Abstract
The objective of this study was to assess usage patterns of ergotamine and sumatriptan over a period of 6 years, primarily to evaluate the impact that sumatriptan has had on the prescription of ergotamine. This study used ergotamine and sumatriptan prescription data representing inhabitants of eight cities in the Netherlands and covering the period of 1991–1997. The yearly incidence of new users between 1991 and 1997 was estimated for both drugs as well as for the drug of first choice to be prescribed to patients initiating specific abortive migraine treatment with either ergotamine or sumatriptan. Intra-individual ergotamine and sumatriptan usage patterns, characterized by single (incidental), continuous (rate of retention) or switch use, were examined for five patient cohorts, each for a follow-up period of 1 year. During the year of sumatriptan introduction (1991–1992), the overall incidence of new use for both drugs was highest (5.4 per 1000 inhabitants). Hereafter, a substantial reduction of more than 50% was observed. From 1992 to 1996, the yearly incidence of ergotamine first-time use was significantly higher than that of sumatriptan and up to 1996 ergotamine was more than twice as likely than sumatriptan to be prescribed to patients initiating specific abortive treatment. Hereafter, sumatriptan was as likely as ergotamine to be prescribed as the drug of first choice, which coincided with the full reimbursement of sumatriptan tablets. Overall, neurologists were more likely than general practitioners (GPs), to prescribe sumatriptan as the drug of first choice. Approximately half of the total study population were identified as single-time users. This phenomonen occurred more frequently in the ergotamine cohorts. The sumatriptan cohorts displayed a slight yet significant stronger retention rate compared with the ergotamine cohorts. The overall impact of sumatriptan on ergotamine use in The Netherlands was marginal, predominantly due to GP's adherence to migraine treatment guidelines and reimbursement policies concerning sumatriptan tablets. Overall, incidental use was relatively high and may reflect the reported difficulties in diagnosing migraine, lack of patient–doctor consultation, or that anticipated benefits of the drug were not achieved. Further study is required to clarify these issues.
Introduction
Before the start of the previous decade, the ergot alkaloids were considered the drugs of choice for the treatment of moderate to severe migraine attacks. Recently, however, a meta-analysis of clinical studies on ergotamine has actually cast doubt on the utility of the drug and suggests that its side-effects may outweigh its benefits (1). The limitations associated with this drug have given rise to heightened research in migraine therapy which first lead to the release of sumatriptan, a selective serotonin agonist, and quite recently the newer ‘triptans’, naratriptan, rizatriptan and zolmitriptan.
However, after the introduction of sumatriptan in 1991, national (2, 3) as well as international (4–6) treatment guidelines were uncertain as to whether ergotamine or sumatriptan should be the first choice after non-specific analgesics were no longer effective for the migraine patient. The therapeutic guidelines of migraine established by the Dutch College of General Practitioners and the Dutch Pharmacotherapeutic Formulary recommended the use of ergotamine in preference to sumatriptan from 1991 to 1997. The relative high acquisition cost of sumatriptan compared with ergotamine, the beneficial effects of ergotamine when used appropriately and lack of clinical documentation in favour of sumatriptan in treating migraine attacks were the main criteria for these recommendations (2, 3, 7). The national health insurance council and government reimbursement body established a similar consensus which resulted in only partial reimbursement of sumatriptan tablets; patients were required to pay approximately $14.00 per 100-mg sumatriptan tablet, from 1992 to 1996. During the first year of introduction of sumatriptan and after 1996, the tablets were fully reimbursed. The far more expensive injectable form of sumatriptan, however, was fully reimbursed since its introduction due to distinct benefits in the treatment of cluster headache.
In contrast, the migraine therapeutic guidelines established by The Dutch Association of Neurology in 1997, consisting primarily of neurologists, considered sumatriptan as the drug of choice following the use of non-specific analgesics due to the high incidence of side-effects associated with ergotamine (8).
