Abstract
The aim of this study was to determine the characteristics of placebo effects in acute migraine treatment trials of triptans performed over 12 years and assess whether the use of placebo controls in trials of acute migraine treatment remains ethically and scientifically appropriate. We conducted a search for all controlled trials published in English between January 1991 and April 2002 in which adult subjects with migraine were randomly assigned to receive an oral triptan or placebo for the acute treatment of a migraine attack. Thirty-one trials met our criteria for inclusion. Placebo results for each study and pooled placebo results were calculated for the endpoints of headache response, pain-free response and adverse events. Heterogeneity was assessed using the Q statistic, and meta-regression using prespecified covariates was performed to investigate heterogeneity. The study results show a significant degree of heterogeneity. Efforts to explain heterogeneity with available data were not successful, with the exception of adverse event rates to placebo, for which study location (Europe vs. North America) partially explained differences in study results. AE rates were lower in European studies than in North American studies. Across all studies, the mean proportion of subjects who experienced a treatment response at two hours to placebo was 28.48 + 8.73% (range 17-50%). The mean proportion of subjects who experienced an adverse event to placebo was 23.40 + 14.05% (range 4.86-74%). The mean proportion of subjects who experienced a pain-free response to placebo at two hours was 6.08 + 4.43% (range 5-17%). Results of studies allowing use of prophylaxis did not differ significantly from those that did not allow prophylaxis. Placebo effects appear to be enhanced in studies involving children and adolescents. In contrast to an earlier, smaller review, our results do not suggest that randomization ratios influence placebo rates. We conclude that placebo effects in published trials of acute migraine medications are highly variable and often substantial. This variability in placebo response means that active control equivalence trials or the use of historical controls will not provide adequate proof of the safety or efficacy of new drugs, and will not differentiate between drugs that are active vs. placebo but of unknown efficacy relative to each other. The potential for approval of ineffective drugs, inability to compare results of studies performed in different locations, and poor characterization of the tolerability and safety profiles of new drugs represent a greater danger to migraineurs than does the limited-duration use of placebo in carefully monitored clinical trials of consenting subjects. These observations support the view that the inclusion of a placebo group remains of major scientific and ethical importance in trials of migraine medications.
Introduction
Studies of triptans for the abortive treatment of migraine usually include a group of subjects who are assigned to receive placebo in the context of a randomized controlled trial and a parallel group design. Based on trials using this design, the United States Food and Drug Administration (FDA) has approved for marketing seven triptans that are widely viewed as the standard of care for acute migraine treatment (1).
The use of placebo in clinical trials is increasingly controversial, particularly once treatment of established efficacy becomes available (2). The Declaration of Helsinki was drafted by the World Medical Association in 1964 to provide guidelines for ethical human subject research, and revised in 2000. It states that
‘a new method should be tested against…the best current prophylactic, diagnostic and therapeutic methods. This does not exclude the use of placebo, or no treatment, in studies where no proven prophylactic, diagnostic or therapeutic method exists.’ (3)
International Headache Society (IHS) guidelines regarding controlled trials of acute migraine medications recommend that trials should be carried out ‘
Because the standards for acceptable use of placebos in clinical trials are changing, it has been suggested that it is now necessary to provide both ethical and scientific justification for the continued routine use of placebo in specific therapeutic areas. Two situations are generally acknowledged to justify the use of placebo:
withholding the best current treatment will result in only temporary discomfort and no serious adverse consequences
a comparative study of two active treatments would not yield reliable scientific results (5)
The use of placebo controls in trials of acute migraine medication satisfies the first criterion; in addition, the typical protocol allowance of rescue medication at two hours for subjects whose headaches are unimproved offers some protection by limiting the duration of placebo exposure. However, it is less clear that active-comparison equivalence trials (ACETs) would not yield reliable scientific results in trials of acute headache treatments. We therefore examined data on placebo effects in controlled trials of triptans in adult subjects, as a way of assessing whether it remains scientifically necessary to use placebo in trials of acute migraine treatment.
