Abstract
Craniofacial nociceptive processing in patients (n = 25) suffering from unilateral cluster headache was assessed by laser-evoked cortical potentials (LEPs). Latencies and amplitudes of late (N2, P2) and middle-latency (N1) LEPs were measured in chronic (CCH, n = 9) and episodic cluster headache (ECH, n = 17). In CCH patients on headache side N1c occurred later and P2 amplitude was smaller than on the healthy control side. In active periods of ECH patients P2 latency was shorter on the headache side. In remission periods of ECH patients the N2P2 ratio was lower on the headache side. In 19 out of 26 examinations in 25 headache patients LEP deviated from normative data in healthy controls (n = 10) without any specific pattern of altered parameters. LEPs document pathological changes in craniofacial nociception in cluster headache. However, there seems to be no pathognomonic deviation pattern that enables reliable diagnosis of cluster headache and application of LEPs in further studies of pathophysiological mechanisms.
Introduction
Severe, strictly unilateral pain attacks in the periorbital region with a duration between 15 and 180 min characterize cluster headache (1). Whereas periods of active headache and remission alternate in episodic cluster headache (ECH; International Classification of Headache Disorders ICHD-II code 3.1.1), attacks recur over >1 year without remission periods in chronic cluster headache (CCH; ICHD-II code 3.1.2) (1).
Basic neuroscience studies stress sustained plastic changes of nociceptive synaptic transmission by repetitive noxious input (2–5). High-frequency burst activity and low-frequency excitation of nociceptive afferents may induce long-term potentiation and long-term depression of nociception, respectively. Both electrophysiological phenomena are models of synaptic plasticity and play important roles in learning and memory processes (6, 7). Sustained alterations of nociceptive processing may be involved in chronification of pain (3). Repetitive headache attacks with episodic or chronic characteristics for years are pathognomonic for cluster headache. Thus, craniofacial nociception may be altered in these patients.
Nociceptive processing in human subjects can be assessed by noxious laser heat stimulation that selectively excites Aδ and C fibre nociceptors in hairy skin (8–11). Laser-evoked cortical potentials (LEPs) in healthy volunteers primarily represent cerebral responses to activation of Aδ nociceptors (12, 13). The earliest cortical LEP component is a small negative bilateral N1 with maximum amplitude at temporal leads (14, 15). The most prominent signal is the negative–positive N2P2 complex with maximum amplitude at the vertex (12, 15, 16). The precise brain generators of LEP are still under debate. However, bilateral operculoinsular cortices, the contralateral primary somatosensory cortex and the anterior cingulate cortex seem to be major sources of vertex LEP in man (15–22). In particular, LEP recordings under laser stimulation of orofacial skin have been performed in healthy volunteers and patients suffering from migraine and tension-type headache (23–28).
The present study addressed craniofacial nociceptive processing in cluster headache patients and healthy volunteers by applying LEP. A preliminary report of the data was presented at the International Headache Congress in Kyoto (29).
Methods
LEPs were recorded in four female and 21 male cluster headache patients (n = 25) between 19 and 69 years of age (mean ± SD 45.7 ± 12.2 years). Diagnoses were made by pain management physicians according to the International Classification of Headache Disorders (ICHD-II) (1). Nine and 16 patients suffered from CCH and ECH, respectively. Within the ECH group, seven patients were investigated in the cluster period (ECHP) and 10 patients in the remission period (ECHR). One ECH patient participated in the remission and cluster periods. All cluster headache patients suffered from unilateral attacks without side shift. Unilateral cluster headache occurred in 11 patients on the right side and in 14 patients on the left side. Normative data were collected in two female and eight male healthy volunteers (n = 10) aged from 23 to 62 years (43.2 ± 11.9 years) (Table 1). Twenty-three patients and nine healthy volunteers were right-handed. All participants gave their informed consent prior to inclusion in the study according to the 1964 Declaration of Helsinki (http://www.wma.net). The protocol was approved by the local ethics committee of the RWTH Aachen University. Each experiment lasted approximately 2.5 h. Volunteers were awake, relaxed with eyes closed and sitting comfortably in a reclined chair.