So far only a few studies have evaluated the impact of sumatriptan on the use of ergotamine and the long-term usage patterns of ergotamine and sumatriptan. Gaist et al. studied the use of sumatriptan in Denmark during 1994–1995 by analysing prescription data representing the total Danish population. A slight reduction in the use of ergotamine and a significant increase in sumatriptan consumption was observed during the observation period (9).
In view of the conflicting (inter)national guidelines regarding ergotamine and sumatriptan use in migraine patients, we were interested in the impact of sumatriptan on the use of ergotamine as well as usage patterns of ergotamine and sumatriptan in The Netherlands over 6 years following the introduction of sumatriptan (1991–1997).
Methods
Study setting
The study used prescription data from the PHARMO-RLS database located at the University of Utrecht, The Netherlands, covering the period 1990–1997. This database has been described in full elsewhere (10). In brief, the system was designed in 1985, providing relevant demographic and prescription data at an individual level for five medium-sized cities in The Netherlands from 1985 up to 1989. Since 1990 it has been further updated, covering a total of six cities (n = 300 000), and from 1993 to 1998 for eight cities (n = 450 000). In view of a high patient–pharmacy registration commitment in The Netherlands, in addition to sophisticated pharmacy software, the prescription medication information for each patient is virtually complete.
Each registered person is identified with an anonymous unique patient identification code which allows for the observation of patient medication use in time. Retrievable information per prescribed medicine includes date of dispensing, drug, dosage regimen, quantity supplied and type of prescriber. Patient information per prescribed medicine is also present and includes gender and date of birth. The database does not provide information concerning the indications of use of the medicines, in this case the diagnosis of migraine vs. cluster headache, or use of non-prescription medicines (e.g. OTC use of salicylates or paracetamol).
Study population
For this study all patients presenting a prescription for either ergotamine, alone or in combination with caffeine and/or cyclizine, or sumatriptan, during 1 June 1991 to 31 May 1997 were included. Sumatriptan was registered in The Netherlands in June 1991. Prescriptions were retrieved using the WHO Anatomical Therapeutical and Classification system for ergotamine, alone (N02CA02) or combination (N02CA52), and sumatriptan (N02CX01). No differentiation was made between users of ergotamine alone and combined ergotamine preparations for this particular study.
Data analysis
For each patient included in the study the first use during the study period of the particular drug was termed ‘first-time use’ and the date of the corresponding prescription was termed the ‘index date’. For each first time use, several characteristics were determined, namely age, gender, type of prescriber (either general practitioner or neurologist), and dosage form of the particular drug.
For the two drugs separately, the incidence (expressed as the number of new users per 1000 inhabitants per year) was estimated for each year following 1 June 1991.
The drug of first choice for patients initiating specific abortive migraine therapy, with either ergotamine or sumatriptan, was determined per year.
In addition, usage patterns were evaluated, characterized by incidental, continuous or switch use. For this examination five patient cohorts were defined, distinguished by the year of the index date. The follow-up period of each cohort was exactly 1 year. For example, cohort one represented those patients with an index date between 1 June 1991 and 31 May 1992 and who were followed during 1 year. This analysis could not be performed for patients with an index date during the period 1 June 1996 to 31 May 1997, as follow-up data corresponding to one year were not available for all patients in this cohort. ‘Incidental use’ of the drug was defined as those patients presenting only one prescription and not switching to the other drug during the follow-up year. Switch use was defined as the change in treatment to either ergotamine or sumatriptan during the cohort follow-up year. Since migraine is a disease in which the frequency of attacks are irregular and unpredictable, we could not define ‘continuous use’ by the number of days of medication use. Instead we defined ‘continuous use’ of the drug as the presentation of more than one prescription for the specific drug during the follow-up period. The rate of continuing use was compared between the two drugs and expressed as a ‘retentiveness ratio’ (relative risk with 95% confidence intervals). In addition, for each cohort the number of defined daily dosages per prescription (DDDs) was calculated during the follow-up of 1 year. One DDD sumatriptan corresponded to one 100-mg tablet or one 6-mg subcutaneous injection. One DDD ergotamine corresponded to one unit of ergotamine 4 mg by any route or one unit of a 2-mg combination preparation by any route (9). The number of DDDs used per month, expressing intensity of drug use, was determined for each cohort and compared between the two drugs (95% confidence intervals).
