Abstract
The following systematic reviews and meta-analyses are presented and the results discussed: the evidence-based American guidelines, five systematic reviews on naratriptan, rizatriptan, eletriptan, sumatriptan and propranolol; a meta-analysis of sumatriptan, a meta-analysis of acute migraine therapy, a meta-analysis of triptans available in Canada and a large meta-analysis of oral triptans. The systematic reviews of several randomized trials of one drug overcome random effects in estimating treatment effect of the reviewed drug. The results from the large meta-analysis of several drugs are compared with head-to-head comparative trials. Results are generally the same in the meta-analysis and in the comparative trials, with some exceptions. Head-to-head comparisons should remain the ‘gold standard’ and meta-analyses are a useful supplement in cases when comparative trials are relatively small and when no comparative trials exist.
Introduction
In 1996 Sackett et al. defined evidence-based medicine as follows: ‘Evidence based medicine is the conscientious, explicit, and judicious use of current best evidence in making decisions about the care of individual patients. The practice of evidence based medicine means integrating individual clinical expertise with the best available external clinical evidence from systematic research (1) … It is when asking questions about therapy that we should try to avoid the nonexperimental approaches, since they routinely lead to false positive conclusions about efficacy. Because the randomised trial, and especially the systematic review of several randomised trials, is so more likely to inform us and so much less likely to mislead us, it has become the “gold standard” for judging whether a treatment does more good than harm’ (1).
Best clinical evidence is now available in migraine therapy and in the following a review by the American Consortium, six systematic reviews and five meta-analyses of drugs used for migraine therapy are presented and discussed.
Review by the American Consortium
In 2000 the US Headache Consortium, consisting of seven member organizations with an interest in improving the quality of care for migraine patients, completed a landmark evidence-based review of the literature concerning the diagnosis and treatment of migraine (2–8). This was based on a literature search in English for controlled trials of treatments for migraine from 1966 to 1996, with additions of some newer randomized clinical trials (RCTs). The Consortium then reached agreement on a scale for quality of the evidence, as shown in Table 1.
In addition to this scale for quality of evidence, A to C, the drugs were graded on scales for scientific effect and for clinical impression of effect.
Acute migraine drugs
On the scale for quality of the evidence ergotamine received a grade B, dihydroergotamine nasal spray received a grade A, butalbital plus aspirin plus caffeine received a grade C, aspirin a grade A, acetaminophen plus aspirin plus caffeine a grade A, naproxen a grade A, ibuprofen a grade A, flurbiprofen a grade B, tolfenamic acid a grade A, paracetamol a grade B, isometheptene-containing compounds a grade B, corticosteroids a grade C, parenteral opiods a grade B, butorphanol nasal spray a grade A and triptans (sumatriptan nasal spray, sumatriptan subcutaneous injection, oral sumatriptan, naratriptan, zolmitriptan and rizatriptan) received a grade A (5).
This grading of acute treatment drugs for migraine is, however, to some extent controversial as discussed in the following.
Ergotamine received a grade B in accordance with a European Consensus (9) which recommended that, if used, ergotamine should be given by the rectal route. Aspirin received a grade A but should be graded in combination with metoclopramide and should in this combination receive a grade A (10). Paracetamol plus aspirin plus caffeine received a grade A. This rating is based on three randomized, placebo-controlled controlled trials published in one paper (11). It should be noted, however, that the most disabled segment of migraineurs, including those who usually required bed rest or who vomited 20% or more of the time, were excluded (11). The results can thus not be generalized to all migraine patients. Butorphanol nasal spray received a grade A based on two RCTs; but in one of the RCTs 57% of patients rated the drug as poor, probably due to frequent adverse events (AEs) (12). Buthorphanol should receive only grade B. The triptans, which have been investigated with up-to-date methodology (13, 14), rightly received a grade A and they are currently the mainstay in the acute management of migraine (15).
Prophylactic migraine drugs
Preventive drugs were similarly ranked A, B and C according to quality of evidence, but were also ranked according to clinical impression of effect and AEs:
Group 1. Medication with proven high efficacy and mild to moderate AEs.
Group 2. Medication with lower efficacy (i.e. limited number of studies, studies reporting conflicting results, efficacy suggesting only ‘modest’ improvement) and mild to moderate AEs.