Normative data of laser-evoked cortical potential parameters in healthy volunteers (n = 10)

Painful heat test stimuli were applied to the supraorbital region by a Thulium-YAG laser that emitted infrared light of 2 µm wavelength for 1 ms (Carl Baasel Lasertechnik, Starnberg, Germany). The stimulation area of about 40 cm2 within the cutaneous innervation territory of the supraorbital nerve (terminal branch of the ophthalmic nerve) was clearly separated from forehead midline (>1 cm distance) and approximated the innervation territory of the infraorbital nerve at the temple. These laser stimuli selectively excite Aδ and C fibre nociceptors in hairy skin inducing pricking painful sensations (9–12, 30). In order to avoid adaptation of nociceptors and skin reddening, the stimulation spot (5 mm diameter) was slightly shifted from stimulus to stimulus within the periorbital region. It is generally accepted that this selective excitation of cutaneous nociceptors generates a typical pattern of cortical potentials that is recorded by electroencephalography (31). LEPs were recorded at positions Fz, Cz (vertex), and Pz vs. a left earlobe (A1) reference and at positions T3 and T4 vs. an Fz reference according to the international 10–20 system (band pass 0.08–30 Hz). T3 and T4 recording positions were renamed as contralateral Tc and ipsilateral Ti according to side of laser stimulation. The electrooculogram of the right eye was recorded in order to monitor possible artefacts due toeye and lid movements (band pass 0.01–100 Hz). Laser-induced detection (I0) and pain (IP) thresholds were determined by the method of limits with four series of descending and ascending intensities (32). Noxious laser stimuli were applied in four stimulation series of 50 stimuli each (Fig. 1). In order to reduce habituation of LEP, two different stimulus intensities were applied with variable interstimulus intervals between 8 and 12 s. The low energy was slightly above the individual pain threshold (+20%) and the high-intensity stimulus was adjusted to energy of about 60% above the individual pain threshold. Individual stimulus intensities were adjusted to identical laser energy on both sides. An audible signal, 1.5 s after each laser stimulus, prompted the volunteer to rate the laser-evoked sensation intensity (rating) according to a common ordinal scale (0, no perception; 1–39, non-painful perception; 40–100, pricking painful). Two laser stimulation series each were performed on each side of the face (periorbital region). The first stimulation series was executed on one side (right or left, cluster headache or control side), the second immediately followed on the contralateral periorbital region. After a break of 15 min, the stimulation series was continued in reverse order, i.e. stimulation series were conducted in the succession left–right–right–left or right–left–left–right (Fig. 1). This stimulation sequence was chosen in order to exclude intrinsic inhibitory effects on LEP by habituation (compare (33)). Electrophysiological recordings were performed by utilizing the EEG bioamplifier VD32 (Schwarzer, Munich, Germany), Power1401 A/D-converter and Signal software (http://www.ced.co.uk). Within one laser stimulation series, 25 LEP sweeps evoked by high-energy laser stimuli were averaged. Latencies of N2 and P2 components and peak-to-peak amplitude between N2 and P2 were determined. Latencies and amplitudes of contralateral and ipsilateral N1 components were measured in temporal leads T4 and T3, respectively. Only LEP components that reproducibly occurred in both series on one side were considered for statistical analysis (Fig. 1). In order to assess tendency of LEP habituation, ratios were calculated (2nd laser series/1st laser series) for bilateral N1 and N2P2 amplitudes (compare (33)).

Trigeminal laser-evoked cortical potentials (LEPs). (a) Noxious laser stimuli were applied in four stimulation series of 50 stimuli each. In order to reduce habituation to LEP, two different stimulus intensities were applied with variable interstimulus intervals between 8 and 12 s. The low energy was slightly above the individual pain threshold (grey) and the high-intensity stimulus was adjusted to energy of about 60% above the individual pain threshold (black). Two laser stimulation series each were performed on each side of the face (periorbital region). The first stimulation series was executed on one side (right or left, cluster headache or control side), the second immediately followed on the contralateral periorbital region. After a break of 15 min, the stimulation series was continued in reverse order. This stimulation sequence was chosen in order to minimize the intrinsic inhibitory effects on the LEP by habituation. (b) LEPs evoked by laser stimulation of the right periorbital region in one specimen (male, 30 years old, high laser energy 550 mJ, pain threshold 370 mJ). LEPs were recorded at positions Cz, T3 (contralateral, Tc) and T4 (ipsilateral, Ti). Within one laser stimulation series, 25 LEP sweeps evoked by high-energy laser stimuli were averaged (first series light grey, second series dark grey). An individual average of both series averages was calculated (black). Latencies of N2 and P2 components and peak-to-peak amplitude between N2 and P2 were determined. Latencies and amplitudes of contralateral N1c and ipsilateral N1i components were measured in temporal leads.