Results
During the study period a total of 5355 patients were identified and responsible for 6383 first-time uses of ergotamine and or sumatriptan. This corresponded to an overall incidence density of 2.5 first-time uses per 1000 person-years; 1028 patients (19%) were using both drugs either simultaneously or had switched from the first drug of choice to the other drug.
The highest overall incidence (5.4 per 1000 persons per year) of first-time use was observed during 1991–1992, the first year of introduction of sumatriptan (Table 1). Thereafter the overall incidence fell to 2.9 first-time users per 1000 person-years during 1992–1993 and remained stable at approximately 2.3 first-time users per 1000 person-years during the remaining study years. The incidence of use of ergotamine prior to the introduction of sumatriptan during 1 June 1990 to 31 May 1991, was 3 per 1000 (data not shown). This also corresponds to the overall incidence, in that year, of commencing specific abortive treatment, since ergotamine was the only specific abortive drug available before the introduction of sumatriptan.
Incidence of ergotamine and sumatriptan first-time use during 1991–1997
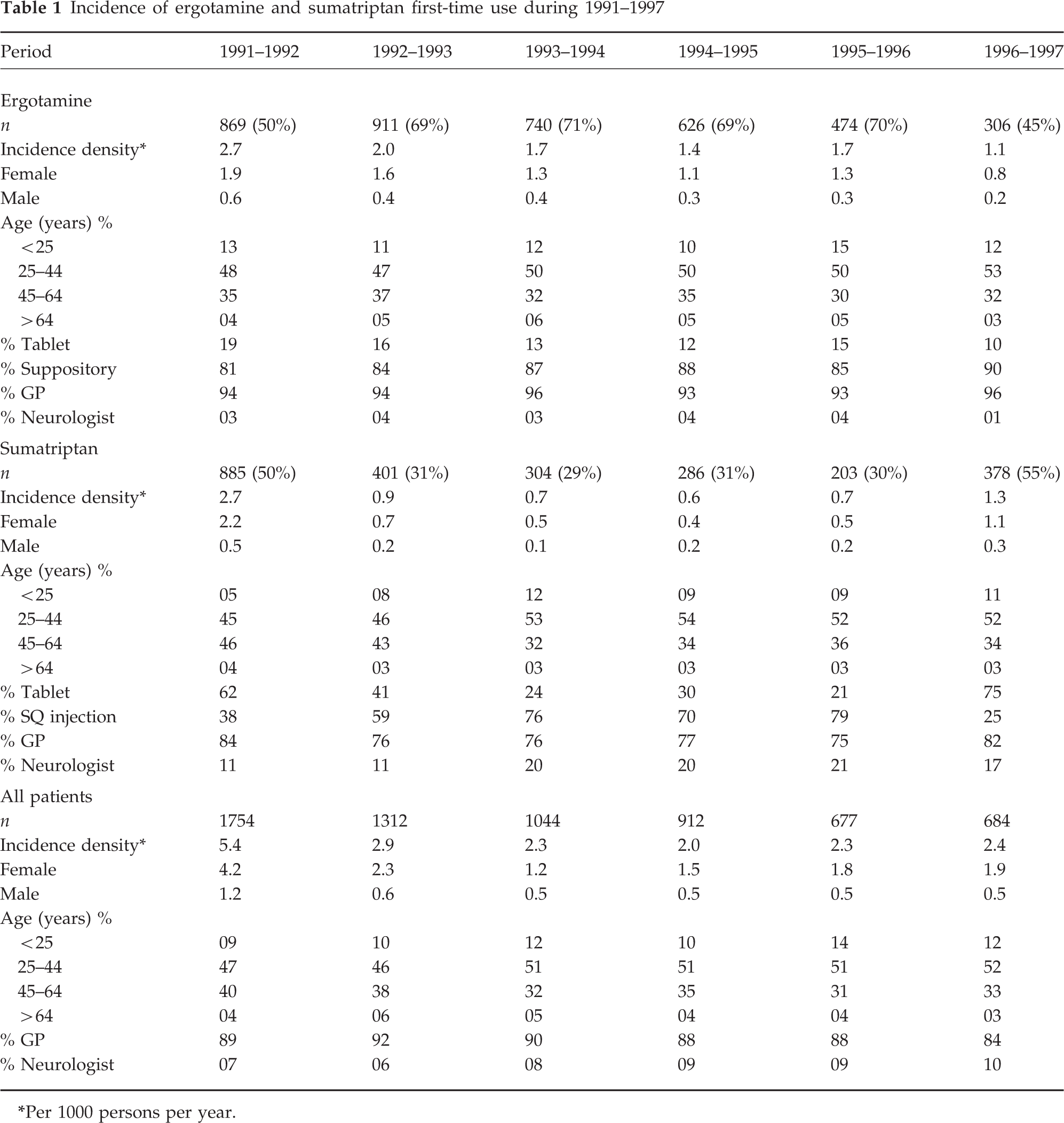
Per 1000 persons per year.
This pattern in the overall incidence was similarly displayed by the two drugs individually; however, the incidence of ergotamine first-time use (1.7 first-time users per 1000 person-years) was twice that of sumatriptan (0.7 first-time users per 1000 person-years) from 1992 until 1996. Hereafter, the incidence of sumatriptan use significantly increased and was slightly higher than that of ergotamine.
