Abstract
This study investigated nuclear factor-kappa B (NF-κB) activity by electrophoresis mobility gel shift assay and IκBα expression by Western blot analysis in monocytes obtained from serial samples of internal jugular venous blood taken from seven migraine patients without aura during attacks. Inducible nitric oxide synthase (iNOS) expression was also assessed by reverse transcription-polymerase chain reaction. An increase in NF-κB activity peaked 2 h after attack onset. This was accompanied by a transient reduction in IκBα expression. Up-regulation of iNOS was evident at 4 h, maintained at 6 h and reduced at the end of the attack. These findings substantiate the hypothesis of transitory delayed inflammation, as suggested by the animal model, and suggest the possibility of using therapeutic approaches to target NF-κB transcription in the treatment of migraine.
Keywords
Introduction
Nitric oxide (NO), which is produced by different isoforms of NO synthase (NOS) and is involved in cell signalling in many cells and tissues, has been recognized as playing a pivotal role in different physiological and pathological processes. In particular, it has been implicated in migraine, based on findings from both experimental animals and humans. Pivotal findings that are widely accepted in migraine are the increased production of NO in migraine patients between and during attacks, hypersensitivity to exogenous NO and improvement of migraine in two-thirds of patients by the administration of NOS inhibitors (1–5).
Because NO has been linked to inflammation and cytokine expression, Reuter et al. (6) investigated the delayed effects of brief glyceryl trinitrate (GTN) infusion (30 min) on the development of meningeal inflammation in a rat model in doses relevant to the human model. The authors found a dose-dependent, type II inducible NOS (iNOS) up-regulation of mRNA in dura mater and an increase in the corresponding protein expression at 4, 6 and 10 h after infusion. Type II NOS immunoreactivity was expressed chiefly within resident meningeal macrophages. Consistent with development of a delayed inflammatory response, Reuter et al. (6) detected induction of interleukin (IL)-1β in dura mater at 2 and 6 h and increased IL-6 in dural macrophages and cerebrospinal fluid at 6 h after GTN infusion.
In a more recent study carried out in 2002, the same authors confirmed in the rat model increased NO production within macrophages following GTN infusion, and demonstrated that L-N(6)-(1-iminoethyl)-lysine, a selective iNOS inhibitor, attenuates the NO signal, emphasizing the importance of enzymatic activity to delayed NO production. The expression of iNOS was preceded by significant activity of nuclear factor-kappa B (NF-κB), as reflected by reduction of the inhibitory protein IκBα and activation of NF-κB after GTN infusion (7).
The transcription factor NF-κB plays a pivotal role in iNOS induction and controls transcription of acute-phase proteins, including cytokines, adhesion molecules and antioxidant enzymes, among others (8). NF-κB is ubiquitously expressed and consists of homo- and heterodimers (e.g. p65-p50) of Rel family proteins. Under basal conditions, NF-κB is sequestered within the cytoplasm by the IκB family of inhibitory proteins. Noxious stimuli, such as oxygen radicals as well as proinflammatory cytokines, indirectly activate the IκB kinase complex (IKKβ) to phosphorylate IκB proteins. Once phosphorylated, IκB family members dissociate from NF-κB, undergoing degradation by ubiquitin-dependent proteosomes. As a result, NF-κB (p65-p50) translocates into the nucleus and binds specific recognition elements in the promoter regions of inflammatory and stress-induced proteins, thereby activating their transcription (9).
Recent data have emphasized NF-κB involvement in iNOS expression in human macrophages and monocytes, as a consequence of a variety of stimuli, such as lipopolysaccharide (LPS), cytokines such as IL-1β, IL-6, tumour necrosis factor (TNF)-α, as well as oxidative stress (10, 11). Until now, no data have been available regarding the above mechanisms involved in migraine.
This study was aimed at verifying involvement of iNOS in the production of NO and the contribution of NF-κB to iNOS expression during spontaneous migraine attacks such as that shown in the animal model developed by Reuter et al. (6, 7). We evaluated the changes in NF-κB activity and protein expression and IκBα protein expression in monocytes from the internal jugular blood of migraine patients assessed at different time points during attacks. Changes in iNOS protein and mRNA expression and in nitrite production were also assessed in the same cells.
