Abstract
Trigeminal and somatic nociceptive systems were studied in controls (n = 15), episodic migraine (n = 16), analgesics (n = 14) and triptan-induced medication overuse headache (MOH) (n = 15) before and after withdrawal. Patients with MOH and comorbid depressive symptoms and depression without headache were studied to investigate the influence of depression. Trigeminal nociception was studied by simultaneous registration of pain-related cortical potentials (PREP) and nociceptive blink reflex (nBR) following nociceptive-specific electrical stimulation of the forehead. Somatic nociception was evaluated using PREP of upper limbs. We found facilitation of both trigeminal and somatic PREP but not of nBR in MOH, which normalized after withdrawal. No differences were found comparing analgesics vs. triptan MOH. No differences were observed between controls and patients with episodic migraine and depression without headache. A transient facilitation was found of trigeminal and somatic nociceptive systems in MOH, which was more pronounced on a supraspinal level.
Introduction
Medication overuse headache (MOH) has been defined by the International Headache Society (IHS) as a chronic headache occurring on more than 14 days per month following overuse of acute headache medication and which vanishes or at least significantly improves after withdrawal (1). The pathophysiology of headache chronification is very complex and involves impairment of trigeminal nociceptive processing, but also psychological and behavioural abnormalities. Epidemiological and clinical studies clearly suggest that overuse of acute headache medication is the main risk factor leading to chronification of migraine. Approximately 30–50% of patients, however, develop chronic headache without overusing acute headache drugs (2). Psychological comorbidities such as depression, anxiety and failure of pain coping abilities may contribute to headache chronification.
The aim of this study was to investigate pathophysiological mechanisms of impaired trigeminal pain processing in patients with medication overuse headache. As the definition of MOH covers two different clinical entities, tension-type headache (TTH) and migraine, which undergo chronification following overuse of different classes of headache substances or even their combination, we studied only patients with migraine as a primary headache who developed MOH following overuse of analgesics or triptans only. We focused on analgesics and triptans induced MOH for two reasons: first, overuse of these two substance groups outnumbers the others by far, and second, a recent clinical study of our group has demonstrated that patients overusing analgesics usually develop tension-type like headache, in contrast to patients who overuse triptans and develop migraine-like daily headache (3). It is plausible, furthermore, that even after these restrictions a variety of different pathophysiological mechanisms involved in the process of headache chronification should be considered. We aimed to investigate the central trigeminal pain processing. Several electrophysiological studies have demonstrated abnormal trigeminal nociceptive processing (‘functional changes’) in patients with chronic migraine or chronic TTH. Two studies demonstrated morphological changes: Welch et al. found increased iron levels in the periaqueductal grey (4). A very recent study used magnetic resonance imaging and voxel-based morphometry to demonstrate a grey matter decrease in patients with chronic TTH (5). The above-mentioned studies did not differentiate and compare, however, MOH following overuse of analgesics and overuse of triptans.
To probe the function of trigeminal nociceptive system we used the ‘nociceptive’ blink reflex (nBR) and pain-related evoked potentials (PREP) elicited with a novel concentric stimulating electrode as proposed recently. By virtue of its concentric design and small anode–cathode distance, this surface electrode produces a high current density at low current intensities. Thus, depolarization is limited to the superficial layer of the dermis containing nociceptive A-δ fibres, but does not reach the deeper, A-β containing layers (6). We were able to demonstrate that the nBR elicited by the concentric electrode reflected trigeminal nociceptive processing in a quantitative way and was highly sensitive to changes in trigeminal nociception (7, 8). By using spatial (simultaneous stimulation with two electrodes) and temporal (train pulse stimulation) effects, we were able to achieve more robust and persistent responses, which were more sensitive to changes in trigeminal nociceptive processing (9). Very recently we simultaneously recorded the nBR and PREP following stimulation of the forehead in order to examine trigeminal nociceptive processing at the brain stem (nBR) and supraspinal (PREP) levels (10).
Hence, in this study we applied the technique of simultaneous nBR and PREP recording following trigeminal electrical nociceptive stimulation to investigate trigeminal pain processing in patients with migraine who developed MOH following overuse of analgesics or triptans. As mentioned previously, patients with MOH often suffer from depression, which could influence the magnitude of PREP. To control for this possible interference, we separated MOH patients into two groups with and without depressive symptoms and added one control group of patients with depression without headache.
Methods
Approval of the study protocol was obtained from the local ethics committee and informed consent was obtained from each subject before enrolment in the study.