Ergotamine and sumatriptan were more commonly used by females than males, which corresponded to an overall incidence ratio of approximately 3:1 for users of both drugs. This relationship remained stable throughout the study period. Approximately 50% of all users were aged between 25 and 44 and were predominant users of either drug throughout all years of the study period.
The vast majority of first-time prescriptions for ergotamine or sumatriptan were prescribed by a general practitioner. Neurologists, though far less involved in the initiation of ergotamine or sumatriptan, were more likely to initiate sumatriptan as the drug of choice.
Throughout the study period the suppository form of ergotamine was by far the major dosage form to be prescribed for ergotamine first-time users. In contrast, a fluctuating pattern concerning the choice of sumatriptan dosage form was observed. During 1991, the tablet form of sumatriptan was the dosage form of choice to be prescribed to first-time users. However, a sudden preference for the subcutaneous form of sumatriptan during 1992–1993 and onwards had taken place. This phenomonen persisted until 1996 when a sudden preference for the tablet form of sumatriptan occurred.
Overall, ergotamine was by far (71%) the drug of first choice to be prescribed to patients commencing specific anti-migraine therapy and was more than twice as likely (RR 2.5, 95% CI 1.8–3.4) to be prescribed than sumatriptan (29%). This preference was maintained until 1996. Hereafter, sumatriptan was as likely to be prescribed as ergotamine to patients commencing specific abortive migraine therapy (Fig. 1).