Data were described by arithmetic mean and SD (e.g. age of volunteers, stimulus intensity) and by median. The statistical analysis was performed by paired t-test and by Wilcoxon signed rank test. The level of significance was set at P < 0.05. Normative data of LEP parameters were calculated by applying the 99% confidence interval (CI). SigmaStat® software 3.0 (SPSS Inc., Chicago, IL, USA) was used.
Results
In 10 healthy volunteers noxious laser radiant heat with an average intensity of 498.0 ± 40.2 mJ (corresponding to 159.6 ± 26.9% of IP and 212.3 ± 46.0% of I0) evoked cortical potentials consisting of bilateral middle-latency N1, late N2 and P2 components (Fig. 1). In three and seven volunteers the laser series started on the right and left side, respectively. Latency and amplitude data were tested for side-to-side differences by paired tests (n = 10). Laser stimulation on the left side generated larger N2P2 peak-to-peak amplitudes (22.4 ± 15.0 µV vs. 19.4 ± 14.3 µV; paired t-test: t = −3.28, P < 0.01) and smaller N2 amplitudes (−11.3 ± 8.4 µV vs. −9.0 ± 6.6 µV; paired t-test: t = −3.00, P < 0.05). LEP parameters without side-to-side differences were pooled in order to calculate normative data (99% CI; Table 1). The contralateral N1c component arose earlier than the ipsilateral N1i (132.0 ± 13.6 ms vs. 134.0 ± 13.6 ms; paired t-test: t = −2.30, P < 0.05). Amplitudes of N1c and N1i differed significantly (−9.1 ± 3.5 µV vs. −7.2 ± 2.9 µV; Wilcoxon signed rank test: P < 0.05).
In cluster headache patients LEPs were evoked with stimulus intensities of 521.2 ± 75.8 mJ corresponding to 150.8 ± 25.4% of individual pain thresholds and 200.5 ± 42.0% of individual detection thresholds (Fig. 2a). Individual stimulus intensities were adjusted to identical laser energy on both sides. In eight and 18 patients, the stimulation series started on the right and left side, respectively. In some patients the LEP pattern was incomplete (Table 2). In one ECHR patient no reproducible LEP could be elicited on either side (patient 21; seeTable 2). Additional measurement of heat pain thresholds with a Peltier type thermode at the forehead showed increased thresholds of 49.6°C and 49.2°C on the right and left sides. This male patient (59 years old) had suffered from ECH since the age of 38. Headache periods typically arose for 3 months in spring and fall with about two attacks per day. Verapamil 240 mg was the prophylactic treatment, acute attacks responded to inhalative oxygen. In one ECHR and two CCH patients N1 components were missing on both sides. On control sides in two ECHP patients laser stimulation did not evoke any N1 component. N2 and P2 did not occur on the headache side of one ECHR patient, while both components were normal on the other side. In one ECHR patient P2 was lacking bilaterally.

Trigeminal laser-evoked cortical potentials (LEPs) in patients suffering from unilateral cluster headache. Grand averages of LEPs evoked by noxious laser stimulation on cluster headache side (black) and healthy control side (grey) in all 26 patients (a), in chronic cluster headache (b), in episodic cluster headache during active periods (c) and during remission periods (d).