Patient population and study design
Inclusion criteria were age 18–65 years and episodic or chronic migraine according to the second version of the IHS criteria (1). Exclusion criteria were suspicion of symptomatic headache, coexisting TTH, combined overuse of two or several different types of acute headache drugs and intake of serotonin reuptake inhibitors or tricyclic antidepressants. All patients underwent in-patient withdrawal. Overused medication was stopped and preventive treatment with β-blockers or valproic acid was initiated. Patients with chronic migraine with medication overuse who did not improve 6–8 weeks after withdrawal were also excluded.
All patients and controls received a standard interview and neurological examination. Depressive symptoms were assessed by using Beck's depression inventory (BDI). Patients with a BDI score >18 were considered as having ‘depressive symptoms’.
The study population was divided into six groups: Group 1, healthy controls (n = 15); Group 2, episodic migraine (n = 16); Group 3, analgesics-induced chronic migraine without depressive symptoms (n = 14); Group 4, triptan-induced chronic migraine without depressive symptoms (n = 15); Group 5, both analgesics- and triptan-induced chronic migraine with comorbid depressive symptoms (n = 14); and Group 6, patients with depression, without migraine (n = 14).
The study design is illustrated in Fig. 1. In all subjects and patients the trigeminal nociceptive system was studied by simultaneous recording of nBR and trigeminal pain-related potentials. Extracranial (non-trigeminal) nociception was investigated by recording somatic pain-related potentials following stimulation of the hands. Patients with analgesics- and triptan-induced chronic migraine with or without depressive symptoms (Groups 3, 4 and 5) were investigated on the first or second days of in-patient withdrawal in the attack-free state [headache intensity verbal rating scale (VRS) ≤3]. Only patients with analgesics- and triptan-induced chronic migraine without depressive symptoms (Groups 3 and 4) were investigated again 6–8 weeks after withdrawal.

PREP and nBR example graphs for analgesic- (a) and triptan-induced (b) chronic migraine before (lower graph) and after (upper graph) withdrawal.
Electrophysiological settings
For stimulation a custom-built planar concentric electrode (CE) assembly of a central metal cathode (diameter 0.5 mm), an isolation insert (diameter 5 mm) and external anode ring (diameter 6 mm) was used, providing a stimulation area of 19.6 mm2. Trigeminal stimulation was performed with two electrodes placed 10 mm above the entry zone of the supraorbital nerve on the forehead with a distance between electrodes of 2 cm. For eliciting somatic PREP two electrodes were placed on the back of the hand between the first and second metacarpal bones with a distance of 2 cm between them. The individual pain threshold was determined by two ascending and descending 0.2-mA stimulation sequences. Finally, blocks of 15 triple-pulses (monopolar square wave, intensity 1.5-fold of the individual pain threshold, duration 0.5 ms, pulse interval 5 ms, interstimulus interval 12–18 s, pseudorandomized) were applied.
nBR and PREP were recorded simultaneously following the trigeminal stimulation and PREP and an electroculogram were recorded during stimulation at the upper limbs. PREPs were recorded with electrodes placed at Cz referred to linked earlobes (A1–A2) of the international 10–20 system. The nBR was recorded using bilateral surface electrodes placed infraorbitally. The following recording parameters were used: bandwidth 1 Hz to 1 kHz, sampling rate 2.5 kHz, sweep length 300 ms (1401plus, Signal; Cambridge Electronic Design, Cambridge, UK).
Signal analysis was performed by an investigator blinded to the study design. The first sweep was rejected to avoid contamination by startle responses. Thus, 14 sweeps were averaged. We analysed N (negative peak) and P (positive peak) latencies, as well as NP (peak-to-peak) amplitudes of PREP. Onset latencies and area under the curves (AUC) of nBR were analysed on rectified traces. We also analysed individual pain thresholds and subjective pain perception score on the VRS 0–10.
Mean values of the stimulation blocks for each subject and subsequent mean values for the group were calculated. PC-based offline analysis used custom-written software (Matlab 5.1; MathWorks, Natick, MA, USA).
Statistical analysis
One factorial
All statistics were calculated with SPSS software (9.0.1; SPSS Inc., Chicago, IL, USA). The level of significance was set at 0.05.
Results
Demography and clinical characteristics of the study population are given in Table 1. Eleven patients with analgesics-induced and 13 patients with triptan-induced chronic migraine were re-examined after withdrawal. All of them had a significant (>50%) improvement of headache frequency.
Demography and clinical characteristics of the study population

BDI, Beck's depression inventory; CM, chronic migraine.
The groups were similar with regard to age and gender. The headache groups had a comparable history of migraine, duration of chronic migraine and intake frequency in days per month. During the electrophysiological recordings up to 50% of patients suffered from light to moderate headache but none of them had a migraine attack.