Methods
We collected peer-reviewed articles describing randomized, placebo-controlled trials of triptans for the abortive treatment of migraine. Studies published between 1991 and March 2002 were identified by several methods. A MEDLINE literature search was conducted using specific key words, including ‘placebo’
written in English
fully published in a peer reviewed medical journal between January 1991 and March 2002 (abstracts did not reliably provide complete information regarding the placebo group and were not included)
composed of primarily adults with migraine according to the diagnostic criteria of the International Headache Society (Two trials with a lower age limit of 12 were included in this analysis, since the average age of the population was consistent with that of trials enrolling adults only, and the number of subjects under 18 was judged to be insignificant)
subjects randomly assigned to receive a triptan or placebo for acute treatment of individual migraine attacks, and assessed treatment response to in a double-blind manner
reported the total number of subjects assigned to triptan and placebo groups and the number who responded to medication for a single or first attack two hours after treatment using a four point scale (0 = no headache; 1 = mild headache; 2 = moderate headache; 3 = severe headache)
defined headache response as improvement from grade 2 or 3 headache at treatment to grade 0 or 1 headache at two hours. In multiple attack studies, only data from the first attack were assessed, to minimize carryover effects on placebo responses
Some studies, especially those done recently, also assessed pain-free status (improvement from grade 2 or 3 headache at treatment to grade 0 headache at two hours). All but one study reported adverse event data for the placebo group.
Data were extracted from the articles using a standardized data abstraction form. We calculated the response, pain-free and adverse event rates for the placebo group for each study and across studies by dividing the number of patients in each group who experienced the outcome of interest by the number of patients assigned to the group. In studies that reported only percentages, we obtained the number of subjects by multiplying the percentage of patients experiencing the outcome of interest by the total number of patients in the group and rounded to the nearest whole number. Randomization ratio, year of study publication, and study location (European or North American) were identified as variables of interest prior to any statistical calculations. Heterogeneity was assessed using the Q statistic, and meta-regression techniques were used to investigate heterogeneity (software: Stata, version 7).
Results
Thirty-one trials of triptans meeting our criteria for review were identified (8–38). Data are presented as mean ± standard deviation. The average number of subjects in the placebo group was 112 (range 12–302; SD ± 71).
Trial methodology was remarkably constant over the time period reviewed. All trials used IHS diagnostic criteria to determine eligibility for trial participation. Most trials required between one and six or eight headaches per month for trial inclusion and excluded patients with chronic daily headache or frequent tension-type headache that could not be reliably distinguished from migraine. No trials used a placebo-run-in period. Twenty-four studies provided specific information on whether the use of prophylactic medication for migraine was permitted during the trial; most, but not all, of these trials excluded specific prophylactic medications. Only one trial provided information on whether subjects were naïve to the use of triptans. 23 trials allowed the use of rescue medication at two hours for patients without a satisfactory clinical response. No trial allowed the use of rescue medication prior to two hours. Twenty-five trials provided information on the number of subjects in the placebo group who were pain-free two hours after treatment.
Patient characteristics
The percentage of women in the placebo group was reported in 24 trials. In this group of trials, the average percentage of women was 80% (range 77–93%). In 5 studies, only the percentage of women in the entire population was reported. In this group of trials the average percentage of women was 81%. Two studies did not report the gender distribution of patients in the placebo group. The average age of the placebo group was 40.1 years (range 37.2–44 years) in the 22 trials that reported this statistic; in five trials reporting only the age of the entire study population, the average age was 37.82 years (range 34.4–40.6 years). Population ages were not reported in three trials. There was no significant change in average age over time.