Cluster headache patients with noticeable laser-evoked cortical potential findings
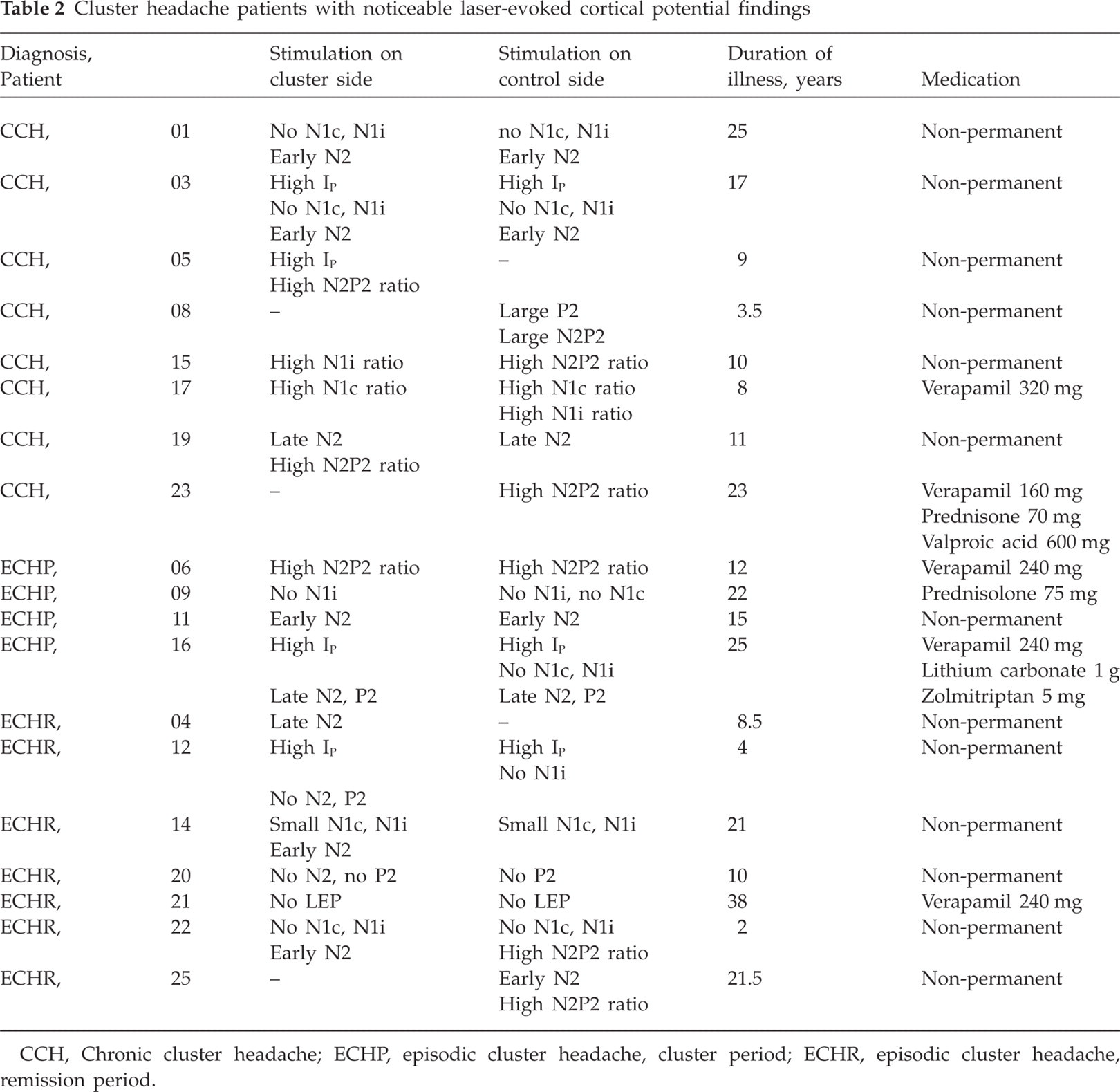
CCH, Chronic cluster headache; ECHP, episodic cluster headache, cluster period; ECHR, episodic cluster headache, remission period.
LEP parameters after left- and right-side laser stimulation were tested statistically for intraindividual side differences. Laser detection thresholds (control 277.1 ± 98.9 mJ, headache 277.9 ± 122.3 mJ) and pain thresholds (control 354.2 ± 98.0 mJ, headache 366.2 ± 123.9 mJ) did not differ statistically between control and cluster headache sides. In CCH patients N1c occurred later after stimulation on the headache side (median 148.4 ms vs. 135.5 ms; Wilcoxon signed rank test: P < 0.05; n = 7) and the P2 amplitude was larger under control side stimulation (8.2 ± 9.7 µV vs. 10.9 ± 11.4 µV; t-test: t = −3.07, P < 0.05; n = 9) (Fig. 2b). In ECHP (n = 7) laser stimulation on the headache side evoked an earlier P2 than contralateral stimulation (257.1 ± 47.4 ms vs. 264.2 ± 48.0 ms; t-test: t = −2.78, P < 0.05) (Fig. 2c). In ECHR patients the N2P2 ratio was lower on the cluster side in comparison with the control side (0.86 ± 0.30 vs. 1.09 ± 0.34; t = −2.749, P < 0.05; n = 8) (Fig. 2d). Side-to-side comparisons of all other LEP components including amplitude ratios in all cluster headache examinations (n = 26), in CCH (n = 9), in ECH (n = 17) and in ECHR (n = 10) patients showed no significant differences.