Trigeminal nociceptive system
Electrophysiological results are summarized in Table 2a and 2b. Pain thresholds and stimulation intensities did not differ between the groups. No differences were observed in PREP (amplitudes and latencies) or nBR (AUC and latencies) between controls, episodic migraine and depression without headache. A significant increase was found in PREP amplitudes in all patients with chronic migraine (analgesics-induced MOH, triptan-induced MOH and MOH with depressive symptoms), which normalized again after withdrawal. No differences were found between analgesics-induced MOH, triptan-induced MOH and MOH with depressive symptoms. Mean PREP latency was the shortest in patients with analgesics-induced MOH, which, however, did not reach significance. Interestingly, no differences were found comparing nBR latencies and AUC. All patients with MOH had increased pain perception scores. Patients with depression without headache tended to have a higher mean VRS score than patients with episodic migraine.
Changes of nociceptive blink reflex (BR) between groups and between periods: before and after withdrawal

CM, Chronic migraine.
Changes of trigeminal pain-related evoked potentials between groups and between periods: before and after withdrawal
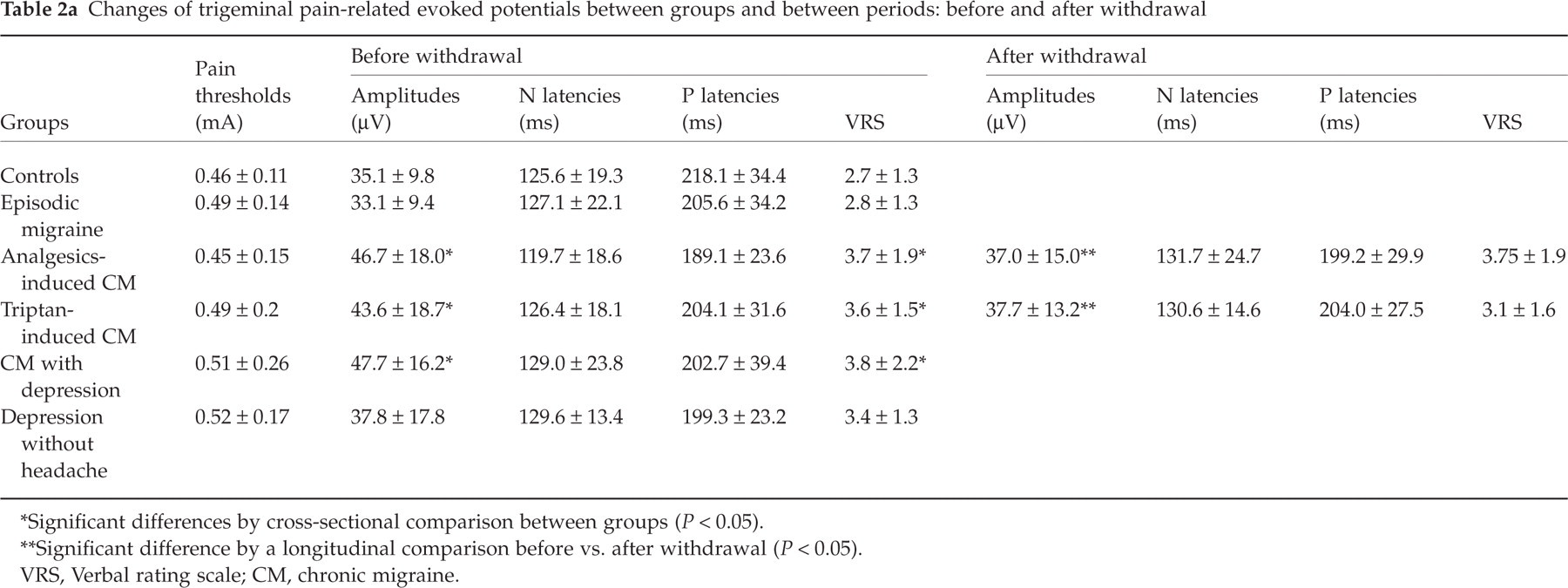
Significant differences by cross-sectional comparison between groups (P < 0.05).
Significant difference by a longitudinal comparison before vs. after withdrawal (P < 0.05).
VRS, Verbal rating scale; CM, chronic migraine.
Somatic nociceptive system
Results are summarized in Table 3. We found no differences comparing pain thresholds and stimulation intensities in any groups. PREP latencies and amplitudes did not differ between controls, episodic migraine and depression without headache. In parallel to changes in the trigeminal nociceptive system, an increase was observed in PREP amplitudes in all patients with chronic migraine, which normalized again after withdrawal. Patients with analgesics-induced MOH had significantly shorter latencies before withdrawal, which returned to normal values after withdrawal.
Changes of somatic pain-related evoked potentials between groups and between periods: before and after withdrawal

Significant difference between groups (P < 0.05).