Jugular blood provides, in fact, much more information than peripheral blood regarding biochemical and neurotransmitter changes in the cerebral circulation, which characterize migraine attacks.
Materials and methods
Patients
The protocol was approved by the local Ethics Committee of Umbria.
After providing written consent, seven migraine without aura (MoA) patients (four female, three male), diagnosed according to ICHD 2nd edition criteria (Headache Classification Subcommittee, 2004) (12), were admitted to our Neurological Clinic as in-patients and assessed during their first spontaneous, non-induced attack occurring during the period of hospitalization (mean hospital stay was 7 days). The mean age of the group was 36.5 ± 5.7 years, duration of migraine was 9.7 ± 3.4 years and frequency of attacks in the last 3 months was 2.8 ± 0.9 attacks/month. Exclusion criteria for patients joining the study were: smoking habit, duration of migraine attack <4 h, presence of systemic comorbid pathologies (renal disease, cardiovascular disease, including hypertension, epilepsy) and pregnancy.
Before inclusion in the study, three patients had been taking simple analgesics and the other four patients triptans for symptomatic treatment of their usual attacks with satisfactory relief of headache. None of the patients had been treated with prophylactic drugs during the previous 2 months.
Central venous procedure
For the central venous procedure, internal jugular blood was first examined with a hand-held sonographic transducer to determine the size, patency and location of the internal jugular vein. Access to the right or left internal jugular vein was subsequently achieved (depending on the laterality of head pain; in three patients the right jugular vein, in the remaining patients the left jugular vein) under sterile conditions, under sonographic guidance. Details of the method are reported in a previous paper (3). Briefly, the internal jugular vein was punctured just cephalad to the clavicle using a 21-G needle from the peripherally inserted central catheter (PICC) kit. Once venous access was obtained, a 0.018-inch wire guide was passed under fluoroscopic guidance through the needle, which was then removed. The wire guide was removed after insertion of a 4 or 5 peel-away sheath (from the PICC kit). Only one puncture was required for five patients, while two punctures were required for the other migraine patients undergoing the procedure. In no case was the common carotid artery inadvertently punctured. No periprocedural complications occurred. Post-procedural chest radiographs showed no evidence of pneumothorax, haemothorax or mediastinal haematoma in any of the migraine patients undergoing central venous catheter insertion.
Sampling of jugular venous blood
Internal jugular venous blood samples were taken from all patients within 30 min (T0) of onset of a moderate to severe level of pain, at 1, 2, 4 and 6 h after attack onset and within 2 h after the end of the attack, when the patients were headache free. Pain intensity was scored at each time point using a 4-point scale: 0, no pain; 1, mild pain, does not interfere with usual activities; 2, moderate pain, inhibits but not wholly prevents usual activities; 3, severe pain, prevents all activities. Within the first hour after catheter insertion, the head pain was severe enough to be regarded as migraine (score = 3 in pain intensity). No symptomatic drugs were administered to stop the migraine attack.
Peripheral blood samples were also taken at the same time as internal jugular blood sampling in all migraine patients.
Monocyte separation
Monocytes were chosen because we believed that they are the blood counterpart of macrophages, in which changes of iNOS were found in previous studies by Reuter et al. (6, 7).
Blood mononuclear cells were separated by a Ficoll–Hypaque gradient and monocytes were obtained by counterflow elution using the JE-6B elutriator system (Beckman Instruments, Milan, Italy). Monocytes obtained were 95% pure as determined by staining with Leu-M3 (anti-CDNA) MAb and from cytofluorographic analysis (FACScan V; Beckton Dickinson, Franklin Lakes, NJ, USA).
Transcription factor-binding activity
Nuclear proteins were prepared and assayed for transcription factor-binding activity by electrophoresis mobility gel shift assay (EMSA), using an NF-κB consensus oligonucleotide. Nuclear proteins were harvested and assayed for transcription factor-binding activity using the NF-κB (p65-p50) consensus sequence by EMSA (13).
