Abstract
Background and purpose
An estimated 2.5–3.1% of people with episodic migraine develop chronic migraine in a year. Several risk factors are associated with an increased risk for this transformation. We conducted a systematic review and meta-analysis to provide quantitative and qualitative data on predictors of this transformation.
Methods
An electronic search was conducted for published, prospective, cohort studies that reported risk factors for chronic migraine among people with episodic migraine. Risk of bias was assessed using the Newcastle-Ottawa Quality Assessment Scale. Quality of evidence was determined according to the Grading of Recommendations Assessment, Development and Evaluation (GRADE) guidelines. Effect estimates were retrieved and summarized using risk ratios.
Results
Of 5695 identified publications, 11 were eligible for inclusion. The pooled analysis (GRADE system) found “high” evidence for monthly headache day frequency ≥ 10 (risk ratio = 5.95), “moderate” evidence for depression (risk ratio = 1.58), monthly headache day frequency ≥ 5 (risk ratio = 3.18), and annual household income ≥ $50,000 (risk ratio = 0.65) and “very low” evidence for allodynia (risk ratio = 1.40) and medication overuse (risk ratio = 8.82) in predicting progression to chronic migraine.
Conclusions
High frequency episodic migraine and depression have high quality evidence as predictors of the transformation from episodic migraine to chronic migraine, while annual household income over $50,000 may be protective.
Introduction
Chronic migraine (CM), a subtype of chronic daily headache (CDH), refers to a diagnosis of migraine with headache on average ≥ 15 days per month of which ≥ 8 days are linked to migraine (1). CM evolved from “transformed migraine” (TM) and has undergone multiple revisions in nomenclature and criteria (2). It has been defined as a complication of migraine, in which headache due to medication overuse (MO) was excluded (3). Most recently, the International Criteria for Headache Disorders, 3rd edition (ICHD-3) (4) criteria include CM with MO again.
CM has an estimated worldwide prevalence between 1–3% (5,6) and accounts for approximately 8% of all migraine cases in population-based samples (5). An estimated 2.5–3.1% of people with EM progress to CM per year (7). CM can be highly disabling, impactful and burdensome. People with CM have lower annual incomes, are less likely to be employed full time, have more comorbidities, and are more likely to be occupationally disabled than people with EM (8). Identifying risk factors for the development of CM is an important target for understanding the natural course of migraine as well as selecting and/or designing interventions.
Previous studies have reported several risk factors for the transformation of EM to CM or CDH including obesity, sleep disorders, depression, anxiety, high monthly headache day frequency, medication overuse, female sex, lower socioeconomic status, comorbid pain disorders, caffeine intake, major life events and cutaneous allodynia (9–12) . These risk factors can be divided into those that may be modifiable and those that are not (13). Identification of modifiable predictors of the new onset of CM is beneficial for developing and targeting interventions that may protect against progression to CM.
The strength of evidence for identified risk factors for progression from EM to CM and CDH were presented in a recent systematic review. This manuscript included studies listing risk factors for CM, TM, and CDH, assessed strength of data differently than the current review and did not conduct a meta-analysis or assess bias (14). We aimed to conduct a systematic review and meta-analysis of published observational cohort studies on the risk factors associated with the new onset of CM among people with EM using both quantitative and qualitative analytic approaches following recommendations for the conduct of systematic reviews and meta-analyses.
Methods
Inclusion and exclusion criteria for included studies
We adopted the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines (15) and included eligible studies which met the following criteria: (i) observational studies; (ii) cohort study designs that reported the risk factors or predictors of CM, and (iii) studies reporting odds ratios (ORs), relative ratios, or hazard ratios (HRs) with 95% confidence intervals (95% CIs) or sufficient data to calculate these figures. Basic science studies, retrospective cohort studies and studies that included participants without explicitly citing diagnosis of or meeting criteria for migraine with an average of 14 or fewer days per month were excluded.
Types of outcome measures
Risk factors for or predictors of CM.