Placebo effects
Response
All 31 studies provided information on the number of subjects reporting a headache response to placebo, based on the definition of response using the 0–3 scale. The average proportion of subjects who responded while receiving placebo was 28.90% (SD 8.55). The percentage of subjects receiving placebo who reported a headache response was highly variable, ranging from a low of 17% to a high of 50%. The
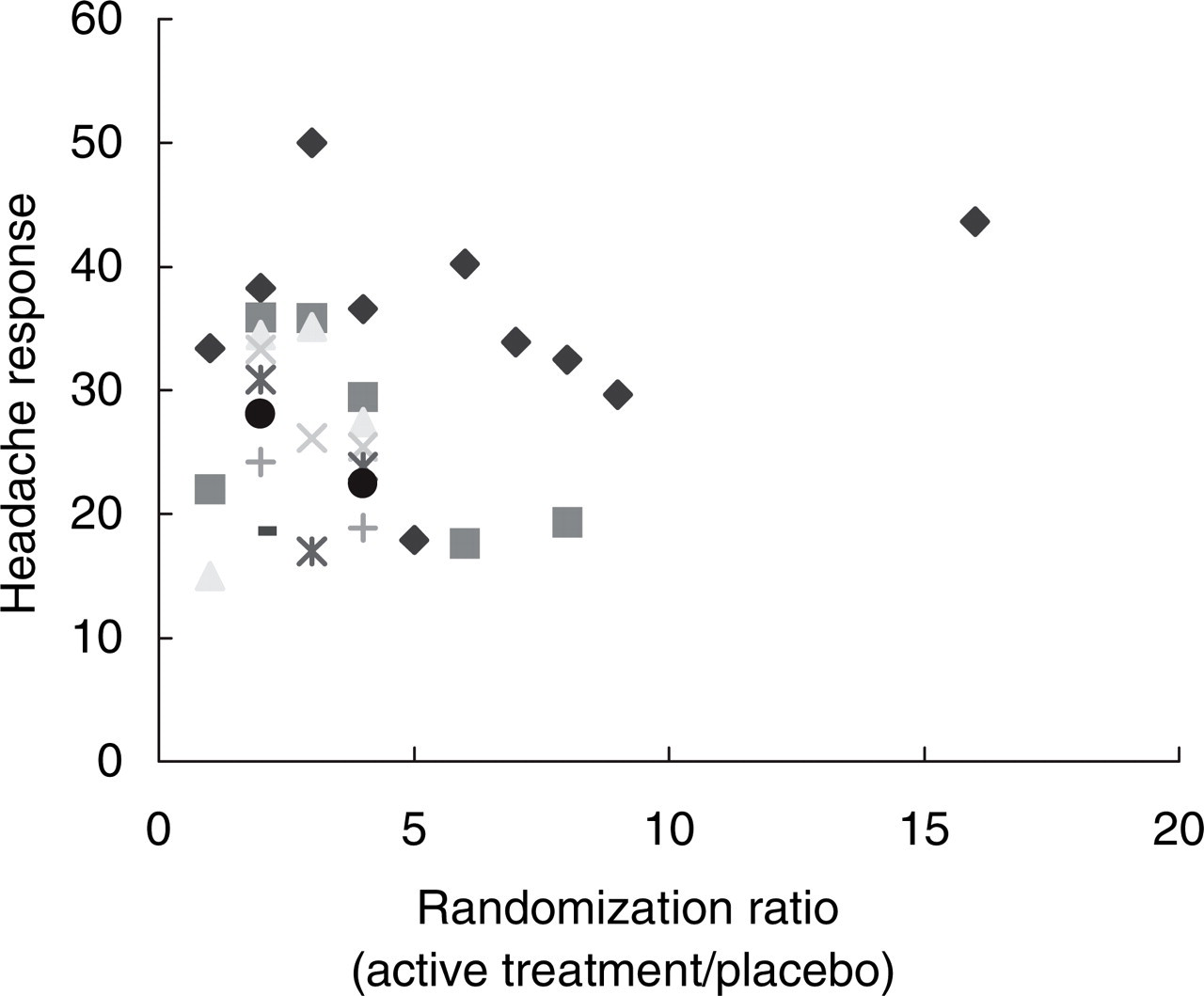
The percentage of patients reporting a headache response 2 h after receiving placebo does not appear to be influenced by patient expectations about the likelihood of receiving placebo. A prior study suggested that patients in studies where most subjects received active treatment (high randomization ratio) were more likely to report a headache response to placebo.