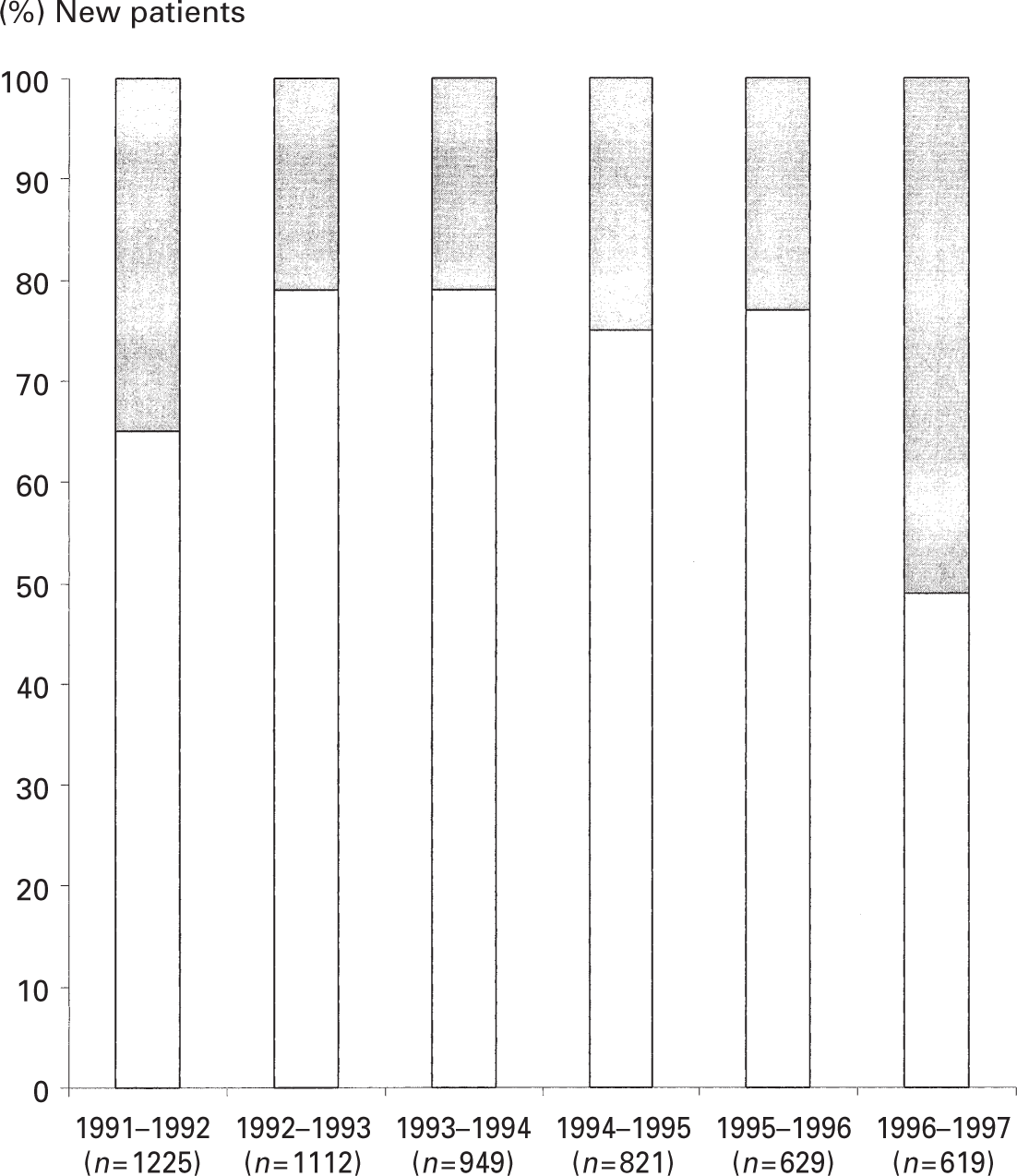
Drug of choice, ergotamine (▲) or sumatriptan (▪), in new patients commencing specific anti-migraine treatment.
Table 2 summarizes the utilization patterns during a follow-up of 1 year for the defined five cohorts in terms of continuing, switching or incidental use. A significant proportion of each drug group received only one prescription of the particular drug (incidental use). Overall, incidental use was higher in the ergotamine group (53%) than in the sumatriptan group (34%). The incidence of switch use for each ergotamine and sumatriptan cohort was low and consistent at around 5%. The incidence of continuous use was higher in the sumatriptan group than in the ergotamine group, as expressed by the overall retentiveness ratio of 1.5 (95% CI 1.1–1.9).