Group 3. Medication use based on opinion, not randomized trials: (i) mild-to-moderate AEs; (ii) frequent or severe AEs (or safety concerns), complex management issue (special diets, high potential for severe adverse drug interactions, or drug holidays).
Group 4. Medication with proven efficacy but with frequent or severe AEs (or safety concerns), complex management issue (special diets, high potential for severe adverse drug interactions, or drug holidays).
Group 5. Medication proven to have little or no efficacy.
For preventive treatment of migraine the following ratings for quality of evidence and total ranking, in brackets, were given: clonidine received a grade B [5], carbamazepine a grade B [5], divalproex and valproate a grade A [1], gabapantin a grade B [2], vigabatrine a grade B [5], amitriptyline a grade A [1], nortriptyline a grade C [3a], fluoxetine a grade B [2], phenelzine a grade C [3b], atenolol a grade B [2], metoprolol a grade B [2], nadolol a grade B [2], propranolol a grade A [1], timolol a grade A [1], flunarizine a grade B [4], nimodipine a grade B [2], verapamil a grade B [2], ketoprofen a grade B [2], tolfenamic acid a grade B [2], naproxen a grade B [2], methysergide a grade A [4], pizotifen a grade A [4] and lisuride received a grade A [1] (8).
I have the following comments on these ratings. Clonidine rightly received a Grade B [5]. Three early double-blind, placebo-controlled RCTs apparently demonstrated the efficacy in migraine prophylaxis but later RCTs vs. placebo failed to confirms this (16). Clonidine is rarely used for migraine prophylaxis. Carbamazepin, grade B [5], has been found superior to placebo in only one RCT (17, 18) and is seldom used in migraine prevention. Amitriptyline received a grade A [1] but AEs, dry mouth and sedation are frequent and often limit the clinical use of amitriptyline. There are four RCTs showing superiority of amitriptyline vs. placebo (16) and the drug should receive grade A [4]. Divalproex sodium and valproate received a grade A [1] based on four RCTs (18–22). They are second-line drugs for migraine prophylaxis but their use is hampered by AEs such as sedation, weight gain and tremor, and a grade A [2] would be more appropriate. Timolol, which was superior to placebo in three RCTs (23–26), was equivalent to propranolol in one RCT (25) and correctly received a grade A [1]. It is, however, not commonly used for migraine prophylaxis. Metoprolol received a grade B [2] but in my view the rating should be A [1]. Metoprolol was superior to placebo or aspirin in three RCTs using the recommended 200 mg of metoprolol (27–29) and somewhat better than placebo in one RCT using the dose of 100 mg metoprolol (26, 30). In one comparative RCT metoprolol was comparable to propranolol (31), although with wide 95% confidence intervals (CIs) (32). Propranolol, grade A [1], was superior to placebo in 10 RCTs and comparable to placebo in two RCTs (26) and remains the standard first-line drug for migraine prophylaxis. Flunarizine, amazingly, received only a grade B [4], but should receive a grade A [2] because it was superior to placebo in six out of seven RCTs (33). Because of the side-effects of flunarizine it is a second-line agent for migraine prophylaxis. Nimodipine received a grade B [2] but two European RCTs have shown that it was not superior to placebo (33). As a consequence of these negative trials, nimodipine has not been registered for migraine prophylaxis in any country. It should be ranked B [5]. The evidence for verapamil (B [2]) is very sparse, 41 patients included in three RCTs (33). Pizotifen (A [4]) was superior to placebo in six RCTs and not superior to placebo in three RCTs (34). Again, AEs, especially weight gain and sedation, hamper its use and I agree with the A [4] rating. The anti-Parkinson agent lisuride received a grade A [1], but in two of the pivotal trials (35, 36) migraine patients with up to 21–30 attacks per month were included. The diagnosis of migraine must therefore be questioned. Lisuride is not in general use for migraine prophylaxis and should receive B [2].
Systematic reviews in migraine
Within recent years several systematic reviews with meta-analyses of acute migraine treatment have been published (37–46). In addition, three systematic reviews of preventive migraine treatment have been published (47–49). One should distinguish between systematic reviews, where several RCTs of a single drug are evaluated by meta-analysis to gain a more precise impression of its merits (37, 42, 43, 46–48), and systematic reviews, where several drugs or administration forms of a drug are compared in a meta-analysis (38–41, 44, 45).
