Abstract
Prophylactic activity of flunarizine in migraine is attributed to its antioxidant properties and to the relief of cerebral vasospasm in which nitric oxide (NO) is involved. We investigated the antimigraine activity of flunarizine and its influence on NO and oxidative marker bioavailability in 25 subjects suffering from migraine without aura and in 25 healthy controls. Urinary samples collected before and after treatment with flunarizine (5 mg orally per day for 6 months) were assayed for NO stable metabolites (NOx) and thiobarbituric acid reactive substances (TBARS). Urinary levels of NOx and TBARS were higher in migraine sufferers before treatment than in healthy controls. No differences were observed in NOx levels in migraine sufferers, before and after flunarizine treatment; urinary TBARS levels were decreased after flunarizine treatment (P < 0.05) and remained persistently higher than in healthy controls (P < 0.05). Our results suggest that flunarizine did not prevent NO-mediated vasodilatation, while it proved effective in limiting the oxidative reactions occurring in migraine sufferers.
Introduction
Migraine treatment is currently focused on pain relief and on long-term reduction of frequency, severity, and duration of the attacks (1). However, it is not clear how prophylactic treatment may improve migraine symptoms that are associated with the underlying vascular and neuronal phenomena (2).
The calcium channel antagonist flunarizine is largely used in migraine prophylaxis because of its effectiveness in limiting frequency of the attacks and pain severity (2). The antimigraine activity of flunarizine, primarily linked to relief of cerebral vasospasm, possibly depends on its influence on nitric oxide (NO) generation through a reduction of neuronal NO-synthase activity and of calcium influx in perivascular cranial nerve terminals (3, 4). NO is considered the key molecule in migraine pathogenesis (5, 6), being involved in the activation of the trigeminovascular system and in the regulation of the cerebrovascular tone (7). Besides, as a non-adrenergic non-cholinergic neurotransmitter, NO facilitates transmission of noxious impulses, sensitizes perivascular sensory nerves, and processes painful stimuli (8, 9).
The vasoactive control of cerebral microcirculation is also modulated by the release of oxygen free radicals, including peroxynitrite, a product of the interaction of superoxide and NO (10, 11). Peroxynitrite and hydrogen peroxide reversibly dilatate the cerebral arterioles through an oxidant mechanism probably activating ATP-sensitive potassium channels while superoxide opens calcium-activated potassium channels dilatating cerebral arterioles (11–13). A widespread increase of cortical blood flow mediated by NO follows periaqueductal grey matter stimulation. Brain-stem structures may be at risk of iron-catalysed free radical injury as a consequence of repeated episodes of hyperoxya during migraine attacks (14). For this reason, migraine sufferers may be considered more vulnerable to the toxic effects of free radicals (15) whose interaction with NO may promote migraine-related cerebral blood flow changes (6, 16). The inhibitory effect of flunarizine on membrane damage and its activity as a free radical scavenger with antioxidant potential might play a role in controlling cerebral vasomotion and in preventing mitochondrial metabolism dysfunction also involved in migraine pathogenesis (17–20).
To investigate whether flunarizine antimigraine action might also depend on its influence on NO and oxidative marker bioavailability, we measured urinary NO stable metabolites, derived from NO plasmatic oxidation (21) and lipid peroxidation by-products, before and after flunarizine prophylactic treatment.
Methods
Twenty-five migraine sufferers, 19 women and six men (mean age ± SD 34 ± 16 years) affected from migraine without aura and 25 matched healthy controls, 19 women and six men (mean age ± SD 35 ± 12 years) were enrolled into the study. Migraine without aura was diagnosed according to the criteria of the Headache Classification Committee of the International Headache Society (22). All subjects were non-smokers, free of cardiovascular, endocrine, renal, hepatic, neurological, psychiatric, and coagulation diseases. Subjects with gastrointestinal and extradigestive infections, pregnancy, and treatment with migraine prophylactic drugs in the previous 6 months were also excluded (23, 24). Migraine sufferers were invited to collect urinary samples throughout 24 h during the headache-free period: before starting flunarizine treatment and 24 h after last dose administration. Patients were carefully instructed to avoid any nitrite/nitrate-enriched diet for at least 4 days before the assessment and during all the study period thereafter (25). In women, urine collection was performed out of the menstrual cycle
Urinary sample processing
Urinary samples were ultrafiltered and centrifuged at 17 089
Determination of urinary NO stable nitrite/nitrate metabolites (NOx)
NO is a free radical rapidly decomposed to nitrite and nitrate (NOx). NOx were measured by reducing nitrate to nitrite by the action of nitrate reductase from Aspergillus niger (26) and measuring nitrite by a sensitive fluorimetric assay based on the reaction of nitrite with 2,3-diaminonaphthalene to form the fluorescent product 1-(H)-naphthotriazole (27). Intensity of the fluorescent signal of the samples was measured and compared with known concentrations of sodium nitrite (excitation wavelength 375 nm, emission wavelength 415 nm). The fluorimetric assay was able to detect amounts in the order of 30 n
NOx corrected by creatinine excretion were expressed as mmol/mmol creatinine. Urinary creatinine was evaluated using a commercial assay kit (Sigma-Aldrich Fine Chemicals, St Louis, MO, USA) based on the reaction of creatinine with picric acid under alkaline conditions.