Search strategy
We searched the PubMed (1966 to November 2018), EMBASE (1980 to November 2018), and Ovid (1946 to November 2018) electronic databases for articles published in English. We also searched the China Biological Medicine Database (CBM-disc, 1979 to November 2018), the Chinese National Knowledge Infrastructure (CNKI, 1979 to November 2018), and Wan Fang Med (1998 to November 2018) for articles published in Chinese. The following subject headings were used for searching: (‘episodic [Title/Abstract]’ OR ‘chronic [Title/Abstract]’ OR ‘chronification [Title/Abstract]’) AND (‘migraine [Title/Abstract]’ OR ‘headache [Title/Abstract]’) AND (‘transmit* [Title/Abstract]’ OR ‘risk factor* [Title/Abstract]’ OR ‘transform* [Title/Abstract]’ OR ‘translate* [Title/Abstract]’ OR ‘predict* [Title/Abstract]’ OR ‘progress* [Title/Abstract]’). We also searched the reference lists of all related manuscripts (e.g. reviews and guidelines). Search strategies are provided in the Supplemental material.
Study selection
Two reviewers (JX and FK) independently scanned titles and abstracts retrieved from the initial searches and selected potentially relevant studies for further consideration. Full texts were obtained when the information given in the title or abstract met the established selection criteria.
Data collection
JX and FK extracted data from the included studies on research country, study design, sample size, diagnostic criteria, adjusted confounders, and identified risk factors or predictors with fully adjusted ORs, HRs and 95% CIs using a data extraction form. Disagreements between the investigators were resolved through discussion or with a third party (DB) to reach consensus if necessary. We contacted authors of included studies if data provided were insufficient for synthesis.
Risk bias and quality of evidence assessment
Risk bias of each included study was accomplished using the Newcastle-Ottawa Quality Assessment Scale (NOS) for cohort studies (16). The NOS was developed to assess the quality of nonrandomized studies using a “star system” in which a study is judged in three areas: the selection of the study groups (four items); the comparability of the groups (two items); and the ascertainment of either the exposure or outcome of interest for case-control or cohort studies respectively (three items). Items that are judged as adequate receive a star and one point. Responses are summed (range 0–9). A sum score of ≥ 5 is defined as low risk of bias and high quality (17).
The overall quality of the evidence for each risk factor in the pooled analysis was assessed with the Grading of Recommendations Assessment, Development and Evaluation (GRADE) guidelines (18). GRADE pro software (Version 3.2.2) was used to transparently grade the strength of evidence and generate a summary table (Table 2).
Statistical analysis
Effect estimates were summarized using risk ratios (RRs) with 95% CI for cohort studies. For the measures of risk estimates reported as OR from multiple logistic analyses in the enrolled studies, we used the equation:
A sensitivity analysis was planned by deleting trials with low quality (NOS score ≤ 4) to evaluate the impact of the study quality on the final results, provided that sufficient data were available. However, the lowest NOS score was 6 and the results provided by that study did not allow for meta-analysis. Therefore, a sensitivity analysis was not performed. We also checked whether data from a study registration were obtained within the same year as data collection and follow-up in order to avoid overlapping data sources. In that case, we deleted repeated data to test whether it would change the tendency of the summarized effect.
We did not specifically address publication bias due to the small number of eligible studies enrolled in the meta-analysis because according to the Cochrane Handbook for Systematic Reviews of Interventions (https://training.cochrane.org/handbook), the minimum number of studies required to perform a publication bias assessment when using funnel plots is 10.
Heterogeneity among studies was tested using inverse-variance methods for combining results across studies based on Cochrane’s I-squared-based Q-test when conducting the meta-analysis. The calculation for Cochrane-Q is listed in statistical algorithms in Review Manager 5.3 (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration) (24). We synthesized data in the meta-analysis with a fixed-effects model unless there was no significant heterogeneity (I2 ≤ 50%), otherwise, a random-effects statistical model was used (I2 > 50%). All statistical analyses were performed using Review Manager 5.3.
Results
Results of study selection
A total of 5695 studies were identified in the initial search, leaving 3563 studies after duplicates were removed. Of these, 3375 were excluded by scanning the title and abstract and 177 were excluded through full-text review, leaving 11 studies. The flow diagram is presented in Figure 1.
Flow diagram of study selection.
Baseline data of included studies
Characteristics of included studies.

Indicates HR and 95% CI.
CM: chronic migraine; EM: episodic migraine; ICHD-II: International Classification of Headache Disorders-II; ICHD-IIR: International Classification of Headache Disorders-II revised version; TM: transformed migraine; ICHD-3b: International Classification of Headache Disorders 3 (beta); BMI: body mass index; PHQ-9: Nine-item Patient Health Questionnaire; SRPD: self-reported physician-diagnosed; MOH: medication overuse headache; NSAIDs: nonsteroidal anti-inflammatory drug; PFN: persistent frequent headache-related nausea; MIDAS: Migraine Disability Assessment Scale.