In the systematic reviews of acute migraine treatment (37–43, 45, 46) migraine was diagnosed according to the criteria of the International Headache Society (IHS) (50) and the same methodology was used (14). Patients treated moderate or severe headache and headache relief was defined as a decrease to none or mild (13). Headache relief was the primary efficacy measure in most RCTs. Pain-free after 2 h, which is now the recommended primary efficacy measure of the Subcommittee of IHS on Clinical Trials (14), was also reported in most studies and was evaluated in some meta-analyses (38, 39, 46). One of the systematic reviews (38, 39) also evaluated sustained pain free, i.e. pain free after 2 h, no use of rescue medication and no recurrence within 24 h, and consistency across attacks. In addition, tolerability vs. placebo was evaluated in these systematic reviews.
Systematic reviews of drugs used for the acute treatment of migraine
Large amounts of data are needed to overcome random effects in estimating treatment effects (51). This is the background for systematic reviews of one drug for acute migraine treatment (37, 42, 43, 46) or migraine prophylaxis (47–49). The systematic reviews, using meta-analysis, showed that naratriptan 2.5 mg was superior to placebo (37); for a comment on the magnitude of the effect of naratriptan, see below. Rizatriptan 5 mg, the dose used in patients treated with propranolol for migraine prophylaxis, and rizatriptan 10 mg were superior to placebo (42). Rizatriptan 10 mg is a first-line drug for the treatment of migraine attacks. Eletriptan 40 mg and 80 mg were superior to placebo (43). Eletriptan 40 mg is a first-line drug for acute migraine attacks and eletriptan 80 mg can be tried in especially severe migraine attacks. In one systematic review the efficacy parameter pain-free response at 2 h was used (46) and 100 mg sumatriptan was superior to placebo with numbers-needed-to-treat of 5.1. Sumatriptan 50 mg was not superior to placebo for this parameter (46). In a recent trial, 50 mg sumatriptan (50%) taken in the mild phase of a migraine attack was superior to placebo (29%) for pain free at 2 h (52).
Meta-analysis of drugs used for the acute treatment of migraine
One systematic review of triptans presented a meta-analysis of the seven oral triptans (sumatriptan, zolmitriptan, naratriptan, rizatriptan, almotriptan, eletriptan and frovatriptan) and of subcutaneous, intranasal and rectal sumatriptan (45). The meta-analysis was based on headache relief and the mean therapeutic gains (TG) (percentage relief with active drug minus percentage relief with placebo) with 95% CIs were calculated. Based on this meta-analysis, subcutaneous sumatriptan 6 mg (TG = 51%; 95% CI 49, 53) and eletriptan 80 mg (TG = 42%; 95% CI 37, 47) were superior to sumatriptan 100 mg (TG = 32%; 95% CI 29, 34%). In contrast, the mean TGs were inferior to sumatriptan 100 mg for naratriptan 2.5 mg (TG = 22%; 95% CI 18, 26) and for frovatriptan (TG = 16%; 95% CI 8, 25).
In another systematic review (41), the meta-analysis was based on number-needed-to-treat (NNT) (the reciprocal of TG) and included oral triptans, subcutaneous sumatriptan, intranasal sumatriptan, intranasal dihydroergotamine and the oral combination of aspirin plus metoclopramide. Again, subcutaneous sumatriptan 6 mg (NNT = 2.0; 95% CI 1.8, 2.2) was superior to sumatriptan 100 mg (NNT = 3.3; 95% CI 3.0, 3.7) for headache relief, and the combination of aspirin plus metoclopramide (NNT = 3.2; 95% CI 2.6, 4.0) and intranasal dihyergotamine (NNT = 2.5; 95% CI 1.9, 3.7) were comparable to sumatriptan 100 mg. Rizatriptan 10 mg (NNT = 2.7: 95% CI 2.4, 2.9) was superior to sumatriptan; naratriptan 2.5 mg (NNT = 5.4; 95% CI 5.4, 9.2) was inferior to sumatriptan. For pain free after 2 h the combination of aspirin plus metoclopramide (NNT = 8.6; 95% CI 6.2, 14) was inferior to sumatriptan 100 mg (NNT = 4.7; 95% 95% CI 4.1, 5.5); and subcutaneous sumatriptan 6 mg (NNT = 2.1; 95% CI 1.9, 2.4) and rizatriptan 10 mg (NNT = 3.1; 95% CI 2.9, 3.5) were superior to sumatriptan 100 mg. In this meta-analysis excedrin (paracetamol 500 mg + aspirin 500 mg + caffeine 200 mg) had a NNT of 3.4 (95% CI 3.1, 3.8), but, as noted above, the patients with severely debilitating migraine were excluded from that study (11).
