%) of patients experiencing a triptan switch returned to a previous triptan therapy (sumatriptan), suggesting that the alternative was less acceptable (24). The significance of our previous findings is difficult to determine. It is possible that patients and physicians too try a new triptan due to dissatisfaction with previous triptan medication, but usually with unsatisfactory results. The fact that the triptans most frequently discontinued are the same as those more frequently prescribed by physicians in new users could be due to the search by physicians to find a more efficacious response by using the triptans most recently introduced.
In conclusion, in agreement with data coming from Italian and world populations (6), the following findings emerge: (i) a very low percentage of migraine patients are treated with triptans; (ii) if triptans are used, they are prescribed only once in a year in a large number of cases; (iii) a high turnover (high percentage of past users and new users) is not associated with a substantial increase in the percentage of users over the years; and (iv) many triptans were prescribed for a few subjects (including abusers). These data clash with both the proven efficacy of these drugs, and studies showing the superior quality of satisfaction of patients on triptan medication compared with non-triptan drugs.
The principal question arising from our findings is how these results can be generalized, because the patient population and treatment prescribers in our country may differ from those in other countries. However, the low percentage of triptan users in our area is very similar to that obtained from a large Italian population (6 million) and many large populations elsewhere in the world (6). Theoretically, the high cost, in comparison with non-triptan drugs, can play a role in the limitation of triptan usage in two ways: (i) patients may prefer drugs that are less expensive than triptans, but in our and many other countries triptans are dispensable free of charge by National Healthy Services; (ii) general practitioners (GPs), who are frequently subjected to control of their pharmacological budget, prefer to prescribe drugs with a lower cost. In fact, there is a difference in the degree of triptan prescription between GPs and headache specialists, as shown by the increased percentage of triptan users after consultation with specialized centres (27). However, data taken from the population are independent of the type of physician prescribing triptans.
Several reasons may explain the low triptan utilization compared with OTC drugs and NSAIDs. The percentage of triptan utilization is very low, perhaps due to a large number of migraine patients who remain undiagnosed and/or undertreated (28). However, low utilization also emerges in studies on migraine subjects (5). Certainly, all headache specialists know that in many migraine patients OTC drugs and NSAIDs can work well. The high percentage of single prescriptions/year of triptans could indicate that many migraine patients had a low frequency of attacks. In fact, it was reported that only 20% patients had > 14 migraine days per year (29). Another plausible explanation of the low percentage and frequency of triptan use is that many migraine patients cannot be completely satisfied by triptan therapy with regard to efficacy and/or side-effects, as suggested also by the high turnover. Side-effects of OTC drugs and NSAIDs are generally subjectively limited to gastric discomfort, whereas triptans have central and other side-effects that can be troublesome. In these situations, patients prefer other medications such as OTC drugs or NSAIDs. Although triptans are undoubtedly very efficacious for migraine attacks, a clear superiority over NSAIDs in comparative studies did not appear on the primary end-point (8,30,31). A recent study has also reported a superiority of an OTC drug in comparison with sumatriptan (32).
It is clear that there is a need for a new approach to headache problems and for closer collaboration between GPs and specialists, which would certainly also provide a better means of obtaining data on the pattern of triptan utilization directly from migraine patients over the years. This task force will allow the emergence of the ‘iceberg’ headache and consequently the optimal use of triptans, an important, but still underused therapeutic tool.
Footnotes
Acknowledgements
The authors thank Marno Institute (Castelnuovo Misericordia, Livorno, Italy) for their valuable collaboration in managing the data on triptan prescription.
