Abstract
Chronic migraine (1.5.1) is burdened with headache-related disability. During noxious stimulation, changes of cerebral blood flow enhance the release of oxygen free radicals that react with nitric oxide (NO). We investigated the role of biofeedback in limiting migraine disability by influencing oxidative stress. Peroxides, NO and superoxide dismutase (SOD) were analysed in 20 female subjects with chronic migraine and in 20 female healthy controls before and after biofeedback sessions. NOx levels (23.7 ± 4.2 vs. 34.9 ± 4.6 μM; P < 0.05) and SOD activity (6.5 ± 1.0 vs. 8.0 ± 0.7 U/ml; P < 0.05) were lower in migraine sufferers before treatment than in healthy controls, whereas peroxide levels (145.8 ± 40.3 vs. 78.0 ± 20.0 μM; P < 0.05) were higher in migraine sufferers before treatment than in healthy controls. In migraine sufferers NOx levels (23.7 ± 4.2 vs. 31.3 ± 7.1 μM; P < 0.05) and SOD activity (6.5 ± 1.0 vs. 7.9 ± 0.9 U/ml; P < 0.05) were lower before than after treatment, whereas peroxide levels (145.8 ± 40.3 vs. 82.4 ± 21.1 μM; P < 0.05) were higher before than after treatment. SOD serum activity correlated positively with NOx serum levels and negatively with peroxide serum levels in healthy controls and in chronic migraine sufferers before and after biofeedback. The mean Migraine Disability Assessment Score before biofeedback sessions was higher than after treatment (36.9 ± 13.9 vs. 18.8 ± 10.4; P < 0.001). The effectiveness of biofeedback in limiting chronic migraine may be related to muscular relaxation associated with decreased oxidative stress accompanied by psychological well-being.
Introduction
The International Classification of Headache Disorders, 2nd Edition (1), identifies chronic migraine (1.5.1) as a pathological entity that occurs on ≥15 days per month, for >3 months, in the absence of medication overuse, thus resulting in a headache-related disability.
Chronic pain is a complex phenomenon mediated by an alteration of serotonin concentration and by central sensitization. Central sensitization, a N-methyl-D-aspartate (NMDA)-mediated phenomenon, is influenced by excitatory amino acids such as glutamate and by nitric oxide (NO), and might be maintained by neuropeptides such as substance P and calcitonin gene-related peptide (CGRP) (2–4). NO is a nonadrenergic non-cholinergic neurotransmitter processing noxious impulses and sensitizing perivascular sensory nerves (5). NO is also involved in activation of the trigeminovascular system and in regulation of the cerebral vessel tone (4). In an animal model, during noxious stimulation cerebral blood flow is adjusted to the increased neuronal activity (6); changes of regional cerebral blood flow may enhance the rate of release of oxygen free radicals (7). NO rapidly reacts with superoxide anions because of their unpaired electrons in the outer orbitals, determining a rapid radical/radical reaction that reduces its effective half-life and biological function (8). In addition, the interaction between NO and superoxide anions causes the production of peroxynitrite, a strong oxidant that influences the mechanism of hyperalgesia in chronic pain and reversibly dilates cerebral arterioles through an oxidant mechanism probably activating ATP-sensitive potassium channels (9–12). Even if the peroxynitrite-mediated vasorelaxation is subject to the development of rapid tachyphylaxis, the peroxynitrite, promoting lipid peroxidation, may trigger the production of nitrotyrosine, nitrosothiols and isoprostanes that are able to modulate vascular tone (13). The interaction between NO and superoxide anions may therefore be involved in inducing and maintaining migraine-related changes in cerebral blood flow (14, 15).
Superoxide dismutase (SOD) is a family of metalloenzymes that catalyses the dismutation of superoxide anions to molecular oxygen and hydrogen peroxide essential to the cellular anti-oxidant defence mechanism (16). SOD lowers the superoxide concentration and prevents NO decomposition by scavenging superoxide anions (17). In migraine sufferers the activity of SOD, a radical-scavenging enzyme, is lower than in healthy controls, suggesting enhanced vulnerability to oxidative stress (18).