Usage patterns of ergotamine and sumatriptan cohorts: continuous, incidental and switch use

Sumatriptan vs. ergotamine.
The overall intensity of use, expressed by DDDs per month, revealed no significant differences between the two drugs. This was similarly observed when comparing the individual cohorts separately.
Discussion
In our study concerning the use of sumatriptan and ergotamine in The Netherlands over a period of 6 years, we observed that the introduction of sumatriptan had a marginal impact on the prescribing practice of ergotamine for the first 5 years after the introduction. However, when sumatriptan tablets were fully reimbursed, the preference for sumatriptan as the drug of first choice suddenly increased. In addition, we observed during the whole study period that almost half of all first-time users of either drug presented only one prescription.
In our study, three times as many women as men were using specific anti-migraine therapy and this predominantly involved patients between the ages of 25 and 45 years. These findings coincide with other migraine epidemiological studies (4, 11, 12). The overall incidence was relatively low in proportion to the prevalence and incidence of migraine in the general population. This may be explained by the increasingly low medical consultation rates of migraineurs, as well as the fact that only a minority of migraineurs are prescribed drugs such as ergotamine or sumatriptan (6, 13–16).
We observed a remarkable peak in the overall incidence of specific anti-migraine treatment during the year following the introduction of sumatriptan. This may be attributed to a surge of enthusiasm of patients and physicians concerning the availability of a novel abortive migraine drug together with an increased awareness concerning migraine and its diagnosis, resulting in an increased rate of migraine diagnosis and prescribing of sumatriptan as well as other non-specific and specific abortive medications, such as ergotamine (13). However, this peak lasted for only approximately 1 year. Hereafter, the overall incidence for the two drugs remained stable yet relatively low. The reason for this overall decrease remains unclear. The decrease could reflect that the high expectations concerning sumatriptan were not fully met. Another reason could have been the frequent reporting in the medical and lay press of harmful effects, mainly cardiovascular-related, possibly associated with sumatriptan. In addition, the reimbursement policy concerning sumatriptan tablets seems to have been a major factor influencing the prescribing practice of sumatriptan. In The Netherlands, sumatriptan tablets were fully reimbursed during the year of introduction but not between 1992 and 1996. Other studies have shown that reimbursement policies can have a major impact on prescribing practices (17).
In our study, a discrepancy was observed between neurologists and general practitioners in the preference for sumatriptan and ergotamine for first-time users of specific anti-migraine treatment. Ergotamine was considered by far the overall drug of choice to be prescribed to first-time users initiating specific anti-migraine treatment. It seemed that general practitioners fully adhered to the guidelines of the Dutch College of Family Practitioners and the Dutch Pharmacotherapeutic Formulary from 1991 to 1997 (2, 3). Furthermore, the substantially higher percentage of neurologists prescribing sumatriptan in this study in preference to ergotamine as the drug of choice, reflects the contrasting guidelines established by the Dutch Association of Neurology (8). This highlights the need to bridge the gap between first- and second-line care in The Netherlands in order to establish one common therapeutic approach in the management of migraine and other chronic headache disorders. Our findings concerning the drug of first choice are in contrast to the findings obtained from the study by Gaist et al. (9). In their study, there was an equal preference for ergotamine and sumatriptan during 1994–1995.
After 5 years of observation in our study, sumatriptan was, suddenly, as likely as ergotamine to be prescribed to patients initiating specific anti-migraine therapy. This is probably largely attributed to the changes in Dutch government and health insurance recommendations concerning sumatriptan tablets: in 1997 sumatriptan was considered equally appropriate to ergotamine as an alternative for migraineurs who no longer responded to non-specific analgesics. As a consequence, full reimbursement of the tablet form was reinstated. These modifications are highlighted by our observations that the subcutaneous dosage form of sumatriptan, being fully reimbursed throughout the study period, had comprised approximately 70% of all new sumatriptan prescriptions per year from 1993 to 1996, dramatically reducing thereafter. After this period the tablet form accounted for 75% of all new sumatriptan prescriptions. Our findings may support those obtained from an American study that, having been conducted prior to the approval of oral forms of sumatriptan, concluded that the availability of only subcutaneous sumatriptan had a marginal impact on the pattern of medication use in migraineurs (16).