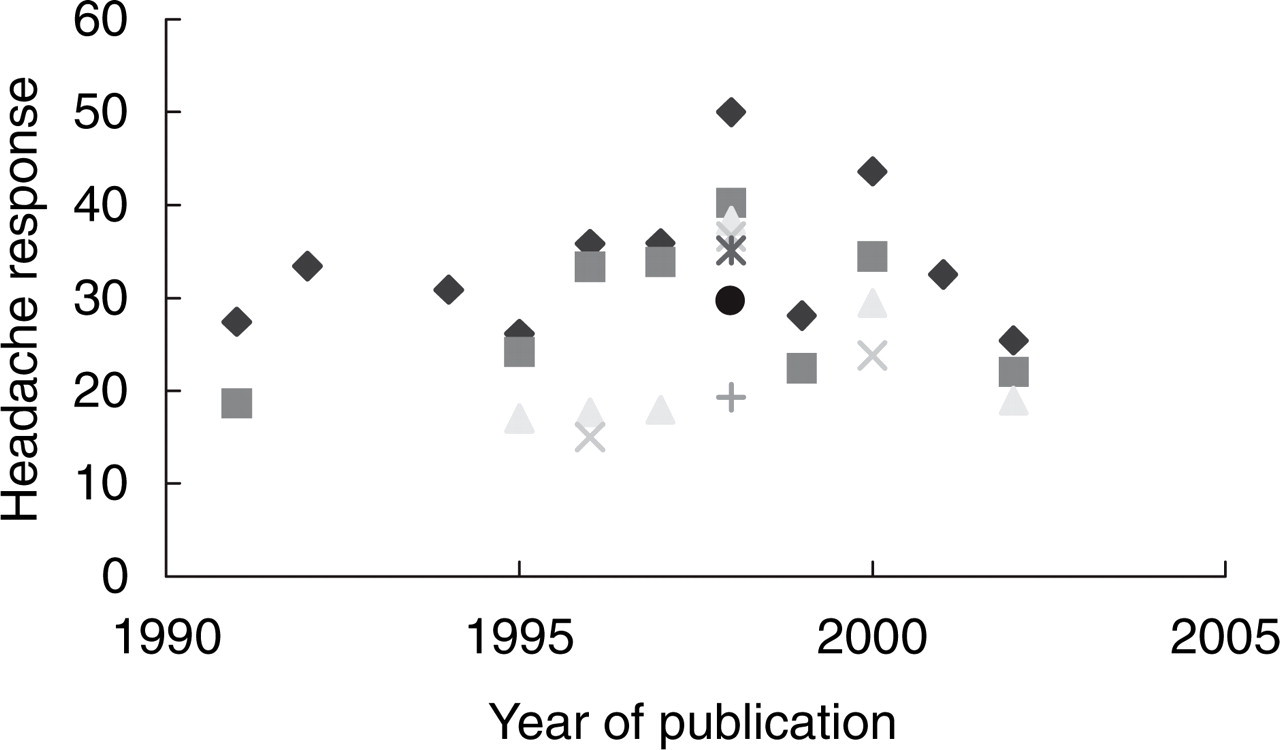
There has been no significant change in the average percentage of patients reporting a headache response to placebo over the period of this study.

The percentage of patients reporting adverse events two hours after taking placebo is significantly higher in studies performed in North America (▪) than in Europe (□). The percentage of patients who are pain-free or report a headache response two hours after taking placebo is not statistically different between the two locations.
Pain-free
Twenty-four studies provided information on the number of subjects who were pain-free two hours after receiving placebo. The average proportion of subjects achieving this endpoint was 6.08 ± 4.83%. Pain-free response ranged from a low of 5% to a high of 17%. Data for the three prespecified covariates (randomization ratio, location and year of publication) were available for meta-regression, but this analysis did not explain the heterogeneity.
Adverse events
All but one study provided information on the number of subjects in the placebo group who reported adverse events. The average proportion of subjects experiencing adverse events in association with placebo administration in these studies was 23.4 ± 14.05%. This endpoint was even more variable than that of headache response, with a range of 4.86% to a high of 74%. Data for the three prespecified covariates (randomization ratio, location and year of publication) were available for meta-regression. This analysis revealed that study location partially explained the heterogeneity, with studies conducted in Europe reporting lower adverse event rates than studies conducted in North America (Fig. 3).
Formulation
Placebo results can be influenced by factors such as the size, shape and colour of pills (39). Triptans formulated as orally dissolving tablets might therefore be associated with different placebo effects than triptans taken as conventional tablets. To examine this possibility, we compared results from two trials of triptan orally dissolving tablets (one using zolmitriptan and one using rizatriptan) (21, 29). The average placebo response rate in these 2 trials was 24.70%, the placebo pain-free rate was 8.55%, and the placebo adverse event rate was 8.94%. The placebo response rate in the 28 studies with conventional tablets was 28.85%, the placebo pain-free rate was 5.41%, and the placebo adverse event rate was 23.74%.
Adolescents and children
We did not include trials involving only children or adolescents in our study. However, since placebo rates in such trials are said to be higher than those involving adult subjects, we made a preliminary examination of results from the only two fully published trials of oral triptans for the acute treatment of migraine in children and adolescents in our identified trials (40, 41). The average placebo response rate in these two trials was 48.48%, the pain-free rate was 25.45%, and the adverse event rate was 32.35%, significantly higher than results from trials in which subjects were primarily or exclusively over the age of 18.