A third systematic review (40) reviewed the triptans available at that time in Canada: sumatriptan, rizatriptan, zolmitriptan and naratriptan. Subcutaneous 6 mg (NNT = 2.0; 95% CI 1.8, 2.3) was superior to oral sumatriptan 100 mg (NNT = 3.1; 95% CI 2.8, 3.4) for headache relief after 2 h. Naratriptan 2.5 mg (NNT = 5.1; 95% CI 4.1, 6.7) was inferior to oral sumatriptan 100 mg for this parameter. For pain free after 2 h rizatriptan 10 mg (NNT = 3.2; 95% CI 2.9, 3.5) was superior to sumatriptan 100 mg (NNT = 4.9; 95% CI 4.2, 5.9).
These three systematic reviews, in which therapeutic gains or NNT are used in the meta-analysis as a ‘correction’ for the variable placebo effect, generally give the same results. There are minor differences most likely due to differences in the RCTs included in the meta-analyses (40, 41, 45). From these systematic reviews one can conclude that subcutaneous sumatriptan 6 mg is the most effective triptan. Eletriptan 80 mg (for headache relief, pain free) and rizatriptan 10 mg (for pain free) are somewhat better than sumatriptan 100 mg, whereas naratriptan 2.5 mg and frovatriptan 2.5 mg are inferior to sumatriptan.
A meta-analysis of 53 RCTs with oral triptans was published in The Lancet (38) and in detail in Cephalalgia (39). The following efficacy measures and tolerability were evaluated: headache relief after 2 h, pain free after 2 h, headache recurrence (headache relief after 2 h with a relapse of moderate or severe headache in the subsequent 22 h post dose), sustained pain free (pain free after 2 h, no recurrence of moderate or severe pain and no use of migraine medication over the subsequent 24 h post dose), intrapatient consistency for headache relief and pain free after 2 h, and AEs, both any AEs and CNS-related AEs. Some of the results of this meta-analysis are shown in Fig. 1a,b. The authors’ interpretation of the meta-analysis combined with evaluated comparator RCTs are shown in Table 2 (for details see (39)). Headache relief at 2 h was the primary per protocol end-point in nearly all triptan RCTs; and, as illustrated in Fig. 1a, headache relief after 2 h, compared with 100 mg sumatriptan, was higher for rizatriptan 10 mg and eletriptan 80 mg, lower for naratriptan 2.5 mg and frovatriptan 2.5 mg. For the placebo-substracted response, the same as TG (see above), only eletriptan 80 mg was superior to sumatriptan 100 mg. Pain free at 2 h, as recommended by the IHS (14), was a secondary end-point in most trials. Compared with sumatriptan 100 mg, naratriptan 2.5 mg showed lower absolute pain-free rates whereas eletriptan 80 mg, almotriptan 12.5 mg and rizatriptan 10 mg showed higher values; however, only eletriptan 80 mg and rizatriptan 10 mg showed higher values than sumatriptan for TG for pain free (placebo-substracted values) (see Fig. 1b). Headache recurrence rates compared with sumatriptan 100 mg (30%; 95% CI 27, 33) were lower for eletriptan 40 mg (22%; 95% CI 19, 24) and 80 mg (20%; 95% CI 17, 22) and higher for rizatriptan 10 mg (37%; 95% CI 35, 39). Sustained pain free (for definition see above) was higher for rizatriptan 10 mg (26%; 95% CI 24, 27), eletriptan 80 mg (25%; 95% CI 23, 27) and almotriptan 12.5 mg (27%; 95% CI 23, 30) compared with sumatriptan 100 mg (20%; 95% CI 18, 21). Intrapatient consistency was evaluated in trials where the patients treated three attacks. Headache relief in two out of three attacks was found in 47–72% of patients (39). In one study (53) with crossover design, with placebo interspersed at random in four of five treatment groups (the fifth group received rizatriptan for four attacks), the consistency for rizatriptan was 86%. For AEs, sumatriptan 100 mg had a mean placebo-subtracted rate of any AEs of 13% (95% CI 8, 18). The rates for the other triptans overlap, except for lower values for almotriptan 12.5 mg and naratriptan 2.5 mg. The rates for almotriptan and naratriptan did not differ from placebo.