Briefly, the monocyte buffy coat was lysed with a buffer containing 10 m
Immunoprecipitation of IκBα
Agarose-conjugated anti-p65 antibody (100 µg of IgG/ml) was incubated with whole-cell lysates (200 µg) or nuclear extracts (100 µg) in 100 µl of immunoprecipitation buffer containing 150 m
Western blot analysis
Proteins were electrophoretically transferred onto Westran polyvinylidene difluoride membranes and incubated overnight at 4°C with blocking solution (5% skim milk in PBS). Affinity-purified rabbit antibodies (0.4 µg of IgG/ml) to NF-κB subunits, IκB and iNOS were incubated with the blots overnight at 4°C in PBS buffer containing 0.1% Tween 20. The blots were washed twice with PBS buffer and then treated with anti-rabbit antibody (1 : 4000 dilution) coupled to horseradish peroxidase. Immunodetection was accomplished using an enhanced chemiluminescent system (ECL Plus; Amersham Biosciences, Little Chalfont, UK) using an immunoblot assay kit (Bio-Rad Laboratories GmbH, Munich, Germany). iNOS protein expression at 12 h by mononuclear cells stimulated with LPS (25 µg/ml) was used as a positive control in Western blot analysis.
Data were expressed as the ratio between NF-κB/β-actin, IκBα/β-actin and iNOS/β-actin densitometric values (14).
Reverse transcription-polymerase chain reaction
Reverse transcription-polymerase chain reaction (RT-PCR) for NF-κB p65, iNOS and β-actin was performed as described by Lernbecher et al. (15)
Reverse transcribed cDNA was obtained from 2 µg of total RNA with oligo-dT primers (Pharmacia), AMV-reverse transcriptase and Taq-polymerase (Promega, Milan, Italy). When RT-PCR was performed from monocytes, Taq-polymerase was replaced by PFU-polymerase (Stratagene, Heidelberg, Germany). Amplification was performed using the following primer pairs: iNOS sense, 5′-TCGGA GGCAACAGCACATTCA-3′; iNOS antisense, 5′-GGGTTGGGGGTGTGGTGATGT-3′; p65 sense-1, 5′-GCTACAAGTGCGAGGGGC-3′; p65 antisense-1, 5′-GGGGTCTGCGTAGGGAGGG-3′; p65 antisense-2, 5′-GGCCTGCCTGATGGGTCCC-3′; actin sense, 5′-AGAGGTATCCTGACCCTGAAGTACC-3′; actin antisense, 5′-CCACCAGACAACACTGTGTTGGC AT-3′. The PCR was carried out in an automated thermal cycler (Perkin-Elmer, Norwalk, CT, USA). The following conditions were used: 1 × (95°C, 360 s), 40 × (94°C, 40 s; 65°C, 60 s; 72°C, 120 s), 1 × (72°C, 600 s) for actin; 1 × (95°C, 360 s), 35 × (94°C, 40 s; 68°C, 60 s; 72°C, 120 s), 1 × (72°C, 600 s) for NF-κB p65; 1 × (95°C, 360 s), 1 × (94°C, 60 s; 52°C, 120 s; 72°C, 120 s), 1 × (94°C, 60 s; 58°C, 60 s; 72°C, 120 s), 32 × (94°C, 40 s; 62°C, 60 s; 72°C, 60 s), 1 × (72°C, 600 s) for iNOS. Verification of PCR products was performed by reamplification with different primers recognizing sequences internal to the amplification products and by Southern blot analysis. Electrophoresis of PCR products was carried out on 1.5–2% agarose gels and PCR products were visualized by ethidium bromide staining. iNOS mRNA expression at 12 h by mononuclear cells stimulated with LPS (25 µg/ml) was used as a positive control in RT-PCR and Western blot analysis.
Bands were visualized, photographed and densitometrically quantified using ImageQuant software (Molecular Dynamics, Sunnyvale, CA, USA) and arbitrary iNOS/β-actin ratio bands were calculated. Reactions that lacked template RNA or AMV-reverse transcriptase served as internal controls.