A non-pharmacological preventive therapeutic approach, such as biofeedback may prove effective in decreasing the severity, frequency and disability of chronic headache (19). Biofeedback is based on a monitoring instrument which detects, amplifies and displays ongoing involuntary physiological processes, inducing the patient to learn how to modify and interact with them (20). This mind–body technique may be used to treat a wide range of mental and physical health problems, forcing patients to take an active role in attaining well-being. We therefore investigated whether, in patients with chronic migraine, a preventive non-pharmacological approach based on biofeedback sessions might influence serum NO stable metabolites, peroxide and SOD serum activity, before and after the training sessions.
Methods
Twenty female patients with chronic migraine (mean age ± SD 25.6 ± 4.6 years) and 20 female healthy controls (mean age ± SD 28.1 ± 6.0 years) were consecutively enrolled between 1 January and 30 April 2005. Chronic migraine was diagnosed according to the criteria of the International Classification of Headache Disorders, 2nd Edition (1). The study was approved by the institutional ethics committee and complies with national rules on informed consent of subjects involved. All female subjects were non-smokers, free of cardiovascular, endocrine, renal, hepatic, neurological, psychiatric and coagulation diseases. Subjects with pregnancy, pacemaker, gastrointestinal and extradigestive infections were excluded (21, 22). No pharmacological preventive treatment was allowed.
Collection of the first and the second blood sample was performed on two different days during a headache-free period, respectively, corresponding to the day of the first biofeedback session and of the last one. The headache-free period was considered as the interval separating two distinct migraine attacks, lasting >72 h. Migraine sufferers were carefully instructed to avoid any nitrite/nitrate-enriched diet for at least 4 days before assessment and during all the study period thereafter (23). Migraine frequency, severity and disability were evaluated by the Migraine Disability Assessment Score (MIDAS) (24) administered before and soon after the scheduled biofeedback sessions.
Blood sample processing
Blood samples were collected in polypropylene tubes and were centrifuged at 1000 g for 15 min to obtain serum samples.
Determination of NO stable nitrite/nitrate metabolites (NOx)
NO is a free radical rapidly decomposed to nitrite and nitrate (NOx). Serum samples were ultrafiltered at 17 000 g for 15 min to remove substances >10 kDa and to separate precipitates (Ultrafree MC microcentrifuge device; Millipore Corp, Bedford, MA, USA). NOx were measured by reducing nitrate to nitrite by the action of nitrate reductase from Aspergillus niger (25) and measuring nitrite by a sensitive fluorimetric assay based on the reaction of nitrite with 2,3-diaminonaphthalene to form the fluorescent product 1-(H)-naphthotriazole (26). The intensity of the fluorescent signal of the samples was measured and compared with known concentrations of sodium nitrite (excitation wavelength 375 nm, emission wavelength 415 nm). The fluorimetric assay was able to detect amounts in the order of 30 n
Determination of peroxides
The oxidative status in serum samples was evaluated measuring the total peroxide concentrations by the Biomedica OxyStat assay (Biomedica Gruppe, Vienna, Austria). The peroxide concentrations were determined by reaction of the biological peroxides with peroxidase by a colour reaction using 3,3′,5,5′-tetramethylbenzidine as substrate and measured photometrically at 450 nm. Inter- and intra-assay variations were 5.1% and 3.1%, respectively.
Superoxide dismutase activity
SOD activity was measured using a tetrazolium salt for detection of superoxide radicals generated by xanthine oxidase and hypoxanthine (Cayman Assay kit; Cayman, Ann Arbor, MI, USA). One unit of SOD was defined as the amount of enzyme needed to exhibit 50% dismutation of the superoxide radical.