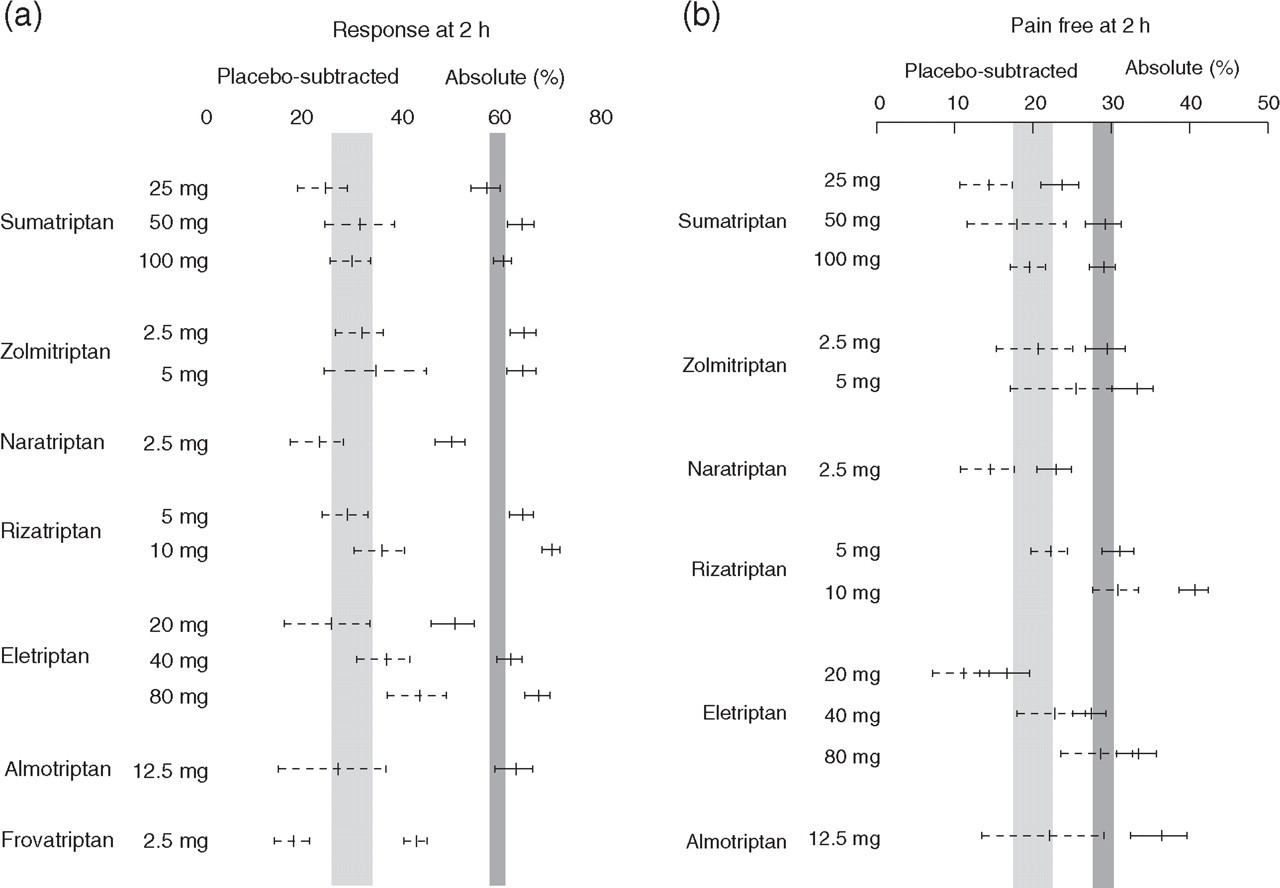
Headache response (a) and pain free after 2 h (b) after seven oral triptans. The shaded area indicates the 95% confidence intervals for sumatriptan 100 mg both for absolute responses and placebo-substracted results (from (38) with permission from the publisher).