LEP data were compared with normative values from healthy volunteers (Table 2). Nineteen of all 26 electrophysiological examinations in 25 cluster headache patients showed noticeable LEP findings. These 19 patients consisted of eight CCH, four ECHP and seven ECHR participants. The laser pain threshold was raised in three patients (03, 16, 12) on both sides (cluster side/control side in mJ: 460/530, 460/490, 860/670) and in one patient (05) only on the headache side (480 mJ vs. 430 mJ). N1c and N1i amplitudes in one ECHR patient (14) were below the lower range after stimulation of both sides. In three (01, 03, 11) and two (19, 16) patients N2 latencies decreased (cluster side/control side in ms: 127.2/134.7, 134.7/132.5, 117/117) and increased (cluster side/control side in ms: 191.2/202.2, 265.4/271.3), respectively, on both sides. In one (04) and two (14, 22) ECHR patients N2 latency increased (187.9 ms) and decreased (140.6 ms, 139.8 ms), respectively, only after laser stimulation on the headache side. N2 latency was reduced in one ECHR patient (25) only on the healthy control side (140.6 ms). In one ECHP patient (16), P2 latency was significantly raised on both sides (cluster side/control side in ms: 348.9/355.6). In one CCH patient (08), P2 and N2P2 amplitudes on the control side increased above upper ranges to 36.3 µV and 60.2 µV, respectively. In seven and two patients, the N2P2 ratio and N1 ratio, respectively, were higher than the upper limit. The deviant LEP findings in cluster headache patients were similarly distributed among headache and the contralateral control sides. The most frequent findings concerned different changes in N1c, N1i and N2 components. The results suggested no common LEP pattern in cluster headache patients.
Discussion
Assessment of craniofacial nociceptive processing by LEP documented alterations in patients suffering from unilateral cluster headache. In CCH patients on the headache side N1c occurred later and the P2 amplitude was smaller than on the healthy control side. In active periods of ECH patients P2 latency was shorter on the headache side. In ECH remission periods the N2P2 ratio was lower on the headache side. Results of side-to-side comparison of LEP data in headache patients are compatible with central changes of nociception and contradict a simple dysfunction of peripheral nociception that usually induces global delay and/or amplitude reduction of all components (12, 31, 33). In one ECHR patient reproducible LEPs did not occur on both sides. A reasonable explanation for this result did not arise from the individual medical history as collected by a pain questionnaire (German Pain Society). The extraordinary long duration of cluster headache of 38 years may suggest an explanation. Additional medication, possibly not reported by the patient, may also be a reason. Whereas 19 out of 26 examinations in 25 patients showed pathological deviation of craniofacial nociceptive processing as assessed by LEP, no typical pattern of altered parameters could be characterized.
Trigeminal somatosensory cortical potentials were evoked by non-painful cutaneous electrical stimulation at the corner of the mouth (34) and of the infraorbital nerve (35) in cluster headache patients (36, 37). During active cluster periods N2 latency of trigeminal potentials was longer on the headache side compared with the non-symptomatic side. N1, P1 and N2 latencies after electrical stimulation of the headache side were longer than in healthy controls. In the remission period N1 latencies of both sides were longer than in healthy controls. Amplitude changes were not demonstrated (34). With infraorbital nerve stimulation all eight patients tested during an attack showed a delayed or absent W2 and W3 component of trigeminal evoked potentials, while only three out of 24 patients tested outside the attack had delayed W2 and W3 (35). In contrast to the present LEP study, electrically evoked potentials in the cited study represent processing of tactile information mediated by thick-myelinated fibres. Furthermore, recorded short-latency potentials correlated to excitation of perioral afferents, but not periorbital sensory receptors where cluster headache attacks usually occur. The electrically elicited corneal reflex has been studied in 21 cluster headache patients (38). A significant reduction of pain thresholds was observed during the active phase, while normal values were recorded during remission. Ten out of 15 patients showed a significantly reduced corneal pain threshold on the headache side, while tactile sensitivity was unaffected. A recent study assessed somatosensory processing in cluster headache by applying quantitative sensory testing (39). Perception of warmth, cold and pressure pain was reduced on the cluster side compared with the contralateral asymptomatic side. No alteration of heat pain perception was demonstrated. Similar laser pain thresholds in control subjects and cluster patients in the present electrophysiological study are compatible with the psychophysical investigation.