Biofeedback
For the biofeedback treatment the Satem Myoexpand Series 400, EMG Module M3 was used. The instrument had three scales in µV (rms): 0–10, 0–100, 0–1000 Hz. Noise in typical entrance was <0.4 µV (rms). The band pass was set to 100–1000 Hz. After skin preparation with alcohol, three circular metal disposable cup-type electrodes (2 cm in diameter) were applied to the frontal muscle and secured with paper tape; the active electrodes were directly centred over each eye, while the reference electrode was centred directly over the bridge of the nose. The operator adjusted the threshold of the auditory feedback signal provided to the patient from session to session depending on the level of muscular tension. The operator's task was to shape successively lower EMG levels; the subject's task was to go below threshold and turn off the auditory signal completely. With this procedure, subjects learned gradually to reduce their muscle tension. The same physiotherapist took care of all patients and was instructed to adopt an encouraging attitude to lessen patient's involvement with relaxation exercises. Migraine sufferers underwent three consecutive sessions of biofeedback per week for a total of 12 sessions, with at least a 1-day interval between sessions. Migraine subjects were seated in comfortable recliners with closed eyes to enhance the effects of relaxation. Each biofeedback session lasted for 20 min.
Statistical analyses
Values of all the considered parameters were expressed as means ± SD. Student's paired t-test was used to compare the mean MIDAS score evaluated before and after the scheduled biofeedback treatment. Two-way
Results
Serum levels of NOx and serum activity of SOD were lower in chronic migraine sufferers before treatment than in healthy controls, whereasperoxide serum levels were higher. In migraine sufferers NOx serum levels and SOD serum activity were lower before than after treatment, whereas peroxide levels were higher in chronic migraine sufferers before than after treatment. No differences were observed in SOD serum activity, NOx or peroxide serum levels in chronic migraine sufferers after the biofeedback training sessions compared with healthy controls (Table 1).
NOx and peroxide serum concentrations and SOD serum activity in migraine sufferers before and after biofeedback training sessions and in healthy controls
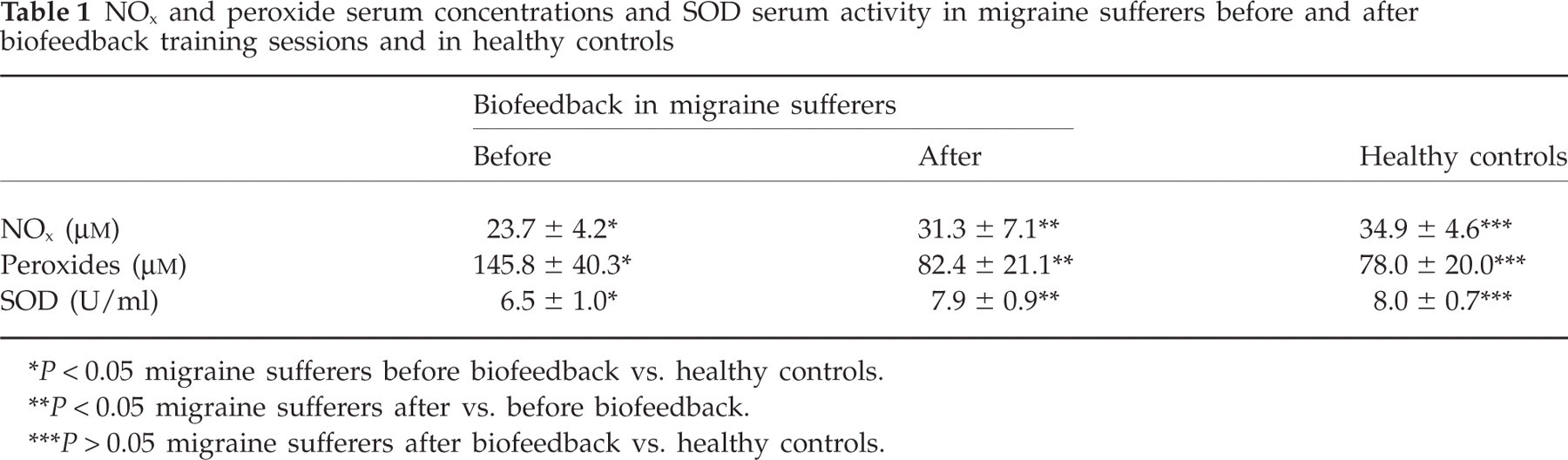
P < 0.05 migraine sufferers before biofeedback vs. healthy controls.
P < 0.05 migraine sufferers after vs. before biofeedback.
P > 0.05 migraine sufferers after biofeedback vs. healthy controls.