Statistical analysis
Data were expressed as means ± standard error of the mean (SEM) of the indicated number of experiments. Statistical analysis was performed by one-way analysis of variance (
Results
Because NF-κB can bind its DNA recognition site only after translocation to the nucleus, NF-κB activation can be easily and accurately determined by EMSA, a fact that has been successfully exploited by others for in vitro analysis. Accordingly, the ability of EMSA to detect NF-κB activation in vivo was determined in monocytes separated from the internal jugular blood of seven MoA patients assessed during attacks. Analysis showed that all patients had marked activation of NF-κB with a peak at 2 h compared with samples taken at the time of catheter insertion and at the end of the attack (

Nuclear factor-kappa B (NF-κB) monitored by electrophoresis mobility gel shift assay (EMSA) in one migraine without aura (MoA) patient at different time points of the study. Variations in the activity of transcription factor NF-κB monitored by EMSA at different time points in monocytes separated from samples of internal jugular blood from one MoA patient are shown in the autoradiogram. NF-κB activity increased 1 h after the start of the attack, reached its maximum at 2 and 4 h, then tended to decrease after the end of the attack.
Mean (±2 SD) of nuclear factor-kappa B (NF-κB) activity, NF-κB/β-actin mRNA and protein ratios, IκBα/β-actin protein ratio and inducible nitric oxide synthase (iNOS)/β-actin mRNA and protein ratios found at different time points in monocytes of the peripheral and internal jugular venous blood of migraine without aura patients
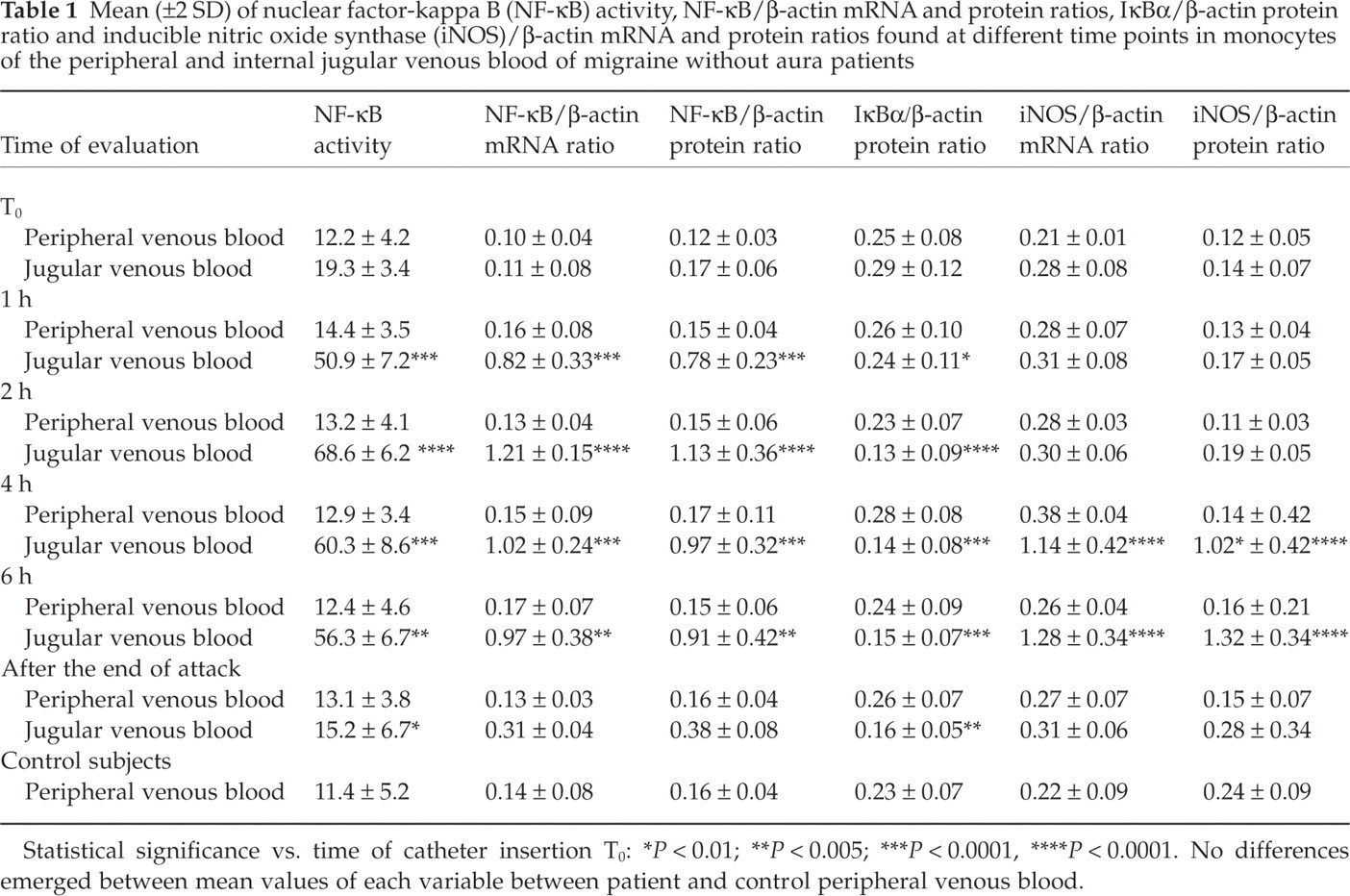
Statistical significance vs. time of catheter insertion T0:
P < 0.01;
P < 0.005;
P < 0.0001,
P < 0.0001.
No differences emerged between mean values of each variable between patient and control peripheral venous blood.
Similar results were obtained for the expression of NF-κB mRNA in migraine patients, as can be seen in Fig. 2, depicting the NF-κB and β-actin bands of a migraine patient during the attack at each time point. The same is true for NF-κB protein, as can be seen from the time course of changes in NF-κB/β-actin protein ratios in Table 1.

Nuclear factor-kappa B (NF-κB) mRNA measured by reverse transcription-polymerase chain reaction (RT-PCR) in one migraine without aura patient at different time points of the study. The expression of NF-κB mRNA measured by RT-PCR in the monocytes of one migraine patient at each time point of internal jugular blood sampling is shown. Total RNA was prepared and each preparation was analysed by RT-PCR with primers specific for NF-κB p65 (top, 680 bp) and β-actin (bottom, 720 bp).