Prophylaxis
To examine whether the use of prophylaxis had an impact on placebo effects, we compared results from five studies in which the use of prophylaxis was specifically allowed (19, 25, 30, 35, 40) with results from eight studies (12, 13, 15, 22–24, 36, 37) in which the use of concomitant prophylaxis of any kind was explicitly prohibited. The remaining 18 studies did not provide clear information about whether prophylaxis was allowed, or excluded the use of some, but not all, prophylactic medications. Average response, pain-free and adverse event rates in the studies permitting concomitant use of prophylactic medication were 25.52%, 6.02% and 19.56%, respectively, and 29.33%, 7.59% and 22.76% in trials where prophylaxis was not permitted. These results were not significantly different.
Discussion
This analysis of 31 placebo-controlled trials of triptans for the acute treatment of migraine in adults published between 1991 and 2002 leads to a number of conclusions relevant to future trials of acute headache medications.
First, the effect of placebo on headache response and adverse event rates varied a great deal across trials, and frequently was considerable. In 14 of 31 studies, 30% or more of the subjects assigned to placebo exhibited a clinically significant improvement in headache. Placebo response in children and adolescents may be especially substantial. Placebo responders to acute headache treatment include subjects whose headaches have spontaneously improved, as well as those with a response based on expectation, conditioning, or other nonspecific factors. Based on the significant variability in placebo rates from study to study, it seems important for future trials of acute migraine therapy to include placebo. Historical or active comparator controls will not provide adequate safeguards against the danger of approving ineffective medications. In addition, the use of ACETs for the evaluation of new acute headache medications raises the question of what the active comparator would be. For strategic reasons, companies might choose to compare their drug with currently approved drugs that are only marginally better than placebo. A new drug that was equivalent to or even better than these compounds would not necessarily represent an important clinical advance. Similarly, a drug that performed less well than subcutaneous sumatriptan, the current ‘gold standard’ for efficacy, might nonetheless represent an important treatment advance, based on a novel mechanism of action, a lower likelihood of drug interactions, or improved tolerability.
A previous review of three headache studies suggested that the endpoint of two hour sustained pain-free is associated with the lowest and most consistent placebo response rates, in addition to best reflecting patient preference and treatment satisfaction (42). Unfortunately, this endpoint was not assessed or reported in enough studies included in our review to allow evaluation of that possibility. One problem with the endpoint of sustained pain-free is that because efficacy rates are lower, placebo effects become an even greater part of the results.
There is significant statistical heterogeneity among triptan studies, both for placebo responses, as demonstrated in this study, but also for active drug response, as demonstrated in other reviews. This is surprising, in view of the high degree of stability in study design. Diagnostic systems have shifted little with time, and criteria for study entry were remarkably similar across trials. In addition, the average subject age and percentage of women in trials has not changed over time. Efforts to explain the heterogeneity by analysis of prespecified potentially relevant covariates were not successful, with the exception of adverse event rates, as noted below. This suggests that other factors, such as study site or subject characteristics, should be explored as potential covariates. Both have been shown in other disease states to exert significant impact on the outcome of clinical trials. Unfortunately, most triptan studies do not report such information. Separating drug from placebo effects is difficult if placebo response rates vary widely, particularly when the reasons for that variability cannot be explained. Unexplained heterogeneity of study results provides support for continued use of placebo in future trials of abortive migraine treatments.
An important exception to the inability to explain heterogeneity is AE rates, which are partly explained by study location. AE rates with placebo were significantly lower in studies conducted in Europe compared with those conducted in North America. The reason for this association is unclear. However, this observation is consistent across a variety of disease states, including hypertension, anxiety and ulcer disease (43). The difference in AE rates with study location suggests that use of a placebo group is important to aid in comparisons of trial results from European sites and trial results from North American sites.
Methods of collecting and reporting AEs in triptan trials have been criticized as nonstandardized (44), though, and there is likely to be residual heterogeneity in AE results that is not accounted for by study location. Adverse event rates for placebo are considerable, and even more variable than response and pain-free effects. In the absence of a placebo group, adverse events may be incorrectly assigned to active drug treatment. For this reason, the use of placebo is necessary to accurately characterize the safety and tolerability of new compounds.
Our results do not confirm those of a previous smaller study that concluded higher randomization ratios were associated with high placebo effects (45). That study examined results of three trials of zolmitriptan, noting that one study with a placebo : active treatment ratio of 1 : 16 was associated with a high placebo rate, whereas two other zolmitriptan trials with the placebo and active treatment groups more closely matched had less pronounced placebo effects.