SOD serum activity correlated positively with NOx serum levels in healthy controls (r = 0.580; P < 0.05), in chronic migraine sufferers before biofeedback (r = 0.550; P < 0.05) and in chronic migraine sufferers after biofeedback training sessions (r = 0.768; P < 0.05) (Fig. 1).

Superoxide dismutase (SOD) serum activity positively correlates with NOx serum levels in healthy controls (a) (r = 0.580; P < 0.05), in chronic migraine sufferers before biofeedback (b) (r = 0.550; P < 0.05) and in chronic migraine sufferers after biofeedback training sessions (c) (r = 0.768; P < 0.05).
SOD serum activity correlated negatively with peroxide serum levels in healthy controls (r = –0.548; P < 0.05), in chronic migraine sufferers before biofeedback (r = –0.646; P < 0.05) and in chronic migraine sufferers after biofeedback training sessions (r = –0.677; P < 0.05) (Fig. 2).

Superoxide dismutase (SOD) serum activity negatively correlates with peroxides serum levels in healthy controls (a) (r = –0.548; P < 0.05), in chronic migraine sufferers before biofeedback (b) (r = –0.646; P < 0.05) and in chronic migraine sufferers after biofeedback training sessions (c) (r = –0.677; P < 0.05).
In chronic migraine sufferers the mean MIDAS score before biofeedback-assisted relaxation training was higher than that found after treatment (36.9 ± 13.9 vs. 18.8 ± 10.4; P < 0.001; Student's paired t-test).
Discussion
Chronic migraine is accompanied by a troubled quality of life and impaired daily activities, both requiring a preventive therapeutic approach.
In chronic migraine sufferers before biofeedback sessions decreased NOx serum levels and SOD activity and increased serum peroxide levels with respect to healthy controls were found. Several studies have shown conflicting results regarding NO levels in migraineur patients, possibly related to the small number of subjects enrolled and to the low reproducibility of results depending on different methodologies to determine NO levels, in patients with different headaches (4, 11, 27, 28). In our study, the lower NO bioavailability in migraine sufferers may be explained by decreased SOD activity, which might have permitted a quicker reaction with species of radicals such as peroxides. Therefore, enhanced vulnerability to oxidative stress, indicated by increased serum levels of lipid peroxidation final products, may promote changes in regional cerebral blood flow and the central sensitization phenomenon (11, 12, 29, 30). After the biofeedback sessions, enhanced NO bioavailability and reduced peroxides levels were observed in migraine sufferers compared with those measured before starting the biofeedback treatment. NO, whose bioavailability depends on the extent of oxidative reactions, is responsible for physiological and psychological relaxing effects, implicated in stress-related diseases. After the biofeedback sessions, increased SOD activity was detected compared with that observed before the relaxation treatment, suggesting that biofeedback may enhance SOD activity, preventing NO decomposition by scavenging superoxide anions and thus limiting oxidative reactions. In fact, a positive correlation was found between NO and SOD and a concomitant negative correlation between peroxides and SOD both in healthy controls and in migraine sufferers before and after biofeedback sessions, indicating that NO and peroxide bioavailability may depend on SOD scavenging activity. Therefore, SOD activity may represent an efficient anti-oxidant defence mechanism that limits the endothelium oxidative injury in chronic migraine sufferers (11). The biofeedback-promoted enhancement of NO bioavailability may be related to activation of the NO pathways, possibly facilitated by the relaxation therapies (31). In chronic migraine sufferers after biofeedback training sessions and in healthy controls, no significant differences in NOx and peroxide serum concentrations or in SOD activity were observed, suggesting the effectiveness of biofeedback in normalizing serum values to the levels found in healthy controls.
The effectiveness of biofeedback in limiting chronic migraine symptoms was also linked to the muscular relaxation obtained by improving affective distress (20, 32). We also found a lower mean MIDAS score after biofeedback relaxation training compared with that measured before treatment, confirming the effectiveness of biofeedback in decreasing headache disability.
In conclusion, biofeedback-promoted increase of SOD activity and NO bioavailability associated with a decrease of peroxides levels suggests a role of biofeedback training sessions as an appropriate therapeutic tool to reduce the vulnerability of chronic migraine sufferers to oxidative stress.