The evaluation of usage patterns revealed that a large proportion of each cohort was a single recipient of the drug (incidental user) during a follow-up of 1 year. Incidental use of sumatriptan, substantially lower compared with that of ergotamine, was remarkably similar to that observed in the study by Gaist et al. in which 41% of sumatriptan users had presented only one prescription during 1 year (9). Incorrect diagnosis of migraine, the drug being prescribed as a diagnostic tool, the occurrence of unpleasant side-effects or low patient–physician consultation rates are interesting yet speculative issues related to the occurrence of incidental use (14, 18, 19). An interview study of such patients may be required to further clarify these issues.
It could be argued that many incidental users in this study were simply suffering a mild form of migraine, presenting ergotamine or sumatriptan prescriptions after relatively long intervals of time, for example after 1 year. These patients could be misclassified as ‘true incidental users’. It could have been possible to test such patients for the presentation of other prescriptions until the end of the study period. Further analysis (data not shown) of patients for whom a longer follow-up was available (cohorts 1–3) revealed that over 90% of the patients classified as incidental users indeed only presented one prescription of the specific drug. Furthermore, epidemiological studies show that patients with a mild form of migraine usually experience one attack per month and therefore would most likely present a prescription at least once a month given the average quantity prescribed per prescription in The Netherlands (4, 11).
A stronger retention, expressed by the ‘retentiveness ratio’, was demonstrated by the sumatriptan cohorts. Since sumatriptan is generally prescribed at lower units than ergotamine, it could be argued that our calculations of continuing use and retentiveness ratios for the sumatriptan cohorts are overestimated. However, our defintion of intensity of use, which is directly related to the total DDDs presented per prescription and period of use, can validate our estimations of ‘retentiveness ratio’. Intensity of use (DDDs used per month) for each drug was similar across all cohort years. A study examining the performance of sumatriptan (primarily the subcutaneous form) demonstrated that 85% of the study population found sumatriptan to be more effective than other conventional therapies (20), prompting continued use. Whether the stronger ‘retentiveness ratio’ estimated for the sumatriptan cohorts is an indication of a more acceptable clinical response to this drug, as observed in other studies (20, 21), cannot be fully confirmed in this study.
A few important limitations should not be ignored. The specific diagnosis, prognosis and severity of the disease according to frequency and intensity of attacks was not provided. An assumption was made that both ergotamine and sumatriptan were indicated for migraine or cluster headache. Our study was purely based on prescription data and could not validate actual patient consumption. However, a large variation between prescribing patterns of sumatriptan and ergotamine and actual patient consumption was not expected. This is likewise confirmed by Gaist et al. (9).
Finally this study was confined to only several geographical areas in the Netherlands. It has been shown, however, that these areas are representative for the entire Dutch population (10).
In conclusion, strict adherence to migraine therapeutic guidelines and economic factors, has largely influenced the prescribing patterns of sumatriptan as well as ergotamine during the 6 years following the introduction of sumatriptan. These factors may have potentially denied our patients in receiving a ‘tailor made’ therapeutic management, an essential requirement in the attempt to further improve the therapeutic outcomes and quality of life of the migraineur (22). This study also highlights the need to examine in detail the potential factors associated with the single use of specific abortive migraine drugs.
Footnotes
Acknowledgements
This research is funded by a grant from the Royal Dutch Society for the Advancement of Pharmacy (KNMP), The Hague, The Netherlands and SIR Institute of Pharmacy Practice Research, Leiden, The Netherlands.