The Grading of Recommendations Assessment, Development and Evaluation (GRADE) of pooled meta-analysis studies.

Result was obtained by fully adjusted regression model.
With the large effect RR > 2.
With the large effect RR > 5.
The study by Seferoǧlu, 2012 did not provided confounder adjustments.
The study by Seferoǧlu, 2012 did not provided confounder adjustments.
There is great heterogeneity among studies in meta-analysis.
The 95% CIs of the studies by Katsarava, 2004 and Seferoǧlu, 2012 are too wide.
The confidence interval ranges from no risk to CM associated with MIDAS.
The following diagnostic criteria for EM and CM were used in the included studies. For EM, six studies used ICHD-II diagnostic criteria for migraine with a headache frequency as < 15 days per month (25–30). Scher et al. (23) used ICHD-3 beta criteria and others defined EM as meeting criteria for migraine with a headache frequency as < 15 days per month (22,31–33). For CM, seven studies employed Silberstein-Lipton diagnostic criteria (25,27–30,32,33). Seferoǧlu et al. (22) used ICHD-IIR criteria. Ashina et al. (26) used both ICHD-II and ICHD-IIR criteria. Scher et al. (23) used ICHD-3 beta and Silberstein-Lipton criteria. In terms of the geographical settings, eight were from the USA (23,25–30,33), two from Turkey (22,32), and one from Germany (31). For outcome measurement, eight studies (23,26–30,32,33) employed CM as a primary outcome, one used TM as an outcome (25) while one study reported CM or medication overuse headache (MOH) (22) as a combined outcome and one used “chronicity of headache” (31) as an endpoint. There are two studies that did not provide adjusted ORs by multiple regression analysis (22,32) (Table 1).
Risk bias of included studies
The overall methodological quality of included studies was assessed. Results indicated an acceptable quality, with NOS sum scores ranging from 6 to 9 (Table 1 and Supplemental Table 1).
Outcome measures
Reported risk factors or predictors of CM in enrolled studies were age, sex, annual household income, body mass index (BMI), migraine symptom severity score, hot and spicy diet, headache days, days of use of specific medication classes, family headache history (father, sibling), poor acute treatment optimization, persistent frequent headache-related nausea, asthma, cutaneous allodynia, number of noncephalic pain locations, and disability measured by the Migraine Disability Assessment Scale (MIDAS) grade (categorical). Definitions of the reported risk factors are provided in Supplemental Table 2. Effect sizes of the risk factors mentioned above are listed in Table 1 and Figure 4.
Predictors for chronic migraine in a fixed-effects model.
In the pooled analysis of a fixed-effect model, depression [assessed by the Patient Health Questionnaire-9, PHQ-9 (34), a validated self-report instrument assessing criteria for major depressive disorder] (RR, 1.58; 95% CI, 1.35–1.85), monthly headache day frequency ≥ 5 (RR, 3.18; 95% CI, 2.65–3.82), monthly headache day frequency ≥ 10 (RR, 5.95; 95% CI, 4.75–7.46), and cutaneous allodynia (RR, 1.40; 95% CI, 1.23–1.59) increased the risk of new onset CM, while annual household income ≥ $50,000 (RR, 0.65; 95% CI, 0.54–0.79) decreased the risk of CM, see Figure 2. In a random-effects model due to substantial heterogeneity (35) among studies, medication overuse (RR, 8.82; 95% CI, 2.88–27) predicted new onset CM, while MIDAS (RR, 1.10; 95% CI, 0.92–1.32) did not. Data are shown in Figure 3.
Predictors for chronic migraine in a random-effects model. MIDAS: Migraine Disability Assessment Scale. Predictors for chronic migraine determined by included studies not applicable for meta-analysis. *HR (hazard ratio) used by author. EM: episodic migraine; BMI: body mass index; NSAID: nonsteroidal anti-inflammatory drug; MIDAS: Migraine Disability Assessment Scale; PFN: persistent frequent headache-related nausea.