Significant difference between periods: before vs. after withdrawal (P < 0.05).
VRS, Verbal rating scale; CM, chronic migraine.
Discussion
In this study we addressed several questions: first, the existence of sensitization of trigeminal nociceptive processing in patients with chronic migraine following overuse of analgesics or triptans. Moreover, by using simultaneous recording of nBR and PREP we aimed to differentiate between the brainstem and supraspinal levels. Finally, we sought to find differences in central trigeminal nociception between analgesics- and triptan-induced MOH, since these two headache types are clinically distinct.
Our findings can be summarized as follows. Both trigeminal and somatic nociceptive systems were activated in patients with chronic migraine and normalized again after withdrawal and consequent reduction of headache frequency. In addition, activation of trigeminal and somatic PREP was found, but not of nBR, indicating that sensitization of central nociceptive mechanisms mainly involved structures outside the trigeminal system and probably occurred at the supraspinal level. We found no evidence that facilitation depended on the class of overused medication or the coexistence of depressive symptoms.
The first finding of activation of trigeminal PREP is in line with previous studies of Fusco et al. (11). Another study by de Tommaso et al., using laser-evoked potentials in patients with chronic migraine without medication overuse, demonstrated reduced pain thresholds in patients with chronic migraine without medication overuse (12). Facilitation of laser-evoked potentials was found in patients with chronic TTH also (13). Interestingly we found facilitation not only of the trigeminal but also of the somatic PREP, indicating that the sensitization of the nociceptive system is a general process not confined to the trigeminal system. This is also in line with results of de Tommaso et al., who reported decreased pain and perception thresholds of the face and hands in patients with MOH. A similar generalized nociceptive hypersensitivity was suggested by Bendtsen et al. in a study demonstrating increased pain sensitivity of both cephalic and extracephalic structures in chronic TTH (14).
Another noteworthy finding was the significant augmentation of both trigeminal and somatic PREP but not of the nBR. A possible explanation could be that reorganization and activation of the nociceptive system occurred mainly on a supraspinal level. Mechanisms of central sensitization at second- or third-order neuron level have been shown to be crucial in the pathophysiology of migraine attack (15). Our group was able to demonstrate this phenomenon by recording nBRs during acute migraine attacks (7, 8). Mechanisms of chronification of migraine could be even more complex, involving a network of cortical and subcortical structures with modulatory nociceptive and antinociceptive functions. Processes of cortical reorganization responsible for pain memory have been well described in other chronic painful conditions such as phantom limb pain (16, 17). The anterior cingulated cortex, especially, seems to involved in the processes of cortical plasticity (18). A new study by Fumal et al. has demonstrated this for MOH in particular. In such patients the authors found impaired metabolism in the anterior cingulate and orbitofrontal cortex which normalized after withdrawal (19).
The fact that the cingulate cortex is the main generator of pain-related cortical potentials (20) may explain why trigeminal but also somatic PREP are electrophysiological markers of the process of cortical plasticity.
Recent clinical studies have clearly demonstrated differences in clinical features between patients with analgesics- or triptan-induced MOH (3). In our electrophysiological study we found no differences between these two groups. At this point it is important to underline that we do not know whether the observed facilitation of trigeminal and somatic PREPs—probably reflecting supraspinal reorganization—was the cause or consequence of the chronification of migrainous head pain. It is also plausible that activation of the trigeminal nociceptive system is the last and common pathway for chronification of migraine.
Chronic migraine is often associated with depression, suggesting that both conditions could be causally related and share common neurobiological mechanisms (21). Depressed patients are vulnerable to pain disorders. However, the nature of this relationship and how depression influences pain processing remains unclear. Previous studies on pain thresholds and pain tolerance have produced mixed results (22–27). To our knowledge, no studies of pain-related cortical potentials in patients with depression exist at present. We could detect no interference of depressive symptoms on central nociceptive mechanisms in our sample. We could demonstrate only a trend toward increased subjective pain perception score in patients with depressive symptoms.
We are aware of the limitations of this study. The small size of the groups underpins the explanatory nature of the study, even though we were able to detect significant differences between groups. It was not possible to evaluate possible influences of the two migraine preventive drugs. Electrical stimulation of the skin directly depolarizes nociceptive fibres bypassing peripheral nociceptors. Hence, we could not evaluate changes in peripheral nociceptive transduction.
With the novel technique of pain-related potentials elicited by nociceptive-specific electrical stimulation of the skin, we have confirmed previous findings suggesting facilitation of nociceptive processing not limited to the trigeminal nociceptive system. Simultaneous registration of the blink reflex and pain-related cortical potentials could be a useful tool for studying painful cranial disorders to evaluate pain processing at different levels.