The expression of the inhibitory protein IκBα measured by Western blot showed in all patients an opposite trend to that of NF-κB, with lower values of the arbitrary IκBα/β-actin ratio just at 1 h. These values continued to be lower at 2, 4 and 6 h (P < 0.001) compared with the time of catheter insertion (Fig. 3). No changes in NF-κB activity and expression or of IκBα expression were found in monocytes separated from the peripheral blood of MoA patients at any time point (Table 1).

IκBα and β-actin protein expression in monocytes from one migraine without aura (MoA) patient at different time points of the study. IκBα and β-actin immunoblots from monocyte extracts of one MoA patient at each time point during the attack. IκBα at different time points in monocytes separated from internal jugular blood samples from one MoA patient assessed during the attack is shown. The signal intensity reached its minimum at 2 and 4 h, i.e. at the time of greater NF-κB activity.
RT-PCR yielded a greater expression of the iNOS-specific amplification product of 462 bp at 4 and 6 h after attack onset (results of one patient are shown in Fig. 4) and these changes in iNOS expression were similar in all migraine patients, as shown by the similar time course of the iNOS/β-actin ratios (P < 0.0002) (Table 1). A similar trend was also observed for iNOS protein at the same time points for all MoA patients (Fig. 5) (Table 1).

Inducible nitric oxide synthase (iNOS) and β-actin mRNA expression in monocytes from one migraine without aura patient at different time points of the study. The bands obtained by the amplification of iNOS mRNA and β-actin with reverse transcription-polymerase chain reaction in monocytes of one migraine patient at each time of jugular blood sampling are shown. The expression of β-actin in monocytes is similar at each sampling time. Greater expression of the iNOS-specific amplification product of 462 bp was found at 4 and 6 h after attack onset.

Inducible nitric oxide synthase (iNOS) and β-actin protein expression in monocytes of one migraine without aura patient at different time points of the study. The immunoblot time course of iNOS and β-actin in monocytes from jugular blood samples taken during an attack is shown. The expression of β-actin protein in monocytes is similar at each sampling time. As with mRNA, an increase in iNOS protein expression was observed at 4 and 6 h after attack onset.