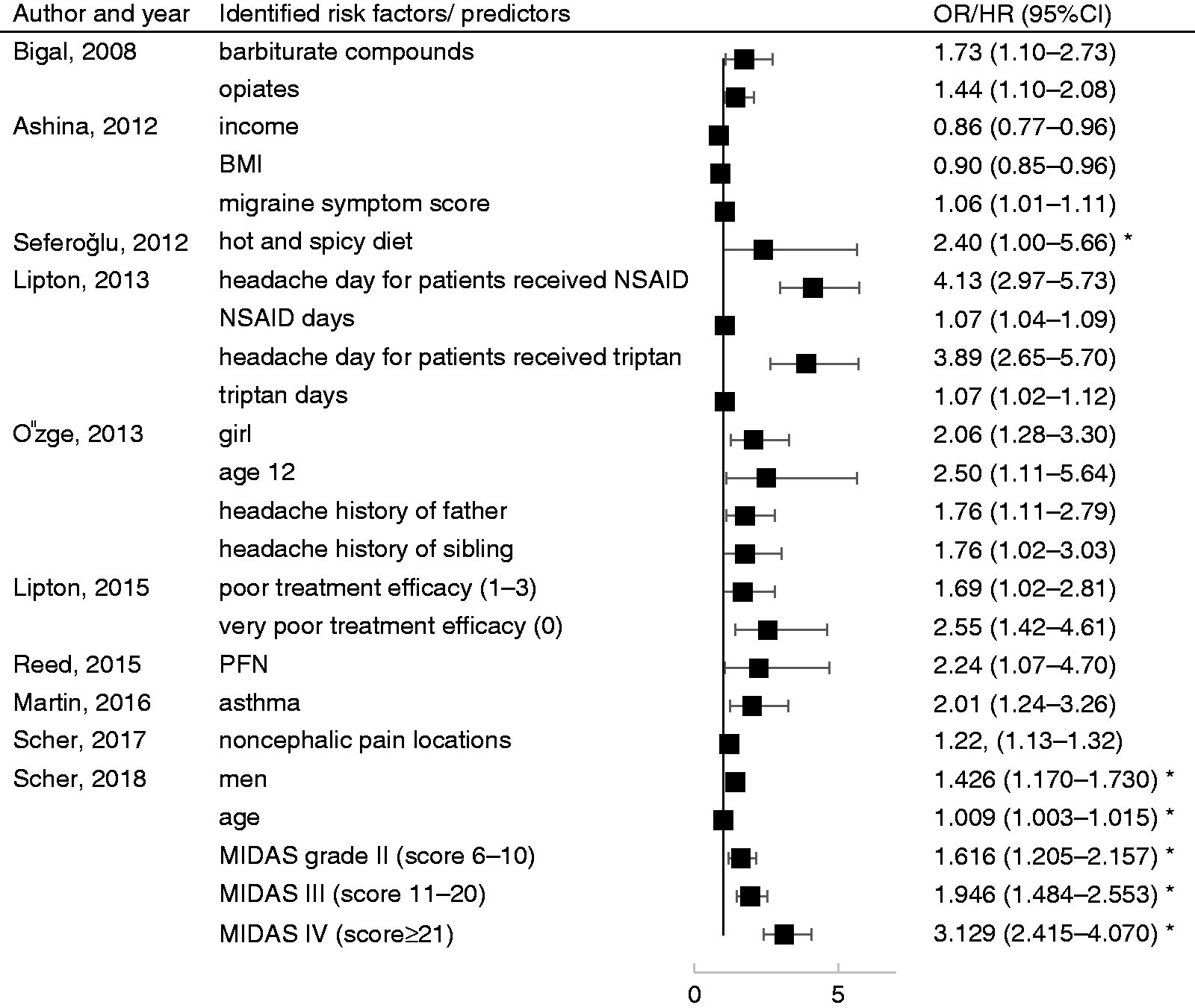
Quality of evidence
Using the GRADE application, monthly headache day frequency ≥ 10 was upgraded as “high” quality evidence for a reduced effect and a very large effect size. The overall quality of evidence was rated as “moderate” for depression, monthly headache day frequency ≥ 5 and annual household income ≥ $50,000. Evidence for allodynia, medication overuse, and MIDAS were graded as “very low”, meaning that estimates of effect were very uncertain for all the predictors considered. The GRADE summary table is provided in Table 2.
Discussion
This meta-analysis of cohort studies found moderate to high quality evidence that high frequency episodic migraine (≥10 headache days per month) increased the risk of CM. Headache ≥ 5 days per month, depression, allodynia, and medication overuse had moderate to low strength of evidence as risk factors for CM, and annual household income ≥ $50,000 was a protective factor against developing CM.