Comparison of the main efficacy and tolerability measures for oral triptans vs. sumatriptan based on the results of the meta-analysis and direct comparative trials, from Ferrari et al. (38)
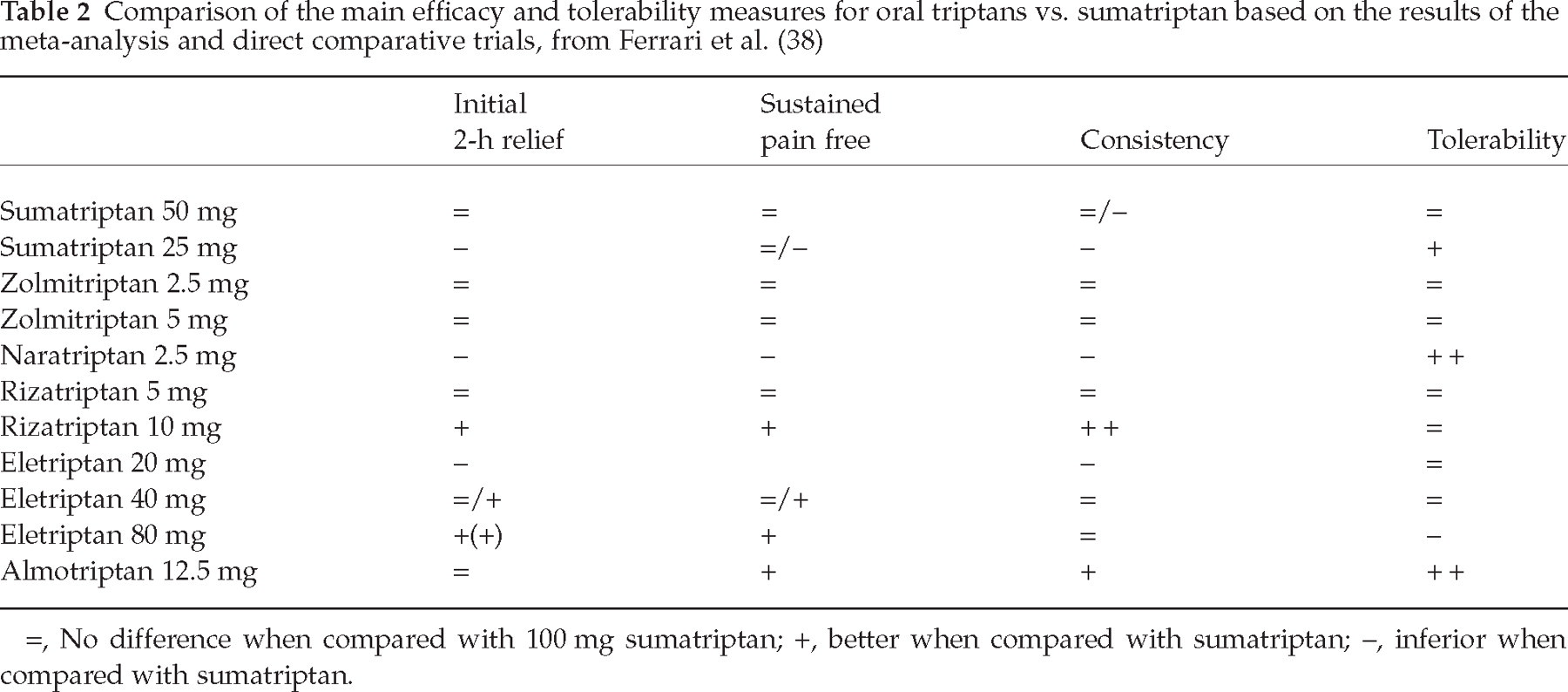
=, No difference when compared with 100 mg sumatriptan; +, better when compared with sumatriptan; –, inferior when compared with sumatriptan.
The authors concluded that at marketed doses, all oral triptans were effective and well tolerated. Rizatriptan 10 mg, eletriptan 80 mg and almotriptan 12.5 mg provide the highest likelihood of consistent success (38).
I agree that all triptans are more effective than placebo but the therapeutic gains for both naratriptan and frovatriptan are low (Fig. 1a), and lower than the therapeutic gain for the combination of aspirin plus metoclopramide (41). These two triptans are also more costly than the combination.