Determination of urinary thiobarbituric acid reactive substance
Lipid peroxidation was measured in urine using the thiobarbituric acid assay which detected thiobarbi-turic acid reactive substance (TBARS) generated by peroxidative process, including lipid peroxides and malondialdehyde. Urinary TBA products were formed at pH 2.5 by heating 1 ml urine with TBA reagent at 100°C, for 30 min (28). Determination of TBARS was carried out fluorimetrically (excitation wavelength 510 nm, emission wavelength 553 nm); the quantitative evaluation was made using a standard curve obtained from a stock solution of 10 m
Statistical analysis
The Wilcoxon paired test was used to compare frequency and intensity of migraine attacks before and after flunarizine treatment. Student's unpaired t-test was used to determine the statistical significance of NOx and TBARS differences between migraine sufferers and healthy controls. Student's paired t-test was carried out to compare mean levels of urinary NOx and TBARS measured before and after flunarizine treatment. Statistical significance was accepted when P < 0.05.
We did not adjust for multiple comparisons, since in this exploratory study statistics were regarded as merely descriptive.
Results
The frequency of migraine attacks during flunarizine prophylactic treatment decreased when compared with that observed before treatment (6.84 ± 2.43 vs 3.36 ± 1.96, P < 0.005; Wilcoxon paired test). A reduction in the frequency of migraine attacks of 50% was observed in 18 of the 25 migraine sufferers (72%). The severity of migraine attacks also decreased during flunarizine treatment with respect to that reported before treatment (2.03 ± 0.40 vs 1.47 ± 0.67, P = 0.001; Wilcoxon paired test). No treatment withdrawals were reported.
As shown in Table 1, urinary levels of NOx and TBARS were higher in migraine sufferers before treatment than in healthy controls (Student's unpaired t-test). No differences were observed in NOx urinary levels in migraine sufferers before and after flunarizine treatment (0.72 ± 0.15 vs 0.75 ± 0.10 mmol/mmol creatinine, P = 0.413; Student's paired t-test).
Urinary nitric oxide stable metabolites (Nox) and thiobarbituric acid reactive substance (TBARS) levels in migraine sufferers before flunarizine treatment and in healthy controls
As shown in Fig. 1, in migraine sufferers TBARS urinary levels were lower when measured after flunarizine treatment than before starting treatment (0.35 ± 0.07 vs 0.43 ± 0.12, P < 0.05; Student's paired t-test). Again, TBARS levels in migraine sufferers after flunarizine treatment (Fig. 2) were persistently higher than in healthy controls (0.35 ± 0.07 vs 0.25 ± 0.08, P < 0.05; Student's unpaired t-test).
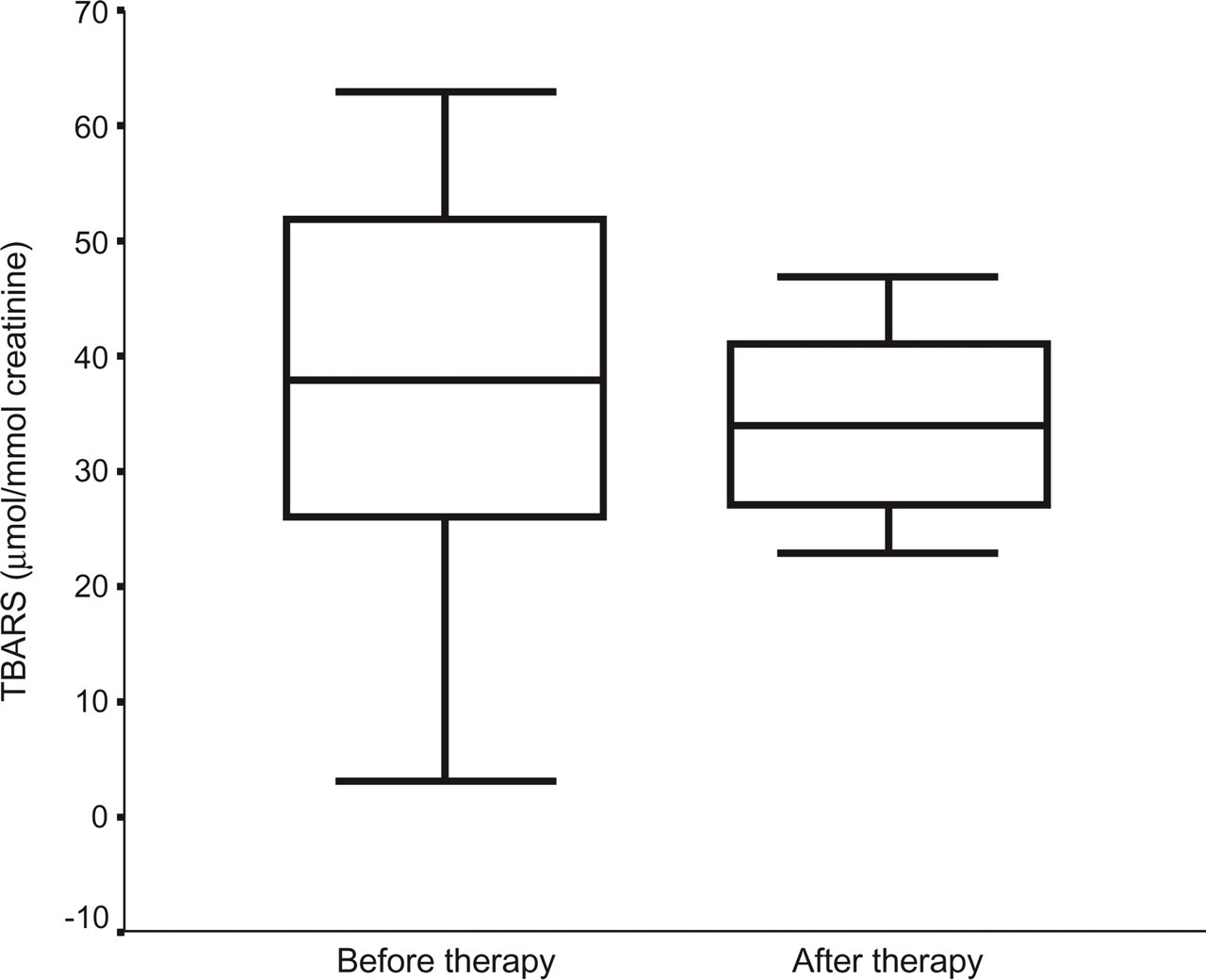
Thiobarbituric acid reactive substance (TBARS) urinary levels before and after flunarizine treatment.

Thiobarbituric acid reactive substance (TBARS) urinary levels in migraine sufferers after flunarizine treatment and in healthy controls.
Discussion