In the pooled analysis, CM was defined differently among the included studies, which may result in a potential bias in evaluating the effect of exposures. However, despite discrepancies, all studies included the criterion of headache on > 15 days per month for ≥ 3 months in the definition of CM. The ICHD-IIR includes the more rigorous criterion of ≥ 8 days of migraine or probable migraine per month in the absence of MO for the definition. This is a difficult criterion to implement both in clinical practice and in large-scale observational studies where individual assessment is not possible. Accurate assessment of this criterion requires prospective daily diaries be kept for at least 3 months. The simplified Silberstein-Lipton criteria provide a useful method for diagnosing CM in clinic. In clinical practice, healthcare professionals would likely provide appropriate acute and preventive therapies to patients meeting the criteria for migraine and experiencing headache on ≥ 15 days per month whether or not they had prospectively documented ≥ 8 headache days with migraine characteristics per month.
Depression is one of the most frequently reported risk factors associated with CM onset across the different research designs (14,36). All studies used the self-reported validated PHQ-9 to assess depression, although some used one of two different severity cut scores (sum score ≥ 15 (26) and ≥ 10 (23,29)). We found that currently meeting criteria for depression as assessed with the PHQ-9 was associated with a 54% increase in relative risk of new onset CM.
We found strong evidence that the higher the frequency of headache days per month, the higher the risk of transformation. Monthly headache day frequency of ≥ 10 had a greater risk than monthly headache day frequency of ≥ 5 in predicting new onset of CM. An average monthly headache day frequency of ≥ 10 days/month is usually considered high frequency episodic migraine, which looks in many ways similar to people with CM (37) and may be managed in similar ways such as considering preventive therapy and behavioral therapies to treat or prevent poor outcomes in functional and emotional disability. An average headache day frequency of ≥ 5 days/month is on the boarder flow and moderate frequency episodic migraine. It is more likely driven extensively by genetic predisposition than environmental risk factors (38–40) and is not associated with the same level of negative outcomes as the high frequency episodic migraine group experiences. For patients with headache day frequency ≥ 5 days/month, acute treatment failure leads to more frequent attacks and contributes to the risk of CM onset (28), which indicates that physicians may be advised to pay more attention to MO and/or acute treatment failure. Recurrent migraine attacks can repetitively activate trigeminovascular neurons and modulatory pain pathways involving the periaqueductal grey, then damage the function or neuronal cell in the periaqueductal grey which is responsible for migraine modulation. This may lead to increased risk of transformation to CM (11). However, the evidence should be considered in light of the fact that there are natural fluctuations that occur in monthly headache frequency, as seen in the CaMEO observational study where monthly headache day frequency was assessed every 3 months over the course of 15 months (41–43). Monthly headache day frequency is also associated with increased headache-related disability (23) and depression, which may share genetic and/or environmental risk factors with CM (26).
Cutaneous allodynia is a marker for central sensitization of nociceptive neurons in the trigeminal nucleus caudalis. Central sensitization of the trigeminal neurons is elicited by persistent pain through activation of meningeal perivascular pain fibers (44). It is reported by 63.2% of people with migraine in the population (45), and has been identified with an increased risk of CM. In our analyses, its strength as a predictor was low; however, this may have been due to limited published data meeting our inclusion criteria. A previous analysis of allodynia from the AMPP study was published only as an abstract presented at a scientific meeting and therefore did not meet the criteria for inclusion in this study. In the abstract, using logistic regression models, cutaneous allodynia was a significant predictor of new-onset CM after adjusting for demographic variables, headache features, and comorbid conditions (OR, 1.7; 95% CI, 1.3–2.3) (46). Louter et al. reported allodynia as a predictor of increasing frequency of headache in people with migraine in the Leiden University Migraine Neuro-Analysis project (11); however, this study was also not included as it did not measure CM as an outcome.