Rizatriptan 10 mg is rated with + for initial 2-h relief compared with sumatriptan 100 mg (see Table 2). The TG for rizatriptan 10 mg was, however, not superior to sumatriptan (Fig. 1a) and, in a rather large comparator RCT, the two drugs were comparable for this efficacy measure (54). For pain free after 2 h, however, the TG was higher for rizatriptan 10 mg than for sumatriptan 100 mg, and the same was found in the comparative RCT (54). In addition, rizatriptan 10 mg (n = 1114) was superior (95% CI 0.6, 9) to sumatriptan 50 mg (n = 1116) for pain free after 2 h in a combined analysis of two trials (55, 56). Rizatriptan 10 mg was superior to sumatriptan 100 mg for sustained pain free (38), but this was not the case in the comparative RCT (39). Consistency was higher for rizatriptan 10 mg than for all other triptans, but it should be noted that this was evaluated with a special design (53) and the results are thus not directly comparable.
After the publication of this systematic review (38, 39) a new, large, placebo-controlled RCT comparing eletriptan 40 mg and sumatriptan was published (57). This study, together with two previous RCTs (58, 59), constitutes a large head-to-head comparator database. A review of data from these three double-blind, placebo-controlled trials included 1132 patients treated with 40 mg eletriptan and 1129 patients treated with 100 mg sumatriptan (60). In these comparative RCTs sumatriptan was encapsulated and it has been questioned whether encapsulation could influence the results against sumatriptan in these comparative RCTs (61). The mean TG for headache relief for 100 mg sumatriptan in these three trials (57, 58, 60) was 31.2% (60), which is equivalent to results of meta-analysis for 100 mg sumatriptan (32%; 95% CI 29, 34 (44); 29%; 95% CI 26, 34 (39)). For pain free after 2 h the mean TG for 100 mg sumatriptan was 20% (60), which is comparable to results from the meta-analysis: 19% (95% CI 17, 22) (39). The sustained pain free for sumatriptan was marginally lower (mean = 15%; 95% CI 13, 17) (60) than the results from meta-analysis (mean = 20%; 95% CI 18, 21) (39). It should be noted that there is no placebo correction for sustained pain free, which makes comparison difficult. In conclusion, encapsulated 100 mg sumatriptan resulted in similar efficacy at 2 h to sumatriptan used in other placebo-controlled RCTs. The headache relief rate after 2 h was higher for eletriptan 40 mg (67%) than for sumatriptan 100 mg (57%) (60). The 2-h pain-free response was also higher for eletriptan (35%) than for sumatriptan (25%) (60). The sustained pain-free response was higher for eletriptan (22%) than for sumatriptan (15%) (60). The frequency of AEs was similar for the two drugs (60). Eletriptan 40 mg is thus superior to sumatriptan 100 mg for several efficacy measures in comparator RCTs. Eletriptan 80 mg, a dose not routinely used, was in the meta-analysis superior to sumatriptan 100 mg for TGs for headache relief, pain free after 2 h (see Fig. 1a,b) and sustained pain free (38, 39) This was also the case in the comparator studies (39).
Almotriptan 12.5 mg had the same initial 2-h relief as sumatriptan 100 mg (see Fig. 1a,b). The headache relief was the same and the TG for pain free after 2 h was the same for almotriptan and sumatriptan. In one comparative RCT (62), however, almotriptan 12.5 mg (18%) was inferior to sumatriptan 50 mg (25%) for pain free after 2 h. Apparently, almotriptan 12.5 mg is better than sumatriptan 100 mg for sustained pain free, based on the meta-analysis (38, 39). This was, however, not the case in a comparative RCT (39) and in a larger crossover RCT (62), published after the meta-analysis, almotriptan 12.5 mg had a lower sustained pain-free response (12.9%) than sumatriptan 50 mg (17.6%) (63). Part of the explanation for the high sustained pain-free response for almotriptan in the meta-analysis may be the relatively high placebo response for this measure in the almotriptan RCTs (39). Sustained pain free, as used in the meta-analysis, is not corrected for the placebo response. The better tolerability of almotriptan 12.5 mg than sumatriptan was confirmed in two trials (62, 64). In one comparative RCT treatment-related AEs occurred more frequently with 50 mg sumatriptan (15.5%) than with 12.5 mg almotriptan (9.1%) (62); and in another comparative RCT the incidence of AEs was higher with 100 mg sumatriptan (22.2%) than with 12.5 mg almotriptan (8.7%) and the incidence of AEs was the same for 12.5 mg almotriptan as for placebo (6.1%) (64).