Although only two fully published studies involved use of orally dissolving tablet formulations and only two had an entirely adolescent study population, placebo effects seemed different in these trials. A notable finding was the lower percentage of subjects receiving orally dissolving tablets who reported adverse events. These are preliminary observations only, but clearly warrant further study and consideration. Particularly intriguing is the apparent inverse correlation of age with placebo effects; in this analysis it appears that younger subjects have high rates of placebo effects; a separate posthoc analysis of age and placebo rates in a zolmitriptan trial suggests that older subjects (over 45) have lower rates of placebo effects (46).
Finally, placebo effects in trials permitting the use of prophylaxis did not differ from trials in which such treatment was prohibited, suggesting that requiring subjects to discontinue migraine prophylaxis in order to participate in trials of abortive migraine treatment might not be justified. Analysis of drug effect rates would be necessary to make a more definitive statement, however, and we did not perform such an analysis. Additionally, those studies that allowed participants to use prophylaxis did not report the number of subjects enrolled who were actually taking prophylactic treatment, nor the type or dose of prophylaxis. Future investigation of this issue is warranted, both to determine whether prophylaxis augments response to abortive therapy, a possibility that has not been systematically investigated, and to determine whether it is ethically justifiable to limit participation in abortive treatment trials to subjects who agree to use no prophylaxis for their disorder.
Several features of this study deserve comment. We examined only fully published data. Many triptan trial results have been reported only in abstract form, and some have never been published. Since all triptan trials to date have been initiated, supported and analysed by pharmaceutical companies, it is possible that studies with undesirably high placebo effects or low rates of response to triptan are disproportionately likely to remain unpublished in comparison to studies with low placebo effects or high response rates to triptan. That would mean that placebo effects in the published studies we examined are lower, and medication response rates higher, than those in unpublished studies. If this is the case, our review may not fully reflect changes in clinical trial results over the years. There is no reason to believe, though, that withholding of such data from publication has changed over the period covered by our study, and our results should therefore be valid for published triptan trials. In addition, if such trials have been withheld from publication, the mean placebo response in the present paper would be an under-estimate of the variability of placebo response; this would strengthen rather than weaken our conclusions, which are based on conservative estimates.
Our report classified studies by year of publication. In most cases, results are published one and a half to three years after the actual trial takes place. This time lag must therefore be taken into account in reviewing data. However, it is likely to be quite similar among studies, owing to the complexities of study design, data analysis and the publication process. Although our results show no significant changes in placebo effects in oral triptan trials over time, such changes have occurred in other disease states (47–49).
Because there is significant heterogeneity among the studies that cannot be explained with meta-regression using prespecified covariates, the results of the random effects model regarding effect sizes must be interpreted with great caution. Other potentially relevant covariates, such as characteristics of the study site, or certain subject characteristics, could not be examined because they were not reported in most trials. The percentage of subjects who are triptan-naïve has repeatedly been suggested as an important covariate, for example, but is rarely reported (50). Similarly, disease severity or duration may also influence study results. More severely affected subjects or those with a longer duration of illness may be less likely to respond to placebo or have a more consistent placebo response, than less severely affected patients. Experience of the study site, investigators and study coordinators may also influence study results (51). Study sites drawing subjects from headache clinics may have different results than those sites whose subjects are not drawn from specialty clinics. Consistent and improved reporting of these and other study characteristics will aid in analysis of future trial results.
Conclusion
In summary, our analysis indicates that placebo effects in trials of oral triptans for the acute treatment of migraine are variable and substantial. The variability cannot be easily explained based on study location, randomization ratio or year of publication. Data on other potentially relevant characteristics, such as duration of illness, actual percentage of subjects using prophylactic medications, prior triptan trial participation or prior use of triptans in a clinical setting, were not reported consistently enough to allow evaluation of their impact on placebo effects. It seems important that future trials collect and report this information.
The variability of the response to placebo across studies, along with the lack of explanation for much of this variability, casts doubt on the validity of any trials of abortive migraine treatment in which there is no placebo group. It also makes meta-analyses difficult. Both trials which use historical controls and those which use active comparators lack assay sensitivity and require large sample sizes to provide the narrow confidence intervals needed to ensure new drugs are not inferior to controls.
The potential for approval of ineffective drugs, inability to compare results of studies performed in different locations, and poor characterization of the tolerability and safety profiles of new drugs represent a greater danger to migraineurs than does the limited-duration use of placebo in carefully monitored clinical trials of consenting subjects. Taken as a whole, our data support the view that inclusion of a placebo group is ethically justifiable and scientifically important in trials of acute migraine treatments.