We found that medication overuse was strongly associated with increasing risk of CM, with a large RR. Among the studies in the pooled analysis, Seferoǧlu et al. (22) reported triptan overuse and analgesic overuse separately; however, the authors did not provide a confounder adjustment in Cox regression, which resulted in limitations when assessed by the GRADE application. An analysis of medication use in the AMPP study also demonstrated that increasing days of use/overuse of some classes of medication was associated with an increased risk of new onset CM. There were interactions by the number of headache days per month and of use of certain classes of medication (25). Using acetaminophen as the reference group, individuals who used medications containing barbiturates (OR, 1.73; 95% CI, 1.10–2.73) or opiates (OR, 1.44; 95% CI, 1.10–2.18) had increased risk of CM (referred to as TM). Triptan use at baseline was not associated with increased risk of CM. They found an interaction with NSAID use, in that it was protective against transition to TM at low to moderate monthly headache days, but associated with increased risk of CM among individuals with higher monthly headache days. The relationship between triptan use and increased risk of CM may be related to the fact that calcium-gene-related peptide (CGRP) induced by sustained exposure to triptans has been found to persist after triptan exposure is discontinued, but sensory thresholds returned to baseline (47). This process creates a state of central sensitization. In addition, headache day frequency and medication use obviously travel together and it can be difficult to disentangle the directionality and relationship between days of headache (a powerful predictor of new onset CM) and days of medication use. In summary, despite receiving a rating of “very low” evidence determined by GRADE, which was largely due to methodological limitations, MO had a large effect size and deserves attention by clinicians.
An annual household income of ≥ $50,000 was found to be protective against the new onset of CM. It is likely that individuals with higher incomes have greater access to healthcare and treatment of migraine as well as other comorbidities, psychological conditions, and healthy lifestyle practices. Lower income has been associated with high headache days among people with migraine (48) and an increased risk ratio of chronic headache (49). A positive effect of income inequality on depression was also observed in people with low income (50,51). Depression increases the risk of new onset of CM; therefore, it is reasonable to conclude that low income, mediated through depression or psychological stress, may also result in a higher risk of CM.
We did not confirm a relationship between headache-related disability (MIDAS) as assessed as a continuous variable and risk of CM in the pooled analyses, but when MIDAS was analyzed as a categorical variable, the more severe the headache-related disability, the higher the risk of CM (23). A possible explanation for the different results may be attributed to heterogeneity among studies and a higher proportion of a study population with low MIDAS scores. It is noteworthy that headache-related disability was only measured by MIDAS in the identified studies. There are other instruments available to measure disability, such as the headache impact test (HIT-6) (52,53) and the WHO-Disability Assessment Schedule, second version (WHO-DAS 2) (54), which may yield different results. Furthermore, the severity of headache-related disability was also related to poor treatment optimization (28) and headache day frequency per month, both of which may account for CM.
The NOS score was used to assess the methodological quality of cohort studies as recommended by the Cochrane Collaboration (55). NOS scores of the 11 studies ranged from 6–9, indicating the high quality of the included studies and low risk of bias in the pooled analysis. The weak points in methodology of the lowest scored study (32) were the unrepresentativeness of the exposed cohort and incomparability of cohorts. However, data provided by the referred study were not applicable in the meta-analysis, which would not influence the quality of evidence in the present systematic review.
When assessing the overall quality of evidence, we detected serious inconsistency and a wide range of the pooled 95% CIs from the random-effects model, which may largely be due to a relatively small sample size compared to similar research. Although findings based on observational studies have inherent limitations due to potential confounding, several deficits or limitations in study design could be avoided in order to preserve or even to upgrade the quality of evidence by following GRADE rules. If full adjustment of confounders and sample size calculations were conducted, stronger evidence for the predictive value of medication overuse, allodynia and MIDAS might have been found in the current review.
The risk factors identified in this manuscript are similar to those in the systematic review on the new onset of CM/CDH among people with EM by Buse et al. (14), although ratings of the strengths of each are different. We followed the most recent rating and reporting guidelines, including using the MOOSE guidelines. Risk of bias was assessed using the NOS. Quality of evidence was determined according to the GRADE guidelines. Buse et al. evaluated the strength of evidence for each risk factor according to a modified version of Hill’s criteria of causation (56). They categorized evidence for a particular risk factor as strong if it was replicated across ≥ 2 studies with a documented dose-response relationship, moderate if it was found in ≥ 1 longitudinal study, and fair if it was reported in a single case control study. They reported the strongest support was for higher headache day frequency, depression and MO/high frequency use (14). These findings are similar to the current analysis, which found “high” evidence for monthly headache day frequency ≥ 10 and “moderate” evidence for depression and monthly headache day frequency ≥ 5. In our manuscript, we summarized effect sizes across eligible studies using RRs. This yielded new, never previously published results. We firstly reported that episodic migraine patients with monthly headache day frequency ≥ 10 had a greater risk than monthly headache day frequency ≥ 5 in transformation to CM. The greatest difference is seen in the different ratings between the two studies for the strength of cutaneous allodynia for predicting new onset CM. Probyn et al. examined a similar clinical topic in their systematic review of predictors of outcomes among people with CDH including CM (36). Interestingly, the majority of the factors that they identified as predictors of poor treatment outcomes among people with CDH are the same factors identified as risk factors for progression to CM, including depression and MO.