Intrapatient consistency as evaluated in the meta-analysis was investigated in only a few RCTs (38) and is difficult to judge because this measure is not placebo-corrected, even though placebo values are presented. The placebo response for this measure is higher for almotriptan 12.5 mg than for sumatriptan 100 mg (38) and if this is taken into account the consistency is most likely the same for almotriptan and sumatriptan, both for headache relief and pain free after 2 h.
My suggestions for the ratings of rizatriptan 10 mg, eletriptan 40 mg and 80 mg, and almotriptan 12.5 mg vs. sumatriptan 100 mg are shown in Table 3, which should be compared with Table 2. Rizatriptan 10 mg should have a +/= rating for initial 2-h relief and for sustained pain free, whereas the rest of the rating is unchanged. Eletriptan 40 mg should have + for initial relief based on the three comparator RCTs (60) and the same is the case for sustained pain free. Eletriptan 80 mg is unchanged. Almotriptan 12.5 mg is in my view comparable to sumatriptan 100 mg for initial relief, sustained pain free and consistency, but its better tolerability, comparable to placebo, has been confirmed in two RCTs (62, 63).
=, No difference when compared with 100 mg sumatriptan; +, better when compared with sumatriptan; –, inferior when compared with sumatriptan.
Consistency for rizatriptan 10 mg was investigated with a different methodology than normally used in the meta-analysis, see text.
In two recently published RCTs (65) with sumatriptan rapid-release tablets, sumatriptan 100 mg (n = 1101) had a TG of 30% (95% CI 26, 34) vs. placebo (n = 1113), the same as in the meta-nalysis (38, 39) (see Fig. 1a). In contrast, for pain free after 2 h the mean TG was 32% (95% CI 28, 36) (65), which is considerable higher than the 20% TG for conventional sumatriptan tablets (see Fig. 1a). Unfortunately, there are no comparative RCTs with this new oral formulation of sumatriptan, and one can thus not draw firm conclusions concerning its merits relative to other triptans.
Systematic reviews of drugs for migraine prophylaxis
In one systematic review of propranolol (47) it was concluded that propranolol is effective for short-term migraine prophylaxis, evidence on long-term effects is lacking, and propranonol seems to be as effective and safe as a variety of other drugs used for migraine prophylaxis. Propranolol is a first-line drug for migraine prophylaxis. In another systematic review the efficacy of feverfew in migraine prophylaxis was judged as unproven (48).
In one systematic review, a group of drugs, anticonvulsants, was evaluated for migraine prophylaxis (49). It was concluded that relatively few robust trials are available for agents other than sodium valproate/divalproex sodium, which is superior to placebo. Topiramate was effective in two RCTs and further RCTs were awaited (49). Valproate/divalproex sodium are second-line, not first-line drugs for migraine prophylaxis because of their AEs. Topiramate has been evaluated in three large RCTs and was superior to placebo in all three (66–68). It was comparable to propranolol (67). There are AEs, sedation and cognitive impairment, with topiramate and its place in migraine prophylaxis depends on how patients in clinical practice will tolerate the drug.
Conclusions
When comparing drugs for the acute treatment of migraine, head-to-head comparative RCTs remain the ‘gold standard’. However, comparative trials are, with a few exceptions (53, 54, 56, 57), relatively small and may overlook differences. In addition, there may be a selection bias in head-to-head RCTs: a patient responding well to the standard drug may be less likely to participate. Not all drugs in a class of drugs will be compared in head-to-head RCTs. Meta-analysis of drugs that underwent placebo-controlled RCTs with similar methodology (13, 14) can therefore be a useful supplement when drugs are compared. The main weakness of the meta-analytic approach is that there is no randomization. In addition, the populations may not be totally comparable: possible bias in time with recruiting over many years and severity of headache (ratio of moderate/severe headaches) (69) may differ in different trials and trial programmes. The main emphasis when judging meta-analytic results should therefore be on therapeutic gains; and these results should be evaluated in context with the findings from head-to-head comparative trials.