LEP studies in primary headache have been performed in patients suffering from migraine and tension-type headache (26–28). In migraine patients, N1, N2 and P2 LEP components showed normal values (27, 28). Reduced habituation behaviour of the N2P2 complex was the remarkable finding in migraineurs (27, 28). In chronic tension-type headache patients N2P2 amplitude was larger when stimulating tender points compared with control subjects (26). While reduced habituation in migraine is suggested to be evidence for abnormal excitability of cortical areas involved in pain processing, larger LEP at tender points in tension-type headache may be an indication of a primary phenomenon that precedes headache (26, 28). In seven of 26 cluster headache patients in the present study, the N2P2 ratio was above the upper limit of normative data. In ECHR patients the N2P2 ratio was significantly smaller on the headache side than on the control side, indicating stronger habituation. Thus, LEP findings in migraine, tension-type headache and cluster headache are quite different.
Latency and amplitudes of laser-evoked vertex potentials in healthy control subjects in the present study were very similar to those of previous experimental studies (23–25, 28). The present mean N2 latency (163 ms), P2 latency (257 ms) and N2P2 amplitude (21 µV) are in the middle of corresponding ranges in the literature (N2 150–179 ms, P2 228–275 ms, N2P2 12–29 µV). The middle-latency N1 component from temporal lead recording evoked by periorbital laser stimulation so far has only been published in one study in healthy subjects. Valeriani and colleagues (28) applied painful radiant heat pulses of 10 ms duration by an infrared CO2 laser stimulator (10.6 µm wavelength). N1 occurred at 112.8 ms and 114.5 ms with amplitudes of 3.7 µV and 4.6 µV after right and left face stimulation, respectively (28). N1 latency was measured in the monopolar electrode montage on the contralateral side, while N1 amplitude was calculated in bipolar temporofrontal deviation as so-called N1–P1 amplitude. In the present study N1 components showed longer latency of about 133 ms and larger amplitude of about −8 µV (Table 1). Methodological differences principally may explain differences in LEP latencies and amplitudes (11, 12). In the present study healthy subjects were older (43.2 vs. 32.5 years), shorter laser pulses (1 ms vs. 10 ms) of shorter wavelength (2 µm vs. 10.6 µm) were applied, and evoked potential measurement of latency and amplitude differed (Fz reference). Whereas Valeriani and colleagues applied laser stimulus intensities of 2.5-fold a sensory threshold (28), in the present study laser intensity was adjusted to 1.5-fold the pain threshold and about twofold the detection threshold. In particular, a lower stimulus intensity would explain longer latencies.
Medication may affect LEPs. Eleven cluster headache patients were permanently treated with various drugs (lithium, corticosteroids, valproic acid, verapamil, etc.), LEPs were affected in only six patients. Of 15 patients without permanent pharmacological treatment, LEPs were pathologically altered in 13. Thus, LEPs were more frequently affected in patients without permanent drug therapy. However, some studies have shown LEP modulation by different medications in man. Laser-evoked vertex potential obtained from UV-irritated skin in healthy volunteers decreased after oral administration of the non-steroidal anti-inflammatory drug ibuprofen (40). LEPs after laser stimulation of untreated human skin were inhibited by sole application of acetylsalicylic acid and by combined treatment with trilithium citrate (41, 42). Paracetamol and codeine administration also reduced LEPs (43). Studies on possible effects of the frequently administered drug verapamil on LEP do not exist.
The present electrophysiologicaI study is the first in cluster headache patients applying laser-evoked cortical potentials. Whereas most of the patients showed alterations of craniofacial nociceptive processing, no typical pattern could be elaborated. Thus, LEPs seem not to be helpful in diagnosis and experimental investigation of cluster headache pathophysiology.
Acknowledgements
This study was supported by grants of the Interdisciplinary Centre for Clinical Research of the Medical Faculty of RWTH Aachen University. We thank the self-help group CSG for their support.