The identification of potentially modifiable risk factors from the present and previous studies provides an important target for interventions. While we do not have proof that we can prevent progression, it is wise and ethical to intervene with these risk factors, which not only may lead to improved migraine outcomes, but also have consequences for improvements in overall health and quality of life.
This review has several limitations. Due to limited reported data on predictors being available and existing confounders in observational studies, our conclusions need confirmation by future studies. Although data on exposures reported as both continuous and categorical variables (e.g. household income, MIDAS, headache day frequency, etc) resulted in there being a small number of studies in the pooled analysis, the trend of the effect in the meta-analysis would not be revised according to the ORs or HRs obtained from the included studies. Due to lack of similar research, the effect of some variables (e.g. nausea, asthma, hot and spicy diet and noncephalic pain locations) require confirmation by future studies. Largely due to the fact that observational cohort studies on migraine are scarce in China, the majority of the included studies in the present systematic review were conducted in the United States and Europe and published in English, which may lead to potential selection bias. Despite the fact that some subjects included were from the same study sample (26–30), results were obtained from two or more measurement occasions and were repeatable. For instance, the effect of depression, medication overuse, MIDAS and monthly headache days were all reported from different analyses. The two studies that employed CM/MOH (22) and chronic headache (31) as endpoints seemed to result in bias in determining predictors of CM, but the criteria of interest did not vary between those criteria, and the combined outcome of CM or MOH is in accordance with the ICHD-3 diagnostic criteria on CM which would offset the possibility of bias. Additionally, it cannot be determined whether weak evidence for a particular risk factor may truly reflect a weak relationship with progression or may be due to methodological challenges in existing studies or lack of published evidence. It is also likely that additional risk factors exist which have yet to be identified. This may be of particular importance for some psychosocial factors that are not commonly addressed in published studies (such as stress, self-efficacy, and social support), but that have been identified as being important factors in both the development and maintenance of chronic migraine with MO. They have also been identified as risk factors for relapse following withdrawal for CM with MO along with depression, disability and headache day frequency (57,58). Finally, most of the variables included in the studies were self-reported, such as depression, which was identified with a patient questionnaire and not a physician diagnosis or other form of validation.
Conclusions
Strong evidence supports ≥ 10 monthly headache days as a predictor of CM progression. Moderate evidence indicates that depression and ≥ 5 monthly headache days increase the risk, while an annual household income ≥ $50,000 is protective. A “very low” quality of evidence was found for allodynia and medication overuse, which may be attributed to methodological limitations. Limitations of future cohort studies should be minimized by applying adapted methodological evaluation criteria and both continuous and categorical variables should be reported where possible and appropriate.
Clinical implications
Healthcare professionals should remain vigilant for factors that may increase risk the risk of progression to CM among people with EM, including high frequency headache, medication overuse and depression, and treat these conditions when they encounter them. Further studies should be conducted to identify additional risk factors from a range of relevant domains, including but not limited to genetics, headache-related features, treatment-related features and psychosocial factors and to determine whether progression can be avoided by treating the risk factors.
Supplemental Material
CEP883355 Supplemental Material1 - Supplemental material for Predictors of episodic migraine transformation to chronic migraine: A systematic review and meta-analysis of observational cohort studies
Supplemental material, CEP883355 Supplemental Material1 for Predictors of episodic migraine transformation to chronic migraine: A systematic review and meta-analysis of observational cohort studies by Jingjing Xu, Fanyi Kong and Dawn C Buse in Cephalalgia
Supplemental Material
CEP883355 Supplemental Material2 - Supplemental material for Predictors of episodic migraine transformation to chronic migraine: A systematic review and meta-analysis of observational cohort studies
Supplemental material, CEP883355 Supplemental Material2 for Predictors of episodic migraine transformation to chronic migraine: A systematic review and meta-analysis of observational cohort studies by Jingjing Xu, Fanyi Kong and Dawn C Buse in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dawn C Buse has received grant support and honoraria from Allergan, Amgen, Avanir, Biohaven, Lilly, Promeius and Teva. She is on the editorial board of Current Pain and Headache Reports.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