In migraine sufferers, prophylaxis is aimed to avoid the attacks and the related symptoms, so improving quality of life. In our patients, flunarizine treatment was clinically effective, decreasing frequency and pain severity of the attacks, after a 6-month treatment. However, the mechanism by which flunarizine, a calcium channel antagonist, is involved in preventing migraine attacks is still uncertain. It has been suggested that flunarizine may act as a migraine prophylactic drug by inhibiting Ca2+-dependent enzymes involved in prostaglandin biosynthesis, in the modulation of cerebral and extracerebral vasomotion, and possibly, in blocking the release of 5-HT (1). Besides, flunarizine, which crosses the blood–brain barrier and modulates several nerve functions (29), may also prevent neuronal hypoxia and neurovascular inflammation (1, 30).
It has recently been reported that the effectiveness of flunarizine in migraine prophylaxis may depend on its inhibitory influence on NO-synthase leading to a decrease in the production and release of NO from the perivascular nerve terminals of intra- and extracranial vessels (4). Therefore, the antimigraine activity of flunarizine might involve its counteraction of the NO production mechanisms, underlying the vascular phenomena taking place in migraine. Our results do not support the hypothesis that flunarizine may work as a molecule inhibiting systemic NO bioavailability (4). Indeed, the lack of any relevant variation of urinary levels of NO stable metabolites measured before and after flunarizine prophylactic treatment suggests that this drug may be ineffective in preventing NO-mediated vasodilatation and in stimulating the trigeminal nerve endings (30, 31). Therefore, as already reported (15), our findings support the difference in NO bioavailability between migraine sufferers and healthy controls during the headache-free period, indicating that the biochemical modification of the L-arginine/NO pathway may be peculiar to migraine headache.
In vitro and ex vivo research indicates that the counteracting protective effect of flunarizine against endothelial and vascular smooth muscle injuries may depend on inhibiting the endothelium-dependent hypoxic facilitation and on its haemorheological effects (32, 33). Flunarizine is also a powerful scavenger of free radicals, namely in the mitochondria, where its effectiveness is three time greater than that of α-tocopherol, a well-known intracellular antioxidant (17). Furthermore, flunarizine antioxidant potential contributes to antagonize peroxidation of membrane lipid bilayer (19, 34, 35). We found a decrease of lipid peroxidation by-products after flunarizine treatment, supporting the hypothesis that the drug may limit oxidative reactions involved in the vasoactive control of cerebral blood flow in migraine sufferers by reducing production and release of hydrogen peroxide and superoxide radicals. However, flunarizine appears ineffective in lowering TBARS at levels comparable to those usually observed in healthy controls. Furthermore, the decreased TBARS values observed after prophylactic treatment may suggest a limited reaction between NO and free radicals, reducing peroxynitrite formation. Therefore, flunarizine, according to our data, may reduce the systemic oxidative stress of migraine sufferers and may possibly balance cerebral blood flow changes, thus protecting the cerebral endothelium against any oxidative insult. Moderation of oxidative stress after flunarizine treatment may avoid any mitochondrial energy metabolism impairment in migraine sufferers.
In conclusion, our results suggest that the antimigraine effects of flunarizine may rely on its antioxidant properties.