Analysis of monocytes separated from the peripheral blood of MoA patients showed no variation either in NF-κB activity and expression or in IκBα and iNOS expression (Table 1).
Discussion
The present study has shown an increased activity of NF-κB during migraine attacks in monocytes from the jugular venous blood of MoA patients at 2 and 4 h after attack onset. Conversely, a significant reduction in expression of the inhibitory protein IκBα was observed at the same times. Up-regulation of iNOS in the same cells was evident later, peaking at 4 and 6 h after the start of the attacks. It was accompanied by an increased basal and LPS-stimulated production of NO metabolites.
Overall, our data suggest increased production of NO by monocytes of MoA patients assessed during attacks due to up-regulation in iNOS expression secondary to the transient increase in NF-κB activity. This could be due to ubiquitination and rapid degradation of the inhibitory protein IκB by the IκB kinase complex (IKKβ), after dissociation of the IκB–NF-κB complex, as supported by the reduction in its expression at the time of greater NF-κB activity.
The IKK KA assay could be helpful in future experiments to verify if the enhancement of NF-κB activity occurs via the IKKs.
These findings parallel those obtained in the animal model developed by Reuter et al. (7), who demonstrated an increase, although transitory, in the activity of transcription factor NF-κB. This was reflected in a reduction of the inhibitory protein IκB and activation of NF-κB in macrophages of rodent dura mater after administration of the NO donor, GTN, which preceded the transcriptional up-regulation of iNOS. If the results of Reuter et al. focused on changes in resident meningeal macrophages, we extended the evidence of inflammatory, though transitory, changes to immune cells, in particular, the circulating counterpart of macrophages, i.e. monocytes, in the jugular venous blood during spontaneous migraine attacks.
The central role in the inflammatory transitory events found in MoA patients during attacks is certainly represented by the transcription factor NF-κB, which has been directly linked to the induction of genes encoding proteins involved in the inflammatory cascade (9). It has been clearly demonstrated that the activation of circulating inflammatory cells is dependent on adequate and appropriately timed increases in the expression of interleukins and cytokines induced by NF-κB. NF-κB is necessary and sufficient to activate transcriptionally monocyte chemoattractant protein-1, vascular cell adhesion molecules and intracellular adhesion molecule-1, which appear critical for vascular wall recruitment of inflammatory cells from the circulation, including the cerebral circulation (16). Metalloproteinase genes are also vigorously induced by NF-κB. Factors capable of activating NF-κB have been identified adjacent to NF-κB within the cerebral vessel wall and in the cerebral circulation. They include the cellular products IL-1β, IL-11, IL-17, TNF-α and intracellular signalling molecules such as protein kinase C and other stimuli, such as oxygen radicals, heparan sulphate, transitory exposure to oxidized low-density lipoprotein and mechanical stretch (17, 18). The capacity for these stimuli to activate NF-κB is cell type- and milieu-specific. Moreover, the degree of NF-κB regulation depends upon cell-specific intracellular and extracellular negative and positive feedback loops. This may render the inflammatory events primed by NF-κB stable and self-maintaining, as in some inflammatory or autoimmune diseases, or transitory, as in the case of migraine, as suggested by our results.
Increased oxidative stress and enhanced endothelial NO production could be responsible for the early activation of NF-κB during migraine attacks; NF-κB, in contrast, can be responsible for a transitory proinflammatory response, including transient up-regulation of the proinflammatory cytokines (IL-1β, IL-6 and TNF-α), as shown in a recent study (19), as well as that of iNOS expression and perhaps cyclooxygenase expression.
It could be postulated that self-limited stimulation of NF-κB initiates the inflammatory response in the ‘primed’ cells of the cerebral circulation, which is, however, a transitory phenomenon. In response to TNF-α, IL-6 and IL-1β, induced by NF-κB activation, monocytes and lymphocytes produce IL-10 and IκB, which significantly limit NF-κB activation. NO produced in greater amounts during attacks could also down-regulate the expression of NF-κB-dependent proinflammatory genes through induction and stabilization of IκB.
NO may have, in fact, dual regulatory effects on NF-κB. Evidence suggests that NO inhibits activation of NF-κB through induction and stabilization of the NF-κB inhibitor, IκB (20). The mechanism for this NO effect seems to be independent of guanylyl cyclase activation, since treatment with the cGMP analogue 8-bromo-cGMP did not affect NF-κB activation. Conversely, there are other studies which reportedly show that NO activates rather than inhibits NF-κB in human peripheral blood mononuclear cells (21). The discrepancy between these results may be due to differences in culture conditions (stimulated or unstimulated cells). In some cases this may lead to activation of other signalling pathways, which may ultimately lead to activation rather than to inhibition of NF-κB.
In the case of migraine, we can hypothesize that NO produced by the cerebral vessel wall, as shown by results from a study of our group (3), can contribute to the earlier marked NF-κB activation in monocytes of the cerebral circulation. However, NO produced later by the same mononuclear cells, secondary to up-regulation of iNOS by NF-κB activation, can contribute with other inflammatory substances, including inflammatory cytokines, to self-limitation of the activation of NF-κB, which is the mechanism by which NO production is ultimately terminated. IκB also regulates expression of the genes encoding itself and IκB, and is thus capable of autoregulation.
Evidence for a role of NF-κB in mediating the cellular effects in response to different stressful conditions that activate the neuroendocrine axis comes from studies carried out in healthy volunteers undergoing laboratory stress testing. Other evidence comes from studies of women stressed by the experience of breast biopsy, an extreme life-threatening stressful situation characterized by anxiety and desperation, not comparable to other forms of psychosocial stress (13, 22–24).
However, it can be argued that the potential mechanism for monocyte activation during transit through the brain in the course of a migraine attack should also be expressed in the peripheral blood, due to the fact that NF-κB and iNOS are not known to be short-lived phenomena and the whole body blood circulates through the brain in roughly 5 min, i.e. more than 70 times in 6 h (the time of our observation).
To explain this discrepancy, it may be hypothesized that intervention of an unknown factor in jugular blood, which is rapidly cleared from systemic blood and activates monocytes in vitro after blood sample collection, is responsible for increased NF-κB and iNOS expression. The nature of this unknown factor needs to be investigated in future research.
One of the major concerns regarding the increase in NF-κB activity found in monocytes from the internal jugular blood of our MoA patients is that it could be due to the trauma induced by catheter insertion. Some experimental data suggest that procedures such as balloon angioplasty-induced activation of NF-κB contribute to lumen loss, probably through induction of an inflammatory response, and that prevention of this NF-κB activation may inhibit neointimal formation and human restenosis (25). However, to our knowledge no data are available using the central venous procedure.
It was impossible for ethical reasons to obtain internal jugular venous blood from healthy subjects, and we therefore could not present the time course of NF-κB activity and expression, IκB/β-actin ratios and iNOS expression related to a physiological condition. Thus, the lack of an adequate control group remains the principal limit of the present study, which cannot be overcome due to the impossibility of applying an invasive procedure to a healthy control group purely for research purposes.
The findings of the present study suggest that targeting the inflammatory response by selective inhibition of NF-κB-driven transcription or downstream proinflammatory gene expression offer promising therapeutic approaches to the treatment of migraine. As already emphasized by Reuter et al. (7), two treatments can be mentioned. Aspirin, the most widely used anti-inflammatory drug for the treatment of migraine attack, has been recognized to block activity of the IκB kinase complex (IKKβ), in addition to inhibiting cyclooxygenase activity. Interestingly, anti-inflammatory levels of acetylated and non-acetylated salicylic acid in culture block the availability of NF-κB for transcriptional activation (26). Parthenolide, which has been demonstrated to reduce the frequency of migraine attacks when administered as a prophylactic drug, also inhibits activation of IκB, similar to other sesquiterpene lactones (27, 28). Furthermore, it can inhibit NF-κB-driven transcription by alkylating p65 homodimers, thereby preventing its transactivation (29).
Acknowledgements
The authors thank John A. Toomey for editing the English and Marisa M. Morson for technical assistance.
